Controlled deposition of Pt nanoparticles on Fe3O4@carbon microspheres for efficient oxidation of 5-hydroxymethylfurfural
Yuwei Zhanga,
Zhimin Xueb,
Jinfang Wanga,
Xinhui Zhaoa,
Yonghui Deng*c,
Wancheng Zhaoa and
Tiancheng Mu*a
aDepartment of Chemistry, Renmin University of China, Beijing 100872, P. R. China. E-mail: tcmu@ruc.edu.cn; Tel: +86-10-62514925
bBeijing Key Laboratory of Lignocellulosic Chemistry, College of Materials Science and Technology, Beijing Forestry University, Beijing 100083, P. R. China
cDepartment of Chemistry, Shanghai Key Laboratory of Molecular Catalysis and Innovative Materials, State Key Laboratory of Molecular Engineering of Polymers, Fudan University, Shanghai, 200433, P. R. China. E-mail: yhdeng@fudan.edu.cn; Tel: +86-21-65643292
First published on 20th April 2016
Abstract
2,5-Furandicarboxylic acid (FDCA) is an important environmentally benign and sustainable chemical which can be derived from biomass and produced by the oxidization of 5-hydroxymethylfurfural (HMF). However, the oxidation of HMF relies highly on catalysts to improve the yield of FDCA. In this study, a series of novel superparamagnetic Pt nanoparticle-containing catalysts with a core–shell structure (Fe3O4@C@Pt) were synthesized and applied for HMF oxidation. These novel spherical catalysts possess a Fe3O4 core and a protective amorphous carbon shell with a surface decorated by Pt nanoparticle clusters. By changing the synthesis temperature, the morphology of the active Pt species on the carbon shell of the microspheres can be transformed from highly dispersed nanoparticles to nanoparticle clusters. The catalytic HMF oxidation results reveal that microspheres decorated with larger nanoparticle clusters (110-Fe3O4@C@Pt) have the best catalytic activity for HMF oxidation, owing to a unique islet morphology coupled with the high-degree crystallization of Pt nanoparticles. The yield of FDCA could reach 100% after 4 h of reaction at 90 °C in water which is superior to previous reports. Furthermore, this catalyst can be reused at least three times without significant performance loss.
1. Introduction
Nowadays, the depletion of non-renewable fossil resources is a major global concern, but the demand for fuels and chemicals derived from fossil energies is still increasing in the long term.1 While considering the consumption of fossil resources and the increasing prices of crude oil,2,3 it becomes more and more attractive to find renewable alternatives to replace fossil ones.4–6 To this end, biomass, a renewable resource which directly or indirectly comes from green plants, should be a good substitute with prominent potential to be converted to a wide variety of valuable compounds which include chemicals, fuels and materials.7–12 Compared with fossil resources, biomass resources, typically lignocellulosic biomass, seafood waste13 and biomass-derived chemicals,14–18 are a kind of abundant, renewable, widely-distributed and green resource in the world.19,20 Therefore, numerous efforts have been made to convert biomass into a variety of valuable chemicals.21–245-Hydroxymethylfurfural (HMF), which can be easily generated from biomass containing glucose, fructose, cellulose and any other carbohydrate, is regarded as a promising chemical platform compound,25,26 holding great potential to be used as a precursor of important industrial chemicals.27–30 In addition, 2,5-furandicarboxylic acid (FDCA) could be easily synthesized through the oxidation of HMF,31 which could be used as a good intermediate to produce a lot of chemicals32 and as an excellent alternative for poly(ethylene terephthalate) (PET).33–35 Conventionally, stoichiometric oxidation of HMF using KMnO4 as an oxidant and homogeneous metal salts (Co/Mn) as catalysts has been employed to produce FDCA and the yield of FDCA was around 60%.36 However, homogeneous catalysts are normally not environmentally friendly and are extremely difficult to recycle. Recently, recyclable heterogeneous catalysts based on precious metal nanoparticles (Au, Pt, Pd) supported on metal oxides like ceria and titania have been proven to facilitate the oxidation of HMF into FDCA.37–39 The oxidation of HMF into FDCA should be divided into two steps in which HMF is converted to 5-hydroxymethyl-2-furancarboxylic acid (HMFCA) or 2,5-diformylfuran (DFF) first, and then HMFCA and/or DFF are further oxidized into FDCA (Scheme 1).40 In consideration of the requirements of green chemistry, heterogeneous catalysts with excellent long-term catalytic performance, high selectivity and especially good recyclability are highly desired. More recently, a magnetically recyclable heterogeneous catalyst based on Pd nanoparticles has been used to convert HMF to FDCA showing a maximum yield of FDCA of around 86.7% in 10 hours.28 However, the yield of FDCA is not 100%, so there is still some space to improve the yield of FDCA and the morphology of catalyst is not good. Therefore, it is still of great interest to explore other kinds of recyclable catalysts for the oxidation of HMF that exhibit a higher conversion, selectivity and reaction rate. As it is a heterogeneous catalytic process, the size of the active phase is one of the most important factors determining the catalytic performance.41 Therefore, exploring effective methods to control the nucleation and growth of the supported metallic particles and eventually to design size-variable catalysts is also crucial for the development of high performance heterogeneous catalysts for the oxidation of HMF.
On the other hand, due to the rapid development of nanotechnology and nanoscience, researchers have been able to design and assemble different kinds of components in the nanoscale. Fine control of the properties of each component enables the resulting composite materials to be applied in diverse applications with boosted performance.42,43 Recently, core–shell structured microspheres as a family of unique composite materials have attracted increasing interest in various fields, including biomedicine, catalysis electronics, controlled drug release, batteries, cell-labeling and so on.44–49 In addition, core–shell microspheres are also important from an economic viewpoint. Microspheres with a magnetic core and functional shell are an important core–shell material, since they can provide unique magnetic properties which allow them to be easily manipulated from a distance. In particular, noble nanoparticles are often loaded on magnetic core–shell microspheres, which provides them with magnetic features and a separable character and means that the valuable metallic species can be reused and recovered during catalysis and adsorption.50–52
Besides the recyclability, the morphological control of size and shape is of great importance for metallic noble nanoparticles since their performance is directly related to their physical properties. As a result, their unique thermal, optical, electronic and catalytic features can be easily tuned by varying the size and shape of metallic nanoparticles.53 Such morphological control of metallic nanoparticles is generally accomplished by controlling the reduction rate of the metal ions at the growth stage of the metallic nanoparticles via different methods, such as the hydrothermal method, microemulsion and sonochemical reduction.54–58
In this regard, here we reported a core/shell structured magnetically recyclable catalyst with a well-defined spherical morphology consisting of a magnetic core of an Fe3O4 particle, a protective shell of amorphous carbon and Pt nanoparticles/clusters as active sites decorated onto the microsphere. By varying the synthetic parameters, the morphologies of the Pt nanoparticles/clusters could be tailored from highly dispersed nanoparticles of below 1 nm to discrete nanoclusters of around 25 nm. Using these core/shell microspheres as catalysts for the oxidation of HMF, a 100% yield of FDCA could be achieved in just 4 h at 90 °C with complete conversion of HMF. Furthermore, this catalyst can be reused at least three times without significant performance loss.
2. Experimental section
2.1. Materials
FeCl3 (≥97%), anhydrous sodium acetate (≥99%), trisodium citrate dehydrate (≥99.5%), ethylene glycol (≥99%), methanol (HPLC grade), potassium hexachloroplatinate (Pt 39.9–40.2%), ethanol (98%), and anhydrous sodium carbonate (99.8%) were purchased from Sinopharm Chemical Reagent Co., Ltd. Glucose (>98%) was purchased from TCI. 5-Hydroxymethylfurfural (98%), 5-hydroxymethyl-furan-2-carboxylic acid (>95%), 2,5-furandicarboxylic acid (97%) and 2,5-diformylfuran (98%) were purchased from J&K Chemical Co. Ltd.2.2. Preparation of Fe3O4 particles
Highly water-dispersible Fe3O4 microspheres were synthesized by a solvothermal reaction.59 Typically, FeCl3 (1.3 g), trisodium citrate (0.5 g) and sodium acetate (2.0 g) were dissolved in ethylene glycol (80 mL) with magnetic stirring. After a yellow solution was obtained, the solution was then transferred to and sealed in a 100 mL stainless-steel autoclave. The autoclave was heated to 200 °C and kept at this temperature for 10 h. After cooling to room temperature, a black product (Fe3O4 particles) was obtained and subsequently washed with ethanol and deionized water separately.2.3. Synthesis of Fe3O4@C core/shell microspheres coated by an amorphous carbon shell
150 mg of the obtained Fe3O4 particles was dispersed in an aqueous solution of glucose (60 mL 0.25 M), then subjected to hydrothermal treatment in a 100 mL stainless-steel autoclave at 180 °C for 6 h. After cooling to room temperature, core/shell microspheres of Fe3O4@C were obtained and washed with ethanol and deionized water separately.2.4. Preparation of Fe3O4@C@Pt catalyst decorated by Pt nanoparticles/clusters with different morphologies
80 mg of Fe3O4@C microspheres was dispersed in 100 mL of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 water/ethanol solution by ultrasonic treatment. Then the resulting solution was refluxed for 1.5 h in the presence of 5 mL 0.1 M K2PtCl6 at 90 °C, 100 °C and 110 °C, respectively. Finally, the obtained Pt nanoparticle immobilized magnetic microspheres (denoted as X-Fe3O4@C@Pt, where X indicates the refluxing temperatures) were obtained. After washing with deionized water and ethanol three times each, these obtained magnetic core/shell microspheres were vacuum dried overnight at 30 °C for the following catalytic reaction.
1 water/ethanol solution by ultrasonic treatment. Then the resulting solution was refluxed for 1.5 h in the presence of 5 mL 0.1 M K2PtCl6 at 90 °C, 100 °C and 110 °C, respectively. Finally, the obtained Pt nanoparticle immobilized magnetic microspheres (denoted as X-Fe3O4@C@Pt, where X indicates the refluxing temperatures) were obtained. After washing with deionized water and ethanol three times each, these obtained magnetic core/shell microspheres were vacuum dried overnight at 30 °C for the following catalytic reaction.
2.5. Characterization of the catalyst
Transmission electron microscopy (TEM) images were taken with a JEOL 1011 transmission electron microscope operated at 100 kV. Samples were first dispersed in ethanol and then collected using carbon-film-covered copper grids for analysis. Scanning electron microscopy (SEM) images were collected on a field emission scanning electron microscope JEOL JSM-7401F operating at 5 kV. X-ray powder diffraction (XRD) patterns of the samples were recorded using an X-ray diffractometer (Rigaku D/max-2500) using Cu Kα as X-ray radiation (λ = 1.5418 Å) under 40 kV and 200 mA. The Pt content in the X-Fe3O4@C@Pt microspheres was determined using inductively coupled plasma optical emission spectrometry (ICP-OES). The magnetic hysteresis loops were recorded using a DKT-4500 vibrating sample magnetometer (VSM) DKT-4500.2.6. Oxidation of 5-hydroxymethylfurfural (HMF)
15 mg of HMF and different amounts of anhydrous sodium carbonate (Na2CO3) were dissolved in water at 90 °C with an oxygen flow rate of 100 mL min−1. Then a certain amount of X-Fe3O4@C@Pt catalyst, equivalent to 0.86 mg of Pt determined by ICP-OES, was introduced to the mixture to initiate the reaction. During the reaction, oxygen was permitted to flow at the bottom of the reactor continuously. After the reaction, the catalyst was collected using an applied magnetic field, washed with ethanol and deionized water separately and dried under vacuum (40 °C) for the next recycling reaction.2.7. Analytical methods
The products were analysed using an Agilent 1100 Series HPLC. Final compounds were separated using a reversed-phase C18 column (4.6 × 250 mm) and detected using a UV detector at a wavelength of 254 nm. The mobile phase was methanol and 0.1 wt% acetic acid aqueous solution with a volume ratio of 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70. The flow rate was 0.03 mL min−1. The conversion of 5-hydroxymethylfurfural (HMF), the yield of 5-hydroxymethyl-furan-2-carboxylic acid (HMFCA), 2,5-furandicarboxylic acid (FDCA) and 2,5-diformylfuran (DFF) were determined using interpolation from calibration curves.
70. The flow rate was 0.03 mL min−1. The conversion of 5-hydroxymethylfurfural (HMF), the yield of 5-hydroxymethyl-furan-2-carboxylic acid (HMFCA), 2,5-furandicarboxylic acid (FDCA) and 2,5-diformylfuran (DFF) were determined using interpolation from calibration curves.
3. Results and discussion
3.1. Synthesis of Fe3O4@C@Pt microspheres
The procedure for synthesizing magnetic Fe3O4@C@Pt microspheres with different morphologies of Pt nanoparticles is shown in Scheme 2. Firstly, the magnetite Fe3O4 particles were synthesized using a solvothermal method by reducing Fe(III) salts with ethylene glycol in the presence of trisodium citrate at a high temperature. Then Fe3O4 particles were coated with a uniform carbon layer in the presence of glucose to generate core/shell Fe3O4@C microspheres via hydrothermal reaction. After that, Pt nanoparticles were introduced and decorated on the surface of these carbon protected microspheres through refluxing in a water/ethanol solution at three different temperatures of 90, 100 and 110 °C. The Pt content of the obtained core/shell structured microspheres 90-Fe3O4@C@Pt, 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt was 14.33%, 17.71% and 15.25%, respectively, examined using ICP-OES.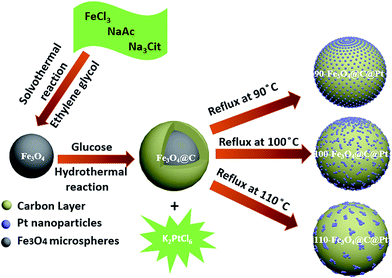 | ||
| Scheme 2 Schematic illustration of the synthesis of superparamagnetic core/shell X-Fe3O4@C@Pt microspheres, where X indicates the refluxing temperature. | ||
It can be seen from the SEM image (Fig. 1a) that the Fe3O4 magnetic particles exhibit a nearly spherical shape. After coating with a carbon layer, the obtained Fe3O4@C microspheres exhibit a more regular spherical shape with a smoother surface compared to the magnetic particles (Fig. 1b). Numerous embossments could be observed from the SEM images (Fig. 1c–e and the red arrow point out embossments and Fig. 1f indicates the model of 110-Fe3O4@C@Pt) after the deposition of Pt nanoparticles on the external surface of the microspheres. When increasing the refluxing temperature from 90 °C to 110 °C (from Fig. 1c–e), the embossments become more and more noticeable indicating the enlargement and the aggregation of the introduced Pt nanoparticles. All of the Fe3O4 magnetic particles (Fig. 1a), Fe3O4@C microspheres (Fig. 1b) and obtained Fe3O4@C@Pt microspheres (Fig. 1c–e) prepared under different temperatures have a discrete spherical morphology.
The elemental composition of X-Fe3O4@C@Pt was determined using energy dispersive X-ray spectroscopy (EDS). The presence of carbon, iron, oxygen and platinum were confirmed using EDS spectroscopy (Fig. 2). TEM images show that the magnetic particles synthesized via solvothermal reaction have a mean diameter of ∼180 nm with polycrystalline features (Fig. 3a). After hydrothermal treatment, a thin carbon layer ∼15 nm in thickness was formed on the surface of the magnetic particles, resulting in core/shell structured microspheres (Fig. 3b and c). In a typical sample after the deposition of Pt nanoparticles at 90 °C, ultrafine Pt nanoparticles with a diameter below ∼1 nm were uniformly decorated on the carbon shell, resulting in a nearly continuous Pt layer (Fig. 3d, inset). When the refluxing temperature increased to 100 °C, Pt nanoparticles began to grow larger and aggregate on the surface of the Fe3O4@C spheres, exhibiting island-like morphology (Fig. 3e and inset). A further increase of the refluxing temperature to 110 °C causes the Pt nanoparticles on 110-Fe3O4@C@Pt microspheres to aggregate severely, resulting in larger clusters of ∼20 nm.
 | ||
| Fig. 2 EDS images of 90-Fe3O4@C@Pt (a), 100-Fe3O4@C@Pt (b) and 110-Fe3O4@C@Pt (c) synthesized at different temperatures. | ||
Fig. 4 shows wide angle XRD patterns of each sample. It can be seen that all of the peaks are well in agreement with the pure Fe3O4 phase. All of the X-Fe3O4@C@Pt samples show a characteristic diffraction peak at 2θ of 40° indexed to the (111) lattice plane of the Pt, confirming the presence of Pt in the composites. Moreover, when the synthesis temperature for depositing Pt on the Fe3O4@C increases, the characteristic peak for Pt (2θ = 40°) becomes stronger, which is due to the enlargement of the particle size and the enhancement of crystallisation.
 | ||
| Fig. 4 XRD patterns of Fe3O4 magnetic particles (a), Fe3O4@C core/shell microspheres (b), and obtained 90-Fe3O4@C@Pt (c), 100-Fe3O4@C@Pt (d) and 110-Fe3O4@C@Pt (e) superparamagnetic microspheres. | ||
The magnetic properties of 90-Fe3O4@C@Pt, 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt microspheres were characterized using a vibrating sample magnetometer (VSM) at room temperature in an applied magnetic field from −5000 to +5000 Oe. All of the Pt-decorated core–shell microspheres show typical hysteresis loops without any detectable remanence or coercivity, reflecting a superparamagnetic property. The magnetization saturation values of 90-Fe3O4@C@Pt, 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt microspheres are 11.95, 7.44 and 6.27 emu g−1, respectively (Fig. 5).
 | ||
| Fig. 5 Magnetic hysteresis loops of the 90-Fe3O4@C@Pt (a), 100-Fe3O4@C@Pt (b) and 110-Fe3O4@C@Pt (c) microspheres. | ||
The successful synthesis of X-Fe3O4@C@Pt microspheres can mainly be attributed to the following aspects. Firstly, the synthesized magnetite particles are coordinated by abundant citrate groups, and thus are highly dispersible in water and ethanol, favouring the interface interaction between magnetic particles and glucose molecules via hydrogen bonding. Thus, a thin carbonaceous layer can be deposited on the magnetite particle under hydrothermal conditions. Secondly, the reductive groups on the carbonaceous shell serving as reducing agents can reduce KPtCl6 in high temperature refluxing conditions, which leads to the controllable deposition of Pt nanoparticles. The temperature effect is very important too. When the temperature of refluxing was set at 90 °C which is high enough for the nucleation of Pt species, extremely tiny Pt nanoparticles were deposited onto the carbon shell. Notably, the reductive groups such as –OH and –CHO, derived from the dehydration of glucose, are evenly distributed on the carbon shell, which makes it possible for Pt nanoparticles to be generated homogeneously and be highly dispersed over the carbon surface. However, a sintering process can occur when the refluxing temperature was elevated from 90 to 100 °C, due to particle migration and growth. As a result, Pt nanoparticles can grow to ∼2 nm and form several slightly separated islands of particle aggregates. Further increase of the refluxing temperature to 110 °C can cause Pt nanoparticles to aggregate into large clusters of ∼25 nm on the carbon surface.
3.2. Oxidation of HMF in mild condition
The aerobic oxidation of HMF towards FDCA under different temperatures in mild conditions was carried out using water as solvent with diverse amounts of base in order to estimate the catalytic performance of all three as-synthesized X-Fe3O4@C@Pt microspheres. The yields of the product, FDCA, were measured and monitored using high performance liquid chromatography (HPLC), and the corresponding results are shown in Fig. 6.The 3D diagram in Fig. 6a shows the yield of FDCA versus reaction time and temperature. The test was carried out with 0.2 mmol anhydrous sodium carbonate Na2CO3 and under an oxygen flow of 100 mL min−1 but with different reaction temperatures which ranged from 30 to 90 °C. When using the obtained Pt-containing magnetic core/shell microspheres as catalyst, low FDCA yields of 29.3, 35.5 and 36.9% are obtained after 10 h at 30 °C for samples 90-Fe3O4@C@Pt, 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt, respectively (Fig. 6a). These values of FDCA yields are however greatly improved to 77.7, 78.3 and 81.1% by increasing the reaction temperature to 60 °C for samples 90-Fe3O4@C@Pt, 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt, respectively (Fig. 6a). Further increasing the reaction temperature to 90 °C results in continuously improved FDCA yields of up to 87.1, 100 and 100% for samples 90-Fe3O4@C@Pt, 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt, respectively (Fig. 6a), which indicates that the high reaction temperature has a positive effect on the yield of FDCA. Compared with other catalysts, Gorbanev et al. reported that a 71% yield of FDCA was obtained at full HMF conversion in the presence of excess base.27 Wang et al. reported that almost a 68.6% yield of FDCA was achieved after 15 h at 80 °C,60 but in this study, we obtained a 100% yield of FDCA after 4 h at 90 °C.
Not only the reaction temperature, but also the amount of Na2CO3 is critical to the yield of FDCA. In order to study the role of base during the oxidation of HMF into FDCA catalyzed by our magnetic catalysts, another test was carried out at the optimal temperature of 90 °C under a constant oxygen flow of 100 mL min−1 but with different amounts of anhydrous Na2CO3 (Fig. 6b). With a decrease of Na2CO3 from 0.2 mmol to 0.1 mmol, the yields of FDCA after 10 hours of reaction over catalysts 90-Fe3O4@C@Pt, 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt show an obvious drop from ∼87.1, 100, and 100% to ∼85.9, 86.1, and 86.5%, respectively. When HMF oxidation was conducted without the addition of any Na2CO3 over all of the employed catalysts, the yield of FDCA exhibited a further decrease down to 34.1, 46.6, and 48.4% over catalysts 90-Fe3O4@C@Pt, 100-Fe3O4@C@Pt, and 110-Fe3O4@C@Pt, respectively. This yield drop, accompanied by the decrease of the Na2CO3/HMF mole ratio, indicates that an appropriate amount of Na2CO3 is necessary to obtain a high performance towards the desired product of FDCA over our synthesized Pt-containing core/shell structured catalysts. During HMF oxidation, the Na2CO3 is used to increase the yield of FDCA and increase its solubility in water, which can effectively prevent the generated FDCA from precipitating on the surface of the catalyst, resulting in high catalyst performance and low by-product formation.16,34 Compared with our reaction system, the base-free oxidation of HMF to FDCA reaction requires a relatively higher reaction temperature and longer reaction time, and low concentrations of HMF should be utilized to keep FDCA within its solubility limit. For example, Wan et al. reported that a 94% yield of FDCA was achieved after 12 h at 100 °C;31 an 88% yield of FDCA was achieved after 10 h at 120 °C in Zhang’s group;61 Zhou et al. reported that a 98% yield of FDCA was obtained after 14 h at 90 °C;62 while Siankevich et al. found that a 95% yield of FDCA was achieved after 24 h at 80 °C.63 However, in this study, a 100% yield of FDCA was obtained in 4 h at 90 °C.
It could be concluded from the above results that the optimized conditions for this HMF oxidation over our synthesized catalysts should be at the temperature of 90 °C with 0.2 mmol Na2CO3. It is worth noting that among these three employed magnetic core/shell microsphere catalysts, both samples 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt, which have large Pt cluster domains, can achieve a full yield of 100% towards the desired product FDCA under optimized conditions within 10 hours of reaction, while 90-Fe3O4@C@Pt with ultra-dispersed Pt nanoparticles gave a yield of only 87.1% (Fig. 6a). Interestingly, the 110-Fe3O4@C@Pt sample with larger Pt clusters shows a faster reaction rate than 100-Fe3O4@C@Pt under the optimized conditions. Using the 100-Fe3O4@C@Pt catalyst, a 100% FDCA yield could be achieved after reaction for 8 h (Fig. 6a); while the 110-Fe3O4@C@Pt catalyst only requires a much shorter time of 4 h to achieve a 100% FDCA yield. To the best of our knowledge, the catalytic performance of 110-Fe3O4@C@Pt is better than the other two catalysts. We assume that the significant difference in the catalytic performance of these catalysts is mainly contributed by the distinct morphology of the Pt cluster. Firstly, 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt microspheres synthesized under higher refluxing temperatures of 100 and 110 °C, respectively, possess Pt nanoparticles with a high degree of crystallization, which can be confirmed from their XRD patterns. Compared to 90-Fe3O4@C@Pt synthesized under a lower temperature of 90 °C, 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt with well-crystallized Pt nanoparticle clusters promote the reaction degree and enhance the yield of FDCA during HMF oxidation. Secondly, different from ultrasmall Pt nanoparticles in the 90-Fe3O4@C@Pt sample, the Pt nanoparticle clusters in 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt possess a lower surface energy, which means that they cannot be covered and isolated easily by reactants or products that lower their catalytic performance during the reaction. Thirdly, the 110-Fe3O4@C@Pt catalyst has large Pt clusters composed of numerous small nanoparticles, which endows these Pt islets with a porous feature due to interparticle porosity favouring the enrichment of reactants that facilitate the activity. This unique islet morphology coupled with the high-degree crystallization of Pt nanoparticles make the catalyst 110-Fe3O4@C@Pt exhibit an outstanding catalytic performance resulting in the highest FDCA yield and fastest reaction rate compared to other catalysts. In addition, different kinds of carbon have different influences on the active phase of metal nanoparticles. For example, carbon and metal nanoparticles can have electronic interactions modifying the properties of metal nanoparticles which make the catalytic activities of Pd–PGO in the oxidation reaction superior to commercial catalysts.64 Compared with commercial Pt/C catalysts, various carbon materials with high surface areas are useful supports in heterogeneous catalysts, which exhibit a higher catalytic ability for the oxidation of HMF than commercial Pt/C catalysts. For example, Zhou et al. used Pt supported on a carbon nanotube as a heterogeneous catalyst for the oxidation of HMF, and a 98% yield of FDCA was achieved after 14 h at 90 °C.62 In our study, reductive groups such as –OH and –CHO, derived from the dehydration of glucose, are evenly distributed on the carbon shell, which makes it possible for Pt nanoparticles to be generated homogeneously and highly dispersed over the carbon surface, and a 100% yield of FDCA was achieved after 4 h at 90 °C.
To obtain a better understanding of the pathway of the HMF oxidation catalyzed by our synthesized magnetic catalysts, the intermediate products of HMFCA and DFF were also recorded during the reaction (Fig. 7). The yield of HMFCA versus reaction time under different temperatures of 30 °C, 60 °C and 90 °C with a base amount of n(Na2CO3) = 0.2 mmol over 90-Fe3O4@C@Pt, 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt was recorded in Fig. 7a. Clearly, the yield of HMFCA increases with the progress of the reaction at a lower temperature of 30 °C. However, it decreased with the progress of the reaction when the reaction temperature was increased to 60 °C or 90 °C. This is because a higher temperature facilitates the oxidation of the intermediate product HMFCA. As a result, HMFCA can be rapidly converted to the final product FDCA at a higher temperature. Notably, only HMFCA was detected without any trace of DFF using the synthesized X-Fe3O4@C@Pt catalysts, which indicates that the oxidation of HMF at our selected temperature with a Na2CO3 amount of n(Na2CO3) = 0.2 mmol was accomplished through the pathway with HMFCA as the intermediate.
The influence of the base amount on the yield of intermediate HMFCA was also investigated by carrying out reactions at 90 °C under an oxygen flow of 100 mL min−1. It is worth noting here that during oxidation a small amount (below ∼1%) of the competitive intermediate product DFF was detected in the absence of Na2CO3. These results clearly indicate that HMF was oxidized into FDCA mainly through the pathway with HMFCA as the only intermediate product when base was employed, whereas competitive pathways through both DFF and HMFCA as intermediates occurred without the use of base. The yield of HMFCA versus reaction time using different amounts of anhydrous sodium carbonate (Na2CO3) (from 0 to 0.2 mmol) over 90-Fe3O4@C@Pt, 100-Fe3O4@C@Pt and 110-Fe3O4@C@Pt was recorded in Fig. 7b. It can be seen that at the initial reaction stage, when the amount of base increased, the yield of HMFCA over all three catalysts increased significantly. With the progress of the reaction, the yield of HMFCA over all three catalysts decreased gradually to below 3% until the end of the reaction, suggesting that the intermediate HMFCA was further oxidized to FDCA quickly. When 110-Fe3O4@C@Pt was used as catalyst with the amount of base being 0.2 mmol, the yield of HMFCA decreased much faster to 0 after only 4 h, which means that the intermediate HMFCA underwent a fast oxidation, and was rapidly and completely oxidized to FDCA by 110-Fe3O4@C@Pt. These results suggest that the partially oxidized product HMFCA is generated as the major intermediate when the base is employed, which also supports the reaction route involving the fast oxidation of the aldehyde groups to carboxyl ones as the only intermediate process. Among the three catalysts synthesized in this work, 110-Fe3O4@C@Pt exhibits a higher catalytic activity than the others in the oxidization. And from the HPLC, during oxidation a small amount (below ∼1%) of the competitive intermediate product DFF was detected in the absence of Na2CO3. These HPLC results clearly indicated that HMF was oxidized into FDCA mainly through the pathway with HMFCA as the only intermediate product with the usage of Na2CO3. Pt was able to effectively oxidize the HMFCA to FDCA, which indicates that Pt can activate the alcohol side chain of HFCA which was proven by the following references38,40,65.
3.3. The recycling of catalysts
To explore the reusability of magnetic core/shell Pt-containing microspheres, these catalysts were recycled from the reaction system using an applied magnetic field at the end of HMF oxidation. After washing with ethanol and water thoroughly, the magnetic nanocatalysts were dried and reused in a new reaction cycle. This reuse process was carried out under optimal reaction conditions which was at 90 °C. The catalysts are allowed to react for 10 h for each cycle. Fig. 8a shows the magnetic separation process of the microsphere catalysts 90-Fe3O4@C@Pt from solvent achieved using a hand-held magnet after the first reaction cycle. It can be seen that the colour of the solvent is brown after the 10 h reaction, which is due to a trace of by-product humins with a brown colour generated during the reaction caused by the addition of base. As shown in Fig. 8b, all three synthesized catalysts can be reused at least three times without significant performance loss and 110-Fe3O4@C@Pt shows the best yield of FDCA.4. Conclusions
In summary, a series of heterogeneous catalysts were prepared by the controllable deposition of Pt nanoparticles/clusters as active sites onto a core–shell structure microsphere, which consisted of a magnetic core of a Fe3O4 particle and a protective shell of amorphous carbon. When changing the refluxing temperature for the in situ reduction of the Pt precursor, the morphology of the generated Pt species on the surface of the microspheres can be varied from highly dispersed ultrasmall nanoparticles to nanoparticle clusters. When these superparamagnetic Pt nanoparticle-containing core–shell microspheres were applied as catalysts for the oxidation of HMF to FDCA, Fe3O4@C@Pt with a Pt morphology of nanoparticle clusters exhibited a much better catalytic activity than others achieving a 100% FDCA yield after only 4 h of reaction, which exceeds other catalysts previously reported. The high catalytic activity of the catalysts might come from the morphological features of the Pt active sites and the synergistic effects between the Pt active sites and the Fe3O4@C microsphere. Furthermore, the synthesized microsphere catalysts can be reused at least three times without significant performance loss. It is believed that these magnetic core–shell microspheres decorated with morphology-controllable Pt nanoparticles catalysts hold great potential for other catalytic processes which are always size-dependent.Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 21473252, 21503016, 51402049, 21173267, 51372041).References
- R. O. M. A. de Souza, L. S. M. Miranda and R. Luque, Green Chem., 2014, 16, 2386–2405 RSC.
- J. A. Melero, J. Iglesias and A. Garcia, Energy Environ. Sci., 2012, 5, 7393–7420 CAS.
- C. H. Zhou, X. Xia, C. X. Lin, D. S. Tong and J. Beltramini, Chem. Soc. Rev., 2011, 40, 5588–5617 RSC.
- D. Carpenter, T. L. Westover, S. Czernik and W. Jablonski, Green Chem., 2014, 16, 384–406 RSC.
- D. L. Dalluge, T. Daugaard, P. Johnston, N. Kuzhiyil, M. M. Wright and R. C. Brown, Green Chem., 2014, 16, 4144–4155 RSC.
- J. Li, F. Qin, L. Zhang, K. Zhang, Q. Li, Y. Lai, Z. Zhang and J. Fang, J. Mater. Chem. A, 2014, 2, 13916–13922 CAS.
- J. Wang, J. Xi and Y. Wang, Green Chem., 2015, 17, 737–751 RSC.
- X. Zhang, H. Lei, L. Zhu, J. Wu and S. Chen, Green Chem., 2015, 17, 4736–4747 RSC.
- L. Wang, Z. Zhang, L. Zhang, S. Xue, W. O. S. Doherty, I. M. O’Hara and X. Ke, RSC Adv., 2015, 5, 85242–85247 RSC.
- C. Liu, H. Wang, A. M. Karim, J. Sun and Y. Wang, Chem. Soc. Rev., 2014, 43, 7594–7623 RSC.
- P. Gallezot, Chem. Soc. Rev., 2012, 41, 1538–1558 RSC.
- D. M. Alonso, J. Q. Bond and J. A. Dumesic, Green Chem., 2010, 12, 1493–1513 RSC.
- N. Yan and X. Chen, Nature, 2015, 524, 155–157 CrossRef CAS PubMed.
- S. Dutta, S. De, B. Saha and M. I. Alam, Catal. Sci. Technol., 2012, 2, 2025–2036 CAS.
- J. Han, S. M. Sen, D. M. Alonso, J. A. Dumesic and C. T. Maravelias, Green Chem., 2014, 16, 653–661 RSC.
- D. M. Alonso, S. G. Wettstein, M. A. Mellmer, E. I. Gurbuz and J. A. Dumesic, Energy Environ. Sci., 2013, 6, 76–80 CAS.
- R. A. Sheldon, Green Chem., 2014, 16, 950–963 RSC.
- Y. Wang, S. De and N. Yan, Chem. Commun., 2016, 52, 6210–6224 RSC.
- D. Shen, R. Xiao, S. Gu and K. Luo, RSC Adv., 2011, 1, 1641–1660 RSC.
- X. Sun, C. Huang, Z. Xue, C. Yan and T. Mu, Energy Fuels, 2015, 29, 2564–2570 CrossRef CAS.
- M. N. Uddin, W. M. A. W. Daud and H. F. Abbas, RSC Adv., 2014, 4, 10467–10490 RSC.
- L. A. Pfaltzgraff, M. De bruyn, E. C. Cooper, V. Budarin and J. H. Clark, Green Chem., 2013, 15, 307–314 RSC.
- H. Kobayashi and A. Fukuoka, Green Chem., 2013, 15, 1740–1763 RSC.
- C. Wen, E. Barrow, J. Hattrick-Simpers and J. Lauterbach, Phys. Chem. Chem. Phys., 2014, 16, 3047–3054 RSC.
- G. Yi, S. P. Teong and Y. Zhang, ChemSusChem, 2015, 8, 1151–1155 CrossRef CAS PubMed.
- T. Dallas Swift, H. Nguyen, A. Anderko, V. Nikolakis and D. G. Vlachos, Green Chem., 2015, 17, 4725–4735 RSC.
- Y. Y. Gorbanev, S. K. Klitgaard, J. M. Woodley, C. H. Christensen and A. Riisager, ChemSusChem, 2009, 2, 672–675 CrossRef CAS PubMed.
- B. Liu, Y. Ren and Z. Zhang, Green Chem., 2015, 17, 1610–1617 RSC.
- X. Sun, Z. Liu, Z. Xue, Y. Zhang and T. Mu, Green Chem., 2015, 17, 2719–2722 RSC.
- Z. Xue, X. Sun, Z. Li and T. Mu, Chem. Commun., 2015, 51, 10811–10814 RSC.
- X. Wan, C. Zhou, J. Chen, W. Deng, Q. Zhang, Y. Yang and Y. Wang, ACS Catal., 2014, 4, 2175–2185 CrossRef CAS.
- J. J. Bozell and G. R. Petersen, Green Chem., 2010, 12, 539–554 RSC.
- T. Pan, J. Deng, Q. Xu, Y. Zuo, Q. X. Guo and Y. Fu, ChemSusChem, 2013, 6, 47–50 CrossRef CAS PubMed.
- J. Pang, M. Zheng, R. Sun, A. Wang, X. Wang and T. Zhang, Green Chem., 2015, 18, 342–359 RSC.
- A. J. J. E. Eerhart, A. P. C. Faaij and M. K. Patel, Energy Environ. Sci., 2012, 5, 6407–6422 CAS.
- W. Partenheimer and V. V. Grushin, Adv. Synth. Catal., 2001, 343, 102–111 CrossRef CAS.
- H. Ait Rass, N. Essayem and M. Besson, Green Chem., 2013, 15, 2240–2251 RSC.
- S. E. Davis, L. R. Houk, E. C. Tamargo, A. K. Datye and R. J. Davis, Catal. Today, 2011, 160, 55–60 CrossRef CAS.
- H. Ait Rass, N. Essayem and M. Besson, ChemSusChem, 2015, 8, 1206–1217 CrossRef CAS PubMed.
- S. E. Davis, B. N. Zope and R. J. Davis, Green Chem., 2012, 14, 143–147 RSC.
- Z. Sun, B. Sun, M. Qiao, J. Wei, Q. Yue, C. Wang, Y. Deng, S. Kaliaguine and D. Zhao, J. Am. Chem. Soc., 2012, 134, 17653–17660 CrossRef CAS PubMed.
- H. Duan, D. Wang and Y. Li, Chem. Soc. Rev., 2015, 44, 5778–5792 RSC.
- N. O. Weiss and X. Duan, Nat. Nanotechnol., 2013, 8, 312–313 CrossRef CAS PubMed.
- X. Sun, D. Li, S. Guo, W. Zhu and S. Sun, Nanoscale, 2016, 8, 2626–2631 RSC.
- Z. Zhou, Y. Li, S. Yao and H. Yan, Ceram. Int., 2016, 42, 2281–2288 CrossRef CAS.
- R. Tan, J. Yang, J. Hu, K. Wang, Y. Zhao and F. Pan, Chem. Commun., 2016, 52, 986–989 RSC.
- P. Chen, Z. Wang, S. Zong, D. Zhu, H. Chen, Y. Zhang, L. Wu and Y. Cui, Biosens. Bioelectron., 2016, 75, 446–451 CrossRef CAS PubMed.
- X. Michalet, F. F. Pinaud, L. A. Bentolila, J. M. Tsay, S. Doose, J. J. Li, G. Sundaresan, A. M. Wu, S. S. Gambhir and S. Weiss, Science, 2005, 307, 538–544 CrossRef CAS PubMed.
- K. Jalaja, D. Naskar, S. C. Kundu and N. R. James, Carbohydr. Polym., 2016, 136, 1098–1107 CrossRef CAS PubMed.
- Z. K. Sun, Q. Yue, Y. Liu, J. Wei, B. Li, S. Kaliaguine, Y. H. Deng, Z. X. Wu and D. Y. Zhao, J. Mater. Chem. A, 2014, 2, 18322–18328 CAS.
- Y. Shao, L. Zhou, C. Bao, J. Ma, M. Liu and F. Wang, Chem. Eng. J., 2016, 283, 1127–1136 CrossRef CAS.
- H. Chen, J. Zhou and J. Deng, Polym. Chem., 2016, 7, 125–134 RSC.
- Z. Niu, Y.-R. Zhen, M. Gong, Q. Peng, P. Nordlander and Y. Li, Chem. Sci., 2011, 2, 2392 RSC.
- K. Byrappa and T. Adschiri, Prog. Cryst. Growth Charact. Mater., 2007, 53, 117–166 CrossRef CAS.
- M. Boutonnet, S. Lögdberg and E. Elm Svensson, Curr. Opin. Colloid Interface Sci., 2008, 13, 270–286 CrossRef CAS.
- J. D. Mackenzie and E. P. Bescher, Acc. Chem. Res., 2007, 40, 810–818 CrossRef CAS PubMed.
- A. Nemamcha, J. L. Rehspringer and D. Khatmi, J. Phys. Chem. B, 2006, 110, 383–387 CrossRef CAS PubMed.
- M. T. Reetz and W. Helbig, J. Am. Chem. Soc., 1994, 116, 7401–7402 CrossRef CAS.
- J. Liu, Z. Sun, Y. Deng, Y. Zou, C. Li, X. Guo, L. Xiong, Y. Gao, F. Li and D. Zhao, Angew. Chem., Int. Ed., 2009, 48, 5875–5879 CrossRef CAS PubMed.
- S. Wang, Z. Zhang and B. Liu, ACS Sustainable Chem. Eng., 2015, 3, 406–412 CrossRef CAS.
- G. Yi, S. P. Teong and Y. Zhang, Green Chem., 2015, 18, 979–983 RSC.
- C. Zhou, W. Deng, X. Wan, Q. Zhang, Y. Yang and Y. Wang, ChemCatChem, 2015, 7, 2853–2863 CrossRef CAS.
- S. Siankevich, G. Savoglidis, Z. Fei, G. Laurenczy, D. T. L. Alexander, N. Yan and P. J. Dyson, J. Catal., 2014, 315, 67–74 CrossRef CAS.
- Y. Gao, X. Chen, J. Zhang, H. Asakura, T. Tanaka, K. Teramura, D. Ma and N. Yan, Adv. Mater., 2015, 27, 4688–4694 CrossRef CAS PubMed.
- S. E. Davis, A. D. Benavidez, R. W. Gosselink, J. H. Bitter, K. P. de Jong, A. K. Datye and R. J. Davis, J. Mol. Catal. A: Chem., 2014, 388–389, 123–132 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2016 |






