Reusable Fe3O4 and WO3 immobilized onto montmorillonite as a photo-reactive antimicrobial agent†
A. I. Mazrad Zihnila,
Insik In*ac and
Sung Young Park*ab
aDepartment of IT Convergence, Korea National University of Transportation, Chungju 380-702, Republic of Korea
bDepartment of Chemical and Biological Engineering, Korea National University of Transportation, Chungju 380-702, Republic of Korea. E-mail: parkchem@ut.ac.kr
cDepartment of Polymer Science and Engineering, Korea National University of Transportation, Chungju 380-702, Republic of Korea
First published on 23rd May 2016
Abstract
Active near-infrared (NIR) tungsten oxide (WO3) and iron oxide (Fe3O4) were immobilized in exfoliated montmorillonite (MMT) by the intercalation of 2-chloro-3′,4′-dihydroxyacetophenone and 1,3-propanesultone quaternized poly(dimethyl amino)ethyl methacrylate (C/S-PDMA) via ionic exchange reaction. UV-Vis spectroscopy, Fourier transform infrared spectroscopy, X-ray diffraction, field-emission scanning electron microscopy and energy-dispersive X-ray spectroscopy were used to confirm the construction of nanocomposites of an enlarged MMT layer with WO3 and Fe3O4 by C/S-PDMA based on catechol chemistry. The metal oxides-immobilized MMT showed the colloidal stability and photothermal effect required for NIR mediated nanocomposites. The NIR mediated nanocomposites showed a high efficiency photothermal effect within 2 min towards both Gram positive (S. aureus) and Gram negative (E. coli) bacteria. Study of the recycled exfoliated MMT by the WO3 and Fe3O4 complexed polymer study disclosed rapid and effective killing for both strains of bacteria within 4 min of NIR irradiation, in which almost 100% of both strains of bacteria were destroyed even after three cycles. This approach is promising for effective, rapid, and stable antibacterial photothermolysis agents.
1. Introduction
The increasing existence of pathogenic microorganisms, including toxic chemical agents and failure of typical antibiotics, has become a global concern in many fields such as medical devices, water purification systems, textiles, and food packaging and storage.1,2 Hence, the development of alternative and reliable methods for rapidly eradicating bacteria have been studied to find promising antibacterial agents as tools. The benefits of nanomaterials for extensive applications have been growing, and one prominent use is to destroy the viability of pathogenic bacteria. Smart nanomaterial based antibacterial agents have shown promising results; they can form a pathogen detection substance to kill or inhibit growth of bacteria.3 Recently, a study demonstrated an alternative antibacterial system based on nanocomposites dispersed within a polymer/inorganic matrix of exfoliated clay; the properties of the resulting material were optimized, and the materials were found to be environmentally friendly. Park et al. reported that the exfoliation of montmorillonite (MMT) by IR825 mediated near-infrared (NIR) irradiation increases its thermal stability.4,5 Exfoliating MMT by such a polymer can enhance its thermal stability and the mechanical properties through an ionic exchange mechanism. Changing of d spacing indicates successful polymer intercalation.6,7 To exfoliate MMT, amino acids, primary aliphatic amines and quaternary ammonium salts were used, because their positive charge allowed for cation exchange in the MMT layer.8,9 Intercalated organic polymers inside MMT clay have been commonly investigated, but organic–inorganic complexed polymer/MMT has been rarely studied, and notably, the stability of exfoliated MMT mediated NIR irradiation has been used in a limited area of antibacterial research.Common ways to combat pathogenic bacteria were so ineffective that there was an urgent demand for further search for a rapid killing system. Expedient means of eradication of bacteria have used NIR as a light source for photothermal agents that can significantly kill pathogenic bacteria by hyperthermia therapy, by changing optical energy to heat.3,6 NIR exposure activated in the range 700–1100 nm effectively penetrates the targeted antibacterial agent, which then releases heat to damage the pathogenic bacterial cell. Lately, photothermal agents, such as carbon derivatives, fluorescent composites, and a group of metal oxides, have been successfully used in photothermal therapy (PTT).10 Metal oxides such as tungsten oxide (WO3) were reported to possess energy levels suitable for d–d transition leading to a localized surface plasmon resonance (LSPR) phenomenon, and therefore they are able to absorb NIR light for PTT.11–13 The extensive use of nanoparticles (NPs) in any scope generate environmental problems, hence recyclable agents need to be fabricated. Park et al. have studied exfoliated MMT composites with sultone group conjugated PDMA to attack microbes for antifouling with excellent stability in an aqueous solution resulting from its zwitterionic character and surface adhesive properties from the catechol moiety.14–17 Several types of iron oxides, such as Fe3O4, α-Fe2O3 and γ-Fe2O3, have magnetic behaviors proven to be useful in bio-applications, including magnetic bioseparation and detection of biological entities, due to their biocompatibility.18 In the antibacterial scenario, our previous study revealed that iron oxide (Fe3O4) coated with catechol-conjugated poly(vinylpyrrolidone) sulfobetaine assembled to poly(3,4-ethylenedioxythiophene) exhibited reusability as a PTT agent for killing bacteria.19
Herein, we report the study of a recyclable MMT exfoliated by the polymer PDMA for bacterial killing system based photothermolysis using the metal oxides WO3 and Fe3O4 based on catechol chemistry. The WO3 and Fe3O4 complexed with PDMA in exfoliated MMT are necessary materials for obtaining superlative conversion of light to thermal energy, and the magnetic properties of Fe3O4 provide the recyclability and good stability in an aqueous solution. NIR light absorption in the hybrid polymer nanocomposites is a promising treatment that rapidly and effectively destroys pathogenic bacteria.
2. Experimental
2.1 Materials
2-(Dimethylamino)ethyl methacrylate (DMA), 2-mercaptoethanol (CTA), azobisisobutyronitrile (AIBN), 2-chloro-3′,4′-dihydroxyacetophenone (CCDP), 1,3-propanesultone, montmorillonite (MMT), tetrahydrofuran (THF), ethanol, nitrogen, deionized water, phosphate-buffered saline (PBS), diethyl ether, hexane, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), MRS, Lysogeny Broth (LB), and agar were purchased from Sigma-Aldrich, Yongin-city, Kyunggi-Do, South Korea. Iron(II,III) oxide (Fe3O4) in nanopowder form with particle sizes of 50–100 nm was purchased from Sigma-Aldrich, Yongin-city, Kyunggi-Do, South Korea. CsWO3 was obtained from the Korea Institute of Industrial Technology (KITECH). Synthesized CsWO3 particles were observed as being 30–100 nm. Propidium iodide and SYTO 9 were purchased from Molecular Probe, Life Technologies (Invitrogen).2.2 Immobilization of Fe3O4 and WO3 with C/S-PDMA/MMT
The exfoliated MMT with (catechol/zwitterionic) quaternized PDMA (C/S-PDMA) was synthesized following our previous method.14 First, to immobilize Fe3O4 on C/S-PDMA/MMT, 0.025 g of the metal oxide Fe3O4 were dispersed in 5 mL of THF. Then, this solution was added dropwise to 0.25 g of C/S-PDMA/MMT dissolved in 30 mL of ethanol. The resulting mixture was stirred for 24 h at room temperature. At the end of the reaction, the solvent was evaporated in a rotary evaporator and hexane was used to precipitate the product. The product was then centrifuged to obtain a nanoparticle pellet. The C/S-PDMA/MMT/Fe3O4 was then filtered and freeze dried.Finally, the immobilization of WO3 with C/S-PDMA/MMT/Fe3O4 was prepared in the ratio of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 WO3 (0.025 g) and C/S-PDMA/MMT/Fe3O4 (0.05 g) were dispersed in pH 8.5 PBS solution. The mixture was stirred for 24 h at room temperature. The product was then centrifuged to obtain a nanoparticle pellet. The resulting C/S-PDMA/MMT/Fe3O4
5 WO3 (0.025 g) and C/S-PDMA/MMT/Fe3O4 (0.05 g) were dispersed in pH 8.5 PBS solution. The mixture was stirred for 24 h at room temperature. The product was then centrifuged to obtain a nanoparticle pellet. The resulting C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 composite was then filtered and freeze dried.
WO3 composite was then filtered and freeze dried.
2.3 Characterization
Scanning electron microscopy (SEM) was carried out with an SNE-3000M (SEC). The UV-Vis-NIR spectra of the samples were obtained with a UV-Vis absorption spectrometer (UV-Vis, Optizen 2120 UV spectrophotometer, Mecasys, Yuseong-gu Daejeon, South Korea). X-ray diffraction spectra were obtained using an XRD Bruker AXS ADVANCES D-8. The NIR laser was 808 nm (PSU-III-LRD, CNI Optoelectronics Tech. Co. LTD, China). Infrared spectra of the polymer were obtained using an FTIR spectrometer (Nicolet iS10 FT-IR spectrophotometer, Thermo Scientific, Madison, WI USA). Photo-thermal heating curves were examined using an infrared camera (NEC Avio, Thermo Tracer TH9100). Field-emission scanning electron microscopy (FE-SEM) micrographs were obtained using a SEM/EDX, JSM-6700F, JEOL, Musashino, Akishima, Tokyo, Japan. Thermogravimetric analysis (TGA, TA Instruments 4100, Lukens Drive, New Castle, USA) was performed using a TGA-DSC1 system (Star; Mettler Toledo) in an inert N2 atmosphere and temperature transfer rate of 10 °C min−1. X-ray photoelectron spectra (XPS) were obtained using an ESCALAB apparatus (Omicrometer, Taunusstein, Germany) and used an system (PHI Quantera-II, Ulvac-PHI, Chigasaki, Kanagawa, Japan). Live/dead bacterial analysis was using a LSM510 confocal laser scanning microscope CLSM (Carl Zeiss, Germany) at a laser wavelength of 405, 488, and 543 nm with an emission filter and 20× magnification. The magnetic properties of the sample were characterized using an alternating gradient magnetometer (AGM; 2900-02 AGIM, PMC Co). The identification and the quantification of the metal component were measured by inductively coupled plasma mass spectrometry (ICP-MS) Bruker 820-MS, Bruker, Fahrenheitstr. 4 D-28359 Bremen, Germany.2.4 Antibacterial activity
Stock solutions of S. aureus (Gram-positive, strain ATCC 25323) and E. coli (Gram-negative, strain ATCC 25922) were prepared in LB and MRS broth (50 mL) medium, respectively, and incubated at 37 °C for 24 h at 108 cells per mL with varying concentrations of C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3. At the end of the incubation period, the nanocomposite treated S. aureus and E. coli strain were irradiated with the 808 nm NIR laser for 1 to 4 min. The bacterial cells were then spread in Petri dishes for evaluation of the growth inhibition. Bactericidal effect was defined as a decrease in the CFU mL−1 after 24 h. For the control, both types of cells were used without adding C/S-PDMA/MMT/Fe3O4
WO3. At the end of the incubation period, the nanocomposite treated S. aureus and E. coli strain were irradiated with the 808 nm NIR laser for 1 to 4 min. The bacterial cells were then spread in Petri dishes for evaluation of the growth inhibition. Bactericidal effect was defined as a decrease in the CFU mL−1 after 24 h. For the control, both types of cells were used without adding C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3.
WO3.
2.5 Recycling antibacterial ability after NIR exposure of the C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) WO3
WO3
The C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 was incubated with a 1 mL cell suspension of both E. coli and S. aureus (105) in a 2 mL Eppendorf (EP) tube at 2 W m−2 for 4 min (808 nm laser). Later, the tube was placed in a magnetic separator for ∼2 min. The supernatant was removed and then metal nanocomposites attracted with a magnetic field were freeze-dried for later uses. Supernatant (0.1 mL) was diluted appropriately and plated onto solid LB and MRS solid media and then incubated at 37 °C for 24 h. The dried metal nanocomposites were incubated with fresh strains of both bacteria in a similar EP conical tube, followed by NIR exposure for 4 min (808 nm laser). The method was repeated thrice.
WO3 was incubated with a 1 mL cell suspension of both E. coli and S. aureus (105) in a 2 mL Eppendorf (EP) tube at 2 W m−2 for 4 min (808 nm laser). Later, the tube was placed in a magnetic separator for ∼2 min. The supernatant was removed and then metal nanocomposites attracted with a magnetic field were freeze-dried for later uses. Supernatant (0.1 mL) was diluted appropriately and plated onto solid LB and MRS solid media and then incubated at 37 °C for 24 h. The dried metal nanocomposites were incubated with fresh strains of both bacteria in a similar EP conical tube, followed by NIR exposure for 4 min (808 nm laser). The method was repeated thrice.
2.6 Live and dead bacteria fluorescence and SEM cell imaging
Bacterial suspension containing 105 CFU mL−1 (1 mL) was centrifuged (4000 rpm for 5 min) and re-suspended in PBS solution. Then, 5 μL of the bacterial suspension was mixed with 20 μL of a fluorescent probe mixture containing 3.0 μM green fluorescent nucleic acid stain SYTO 9 (Invitrogen, USA) and 15.0 μM red fluorescent nucleic acid stain PI (Sigma-Aldrich, USA). C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 (1 mg) was incubated with 1 mL of bacterial suspension of both S. aureus and E. coli (105) in a 2 mL EP tube at 2 W m−2 for 4 min (808 nm laser). A 5 μL aliquot was incubated in the dark for 15 min and then placed on a glass slide, which was then covered with a cover slip, sealed, and examined under an LSM510 confocal laser scanning microscope (Carl Zeiss, Germany) equipped with 405, 488, and 543 nm He Ne lasers. The abovementioned method was followed for fluorescence cell imaging.
WO3 (1 mg) was incubated with 1 mL of bacterial suspension of both S. aureus and E. coli (105) in a 2 mL EP tube at 2 W m−2 for 4 min (808 nm laser). A 5 μL aliquot was incubated in the dark for 15 min and then placed on a glass slide, which was then covered with a cover slip, sealed, and examined under an LSM510 confocal laser scanning microscope (Carl Zeiss, Germany) equipped with 405, 488, and 543 nm He Ne lasers. The abovementioned method was followed for fluorescence cell imaging.
For SEM imaging, the bacterial suspensions were centrifuged followed by washing and re-suspending in PBS buffer. A few microliters (μL) of the untreated and treated bacteria were then dropped onto a silicon wafer and incubated for 3–4 h for drying. The dried treated bacteria on the silicon wafers were used to obtain SEM micrographs.
3. Results and discussion
The utilization of NIR irradiation has become essential in photothermal treatment (PTT), owing to its competence in penetrating biological tissue and it can trigger some materials to release heat after absorption of its light.20 In this study, organic–inorganic polymers scattered in MMT were examined to get photothermolysis material for antibacterial agents by utilizing NIR irradiation of CCDP/sultone group quaternized PDMA (C/S-PDMA).14 The adhesive property of the modified PDMA was obtained from catechol moieties within 2-chloro-3′,4′-hydroxyacetophenone (CCDP) after the quaternization reaction to incorporate CCDP into the polymer backbone, and the zwitterionic structure of the 1,3-propane sultone to enhance the chain mobility in an aqueous medium was subsequently introduced through a ring opening reaction.14 The exfoliation of MMT by the intercalation of (C/S)-q-PDMA in the interlayer of the MMT was accomplished through an ion exchange reaction. The high polarity of water as the solvent to disperse MMT caused swelling and cracking of the MMT sheets, and the prepared cationic polymer displaced and intercalated into that silicate interlayer through cationic exchange. Moreover, the presence of the sultone group in the polymer backbone increased interaction between the clay particles and polymer through its mechanical properties.21 The remaining catechol moieties were used to immobilize both metal oxides Fe3O4 and WO3 in the exfoliated MMT layer at basic conditions (pH 8.5) through the metal complex interaction. To establish the recyclable antimicrobial PTT agent of the MMT composite, WO3 was used as a strong localized surface plasmon resonance (LSPR) agent to get optimum PTT effect and the Fe3O4 gathered in PDMA activated the mussel-inspired adhesive catechol groups.16,17 The detailed experimental system is illustrated in Scheme 1.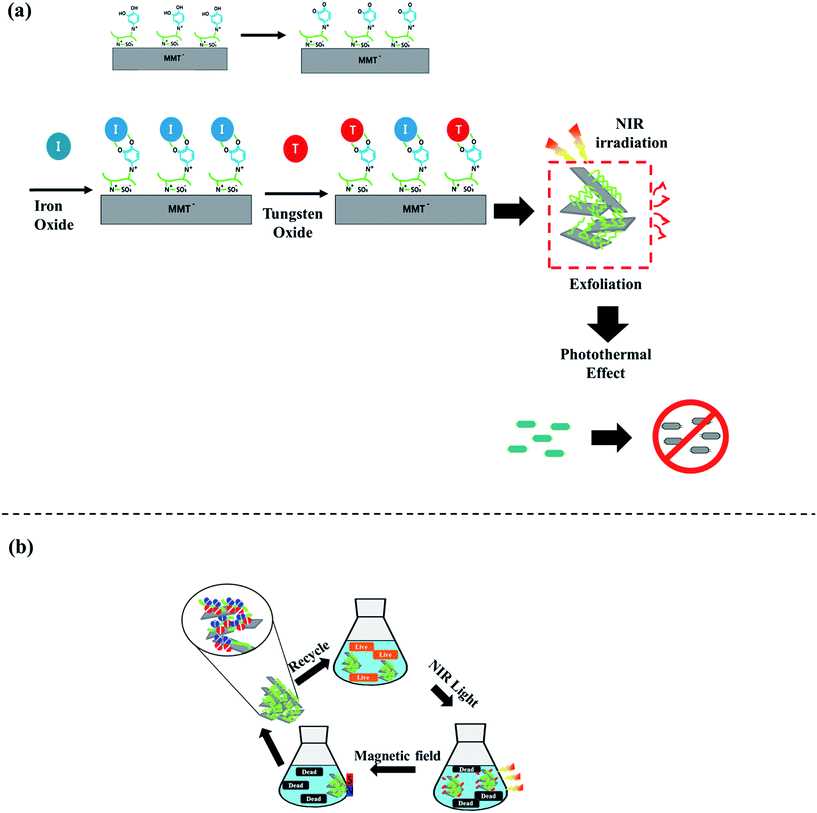 | ||
Scheme 1 (a) The synthesis routes of the C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 and (b) schematic of recycling of C/S-PDMA/MMT/Fe3O4 WO3 and (b) schematic of recycling of C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3. WO3. | ||
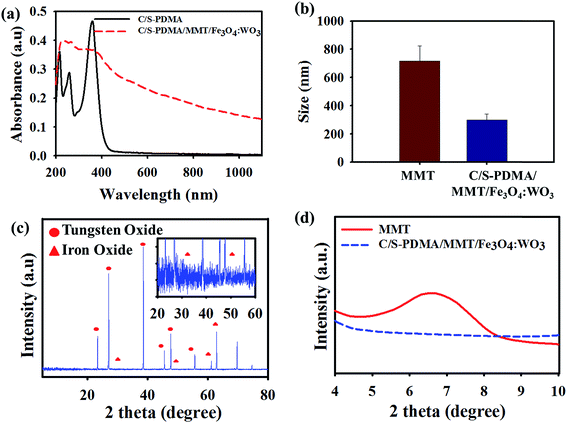
The immobilization of Fe3O4 and WO3 with C/S-PDMA/MMT formation was proved by the UV-Vis, FT-IR, XRD, and X-ray photoelectron spectroscopy (XPS) analyses. Light absorption properties of C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 were recorded by UV-Vis-NIR spectroscopy, assuring the presence of catechol groups through peaks in the range 250–300 nm, which indicate π–π* electron transitions in phenolic compounds.22 Furthermore, the wide spectrum from UV-Vis to NIR area agree with the characteristic of strong LSPR of WO3 in the prepared antibacterial agents, which was able to absorb NIR irradiation in the range of ∼780–1000 nm (Fig. 1a).23 Dynamic light scattering (DLS) measurement in aqueous solution (Fig. 1b) revealed that the average size distribution of exfoliated MMT in the range of 220–250 nm is decreased compared to the size distribution of pure MMT. In addition, SEM was conducted to investigate the differences of surface morphology between pure MMT and exfoliated MMT (Fig. S2†). Fig. S2b† shows that the MMT layered structure formed separated layers, indicating that MMT was easily exfoliated by the C/S-PDMA polymer. To observe the shape of the WO3 and Fe3O4 deposited in the MMT layer, we are also taking SEM images for both pure metal oxides. As shown in Fig. S2c and d,† most of the surface morphologies of both pure WO3 and Fe3O4 showed nearly spherical shapes with size below 100 nm. After the immobilization of Fe3O4 and WO3 in the MMT layer, the morphology dramatically unraveled into separated layers with the adhered Fe3O4 and WO3. The EDX spectrum demonstrated more evidence of the presence of immobilized Fe and W in the polymer chains–MMT nanocomposites (Fig. S3e and f†). As clearly observed in Fig. S2,† the surface of the exfoliated MMT had a plate shape with the average size single plate of about 200 nm, which describes the length from the left end to the right end side of those plates.
WO3 were recorded by UV-Vis-NIR spectroscopy, assuring the presence of catechol groups through peaks in the range 250–300 nm, which indicate π–π* electron transitions in phenolic compounds.22 Furthermore, the wide spectrum from UV-Vis to NIR area agree with the characteristic of strong LSPR of WO3 in the prepared antibacterial agents, which was able to absorb NIR irradiation in the range of ∼780–1000 nm (Fig. 1a).23 Dynamic light scattering (DLS) measurement in aqueous solution (Fig. 1b) revealed that the average size distribution of exfoliated MMT in the range of 220–250 nm is decreased compared to the size distribution of pure MMT. In addition, SEM was conducted to investigate the differences of surface morphology between pure MMT and exfoliated MMT (Fig. S2†). Fig. S2b† shows that the MMT layered structure formed separated layers, indicating that MMT was easily exfoliated by the C/S-PDMA polymer. To observe the shape of the WO3 and Fe3O4 deposited in the MMT layer, we are also taking SEM images for both pure metal oxides. As shown in Fig. S2c and d,† most of the surface morphologies of both pure WO3 and Fe3O4 showed nearly spherical shapes with size below 100 nm. After the immobilization of Fe3O4 and WO3 in the MMT layer, the morphology dramatically unraveled into separated layers with the adhered Fe3O4 and WO3. The EDX spectrum demonstrated more evidence of the presence of immobilized Fe and W in the polymer chains–MMT nanocomposites (Fig. S3e and f†). As clearly observed in Fig. S2,† the surface of the exfoliated MMT had a plate shape with the average size single plate of about 200 nm, which describes the length from the left end to the right end side of those plates.
The structure of the antibacterial composite was investigated through the XRD profile in the 2θ range from ∼30° to 65° and 25° to 55°, describing the crystalline properties of Fe3O4 and WO3 of C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3, respectively.24,25 The successfully C/S-PDMA exfoliated MMT with Fe3O4 and WO3 is shown in the XRD pattern at the 2θ value in the range 6–8°, due to the disappearance of the d spacing of pure MMT at 6.92°, corresponding to an interlayer d(100) of 1.27 nm measured by the Bragg equation [d = λ/2
WO3, respectively.24,25 The successfully C/S-PDMA exfoliated MMT with Fe3O4 and WO3 is shown in the XRD pattern at the 2θ value in the range 6–8°, due to the disappearance of the d spacing of pure MMT at 6.92°, corresponding to an interlayer d(100) of 1.27 nm measured by the Bragg equation [d = λ/2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin(θ)].26,27 No emergence of C/S-PDMA/MMT/Fe3O4
sin(θ)].26,27 No emergence of C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 peaks in the area of 2θ was an evidence of the complete intercalation of the polymer into silicate layers of MMT, exfoliation of MMT, and immobilization of Fe3O4/WO3 within the MMT composite (Fig. 1c and d). The FT-IR transmittance showed the stretching absorption peaks of Al–O and Si–O in MMT at ∼1067 and ∼610 cm−1, respectively. The MMT intercalated with polymer showed C–H stretching at 2850–3000 cm−1, –OH stretching at 3626 cm−1, C
WO3 peaks in the area of 2θ was an evidence of the complete intercalation of the polymer into silicate layers of MMT, exfoliation of MMT, and immobilization of Fe3O4/WO3 within the MMT composite (Fig. 1c and d). The FT-IR transmittance showed the stretching absorption peaks of Al–O and Si–O in MMT at ∼1067 and ∼610 cm−1, respectively. The MMT intercalated with polymer showed C–H stretching at 2850–3000 cm−1, –OH stretching at 3626 cm−1, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching at ∼1735–1750 cm−1 and –COOH stretching at ∼1150 cm−1.28,29 The W–O and Fe–O stretching were shown at 620 and ∼462–560 cm−1, clearly indicating the immobilization of WO3 and Fe3O4 in the MMT–polymer composites, as shown in Fig. S1.†30,31
O stretching at ∼1735–1750 cm−1 and –COOH stretching at ∼1150 cm−1.28,29 The W–O and Fe–O stretching were shown at 620 and ∼462–560 cm−1, clearly indicating the immobilization of WO3 and Fe3O4 in the MMT–polymer composites, as shown in Fig. S1.†30,31
To prove the interactions among each element in the composites, the C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 was analyzed by XPS measurement, as shown in Fig. 2. The wide-scan spectrum (Fig. 2a) confirmed the signal binding energy of C 1s, N 1s, O 1s, Si 2p, Fe 2p, W 4d, and W 4f. A noticeable peaks of Fe and W in the wide scan proved their immobilization in the MMT layer. The narrow scan of the signals for Fe 2p, W 4d, and W 4f revealed two peaks (Fig. 2b–d). The peak centered at 529.8 and 532 eV reflected the O 1s level (Fig. 2e). The C 1s XPS signal shown in Fig. 2f revealed three peaks at 283.9, 285.9, and 287.8 eV, indicating C–C, C–N and C
WO3 was analyzed by XPS measurement, as shown in Fig. 2. The wide-scan spectrum (Fig. 2a) confirmed the signal binding energy of C 1s, N 1s, O 1s, Si 2p, Fe 2p, W 4d, and W 4f. A noticeable peaks of Fe and W in the wide scan proved their immobilization in the MMT layer. The narrow scan of the signals for Fe 2p, W 4d, and W 4f revealed two peaks (Fig. 2b–d). The peak centered at 529.8 and 532 eV reflected the O 1s level (Fig. 2e). The C 1s XPS signal shown in Fig. 2f revealed three peaks at 283.9, 285.9, and 287.8 eV, indicating C–C, C–N and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds, respectively.
O bonds, respectively.
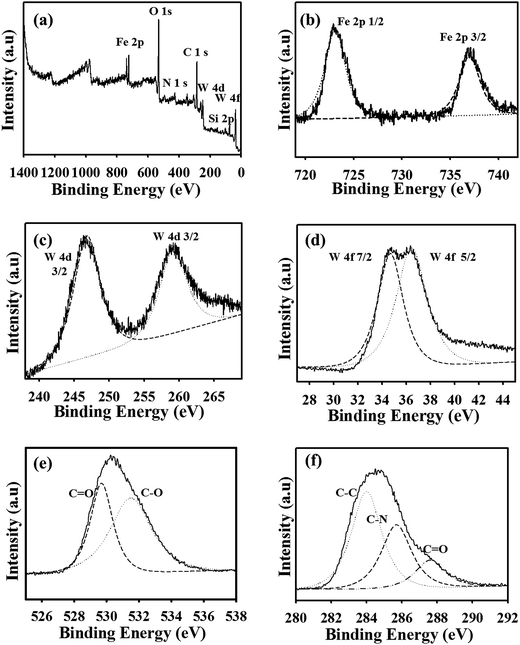 | ||
Fig. 2 XPS spectra of C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3: (a) wide scan and narrow scale of (b) Fe 2p, (c) W 4d, (d) W 4f, (e) O 1s, and (f) C 1s peaks. WO3: (a) wide scan and narrow scale of (b) Fe 2p, (c) W 4d, (d) W 4f, (e) O 1s, and (f) C 1s peaks. | ||
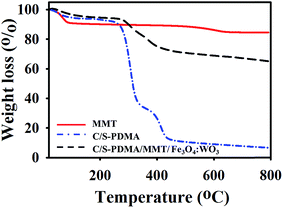
The thermal stability of organic–inorganic polymer MMT was verified by TGA. Fig. 3 shows the TGA analysis of MMT, C/S-PDMA, and C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3, indicating that the pristine MMT has great thermal stability even at high temperature, whereas C/S-PDMA has poor thermal stability and almost 90% of the polymer decomposed. As expected, the incorporated polymer with MMT demonstrated ∼25% reduction upon heating at 800 °C. This is due to the existence of the silicate layer from clay loading, which delayed decomposition.32,33
WO3, indicating that the pristine MMT has great thermal stability even at high temperature, whereas C/S-PDMA has poor thermal stability and almost 90% of the polymer decomposed. As expected, the incorporated polymer with MMT demonstrated ∼25% reduction upon heating at 800 °C. This is due to the existence of the silicate layer from clay loading, which delayed decomposition.32,33
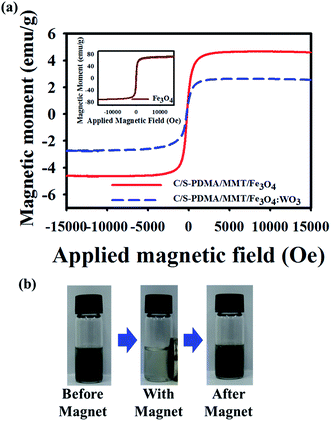
The vibrating sample magnetometer profile showed the strong paramagnetic properties of the C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3, as evidenced by the field-dependent magnetization curve appearing as a sigmoid shape (Fig. 4a), indicating enhancement of magnetization after diverse applied magnetic fields. Thereafter, the magnetic behavior of C/S-PDMA/MMT/Fe3O4
WO3, as evidenced by the field-dependent magnetization curve appearing as a sigmoid shape (Fig. 4a), indicating enhancement of magnetization after diverse applied magnetic fields. Thereafter, the magnetic behavior of C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 was tested in aqueous solution under exposure to an external magnet; the permanent magnet led all particles to move to the magnetic side, and after removing the magnetic exposure, the particles returned to normal positions (Fig. 4b). Therefore, the recyclability of the magnetic nanocomposites emerges from the externally controlled applied magnetic fields.34–36 For applicability, the stability of colloidal particles was examined from 0 to 5 days at four different concentrations, showing good stability after five days. In the same period probe, the stability was investigated by UV-Vis spectroscopy and DLS measurements. After five days, the DLS histogram showed similar size distribution and barely changed absorption at two different concentrations, indicating the formation of stable C/S-PDMA/MMT/Fe3O4
WO3 was tested in aqueous solution under exposure to an external magnet; the permanent magnet led all particles to move to the magnetic side, and after removing the magnetic exposure, the particles returned to normal positions (Fig. 4b). Therefore, the recyclability of the magnetic nanocomposites emerges from the externally controlled applied magnetic fields.34–36 For applicability, the stability of colloidal particles was examined from 0 to 5 days at four different concentrations, showing good stability after five days. In the same period probe, the stability was investigated by UV-Vis spectroscopy and DLS measurements. After five days, the DLS histogram showed similar size distribution and barely changed absorption at two different concentrations, indicating the formation of stable C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 nanocomposites (Fig. S3†).
WO3 nanocomposites (Fig. S3†).
Fig. 5 demonstrates the photothermal effect of C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 mediated by NIR irradiation. The appearance of thermal elevation means that increasing the concentration of MMT composites from 0.1 to 1 mg mL−1 generated temperature changes by ∼57 °C. The C/S-PDMA, C/S-PDMA/MMT, and C/S-PDMA/MMT/Fe3O4 showed very poor photothermal effect under NIR irradiation, clarifying the absence of NIR responsiveness, as shown in Fig. S4.†
WO3 mediated by NIR irradiation. The appearance of thermal elevation means that increasing the concentration of MMT composites from 0.1 to 1 mg mL−1 generated temperature changes by ∼57 °C. The C/S-PDMA, C/S-PDMA/MMT, and C/S-PDMA/MMT/Fe3O4 showed very poor photothermal effect under NIR irradiation, clarifying the absence of NIR responsiveness, as shown in Fig. S4.†
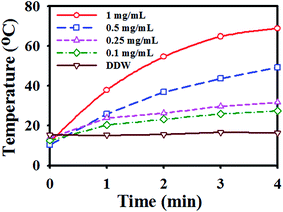 | ||
Fig. 5 Photothermal heating curves of pure water (DDW) and C/S-DMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 (0.1–1 mg mL−1 concentration in water) under 808 nm laser irradiation at a power density of 2 W cm−2. WO3 (0.1–1 mg mL−1 concentration in water) under 808 nm laser irradiation at a power density of 2 W cm−2. | ||
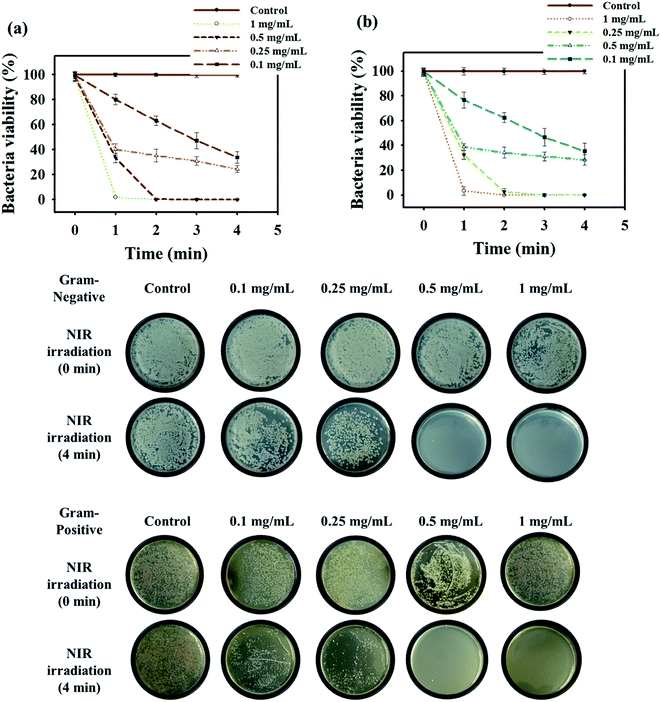
The hyperthermia effect of the stable photothermal composite was tested towards E. coli (Gram-negative bacteria) and S. aureus (Gram-positive bacteria) through the spread plate method. Time-dependent NIR exposure was applied to the bacteria solution treated by the inorganic–organic polymer–MMT composite. Fig. 6 shows the antibacterial activity of the colloidal particle of C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 at different concentrations with both strain bacteria; the higher concentration of immobilized WO3 in the exfoliated MMT resulted in shorter killing time of bacteria. These results suggest that the minimum concentration for killing bacteria is 0.5 mg mL−1 within 2 min NIR exposure, showing similar performance against both bacteria.4 As further evidence, we tested bacteria treated with the addition nanocomposites but without NIR irradiation to check the non-antibacterial activity of these nanocomposites towards both bacteria strains at different starting incubation times. The tests generated almost 100% of all strains of bacteria, i.e., S. aureus, E. coli, treated S. aureus and treated E. coli were alive. These results indicated that the prepared nanocomposites have no antibacterial activity without NIR exposure (Fig. S6†).
WO3 at different concentrations with both strain bacteria; the higher concentration of immobilized WO3 in the exfoliated MMT resulted in shorter killing time of bacteria. These results suggest that the minimum concentration for killing bacteria is 0.5 mg mL−1 within 2 min NIR exposure, showing similar performance against both bacteria.4 As further evidence, we tested bacteria treated with the addition nanocomposites but without NIR irradiation to check the non-antibacterial activity of these nanocomposites towards both bacteria strains at different starting incubation times. The tests generated almost 100% of all strains of bacteria, i.e., S. aureus, E. coli, treated S. aureus and treated E. coli were alive. These results indicated that the prepared nanocomposites have no antibacterial activity without NIR exposure (Fig. S6†).
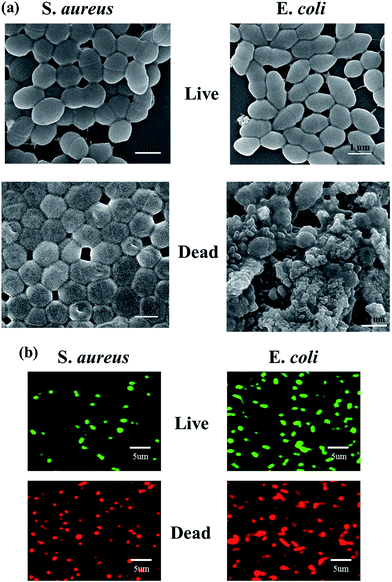
To prove the efficiency of a bacteria killing system with these MMT composites, the fluorescence labeling of SYTO 9 (green color) for live cells and propidium iodide (red color) for dead cells was conducted.37 The fluorescence microscopic images clearly revealed that bacteria treated with the organic–inorganic polymer–MMT composite was destroyed after NIR irradiation (red color), whereas bacteria treated with organic–inorganic polymer–MMT without NIR exposure showed live cells (green color) (Fig. 7b). As further evidence, the morphology of the treated bacteria cells after NIR exposure observed by SEM revealed untidy cell walls, indicating cell lysis, due to heat released by the photothermal based exfoliated MMT agents (Fig. 7a).
The recyclability of C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 demonstrated that the bacterial efficiency almost unchanged even after three times use (Fig. 8). The UV-Vis spectra and particle size of this agent in each number of cycles towards both strains of bacteria demonstrated consistent behaviors, representing colloidal particle stability (Fig. S5†). The identification and quantification of the metal component were further measured by inductively coupled-plasma mass spectroscopy (ICP-MS). After three cycles, ∼90% of Fe and W were detected in both samples used for killing E. coli and those for S. aureus, respectively (Table S1†), suggesting that the organic–inorganic polymer–MMT composite is capable of being an efficient and rapid killing agent in photothermal agent fields because of colloidal particle stability.
WO3 demonstrated that the bacterial efficiency almost unchanged even after three times use (Fig. 8). The UV-Vis spectra and particle size of this agent in each number of cycles towards both strains of bacteria demonstrated consistent behaviors, representing colloidal particle stability (Fig. S5†). The identification and quantification of the metal component were further measured by inductively coupled-plasma mass spectroscopy (ICP-MS). After three cycles, ∼90% of Fe and W were detected in both samples used for killing E. coli and those for S. aureus, respectively (Table S1†), suggesting that the organic–inorganic polymer–MMT composite is capable of being an efficient and rapid killing agent in photothermal agent fields because of colloidal particle stability.
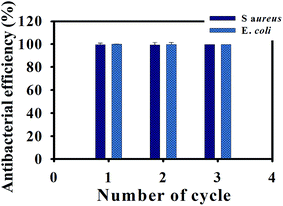 | ||
Fig. 8 Antibacterial ability of the recycled C/S-PDMA/MMT/Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WO3 (1 mg mL−1 concentration in water) towards E. coli and S. aureus. WO3 (1 mg mL−1 concentration in water) towards E. coli and S. aureus. | ||
4. Conclusions
In this study, we developed a method to immobilize active NIR WO3 and recyclable Fe3O4 in the exfoliated MMT layer, employing C/S-PDMA as the intercalation agents based on the catechol chemistry, affording recyclable photothermal antibacterial nanoparticles. The XRD, XPS, and FT-IR analyses confirmed the chemical incorporation of MMT with metal oxides as promising recyclable antibacterial materials. The resulting agent demonstrated good colloidal stability, shown by the constant absorption property and similar size distribution even after five days of storage. This hyperthermal composite material demonstrated excellent heat release for killing Gram-negative and Gram-positive bacteria in a short period time, i.e., within 2 min. The SEM and confocal images of the treated bacteria obviously illustrated the expected NIR photothermal antibacterial agents. The constructed nanocomposites showed paramagnetism, enabling repeated uses, and exhibited effective recyclable antibacterial ability after treatment towards both bacteria three times, indicating a recyclable stable material, with ∼90% Fe and W atoms remaining in the MMT substituted with catechol groups by C/S-PDMA. This approach presents the potential of thermally stable MMT nanocomposites as NIR irradiation mediated photothermal agents.Acknowledgements
This study was supported by the Grant No. 10048377, 10062079 and R0005237 from the Ministry of Trade, Industry & Energy (MOTIE), and Fusion Research R&D Program from the Korea Research Council for Industrial Science & Technology (No. G02054), Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (No. 2014055946).References
- A. M. Bonilla and M. F. García, Prog. Polym. Sci., 2012, 37, 281–339 CrossRef.
- S. H. Kim, E. B. Kang, C. J. Jeong, S. M. Sharker, I. In and S. Y. Park, ACS Appl. Mater. Interfaces, 2015, 7, 15600–15606 CAS.
- J. Hasan, R. J. Crawford and E. P. Ivanova, Trends Biotechnol., 2013, 31, 295–304 CrossRef CAS PubMed.
- S. J. Park, E. B. Kang, S. M. Sharker, G. Lee, I. In and S. Y. Park, Macromol. Mater. Eng., 2016, 301, 141–148 CrossRef CAS.
- T. Pongprayoon, R. Nuangchamnong and N. Yanumet, Appl. Clay Sci., 2013, 86, 179–184 CrossRef CAS.
- C. C. Chu, M. L. Chiang, C. M. Tsai and J. J. Lin, Macromolecules, 2005, 38, 6240–6243 CrossRef CAS.
- K. Prabakaran, S. Mohanty and S. K. Nayak, New J. Chem., 2015, 39, 8602–8613 RSC.
- M. Liu, P. Lai, H. Lin, Y. Luo, H. Li, X. Wang and R. Sun, Carbohydr. Polym., 2016, 137, 375–381 CrossRef PubMed.
- G. B. B. Varadwaj, S. Rana and K. M. Parida, RSC Adv., 2013, 3, 7570–7578 RSC.
- E. B. Kang, J. E. Lee, J. H. Jeong, G. Lee, I. In and S. Y. Park, J. Ind. Eng. Chem., 2016, 33, 336–344 CrossRef CAS; Y. C. Ling, ACS Nano, 2013, 7, 1281–1290 CrossRef PubMed.
- S. M. Sharker, S. M. Kim, J. E. Lee, K. H. Choi, G. Shin, S. Lee, K. D. Lee, J. H. Jeong, H. Lee and S. Y. Park, J. Controlled Release, 2015, 217, 211–220 CrossRef CAS PubMed.
- J. Liu, J. Han, Z. Kang, R. Glamaully, N. Xu, H. Li and X. Han, Nanoscale, 2014, 6, 5770–5776 RSC.
- O. Khantamat, C. H. Li, A. C. Jamison, W. C. Shih, C. Cai and T. R. Lee, ACS Appl. Mater. Interfaces, 2015, 7, 3981–3993 CAS.
- C. J. Jeong, E. B. Kang, S. J. Park, K. H. Choi, G. Shin, I. In and S. Y. Park, Polym. Eng. Sci., 2015 DOI:10.1002/pen.
- S. Y. Lee, S. H. Kim, S. H. Kim, H. Lee, G. Lee and S. Y. Park, New J. Chem., 2014, 38, 2225–2228 RSC.
- G. Yeroslavsky, O. Girshevrtz, J. F. Frey, D. M. Donovan and S. Rahimipour, Langmuir, 2015, 31, 1064–1073 CrossRef CAS PubMed.
- H. Lee, Y. Lee, A. R. Statz, J. Rho, T. G. Park and P. B. Messersmith, Adv. Mater., 2008, 20, 1619–1623 CrossRef CAS PubMed.
- W. Wu, Q. He and C. Jiang, Nanoscale Res. Lett., 2008, 3, 397–415 CrossRef CAS PubMed.
- C. J. Jeong, S. M. Sharker, I. In and S. Y. Park, ACS Appl. Mater. Interfaces, 2015, 7, 9469–9478 CAS.
- Y. W. Wang, Y. Y. Fu, L. J. Wu, J. Li, H. H. Yang and G. N. Chen, J. Mater. Chem. B, 2013, 2, 2496–2501 RSC.
- S. S. Ray and M. Okamoto, Prog. Polym. Sci., 2003, 28, 1539–1641 CrossRef CAS.
- R. M. B. O. Duarte, E. B. H. Santos and A. C. Duarte, Water Res., 2003, 37, 4073–4080 CrossRef CAS PubMed.
- J. M. Luther, P. K. Jain, T. Ewers and A. P. Alivisatos, Nat. Mater., 2011, 10, 361–366 CrossRef CAS PubMed.
- C. Y. Su, H. C. Lin, T. K. Yang, C. H. Chang and K. L. Lin, Mater. Trans., 2009, 50, 2593–2597 CrossRef CAS.
- T. J. Daou, G. Pourroy, J. M. Greneche, A. Bertin, D. F. Flescha and S. B. Colin, Dalton Trans., 2009, 23, 4442–4449 RSC.
- M. Jin and Q. Zhong, J. Agric. Food Chem., 2012, 60, 11965–11971 CrossRef CAS PubMed.
- R. M. Mariano, P. H. de S. Picciani, R. C. R. Nunes and L. L. Y. Visconte, J. Appl. Polym. Sci., 2011, 120, 458 CrossRef CAS.
- A. Hassani, R. D. C. Soltani, S. Karaca and A. Khataee, J. Ind. Eng. Chem., 2015, 21, 1197–1207 CrossRef CAS.
- Y. S. Choi, M. H. Choi, K. H. Wang, S. O. Kim, Y. K. Kim and I. J. Chung, Macromolecules, 2001, 34, 8978–8985 CrossRef CAS.
- B. Zou, S. Gong, Y. Wang and X. Liu, J. Nanomater., 2014 DOI:10.1155/2014/813120.
- M. M. Rahman, S. B. Khan, A. Jamal, M. Faisal and A. M. Aisiri, Nanomaterials, 2011 DOI:10.5772/27698.
- P. Singla, R. Mehta and S. N. Upadhyay, Green Sustainable Chem., 2012, 2, 21–25 CrossRef CAS.
- P. Uthirakumar, Y. B. Hahn, K. S. Nahm and Y. S. Lee, Polym. Compos., 2008, 29, 142–148 CrossRef CAS.
- M. D. Adhikari, S. Mukherjee, J. Saikia, G. Das and A. Ramesh, J. Mater. Chem. B, 2014, 2, 1432–1438 RSC.
- H. Dong, J. Huang, R. R. Koepsel, P. Ye, A. J. Russel and K. Matyjaszewski, Biomacromolecules, 2011, 12, 1305–1311 CrossRef CAS PubMed.
- M. Mahmoudi and V. Serpooshan, ACS Nano, 2012, 6, 2656–2664 CrossRef CAS PubMed.
- C. Ghosh, G. B. Manjunath, P. Akkapeddi, V. Yarlagadda, J. Hoque, D. S. S. M. Uppu, M. M. Kona and J. Haldar, J. Med. Chem., 2014, 57, 1428–1436 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra08180k |
| This journal is © The Royal Society of Chemistry 2016 |