Adsorption behavior of magnetic amino-functionalized metal–organic framework for cationic and anionic dyes from aqueous solution†
Haochi Liua,
Ligang Chen*a and
Jie Ding*b
aDepartment of Chemistry, College of Science, Northeast Forestry University, 26 Hexing Road, Harbin 150040, China. E-mail: ligangchen2010@aliyun.com; Tel: +86-451-82190679
bState Key Laboratory of Urban Water Resource and Environment, Harbin Institute of Technology, Harbin 150090, China. E-mail: hitdingjie@163.com; Tel: +86-451-86289113
First published on 6th May 2016
Abstract
As organic dyes are a major group of water pollutants, the development of materials for removal of dyes is of great significance for the environment. Here we report a kind of magnetic metal–organic framework (MOF) for the adsorption of different kinds of anionic and cationic dyes. Magnetic NH2-MIL-101(Al) was synthesized and characterized with Fourier-transform infrared spectrometry (FT-IR), physical property measurement system (PPMS), X-ray diffraction (XRD), a transmission electron microscope (TEM), a thermogravimetric analyzer (TGA) and nitrogen adsorption/desorption measurements. Adsorption isotherms and thermodynamic studies indicated that the maximum adsorption capacities of malachite green (MG) and indigo carmine (IC) were 274.4 and 135 mg g−1, respectively. The mechanisms of interaction such as electrostatic interaction, hydrogen bonding, π–π stacking interaction, and hydrophobic interaction are discussed for the adsorption of cationic and anionic dyes onto magnetic NH2-MIL-101(Al). Also, magnetic separation in this method shortened separation time, and the magnetic material can be reused at least five times without obvious decrease in the removal efficiency. Moreover, the synthesis of the magnetic NH2-MIL-101(Al) is easy to control and the good solvent stability makes this material promising as a novel adsorbent for the adsorption and removal of dyes from aqueous solution.
1. Introduction
Organic dyes are important products, widely used in textile, paper, rubber and plastics, etc. They have posed a significant threat to the environment and creatures' health due to the fact that they have a certain toxicity and can even be carcinogenic.1 It is thus of vital importance to solve the problem of pollution caused by organic dyes. However, wastewater containing dyes is very difficult to treat, since the dyes are recalcitrant organic molecules, resistant to aerobic digestion, and are stable to light, heat and oxidizing agents.2 There are numerous conventional methods that can be used for removing organic dyes from wastewater, including biotransformation,3 coagulation,4 membrane filtration,5 adsorption6 and advanced oxidation.7 Currently, adsorption is the most common treatment for effective dyestuff removal because it is simple to operate, high performing and cost-effective. In the past few years, a number of adsorbents have been reported to eliminate dyes, such as agricultural wastes,8 inorganic materials9 and polymers.10Recently, metal–organic frameworks (MOFs) have met with great interest, owing to their enormous variety of interesting molecular topologies11 and a wide range of potential applications as functional materials. MOFs have some advantages as adsorbents in that their structures and pore sizes are tunable.12 Further, they contain polar and polarisable bonds, and can even bear open (or, in aqueous solution, water-coordinated) coordination sites on metal atoms.13 Thus, the highly regular channel structures and controllable pore sizes of MOFs permit their applications in adsorption of guest molecules/ions, such as dyes or lanthanide ions. As potential organic dye adsorbents, however, porous MOFs have received much less attention than the well-known activated carbons. As far as we know, only a few examples of porous MOFs being used for organic dye adsorption have been reported to date.14,15
Among the numerous MOFs reported so far, the most widely used mesoporous MOFs are represented by the MIL-100 and MIL-101 types (MIL stands for Material of Institute Lavoisier). The huge variety of possible applications16 of these MOFs is based upon their mesoporous cavities (Fig. S1†), large surface areas, the presence of accessible metal sites and their comparatively high thermal and chemical robustness.17–19 For example, the unfunctionalised MIL-101(Cr) has been used as a sorbent for the cationic dye malachite green9 and anionic dye xylenol orange.20 In addition, the presence of coordinatively unsaturated metal sites in MIL-101 allows its use as a mild Lewis acid21 and, more importantly, allows its postfunctionalization via grafting of active species.22 NH2-MIL-101(Fe) has been reported and applied in drug delivery23 and medical imaging,24 where the relatively low thermal and chemical stability shown by this framework does not seem to affect its outstanding performance. Considering other types of application, the development of materials with a similar topology and stability as MIL-101(Cr) but presenting functional organic sites is needed.
Meanwhile, chemically synthesized magnetic composites are of particular interest for their unique magnetic properties which mean that they can be effectively separated under an external magnetic field and recycled for reuse. Magnetic materials based on SiO2,25 zeolites,26 mesoporous CeO2 (ref. 27) and activated carbon28 have been successfully investigated. Considering the superior properties of MOFs and their successful development in a wide range of research fields, the design and synthesis of magnetic MOFs are especially desirable.
Herein, we demonstrate a novel scheme to fabricate magnetic NH2-MIL-101(Al), which combines the magnetic properties of Fe3O4 nanoparticles with the MOF's functional properties. The resulting material shows a high chemical and thermal stability. Moreover, owing to the presence of amines, magnetic NH2-MIL-101(Al) displays outstanding adsorption properties and has been found to be a versatile adsorbent for removing various dyes in aqueous solution. We chose different kinds of dyes (Fig. S2†) to investigate the mechanisms of interactions, such as electrostatic interaction, hydrogen bonding, π–π stacking interaction and hydrophobic interaction. The influence of the molecular size for the dye's adsorption is also investigated. The prepared magnetic NH2-MIL-101(Al) composites offer superparamagnetic properties and excellent adsorption capability for the fast magnetic removal of dyes from water samples.
2. Experimental
2.1. Reagents and instruments
The standards of methyl orange (MO), methylene blue (MB), malachite green (MG), and indigo carmine (IC) were purchased from Aladdin (Shanghai, China). Iron(III) chloride hexahydrate (FeCl3·6H2O), iron(II) chloride tetrahydrate (FeCl2·4H2O), ammonia (NH3·H2O), tetraethyl orthosilicate (TEOS), 3-aminopropyltriethoxysilane (KH550), sodium hydroxide (NaOH), and hydrochloric acid (HCl) were obtained from Kermel (Tianjin, China). Aluminum chloride hexahydrate (AlCl3·6H2O) and 2-aminoterephthalic acid were purchased from Sigma (St. Louis, MO, USA). N,N-Dimethylformamide (DMF), ethanol, and methanol were obtained from Fuyu (Tianjin, China). Stock solutions of cationic and anionic dyes (1000 mg L−1) were prepared by dissolving the solid in water, and keeping it at 4 °C in a refrigerator. High purity water was obtained from a Milli-Q water system (Millipore, Billerica, MA, USA). All other reagents were of analytical grade.The magnetic MOFs were characterized by a transmission electron microscope (TEM, H7650, Hitachi, Japan), physical property measurement system (PPMS, Quantum Design Instrument, San Diego, CA, USA), Fourier-transform infrared spectrometry (FT-IR360, Nicolet, Madison, WI, USA), XRD-600 diffractometer (Shimadzu, Kyoto, Japan), thermogravimetric analyzer (TGA; Pyris 1, PerkinElmer, Waltham, MA, USA) and a Brunauer–Emmett–Teller (BET) surface area instrument (Quadrasorb SI-MP, Quantachrome, Florida, UK). The dye concentrations were determined using the absorbance of the solutions after getting the UV spectra of the solution with a spectrophotometer (Shimadzu UV spectrophotometer, UV-1800). The adsorption experiments were carried out at constant temperature controlled by a SHA-B shaking table (Shengtang, Tianjin, China). A KQ5200E ultrasonic apparatus (Kunshan Instrument, Kunshan, China) was used to ensure uniform dispersion.
2.2. Preparation of the amino-functionalized Fe3O4 nanoparticles
The Fe3O4 nanoparticles were prepared by a coprecipitation method. FeCl2·4H2O (0.01 mol) and FeCl3·6H2O (0.02 mol) were dissolved in 100 mL water. The mixture was stirred vigorously while the temperature increased to 80 °C, and then 10 mL of ammonia was added into the solution. The mixture was stirred for 1 h at 500 rpm under an N2 stream. After that, the magnetic particles were separated by a permanent magnet and washed with water until the pH was 7. The product was then dried at 60 °C for further use.The magnetic Fe3O4 (0.5 g) obtained was dispersed into a mixed solution of water and ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v). Then 3 mL of NH3·H2O and 5 mL of TEOS were added under stirring at 40 °C for 24 h. The products were reacted with 3-aminopropyltriethoxysilane (2 mL) in 100 mL of water to obtain amino-functionalized Fe3O4 nanoparticles under continuous agitation for 6 h at 60 °C. The resultant nanoparticles were collected and washed with ethanol and water. Then the solid was finally dried at 80 °C under vacuum overnight.
1, v/v). Then 3 mL of NH3·H2O and 5 mL of TEOS were added under stirring at 40 °C for 24 h. The products were reacted with 3-aminopropyltriethoxysilane (2 mL) in 100 mL of water to obtain amino-functionalized Fe3O4 nanoparticles under continuous agitation for 6 h at 60 °C. The resultant nanoparticles were collected and washed with ethanol and water. Then the solid was finally dried at 80 °C under vacuum overnight.
2.3. Preparation of magnetic MOFs
The 2-aminoterephthalic acid (0.56 g) was dissolved in 30 mL of DMF and AlCl3·6H2O (0.51 g) was dissolved in 40 mL of DMF. The 2-aminoterephthalic acid solution was dropped into the solution of Al3+ to prepare the pre-assembly solution. Then the amino-functionalized Fe3O4 nanoparticles were immediately added under ultrasound and maintained for 30 min to ensure uniform dispersion. The resulting mixture was placed in a Teflon-lined reactor and heated for 10 h at 120 °C. The powder obtained was washed several times with DMF, ethanol and water in order to remove the impurities. To remove organic species trapped within the pores, the product was activated in methanol at 80 °C for 10 h and then dried under vacuum at 60 °C for further use.2.4. Adsorption experiments
In a typical adsorption experiment, the adsorbent (10 mg) was weighed precisely and put in the aqueous dye solutions (5 mL) with concentrations from 10 to 1000 mg L−1. The dye solutions containing the adsorbents were mixed well by a shaking table and maintained for a fixed time (10 min to 120 min) at room temperature. After adsorption, the solutions were separated from the adsorbents with a magnet, and the dye concentrations were detected. To get the thermodynamic parameters of adsorption, such as ΔG0 (free energy change), ΔH0 (enthalpy change) and ΔS0 (entropy change), the adsorptions were further carried out at 25, 35 and 45 °C. To determine the adsorption capacity at various pH values, the pH of the dye solutions was adjusted with 0.1 M HCl or 0.1 M NaOH aqueous solution.3. Results and discussions
3.1. Fabrication of magnetic NH2-MIL-101(Al)
The framework of the MOF is based on the coordination interaction between the carboxyl group of 2-aminoterephthalic acid and the Al(III) of AlCl3·6H2O. S. Jung et al.29 confirmed an assumption that a certain number of the carboxyl groups are exposed on the crystalline surface and can be conjugated with other compounds via chemical interaction. Accordingly, in our research, the Fe3O4 nanoparticles were first functionalized with amino groups, which can form chemical bonds with carboxyl groups exposed on the surface of MOF.Previous studies proved that the functionalization of an MOF could improve its adsorption ability to target molecules. MOF functionalized with ethylenediamine sourced from NH2 was used for the adsorption of naproxen and clofibric acid.30 In this study, NH2-MIL-101(Al) is built from supertetrahedral building units formed by aminoterephthalate ligands and trimeric Al(III) octahedral clusters and thus bears an amino group on every linker,31 rather than using the post synthesis grafting method that was demonstrated by Zubair Hasan and co-workers.30
When the MIL-101(Al) has been endowed with magnetism and amine functionalization, the resulting composite material, magnetic NH2-MIL-101(Al) not only has a porous structure, it also has rapid separation and good adsorption ability.
3.2. Characterization of magnetic NH2-MIL-101(Al)
The FT-IR spectrum of magnetic MOF is shown in Fig. 1a. The characteristic peak at 602 cm−1 was assigned to the Fe–O bond. The observed feature around 1089 cm−1 was the Si–O–Si bond of magnetic MOF. The peak at 3431 cm−1 corresponded to an O–H group. The peak at 1398 cm−1 was attributed to the existence of O–H bending vibration. The adsorption band around 2973 cm−1 unveiled the stretching vibration of the C–H group in CH3. The characteristic band of the amino group at 1623 cm−1 was attributed to the N–H bending (scissoring) vibration. | ||
| Fig. 1 FT-IR spectrum (a), magnetization curve (b), TEM images (c and d) and thermogravimetric curve (e) of magnetic NH2-MIL-101(Al). | ||
The saturation magnetization of the composites was performed in an applied magnetic field at room temperature. As shown in Fig. 1b, the magnetization curve showed a superparamagnetic property. The magnetic saturation value of the magnetic NH2-MIL-101(Al) composites is 18.73 emu g−1. This value demonstrated that the composite has strong magnetism.
The TEM image of magnetic NH2-MIL-101(Al) (Fig. 1c) showed a homogeneous surface morphology with uniform particle size. Moreover, the diameter of magnetic NH2-MIL-101(Al) (Fig. 1c) increases markedly compared with crude Fe3O4 nanoparticles (Fig. 1d), which revealed that the NH2-MIL-101(Al) layer was successfully attached to Fe3O4.
To reveal the thermal stability of magnetic NH2-MIL-101(Al), a thermo-gravimetric experiment was carried out with pure single crystal samples under N2 atmosphere conditions at a rate of 10 °C min−1 over a range of 40–850 °C. As shown in Fig. 1e, the first main weight-loss was due to the dehydration of the material up to 120 °C. These weight losses, starting at roughly 380 °C for magnetic NH2-MIL-101(Al), mark the degradation of the framework. This degradation temperature compares well to the value reported in the literature.32
The XRD patterns of the parent materials (NH2-MIL-101(Al), Fe3O4@SiO2) and the magnetic NH2-MIL-101(Al) are presented in Fig. 2a. The experimental XRD pattern shows the as-prepared magnetic NH2-MIL-101(Al) composites match well with those of both NH2-MIL-101(Al) and Fe3O4@SiO2 nanospheres, confirming that the magnetic NH2-MIL-101(Al) composites obtained are composed of crystalline NH2-MIL-101(Al) and Fe3O4@SiO2 nanospheres. The existence of Fe3O4@SiO2 nanospheres and NH2-MIL-101(Al) was further confirmed by energy-dispersive X-ray microanalysis (Fig. 2b). Elemental C, O, N, Al, Si and Fe were detected in the magnetic NH2-MIL-101 (Al) composites.
 | ||
| Fig. 2 Powder XRD pattern (a), energy-dispersive X-ray microanalysis (b), BJH pore size distributions (c) and N2 adsorption–desorption isotherms (d) of magnetic NH2-MIL-101(Al). | ||
To further characterize the porosity of the magnetic NH2-MIL-101(Al), nitrogen-adsorption experiments were performed at 77 K. As can be seen from Fig. 2c, the pore-size distribution of the as-prepared magnetic NH2-MIL-101(Al) samples calculated from the desorption branch of the N2 isotherm by the Barrett–Joyner–Halenda (BJH) model was 3.88 nm. As shown in Fig. 2d, NH2-MIL-101(Al) and magnetic NH2-MIL-101(Al) exhibited a type IV isotherm with a distinct hysteresis loop in the relative pressure P/P0 range of 0.5–1.0, suggesting a mesoporous structure. The BET surface area measured from the nitrogen isotherms was 285.61 m2 g−1. Compared with the NH2-MIL-101(Al) (392.14 m2 g−1), the BET surface areas of the composites significantly decreased due to the incorporation of Fe3O4 nanospheres.
3.3. Adsorption isotherms
The adsorption isotherms of dyes onto magnetic NH2-MIL-101(Al) were measured at three different temperatures. We take MG and IC as the representatives to study the adsorption properties of cationic and anionic dyes. When the MG and IC concentrations were at 1000 mg L−1, the adsorption capacities of magnetic NH2-MIL-101(Al) for MG and IC were 274.4 mg g−1 and 135 mg g−1 at 298 K (Fig. 3a), respectively. The adsorption isotherm models are fundamental for describing the interactive behavior between adsorbate and adsorbent and important for investigating mechanisms of adsorption. In this study, Langmuir,33 Freundlich,34 and Dubinin–Radushkevich35 models were used to describe the adsorption equilibrium data derived from the adsorption of dyes onto magnetic NH2-MIL-101(Al) at different temperatures (298, 308 and 318 K). Equilibrium adsorption isotherms were studied with dye concentrations ranging from 10 to 1000 mg L−1 with a fixed adsorbent mass and pH. | ||
| Fig. 3 Adsorption isotherms (a), Langmuir (b), Freundlich (c) and Dubinin–Radushkevich (d) plots of the isotherms for MG and IC adsorption onto magnetic NH2-MIL-101(Al) at 298 K. | ||
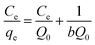 | (1) |
From the value of b deduced from the Langmuir model, the equilibrium parameter (RL) was calculated using the following equation:39
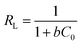 | (2) |
| Dyes | T (K) | Langmuir model | Freundlich model | Dubinin–Radushkevich model | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Q0 (mg g−1) | RL | R2 | n | KF (mg g−1) | R2 | E (kJ mol−1) | QD–R (mg g−1) | R2 | ||
| IC | 298 | 137.0 | 0.03–0.73 | 0.997 | 4.60 | 30.14 | 0.948 | 0.381 | 98.16 | 0.659 |
| 308 | 131.6 | 0.02–0.72 | 0.998 | 4.66 | 29.61 | 0.948 | 0.366 | 96.02 | 0.668 | |
| 318 | 128.2 | 0.02–0.71 | 0.998 | 4.70 | 29.17 | 0.947 | 0.360 | 94.20 | 0.675 | |
| MG | 298 | 285.7 | 0.02–0.68 | 0.993 | 3.05 | 38.63 | 0.976 | 1.25 | 156.19 | 0.561 |
| 308 | 277.8 | 0.02–0.68 | 0.994 | 3.08 | 38.16 | 0.977 | 1.30 | 154.02 | 0.562 | |
| 318 | 270.3 | 0.02–0.68 | 0.994 | 3.12 | 37.51 | 0.977 | 1.35 | 151.00 | 0.562 | |
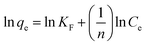 | (3) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe vs. ln
qe vs. ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ce (Fig. 3c). The Freundlich constants and correlation coefficients at different temperatures are listed in Table 1.
Ce (Fig. 3c). The Freundlich constants and correlation coefficients at different temperatures are listed in Table 1.In general, as the KF increases, the adsorption capacity of the adsorbent increases. If n < 1, this means poor adsorption, from one to two means moderately difficult adsorption, and from two to ten means good adsorption.41 All of the n values obtained from the Freundlich model are more than unity, indicating that adsorption of dyes onto the magnetic NH2-MIL-101(Al) is favorable (Table 1).
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe = ln qe = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) QD–R − βε2 QD–R − βε2
| (4) |
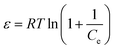 | (5) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe versus ε2 is obtained from the model, as shown in Fig. 3d.
qe versus ε2 is obtained from the model, as shown in Fig. 3d.The energy of adsorption is the free energy of the transfer of 1 mol of solute from infinity (in solution) to the surface of the adsorbent. The mean value of adsorption energy E, can be calculated from the constant of the sorption energy (D–R pattern) as follows:35
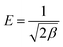 | (6) |
The magnitude of E (kJ mol−1) is used for estimating the type of adsorption mechanism. If this value is between 8 and 16 kJ mol−1, the adsorption process is controlled by a chemical mechanism, while for E < 8 kJ mol−1, the adsorption process proceeds through a physical mechanism.42 The calculated values of E (Table 1) suggested that the adsorption of dye occurs via physical adsorption. However, the correlation coefficients of the Dubinin–Radushkevich isotherm were lower than 0.68, which means that this mode is not suitable for the study of dye adsorption onto magnetic NH2-MIL-101(Al).
In conclusion, the correlation coefficients of the Freundlich isotherm and the Dubinin–Radushkevich isotherm are much lower than those for the Langmuir isotherm, suggesting that the Langmuir isotherm model is the best model to fit the experimental data.
3.4. Adsorption kinetics
The adsorption kinetics is an important parameter for designing adsorption systems and is required for selecting the optimum operating conditions for a pilot-scale process. In this work, as can be seen from Fig. 4a, the adsorption capacity was enhanced rapidly in the beginning, and then slowed down and reached equilibrium. In order to investigate the adsorption kinetics of dyes onto magnetic NH2-MIL-101(Al), two models have been studied: the pseudo-first-order kinetic model and pseudo-second order kinetic model. The linear form of the pseudo-first-order equation is given as follows:43
ln(qe − qt) = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe − k1t qe − k1t
| (7) |
 | ||
| Fig. 4 Kinetics adsorption (a), pseudo-first-order (b) and pseudo-second-order kinetics (c) for the adsorption of MG and IC onto magnetic NH2-MIL-101(Al). | ||
| Sample | Pseudo-first-order | Pseudo-second-order | ||||
|---|---|---|---|---|---|---|
| qe,cal (mg g−1) | k1 (min−1) | R2 | qe,cal (mg g−1) | k2 (g mg−1 min−1) | R2 | |
| IC | 13.01 | 1.0 × 10−2 | 0.958 | 38.91 | 5.27 × 10−3 | 0.999 |
| MG | 16.48 | 3.01 × 10−2 | 0.979 | 48.78 | 4.05 × 10−3 | 0.999 |
The experimental data was also examined by the pseudo-second-order kinetic model which is given by the following equation:43,44
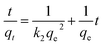 | (8) |
3.5. Thermodynamic analysis
The thermodynamic parameters that must be considered to determine the process are Gibbs free energy change (ΔG0) (eqn (10)), enthalpy change (ΔH0) and entropy change (ΔS0) (eqn (11)) due to the transfer of a unit mole of solute from solution on the solid–liquid interface. These parameters were calculated using the following equations:
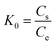 | (9) |
ΔG = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0 K0
| (10) |
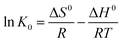 | (11) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0 at a certain temperature was obtained by plotting ln(Cs/Ce) against Cs and extrapolating Cs to zero based on eqn (9).9 The negative values of change in Gibbs free energy (ΔG0) shown in Table 3 indicated the feasibility of the process and the spontaneous nature of the adsorption under the experimental conditions used. ΔH0 and ΔS0 can be determined from the slope and the intercept of the linear plot of ln
K0 at a certain temperature was obtained by plotting ln(Cs/Ce) against Cs and extrapolating Cs to zero based on eqn (9).9 The negative values of change in Gibbs free energy (ΔG0) shown in Table 3 indicated the feasibility of the process and the spontaneous nature of the adsorption under the experimental conditions used. ΔH0 and ΔS0 can be determined from the slope and the intercept of the linear plot of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0 vs. 1/T, as shown in Fig. 5, and the results are also shown in Table 3. The enthalpy change (ΔH0) in Table 3 was positive, which suggested that the adsorption process was an endothermic reaction. The disruption of the magnetic NH2-MIL-101(Al) structure upon exposure to the dyes explains the significantly unfavorable enthalpy upon adsorption, as at least some of the metal–carboxylate bonds that held the structure together were broken and weakened. However, a positive ΔS0 is favorable for spontaneous adsorption of dyes onto magnetic NH2-MIL-101(Al), which may result from the release of more water molecules desolvated from the dye molecules after adsorption onto magnetic NH2-MIL-101(Al).9 Based upon the values of ΔH0 and ΔS0 for the reaction, the destruction of the ordered MOF structure, and the dissolution of Al3+ ions, appear to be the driving force for the interaction.13
K0 vs. 1/T, as shown in Fig. 5, and the results are also shown in Table 3. The enthalpy change (ΔH0) in Table 3 was positive, which suggested that the adsorption process was an endothermic reaction. The disruption of the magnetic NH2-MIL-101(Al) structure upon exposure to the dyes explains the significantly unfavorable enthalpy upon adsorption, as at least some of the metal–carboxylate bonds that held the structure together were broken and weakened. However, a positive ΔS0 is favorable for spontaneous adsorption of dyes onto magnetic NH2-MIL-101(Al), which may result from the release of more water molecules desolvated from the dye molecules after adsorption onto magnetic NH2-MIL-101(Al).9 Based upon the values of ΔH0 and ΔS0 for the reaction, the destruction of the ordered MOF structure, and the dissolution of Al3+ ions, appear to be the driving force for the interaction.13
| Sample | Temperature (K) | ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0 K0 |
ΔG0 (kJ mol−1) | ΔH0 (kJ mol−1 K−1) | ΔS0 (J mol−1 K−1) |
|---|---|---|---|---|---|
| IC | 298 | 6.259 | −15.51 | 2.61 | 60.80 |
| 308 | 6.295 | −16.12 | |||
| 318 | 6.325 | −16.72 | |||
| MG | 298 | 5.103 | −12.64 | 2.20 | 49.80 |
| 308 | 5.129 | −13.13 | |||
| 318 | 5.159 | −13.64 |
 | ||
| Fig. 5 Van't Hoff plot to get the ΔH0 and ΔS0 of IC (a) and MG (b) onto the magnetic NH2-MIL-101(Al). | ||
3.6. Effect of pH
The pH value of the dye solution is recognized as an important factor in the adsorption process, which influences the surface charge, dissociation of functional groups on the active sites, and degree of ionization of the adsorbents and dyes. In order to further investigate the adsorption mechanism, we chose four kinds of dyes to discuss the effect of pH. The existing forms and distribution curves of these dyes under different pH conditions are shown in Table S1.†The zeta potential of the absorbent is the one of the factors which might have an important influence on the adsorption capacity. The zero point charge (pHPZC) is defined as the point at which the zeta potential on the surface of the adsorbent is zero. In aqueous solutions, when pH < pHPZC, the surface charge of a solid is positive. In contrast, it is negative at pH > pHPZC. The pHPZC of magnetic NH2-MIL-101(Al) shown in Fig. 6a was 10. We investigated the effect of pH on the adsorption of dyes onto magnetic NH2-MIL-101(Al) (Fig. 6b and c) and studied the adsorption mechanism.
 | ||
| Fig. 6 The pH effect on the zeta potential of magnetic NH2-MIL-101(Al) (a), effect of the solution pH on the amount of adsorbed anionic (b) and cationic (c) dyes onto magnetic NH2-MIL-101(Al). | ||
The efficiency of removal of adsorbed MO leveled off in the pH range of 4–9, and decreased with further increase of pH from 9 to 12. When pH > 4, MO mainly exists in the form of anions, has an electrostatic interaction with magnetic NH2-MIL-101(Al) carrying positive charge at pH 4–10, but has electrostatic repulsion with adsorbents carrying negative charge at pH > 10. At pH = 3, a part of the MO mainly exists as amphoteric molecules, which reduces the electrostatic interaction with materials. The interaction between IC carrying negative charge and magnetic NH2-MIL-101(Al) was similar to MG.
Compared with the anionic dyes, the cationic dyes showed a contrasting trend. The adsorption efficiency of MG onto magnetic NH2-MIL-101(Al) increased as the pH increased from 3 to 5, then leveled off in the pH range of 5–9. Because of the hydrolysis of MG dye in basic solution, the influence of pH (10–12) on the adsorption capacity of MG was not evaluated further. MG (pKa = 10.3) is protonated in an acidic medium and deprotonated at higher pH.45,46 The removal of MG from the solution showed a fast increase between pH 3 and 5 due to the decreasing positive charge on the surface of magnetic NH2-MIL-101(Al), which weakens the electrostatic repulsion between the cationic MG molecules and the adsorbent surface. At further pH, MG showed high adsorption under conditions with pH > 5, which might be explained by π–π stacking because both the adsorbate and the adsorbent have benzene rings.
MB is another kind of cationic dye existing in the form of two species in the pH range from 3 to 12. Around pH 3, the main form existing is MBH2+. As the pH increases, the main form existing is MB+. When pH < 10, the removal efficiency of MB leveled off. As the solution pH increased from 10 to 12, the negative charges on the surface of magnetic NH2-MIL-101(Al) increased, which was favorable for the adsorption of positively charged MB due to electrostatic attraction.
3.7. Adsorption mechanism
According to the analysis of adsorption kinetics, isotherms, and thermodynamics, it can be considered that the adsorption was related to the chemical action between the dyes and the magnetic NH2-MIL-101(Al). In addition to the electrostatic interaction, there may be other forces between adsorbent and adsorbate.First, due to the sphere of influence of –NH2 involved in the surface of magnetic NH2-MIL-101(Al), it can easily combine with the adsorbate through hydrogen bonding,47,48 which promotes the adsorption of dyes onto magnetic NH2-MIL-101(Al). In addition, there was a potentiality in the structure of magnetic NH2-MIL-101(Al), where the –OH in an octahedral cluster of three AlO5(OH) units can combine with dimethylamino (–N(CH3)2) to form hydrogen bonds.49
In this research, the dyes are compounds endowed with π–π conjugate structure and would have strong affinity with the magnetic NH2-MIL-101(Al) which is rich in aromatic rings. The π–π accumulation force benefits the adsorption of dyes, which resulted in the high adsorption capacity of the dyes.
Hydrophobic interactions are often observed during adsorption of organics from aqueous media. In this work, with the same kind of dye, the adsorption of MO/MG is higher than that of IC/MB, which may contribute to the difference in the partition coefficient P (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P) (Table S2†). The adsorption amounts increased with an increase in the log
P) (Table S2†). The adsorption amounts increased with an increase in the log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P values, indicating the potentially main role of hydrophobic interactions.
P values, indicating the potentially main role of hydrophobic interactions.
Based on the above discussion, we think that the electrostatic interaction is the main force between adsorbent and adsorbate, because when they have opposite charges, the dye adsorption was satisfied. However, forces such as hydrogen bonds, π–π accumulation force, and hydrophobic interactions also exist, because when adsorbent and adsorbate have the same charge, magnetic NH2-MIL-101(Al) also has a certain interaction with dyes.
MOFs are well ordered and highly porous crystalline materials and one of their unique characteristics is the ability to tune their pore size without altering the structural properties. To check the effect of mesoporosity, the adsorption of MG was conducted over virgin materials and meso-structured magnetic NH2-MIL-101(Al). As shown in Fig. 7, it was observed that MG was completely adsorbed in 2 h by the meso-structured NH2-MIL-101(Al) and magnetic NH2-MIL-101(Al), whereas no apparent adsorption of MG onto Fe3O4, Fe3O4@SiO2 was observed after 2 h of adsorption; this clearly indicates the effect of mesopores in accelerating adsorption. The extended structure of amino-MIL-101(Al) is composed of two types of quasi-spherical mesoporous cages formed by 12 pentagonal and 16 hexagonal faces with windows 1.2 and 1.6 nm, respectively, in diameter.29 The measured dimensions of organic dye molecules from the calculation are shown in Table S2.† For the positively charged dye MG+, its two relatively short molecular dimensions along the x and y directions are 9.9 and 4.2 Å, respectively, which are much smaller than the diameter of 12.0 Å. As a result, the access of dye MG+ into the channel of magnetic NH2-MIL-101(Al) is allowed. The same mechanism is suitable for the other kinds of dyes.
3.8. Effect of ionic strength on the adsorption of dyes
Wastewater containing dyes commonly has a high electrolyte concentration, and thus the effect of the electrolyte on the removal of the dyes needed to be clarified to evaluate the real value of magnetic NH2-MIL-101(Al) in the removal of dyes.To investigate the salt effect on the adsorption of dyes, NaCl was used to adjust the solution salinity. The salt could lead to a high ionic strength, which might affect the adsorption of dyes onto the adsorbent. As shown in Fig. 8, the equilibrium adsorption ability for the MB, MO, and IC decreased slightly as the concentration of NaCl increased from 0 to 0.1 mol L−1. The result indicated that there is some competitive adsorption between these dyes and metal ions, but the effect is very small.
 | ||
| Fig. 8 Effect of the concentration of NaCl on the adsorption of MG, MB, MO and IC onto magnetic NH2-MIL-101(Al). | ||
In contrast, for MG, the equilibrium adsorption ability increased slightly as the concentrations of NaCl increased from 0 to 0.1 mol L−1 (Fig. 8). One of the investigation conditions for ionic strength is that the solution is neutral, thus the MG also existed as neutral molecules. The increase in MG adsorption capacity may be due to the salting out effect.
3.9. Recycling of adsorbents
Repeated availability is an important factor for adsorbents. The desorption of cationic dyes (MG and MB) from loaded magnetic NH2-MIL-101(Al) was achieved using 0.1 mol L−1 of HCl/ethanol (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90, v/v), and the desorption of anionic dyes (IC and MO) from loaded magnetic NH2-MIL-101(Al) was achieved using 0.01 mol L−1 of NaOH/methanol (10
90, v/v), and the desorption of anionic dyes (IC and MO) from loaded magnetic NH2-MIL-101(Al) was achieved using 0.01 mol L−1 of NaOH/methanol (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90, v/v).50 In this study, the reusability of the magnetic NH2-MIL-101(Al) was estimated after solvent washing of the used magnetic material. As shown in Fig. 9, the removal rates of MG, IC, MO and MB by magnetic NH2-MIL-101(Al) slightly decrease in successive cycles, gradually stabilizing at about 93%, 88%, 90% and 92%, respectively. The good reusability of spent magnetic NH2-MIL-101(Al) demonstrates the potential of magnetic NH2-MIL-101(Al) for the adsorption and removal of dyes.
90, v/v).50 In this study, the reusability of the magnetic NH2-MIL-101(Al) was estimated after solvent washing of the used magnetic material. As shown in Fig. 9, the removal rates of MG, IC, MO and MB by magnetic NH2-MIL-101(Al) slightly decrease in successive cycles, gradually stabilizing at about 93%, 88%, 90% and 92%, respectively. The good reusability of spent magnetic NH2-MIL-101(Al) demonstrates the potential of magnetic NH2-MIL-101(Al) for the adsorption and removal of dyes.
3.10. Comparison of magnetic NH2-MIL-101(Al) with other adsorbents for the adsorption of dyes
Table 4 shows the adsorption capacities for MG and IC using different adsorbents. The adsorbent used in this study shows a higher adsorption capacity than the most adsorbent materials reported in the literature. Meanwhile, the materials obtained can also be adjusted by pH to adsorb different kinds of dyes from aqueous solution. Moreover, because of the magnetic properties, magnetic NH2-MIL-101(Al) is attractive for its rapid and efficient adsorption, which indicated that the material is superior to other adsorbents.| Adsorbent | Qm (mg g−1) | Temperature (K) | References | |
|---|---|---|---|---|
| MG | MIL-100 | 485 | 323 | 9 |
| MIL-100 | 266 | 303 | 9 | |
| MIL-53 | 34.9 | 303 | 9 | |
| Commercial powder activated carbon | 149 | 303 | 9 | |
| Bamboo-based activated carbon | 264 | 303 | 51 | |
| Cyclodextrin-based material | 91.9 | 298 | 45 | |
| Oil palm trunk fiber | 149 | 303 | 52 | |
| Natural zeolite | 24.5 | 298 | 53 | |
| Chitosan bead | 93.6 | 303 | 54 | |
| Magnetic NH2-MIL-101(Al) | 274.4 | 298 | This work | |
| IC | Rice husk ash | 29.3 | 303 | 55 |
| Zeolitic material | 32.8 | 303 | 56 | |
| Activated sewage sludge | 60.0 | 298 | 57 | |
| Pyrolysis of sewage sludge | 92.8 | 303 | 56 | |
| Nanocomposite hydrogels | 370.4 | 298 | 58 | |
| Magnetic NH2-MIL-101(Al) | 135.0 | 298 | This work |
4. Conclusions
In summary, we have reported the adsorption behavior of magnetic amino-functionalised MOF for cationic and anionic dyes from aqueous solution in view of the adsorption isotherms, thermodynamics, kinetics, and the regeneration of the sorbent. The adsorption follows the pseudo-second-order kinetics model. The best-fit adsorption isotherm was achieved with the Langmuir model. Magnetic NH2-MIL-101(Al), compared with other adsorbents, shows relatively high adsorption amounts for cationic and anionic dyes, probably mainly due to the electrostatic interaction; however, π–π stacking interaction and hydrogen bonding cannot be ruled out. The high adsorption capacity, good solvent stability, and excellent reusability mean that magnetic NH2-MIL-101(Al) is promising as a novel adsorbent for the adsorption and removal of cationic and anionic dyes from aqueous solution.Acknowledgements
This work was supported by Open Project of State Key Laboratory of Urban Water Resource and Environment, Harbin Institute of Technology (No. ES201607) and the National Natural Science Foundation of China (No. 21205010).References
- S. B. Wang, Y. Boyjoo, A. Choueib and Z. H. Zhu, Water Res., 2005, 39, 129–138 CrossRef CAS PubMed.
- Q. Sun and L. Yang, Water Res., 2003, 37, 1535–1544 CrossRef CAS PubMed.
- A. N. Kulkaini, A. A. Kadam, M. S. Kachole and S. P. Govindwar, J. Hazard. Mater., 2014, 276, 461–468 CrossRef PubMed.
- B. Y. Shi, G. H. Li, D. S. Wang, C. H. Feng and H. X. Tang, J. Hazard. Mater., 2007, 143, 567–574 CrossRef CAS PubMed.
- J. W. Lee, S. P. Choi, R. Thiruvenkatachari, W. G. Shim and H. Moon, Water Res., 2006, 40, 435–444 CrossRef CAS PubMed.
- D. Mahanta, G. Madras, S. Radhakrishnan and S. Patil, J. Phys. Chem. B, 2008, 112, 10153–10157 CrossRef CAS PubMed.
- W. X. Chen, W. Y. Lu, Y. Y. Yao and M. H. Xu, Environ. Sci. Technol., 2007, 41, 6240–6245 CrossRef CAS PubMed.
- D. S. Sun, X. D. Zhang, Y. Wu and X. Liu, J. Hazard. Mater., 2010, 181, 335–342 CrossRef CAS PubMed.
- S. H. Huo and X. P. Yan, J. Mater. Chem., 2012, 22, 7449–7455 RSC.
- Z. L. Xie, X. Huang, M. M. Titirici and A. Taubert, RSC Adv., 2014, 4, 37423–37430 RSC.
- H. Y. Zhao, Z. Jin, H. M. Su, J. L. Zhang, X. D. Yao, H. J. Zhao and G. S. Zhu, Chem. Commun., 2013, 49, 2780–2782 RSC.
- E. D. Bloch, W. L. Queen, R. Krishna, J. M. Zadrozny, C. M. Brown and J. R. Long, Science, 2012, 335, 1606–1610 CrossRef CAS PubMed.
- E. Haque, V. Lo, A. I. Minett, A. T. Harris and T. L. Church, J. Mater. Chem. A, 2014, 2, 193–203 CAS.
- H. N. Wang, F. H. Liu, X. L. Wang, K. Z. Shao and Z. M. Su, J. Mater. Chem. A, 2013, 1, 13060–13063 CAS.
- M. L. Ma, J. H. Qin, C. Ji, H. Xu, R. Wang, B. J. Li, S. Q. Zang, H. W. Hou and S. R. Batten, J. Mater. Chem. C, 2014, 2, 1085–1093 RSC.
- W. Xuan, C. Zhu, Y. Liu and Y. Cui, Chem. Soc. Rev., 2012, 41, 1677–1695 RSC.
- T. Wittmann, R. Siegel, N. Reimer, W. Milius, N. Stock and J. Senker, Chem.–Eur. J., 2015, 21, 314–323 CrossRef CAS PubMed.
- G. Férey, C. Mellot-Draznieks, C. Serre, F. Millange, J. Dutour, S. Surblé and I. Margiolaki, Science, 2005, 309, 2040–2042 CrossRef PubMed.
- S. Bernt, V. Guillerm, C. Serre and N. Stock, Chem. Commun., 2011, 47, 2838–2840 RSC.
- C. Chen, M. Zhang, Q. X. Guan and W. Li, Chem. Eng. J., 2012, 183, 60–67 CrossRef CAS.
- A. Henschel, K. Gedrich, R. Kraehnert and S. Kaskel, Chem. Commun., 2008, 35, 4192–4194 RSC.
- D. Y. Hong, Y. K. Hwang, C. Serre, G. Ferey and J. S. Chang, Adv. Funct. Mater., 2009, 19, 1537–1552 CrossRef CAS.
- P. Horcajada, T. Chalati, C. Serre, B. Gillet, C. Sebrie, T. Baati, J. F. Eubank, D. Heurtaux, P. Clayette, C. Kreuz, J. S. Chang, Y. K. Hwang, V. Marsaud, P. N. Bories, L. Cynober, S. Gil, G. Ferey, P. Couvreur and R. Gref, Nat. Mater., 2010, 9, 172–178 CrossRef CAS PubMed.
- K. M. L. Taylor-Pashow, J. Della Rocca, Z. G. Xie, S. Tran and W. B. Lin, J. Am. Chem. Soc., 2009, 131, 14261–14263 CrossRef CAS PubMed.
- H. Chen, C. Deng and X. Zhang, Angew. Chem., Int. Ed., 2010, 49, 607–611 CrossRef CAS PubMed.
- S. Mendoza-Bello, R. A. Morales-Luckie, L. Flores-Santos, J. P. Hinestroza and V. Sanchez-Mendieta, J. Nanopart. Res., 2012, 14, 1242–1251 CrossRef.
- T. Tsoncheva, J. Roggenbuck, D. Paneva, M. Dimitrov, I. Mitov and M. Fröba, Appl. Surf. Sci., 2010, 257, 523–530 CrossRef CAS.
- Z. C. Kadirova, K. Katsumata, T. Isobe, N. Matsushita, A. Nakajima and K. Okada, Appl. Surf. Sci., 2013, 284, 72–79 CrossRef CAS.
- S. Jung, Y. Kim, S. Kim, T. H. Kwon, S. Huh and S. Park, Chem. Commun., 2011, 47, 2904–2906 RSC.
- Z. Hasan, E. J. Choi and S. H. Jhung, Chem. Eng. J., 2013, 219, 537–544 CrossRef CAS.
- E. Stavitski, M. Goesten, J. Juan-Alcaniz, A. Martinez-Joaristi, P. Serra-Crespo, A. V. Petukhov, J. Gascon and F. Kapteijn, Angew. Chem., Int. Ed., 2011, 50, 9624–9628 CrossRef CAS PubMed.
- P. E. Serra-Crespo, V. Ramos-Fernandez, J. Gascon and F. Kapteijn, Chem. Mater., 2011, 23, 2565–2572 CrossRef CAS.
- I. Langmuir, J. Am. Chem. Soc., 1916, 38, 2221–2295 CrossRef CAS.
- H. M. F. Freundlich, J. Phys. Chem., 1906, 57, 1100–1107 Search PubMed.
- M. M. Dubinin, E. D. Zaverina and L. V. Radushkevich, J. Phys. Chem., 1947, 2, 1351–1362 Search PubMed.
- Y. F. Huang, Y. Q. Wang, Q. S. Zhao, Y. Li and J. M. Zhang, RSC Adv., 2014, 4, 47921–47924 RSC.
- M. R. Malekbala, S. Hosseini, S. Kazemi-Yazdi, S. Masoudi Soltani and M. R. Malekbala, Chem. Eng. J., 2013, 229, 257–266 CrossRef.
- T. Wang, K. Kailasam, P. Xiao, G. Chen, L. Chen, L. Wang, J. Li and J. Zhu, Microporous Mesoporous Mater., 2014, 187, 63–70 CrossRef CAS.
- K. Hall, L. Eagleton, A. Acrivos and T. Vermeulen, Ind. Eng. Chem. Fundam., 1966, 5, 212–223 CAS.
- Y. Yu, B. N. Murthya, J. G. Shapter, K. T. Constantopoulos, N. H. Voelcker and A. V. Ellis, J. Hazard. Mater., 2013, 260, 330–338 CrossRef CAS PubMed.
- M. S. Chiou and H. Y. Li, J. Hazard. Mater., 2002, 93, 233–248 CrossRef CAS PubMed.
- M. Kousha, E. Daneshvar, M. Salar-Sohrabi, M. Jokar and A. Bhatnagar, Chem. Eng. J., 2012, 192, 67–76 CrossRef CAS.
- B. Sarkar, Y. Xi, M. Megharaj and R. Naidu, Appl. Clay Sci., 2011, 51, 370–374 CrossRef CAS.
- Y. S. Ho and G. McKay, Process Biochem., 1999, 34, 451–465 CrossRef CAS.
- G. Crini, H. N. Peindy, F. Gimbert and C. Robert, Sep. Purif. Technol., 2007, 53, 97–110 CrossRef CAS.
- I. D. Mall, V. C. Srivastava, N. K. Agarwal and I. M. Mishra, Colloids Surf., A, 2005, 264, 17–28 CrossRef CAS.
- Q. Chen, Q. He, M. Lv, Y. Xu, H. Yang, X. Liu and F. Wei, Appl. Surf. Sci., 2015, 327, 77–85 CrossRef CAS.
- B. Liu, F. Yang, Y. Zou and Y. Peng, J. Chem. Eng. Data, 2014, 59, 1476–1482 CrossRef CAS.
- I. Ahmed and S. H. Jhung, J. Hazard. Mater., 2015, 283, 544–550 CrossRef CAS PubMed.
- Y. Y. Jia, Q. Jin, Y. Li, Y. X. Sun, J. Z. Huo and X. J. Zhao, Anal. Methods, 2015, 7, 1463–1470 RSC.
- B. H. Hameed and M. I. El-Khaiary, J. Hazard. Mater., 2008, 157, 344–351 CrossRef CAS PubMed.
- B. H. Hameed and M. I. El-Khaiary, J. Hazard. Mater., 2008, 154, 237–244 CrossRef CAS PubMed.
- R. P. Han, Y. Wang, Q. Sun, L. L. Wang, J. Y. Song, X. T. He and C. C. Dou, J. Hazard. Mater., 2010, 175, 1056–1061 CrossRef CAS PubMed.
- K. Yurdakoc, Z. Bekci, C. Ozveri and Y. Seki, J. Hazard. Mater., 2008, 154, 254–261 CrossRef PubMed.
- U. R. Lakshmi, V. C. Srivastava, I. D. Mall and D. H. Lataye, J. Environ. Manage., 2009, 90, 710–720 CrossRef CAS PubMed.
- E. Gutiérrez-Segura, M. Solache-Rios and A. Colin-Cruz, J. Hazard. Mater., 2009, 170, 1227–1235 CrossRef PubMed.
- M. Otero, F. Rozada, L. F. Calvo, A. I. García and A. Morán, Dyes Pigm., 2003, 57, 55–65 CrossRef CAS.
- M. Dalaran, S. Emik, G. Güçlü, T. B. İyim and S. Özgümüş, Desalination, 2011, 279, 170–182 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra07567c |
| This journal is © The Royal Society of Chemistry 2016 |


