Exploring the hydrated microstructure and molecular mobility in blend polyelectrolyte membranes by quantum mechanics and molecular dynamics simulations
Ghasem Bahlakeh*a,
Mohammad Mahdi Hasani-Sadrabadib and
Karl I. Jacobbc
aDepartment of Engineering and Technology, Golestan University, Aliabad Katool, Iran. E-mail: gh.bahlakeh@gu.ac.ir; Fax: +98 1734266235; Tel: +98 1734266235
bParker H. Petit Institute for Bioengineering and Bioscience, G.W. Woodruff School of Mechanical Engineering, Georgia Institute of Technology, Atlanta, 30332-0295 GA, USA
cSchool of Materials Science and Engineering, Georgia Institute of Technology, Atlanta, 30332-0295 GA, USA
First published on 6th April 2016
Abstract
Quantum mechanics and molecular dynamics simulations were employed to examine several structural and dynamical characteristics in blend SPEEK–SPPO based membranes at varied water content and temperature values. QM results showed that water molecules were localized around the SPEEK and SPPO sulfonate groups due to the hydrogen bonding interactions, which caused proton dissociation at the increased hydrations. By increasing the hydration level, more water molecules occupied the sulfonate fragments because of the improved sulfonate–water interactions, whereas the hydrogen bond interaction of sulfonate–hydronium ion was weakened, enabling the hydronium ions to be away from the sulfonate groups. Based on water cluster size distribution and structure factor evaluations, it seemed that by improving the water content, isolated smaller aqueous clusters appeared under lower hydration levels, which merged together to form larger clusters comprising almost all molecules. Diffusivities for water and hydronium ion were observed to be enhanced by an increase in water uptake, which were attributed to the fact that larger hydrophilic clusters across the swollen blend SPEEK–SPPO PEMs promoted molecular mobility. Similarly, enhancing operational temperature gave rise to an enhancement in the membrane transport dynamics. Finally, predicted water and hydronium ion diffusion coefficients were noted to be smaller in hydrated SPEEK–SPPO membrane as compared to Nafion under identical conditions, which was in agreement with the experimental results.
1. Introduction
During recent years, polymer electrolyte membranes (PEMs) have been the subject of intensive scientific investigations thanks to their potential usage in both polymer electrolyte membrane fuel cells (PEMFCs) and direct methanol fuel cells (DMFCs)1,2. PEMs are a crucial component in PEMFCs and DMFCs, and fuel cell performance is greatly dependent on their physicochemical properties. Because of their appropriate characteristics of excellent proton conductivity and good chemical and mechanical stability, perfluorosulfonic acid PEMs like Nafion ionomer are the most widely utilized membranes in these fuel cells.3–6 However, major drawbacks associated with Nafion PEMs including high production cost, significant reduction in proton conductivity at higher fuel cell operating temperatures as well as high methanol crossover (in DMFCs) limited their widespread usages and motivated the development of new hydrocarbon-based PEM materials, such as sulfonated poly(styrene) (SPS), sulfonated poly(2,6-dimethyl-1,4-phenylene oxide) (SPPO), sulfonated poly(ether ether ketone) (SPEEK), sulfonated poly(ether sulfone) (SPES), and poly(benzimidazole) (PBI).7–11 These PEMs have been applied in different forms like pure, nanocomposite and blend.Blended polymeric membranes consist of acid–base and acid–acid types in which an acidic material (e.g., Nafion) is blended with either a basic (e.g., PBI) or another acidic material. Tsai et al. investigated the blend PEMs of Nafion-SPEEK and Nafion-nitrated SPEEK with various SPEEK (or nitrated SPEEK) contents for DMFCs and reported a lower swelling and methanol permeability in the blend membranes compared to the recast Nafion membrane.12,13 By using acid–base SPEEK–PBI blend membrane, Pasupathi et al. measured better DMFC performance in comparison with Nafion 117.14 Jung et al. studied the proton and methanol transport features in the blend membranes based on SPS and SPPO, and observed a higher proton conductivity and methanol permeability in the blend membrane compared with the plain SPS and SPPO membranes.15 In another study, Hasani-Sadrabadi et al. demonstrated that proton conductivity, methanol crossover and water swelling properties of SPPO–PBI based acid–base blend membranes reduced with enhancing the PBI content.16 The same authors also investigated the acid–acid blend PEMs made from SPPO and SPEEK, which was incorporated with organically modified montmorillonite (MMT). Their results showed a lower proton conductivity and methanol permeability in the blend system than in the Nafion membrane.17 In a similar study, Sengül et al. found that acid–acid blend PEMs of SPEEK and SPPO exhibited a better swelling behavior compared to the plain SPEEK materials. But, the proton conductivity of the blend membrane was lower than that of pure SPEEK and Nafion membranes.18
Molecular dynamics (MD) simulations are very useful tools that help us to get detailed insights at molecular scales, which are difficult to obtain from experiments. During recent years, MD simulations have been successfully applied to assess the morphology and permeant transport in perflourinated Nafion membranes19–27 and alternative pure membranes like SPEEK, SPPO, and so on.28–36 Along with MD simulations, computational quantum mechanics (QM) studies were conducted as well with the aim of attaining information based on electronic structure calculations.37–45 Despite the extensive experimental attempts reported for blend PEMs, very few theoretical studies of these membranes have been implemented in all of the relevant conducted studies so far.
Hence, in the present study, QM and all-atom MD simulation tools were applied to probe various characteristics of hydrated SPEEK–SPPO based acid–acid blend PEM. Since both experimental and simulation observations have shown that the amount of water absorbed by acidic PEM materials is a crucial factor that affects fuel cell membrane performance to a large extent, which was also confirmed in our previous MD simulation studies on alternative membranes,35,36 the present work was directed toward assessing microstructure and molecular mobility in blend SPEEK–SPPO membrane at varied hydration levels. In addition, the MD simulations were carried out under different thermal conditions to evaluate effects associated with operational temperature upon membrane dynamics. This study is a subsequent step that was taken after our previous experimental characterization of the blend SPEEK–SPPO membranes.17
2. Computational details
2.1. QM calculations
The QM calculations were executed in order to thoroughly assess the water solvent interactions and phase segregation of the hydrated blend polymers from the electronic structure viewpoint. Indeed, in hydrated blend SPEEK–SPPO membrane, water molecules can simultaneously interact with both SPEEK and SPPO polymers. Therefore, both polymers were chosen for computations. Since QM calculations are computationally time-consuming, only one SPEEK and SPPO monomer was taken into account, as their structures were displayed in Fig. 1. The QM measurements of the blend membrane were performed under the dry and wet conditions, and a number of water molecules ranging from 0–5 were considered for this purpose (SPEEK–SPPO + nH2O, n = 0–5). | ||
| Fig. 1 Molecular structures of (a) SPPO polymer, (b) SPPO QM model, (c) SPEEK polymer, and (d) SPEEK QM model. | ||
The calculations were initially performed for the dry SPEEK–SPPO blend model where the sulfonic acid groups in the SPEEK and SPPO monomers were placed near each other. This initial structure seemed appropriate for the calculations as stronger interactions of PEMs possessing acidic moieties most likely happen by their hydrophilic sulfonic acid side. Furthermore, the SPEEK–SPPO model with the minimum energy structure resulted from QM computations were gradually hydrated and subjected to the QM geometry optimization. Upon stepwise hydration, water molecules were positioned in vicinity of the sulfonic acid groups of SPEEK and SPPO models.
For the electronic structure computations, the prepared initial geometries of the dry and hydrated SPEEK–SPPO blend models were optimized by means of Hartree–Fock (HF) theory using 6-31G(d,p) basis functions,46 which was followed by density functional theory (DFT) methods employing B3LYP hybrid functional with 6-31G(d,p) basis set.47–49 All these computations were conducted using Gaussian 09 package.50
2.2. Amorphous cell construction
In order to examine the impact of the absorbed water on the solvated structure and dynamics of blend PEM, the MD simulations were performed for a range of water content values. During the MD simulations, the membrane water content was defined with λ parameter, which is the number of water molecules to the number of sulfonic acid groups in the chemical structure of SPPO and SPEEK polymers (Fig. 1). Five different water content levels of λ = 3, 6, 9, 12 and 15 were selected for the acid–acid blend SPEEK–SPPO membrane. Therefore, five three-dimensional (3D) cubic amorphous cells, which contained the SPEEK and SPPO polymeric chains, hydronium ions (H3O+), and different amounts of water molecules, needed to be constructed.A SPEEK/SPPO weight percent of 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75 and degrees of sulfonation of 60 and 25% were used for SPEEK and SPPO polymers, respectively. This is because they were the optimum composition and degrees of sulfonation according to the results of our previous experimental study on SPEEK–SPPO membranes. To achieve this composition during the simulation, three SPEEK polymeric chains and seven SPPO chains both with degree of polymerization of 40 (i.e., total number of sulfonated and non-sulfonated PEEK or PPO monomers) were used for simulations.51 It is better to notice that the sulfonated and non-sulfonated monomers were randomly arranged along the SPEEK and SPPO chains. With the above-mentioned sulfonation levels, every SPEEK and SPPO chain included 24 and 10 sulfonated PEEK and PPO monomers, respectively. Moreover, all protogenic sulfonic acid groups in both sulfonated PEEK and PPO monomers were considered ionized during the MD simulations, as it has already been used in MD studies of various acid-type polymeric PEMs. Consequently, to maintain the charge neutrality of the 3D simulation cells, the number of hydronium ions inside the simulation cells was set the same as that of the sulfonic acid groups. Table 1 lists the composition of simulation cells for each level of water content that was considered here. Using the compositions provided in Table 1, starting 3D amorphous simulation cell for the maximum hydration level (λ = 15) was first constructed at a very low initial density of 0.005 g cm−3 by means of amorphous cell builder module in Materials Studio software.51 Then, for the lower water content values, some starting structures were generated by removing a number of water molecules from the previously constructed 3D cell for λ = 15.
75 and degrees of sulfonation of 60 and 25% were used for SPEEK and SPPO polymers, respectively. This is because they were the optimum composition and degrees of sulfonation according to the results of our previous experimental study on SPEEK–SPPO membranes. To achieve this composition during the simulation, three SPEEK polymeric chains and seven SPPO chains both with degree of polymerization of 40 (i.e., total number of sulfonated and non-sulfonated PEEK or PPO monomers) were used for simulations.51 It is better to notice that the sulfonated and non-sulfonated monomers were randomly arranged along the SPEEK and SPPO chains. With the above-mentioned sulfonation levels, every SPEEK and SPPO chain included 24 and 10 sulfonated PEEK and PPO monomers, respectively. Moreover, all protogenic sulfonic acid groups in both sulfonated PEEK and PPO monomers were considered ionized during the MD simulations, as it has already been used in MD studies of various acid-type polymeric PEMs. Consequently, to maintain the charge neutrality of the 3D simulation cells, the number of hydronium ions inside the simulation cells was set the same as that of the sulfonic acid groups. Table 1 lists the composition of simulation cells for each level of water content that was considered here. Using the compositions provided in Table 1, starting 3D amorphous simulation cell for the maximum hydration level (λ = 15) was first constructed at a very low initial density of 0.005 g cm−3 by means of amorphous cell builder module in Materials Studio software.51 Then, for the lower water content values, some starting structures were generated by removing a number of water molecules from the previously constructed 3D cell for λ = 15.
| Hydration level (λ) | |||||
|---|---|---|---|---|---|
| 3 | 6 | 9 | 12 | 15 | |
| No. of SPEEK chains | 3 | 3 | 3 | 3 | 3 |
| No. of SPPO chains | 7 | 7 | 7 | 7 | 7 |
| No. of H2O | 426 | 852 | 1278 | 1704 | 2130 |
| No. of H3O+ | 142 | 142 | 142 | 142 | 142 |
| Total no. of atoms | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 132 132 |
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 410 410 |
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 688 688 |
14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 966 966 |
16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 244 244 |
| Cell size (Å) | 52.03 | 52.28 | 53.33 | 54.55 | 55.92 |
| Density (g cm−3) | 1.06 | 1.13 | 1.15 | 1.15 | 1.14 |
2.3. Force field selection
For the SPEEK and SPPO polymers, the DREIDING force field that was developed by Mayo et al. was employed.52 DREIDING is a generic force field that is easily applicable for various materials, as it has already been used successfully in the MD simulations of Nafion and alternative PEMs.20,24,28,29,34,53 The partial atomic charges for SPEEK and SPPO were determined by charge equilibration (QEq) method from Rappe et al.54 Interaction parameters for water molecules were adopted from flexible 3-centered (F3C) force field of Levitt et al.55 For the case of hydronium ions, partial charges and potentials were chosen from the work carried out by Jang et al.20 The total potential energy of the simulation systems is sum of the bonded (i.e., bond, angle, dihedral and improper) and non-bonded (i.e., van der Waals and coulombic) interactions. Functional forms used to express these interactions are the same as those in our earlier MD studies.56,572.4. MD simulation
MD simulations of constructed 3D cells for acid–acid blend SPEEK–SPPO membranes were performed in the two phases of equilibration and production. To ensure the equilibration of the amorphous cells, which contained complex aromatic SPEEK and SPPO structures, all 3D cells were first minimized by conjugate gradient algorithm to remove any overlapping between atoms inside the cells. Then, the equilibration MD simulations were continued using shrinking box procedure as it has been successfully used in our previous MD simulations of solvated PEMs,36,56,57 and also in MD simulation of hydrated SPEEK based PEMs and other polymeric membranes.29,58,59 Details of this procedure have been described previously. Thereafter, the MD simulations were continued for 10 ns under NPT ensemble to get fully equilibrated amorphous cells. The structures resulted from equilibration phase were used as starting cells for the production phase MD simulations, which were conducted under NVT ensemble for time period of 2 ns. Both MD simulation phases were performed at 353 K. Molecular trajectories were saved every 1 ps during the production simulation for later analyses of membrane properties.In order to examine the thermal effects, the simulations were continued for the cell with λ = 12 hydration at lower temperatures. The final structure resulted from the MD simulations at 353 K was chosen as initial structure, which was subjected to a 1.5 ns NPT simulation followed by a 100 ps NVT run before being used for the production simulations.
The MD simulations were carried out using LAMMPS (large-scale atomic/molecular massively parallel simulator) simulation code60 with the use of periodic boundary conditions imposed on all dimensions of the cells. The non-bonded interactions were evaluated with a 12 Å cutoff radius, and Particle–Particle Particle–Mesh (PPPM) summation method was adopted to treat the long-range coulombic interactions.61 For solving Newton's equation of motion, velocity Verlet integrator with a time step of 0.5 fs was used.62 Temperature and pressure during simulations were monitored by means of Nose–Hoover thermostat and barostat with 0.1 and 1 ps as coupling constants, respectively.63–65
3. Results and discussion
3.1. QM optimized structures
The minimum energy structures of the SPEEK–SPPO blend models under the dry and hydration conditions attained from the accurate electronic structure studies were presented in Fig. 2.QM studies were specifically conducted to derive QM-based insights concerning protogenic group-water interactions responsible for nanophase segregation and hydrogen-bonded network formation across the membrane matrix. It is notable that without addition of any water molecules, SPEEK and SPPO interacted mainly by their sulfonic acid groups via forming double H-bonds. In addition, the benzene ring in SPPO rotated to become parallel relative to non-sulfonated rings in SPEEK due to π–π interactions. When one water molecule was added to energy minimized dry SPEEK–SPPO, models of SPEEK and SPPO preserved only single H-bond compared to dry state, and involved in formation of three H-bonds with water molecules. This means that hydration of blend membrane weakens the intermolecular sulfonic acid–sulfonic acid interactions. It is better noting that a single water molecule added to blend model may form more H-bonds with SPEEK rather than SPPO or interact only with either SPEEK or SPPO if computation with different initial positions of water molecule performed.
By further hydration of SPEEK–SPPO (i.e., n = 2 and 3), as shown in panels (c) and (d) in Fig. 2, sulfonic acid side groups in SPEEK and SPPO formed higher number of H-bonds with water molecules while maintaining single H-bond between each other. In case of SPEEK–SPPO–3H2O, ether oxygen in SPEEK also contributed to the H-bond interaction. At even further hydrations, when four and five water molecules were added [Fig. 2(e) and (f)], proton dissociation phenomenon happened in contrast to lower hydration situations, and water molecules showed intermolecular H-bonds not only with sulfonic acid groups but also with other water molecules, which indicates that the network of H-bonds amongst water molecules and protogenic sulfonic acid attached to SPEEK and SPPO models. Under such conditions, neighboring SPEEK and SPPO sulfonic acid functional groups sustained single H-bond, which existed at lower levels of water content.
The behavior of dissociated proton (or water molecule to which proton attached, that is, hydronium ion H3O+) changes against an increase in water uptake, so that in SPEEK–SPPO–4H2O model the hydronium ion remained in contact with sulfonic acid groups in both SPEEK and SPPO models by sharing its two hydrogen atoms in H-bond interaction. Whereas in optimized SPEEK–SPPO–5H2O case the hydronium ion is in direct contact with only sulfonic acid group of SPPO model, and donated only one hydrogen atom in forming H-bond interaction with protogenic group. Furthermore, the calculated average hydronium ion distance from sulfonic acid moieties, represented as the distance between sulfonic acid sulfur and hydronium ion oxygen atoms (S–Oh), as well as average length of its H-bond with acidic groups increased from 3.42 to 4.07 Å, and from 1.65 to 1.73 Å, respectively, with water content enhancement. These quantitative insights shed light on the fact that increasing hydration of proton-dissociated sulfonic acid groups enforces the separated proton to move away from protogenic fragments.
In summary, from the fundamental observations acquired using the QM calculation of hydrated blend SPEEK–SPPO structures, it can be stated that absorbed water molecules in blend acidic polymer bring about phase separation by occupying protogenic fragments and form continuous network of H-bonds at higher water content values. As a result, they promote dissociation and subsequent transport of proton across the hydrophilic parts of the membrane.
3.2. Amorphous cell equilibrium
Prior to analyzing the results of production MD simulations, it is necessary to check the equilibration of simulated 3D cells. Equilibration is vital for suitable distribution of molecules within the hydrated membrane. For this purpose, the widely used thermodynamic properties, including potential energy, temperature and density were monitored during the simulations. The resulting average size and density of equilibrated cells were collected in Table 1. As listed there, an increase in hydration degree brings about an increase in average size of the simulation cells, which is reflective of membrane swelling behavior against hydration. It can be seen that by increasing hydration level, average density increases and then decreases, which has almost a similar trend to what observed by Lins et al. in MD simulation of phenylated sulfonated poly(ether ether ketone) (Ph-SPEEKK) membranes at lower hydration levels (λ < 16).66 There was no experimental density for the blend SPEEK–SPPO membrane in the literature for comparison. The calculated densities seem acceptable for the solvated SPEEK–SPPO blend membrane, especially considering the densities reported by Ohkubo et al. for hydrated pure SPPO membranes,31 densities obtained in our previous MD study, and also those reported by Brunello et al. for hydrated pure SPEEK membranes.28,35 Consequently, the predicted densities can be used as the first evidence for achieving cell equilibration. Furthermore, both potential energy and temperature of all simulated cells were stabilized (data not shown here) with negligible fluctuations throughout the 2 ns production phase of the MD simulations for all water contents studied here. As a result, they can be served as another indication of reaching the equilibrium condition. Fig. 3, indicates the final equilibrated snapshots corresponding to the hydrated SPEEK–SPPO blend membranes with water the uptakes of λ = 3 and 9, which were obtained at the end of the production MD runs using Visual Molecular Dynamics (VMD) software.67The figure generally shows the hydrophobic–hydrophilic phase segregation in the structure of the blend membrane upon solvation by water molecules. While the hydrophilic phase contains water molecules and hydronium ions as well as anionic sulfonic acid groups, the hydrophobic part includes a backbone of SPEEK and SPPO polymers. At a low hydration level, isolated small water clusters are observed, which become better connected with enhancing hydration levels to form larger aqueous clusters. To further explore these observations and get molecular scale information associated with membrane morphological characteristics, diverse structural and dynamical analyses were executed. For the structural characterization, the radial distribution function, coordination number, hydrogen bond interactions, and water cluster size distribution and structure factor in the hydrated blend membrane were analyzed.
3.3. Radial distribution function
Radial distribution function (RDF), gA–B(r) refers to the probability distribution of finding B atoms at distance r from the given A atoms as reference, and is defined as follows
 | (1) |
In order to evaluate dependency of distribution of water molecules and hydronium ions around sulfonic acid groups, their RDFs against sulfonic acid groups were examined as a function of hydration level. Fig. 4(a) and (b) presents the RDFs of oxygen atoms in sulfonic acid (Os) with respect to water oxygen (Ow) and hydronium ions oxygen (Oh) atoms, respectively.
 | ||
| Fig. 4 RDFs of (a) oxygen (sulfonic acid group)–oxygen (water molecules) and (b) oxygen (sulfonic acid group)–oxygen (hydronium ions) for water contents with λ = 3, 6, 9, 12 and 15. | ||
All Os–Ow RDFs show an intensified peak accompanied by smaller peak occurred at greater Os–Ow distances. Appearance of the first great peak implies the formation of first water solvent shell around sulfonic acid groups due to strong non-bonded sulfonic acid groups–water molecules interactions. The RDFs of Os–Oh also show a first sharp peak with a greater height compared to the Os–Ow RDFs thanks to stronger non-bonded electrostatic interactions of cationic hydronium ions with negatively charged sulfonic acid groups, which holds hydronium ions close to sulfonic acid groups. By an increase in λ value, the peak height in Os–Ow RDFs gradually decreases, a phenomenon which is described by the fact that gradual addition of water molecules weakens their interactions with regards to sulfonic acid groups. Compared to the Os–Ow RDFs, the Os–Oh RDFs against water hydration level shows somewhat a non-monotonic behavior, which is likely because of the presence of two different sulfonated aromatic polymers. The position of first peak is occurred at a distance of about 2.82 Å for Os–Ow RDFs, and in the case of Os–Oh RDFs, this is observed within 2.46–2.70 Å, which are both within the distance required for hydrogen bond formation discussed later.
In order to have a quantitative inspection of water molecules and hydronium ions surrounding sulfonic acid groups, coordination number was determined from the integration of corresponding RDFs. The coordination numbers were computed at distance of 4 Å corresponding to location for the first minimum RDFs. The calculated coordination numbers of water around sulfonic acid oxygen atoms are 1.16, 1.85, 2.42, 2.87, and 2.87 for water contents of 3, 6, 9, 12 and 15, respectively. It is clear that increasing hydration leads to an increase in water coordination number. This phenomenon suggests that sulfonic acid groups become more solvated as the membrane hydration degree is raised. At larger levels of hydration, water coordination shows no change since sulfonic acid groups at such hydration amounts of water molecules less affect water molecules. On the contrary, the hydronium ion coordination number in the vicinity of sulfonic acid group oxygen was calculated to be 0.71, 0.65, 0.49, 0.52 and 0.43 for λ values of 3, 6, 9, 12 and 15, respectively. This trend in hydronium ion coordination number is rationalized with the fact that increasing water content weakens the electrostatic sulfonic acid–hydronium ion interactions, and thereby hydronium ions are allowed to move away from sulfonic acid side chains in SPPO and SPEEK polymers.
The structure of water solvent molecules and hydronium ions across the hydrated SPEEK–SPPO blend PEM was examined using RDFs of water and hydronium ions with regards to water molecules as a function of hydration level. Fig. 5(a) and (b) display the MD simulation results for the Ow–Ow and Oh–Ow RDFs. It is apparent that both RDFs give heightened peaks with descending trend in height against water content. Such RDF behaviors are pointing out the strong correlation and interactions among water molecules and hydronium ions, and their tendency to from aqueous clusters inside the hydrated morphology of acid–acid blend PEM. Significant peaks appeared at 2.82 Å for the Ow–Ow RDFs, and 2.58 Å in the case of Oh–Ow RDFs, again showing the water and hydronium ion capability to take part in H-bond network formation within the aqueous clusters, which is in agreement with the QM consequences. The coordination number for water and hydronium ion around water solvent molecules were determined as a function of atomic pair distances, and related results were depicted in parts (c) and (d) in Fig. 5 for all water uptakes considered here. Indeed, an increase in the level of hydration gives rise to improvement of coordination numbers, which indicates that water molecules and hydronium ions are encompassed by more water molecules, and aqueous phase formed along solvated blend PEM becomes larger as the hydration degree is enhanced.
3.4. Hydrogen bonding interactions
Apart from the qualitative assessment of H-bonding interaction occurrence in terms of RDF peak position, the number of H-bonds among constituents of hydrophilic phase (i.e., polymeric protogenic groups and water as well as hydronium ion) was quantitatively probed using geometric criteria. Pairs of hydrogen donor (D) and hydrogen acceptor (A) atoms were assumed to participate in H-bond formation via a hydrogen (H) atom if the length of D⋯A < 3.5 Å and the angle of D–H⋯A > 120°.The average number of H-bonds between different atomic pairs was given in Fig. 6. As displayed in parts (a) and (b) in Fig. 6, by a change in hydration level from λ = 3 to λ = 15, the average number of H-bonds (per sulfonic acid group) for sulfonic acid oxygen–hydronium ion hydrogen (Os–Hh) declined while that of sulfonic acid oxygen–water hydrogen (Os–Hw) increased. The descending trend of Os–Hh H-bond number against level of hydration when compared to the case of Os–Hw interaction is assigned to competition between water and hydronium ion to interact with sulfonic acid fragment. Upon gradual water addition, sulfonic acid groups become more water-surrounded, as understood from QM and MD results, which weakens the hydronium ion H-bond interaction with the sulfonic acid moiety. Under situations of highly enhanced hydration, the number of both Os–Hh and Os–Hw H-bond interactions scarcely changes because at such levels of hydration sulfonic acid groups stay completely solvated, and therefore they are least influenced by the interactions of newly absorbed water molecules.
The average number of H-bonds (per H3O+) that hydronium ion formed with water molecules by donating its hydrogen atoms (Ow–Hh) was noted to increase as a function of λ [Fig. 6(c)]. This result again reflects a stronger hydronium ion solvation at higher values of λ in close accordance with water coordination number results around H3O+ ions that are shown in Fig. 5(d). At a higher water uptake of the blend membrane, the hydronium ion–water interaction hardly changes, which in turn discloses the fully hydrated structure of the hydronium ion. Furthermore, as Fig. 6(d) clearly exhibits, with an increase in the degree of membrane hydration, the average number of H-bonds (per water) amongst water molecules is steadily improved, which further implies the uninterrupted H-bond network appeared within the acidic blend membrane.
3.5. Cluster size distribution
As with QM observations, structural results from MD simulations (RDF, coordination number and hydrogen bonding interactions) demonstrated the water and hydronium ions strong tendency to participate in an aqueous phase formation across the hydrated SPEEK–SPPO blend PEM. To quantitatively examine aqueous clusters, the cluster size was quantified using cutoff radius (Rc) values of 3.5 and 4.5 Å according to the Ow–Ow and Oh–Ow RDFs. Using these cutoff values, water molecules (and hydronium ions) in the first solvation shells of water and hydronium ion will be involved in cluster formation. Any two molecules are in the same cluster if the Ow–Ow distance is less than the given Rc value. To better describe this condition, clusters containing less than 100 molecules were recognized as a small cluster, and those with more than 400 molecules were classified as a large cluster. The average number of molecules (H2O plus H3O+) constituting a specific cluster is computed as a product of cluster size and its average occurrence number.Fig. 7 presents the profile of simulated small and large water clusters size distribution within the swollen SPEEK–SPPO blend membrane for all water contents taken into account here.
As Fig. 7(a) and (b) prove, clusters of small size almost disappear by enhancing the membrane water uptake irrespective of Rc value. In addition, it is found that at the lowest hydration level (λ = 3), nearly all molecules are found in very small clusters. At the same time, as illustrated in panels (c) and (d) of Fig. 7, in the case of large clusters, which occurred for λ > 3, an enormous number of molecules contribute to large cluster formation, especially in the acidic blend PEM swollen with hydration level of λ = 15. In case of large clusters, it is also noted that when the degree of hydration is improved distribution becomes less broaden with a sharp height, particularly, for hydration with λ = 12 (Rc = 4.5 Å) and 15. This manifests the fact that in increasingly swollen SPEEK–SPPO membrane, water molecules are found in single large aqueous clusters. Consequently, according to the MD simulated cluster size distribution results, it is deduced that as blend SPEEK–SPPO membranes uptake more water molecules, the aqueous clusters grow and the smaller clusters existed at lower hydrations were merged to form larger clusters comprising considerable amounts of absorbed water molecules.
To obtain more information regarding the membrane hydrophilic clusters, structure factor, S(q), was calculated from the Fourier transform of Ow–Ow RDFs as below:
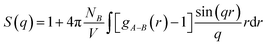 | (2) |
The quantitative insights derived from the cluster size distribution together with structure factor examinations of water clusters are in close agreement with the final hydrated SPEEK–SPPO blend membrane snapshots displayed in Fig. 3, and supports the molecular water and hydronium ion distribution within the hydrophilic phase of the hydrated acid based blend PEM.
3.6. Transport dynamics
To assess the transport dynamics associated with the hydrated SPEEK–SPPO blend membrane as a function of hydration degree, water and hydronium ion diffusivities were estimated from the slop of their mean squared displacement (MSD) (or Einstein relation) expressed according to the following equation:
 | (3) |
Fig. 8(a) and (b) illustrate the hydronium ion and water solvent mobility by evaluating the corresponding MSDs as a function of level of hydration during the last 2 ns simulation time. First, it is seen that the MSD curves showed a linear trend, especially in case of water molecules, indicating that the molecular diffusion through solvated blend PEM reached a continuous state. In addition, it is obvious that water and hydronium ion experienced MSDs with a sharper slope at increased water uptake amounts of membrane. This finding indicates that an improvement in hydration gives rise to rapid molecular movement. The dependency of water and hydronium ion dynamics on thermal influences was also analyzed and the results were given in panels (c) and (d) in Fig. 8. The water and hydronium ion mobility was noted to enhance at elevated membrane operational temperatures.
The diffusion coefficients for water and hydronium ion, as predicted by making use of MD simulations, were provided in Fig. 8(e) for all hydration degrees that were studied. The calculated diffusion coefficients were found to enhance by changing the water content from λ = 3 to λ = 15. The improved molecular diffusivities inside increasingly hydrated blend membrane could be rationalized by potential solvent interactions with respect to polymeric materials. According to the finding obtained from the QM and MD simulations, as the degree of hydration is increased, water molecules occupied protogenic sulfonic acid groups on account of intermolecular H-bonds, and resulted in hydrophobic–hydrophilic phase separation with larger hydrophilic clusters through which water as well as proton transfer take place. At the same time, as evidenced by both the QM and MD simulation measurements, the interactions of water with the SPEEK and SPPO sulfonic acid fragments facilitated proton transfer from the sulfonic acid into an aqueous phase by pushing proton away from anionic sulfonic acid as a result of weakened hydronium ion–sulfonic acid interactions.
The results for water and hydronium ion diffusion coefficient features as a function of temperature were depicted in Fig. 8(f). It is apparent that an increase in operational temperature leads to improved diffusion coefficients for both water and hydronium ion, which is explained by the fact that increasing the temperature can enhance the dynamics of water and hydronium ion. The dynamical characteristics of SPEEK–SPPO blend membrane containing varied amounts of water uptake were compared with the earlier simulation results for Nafion PEM as a reference. Devanathan et al.68 reported the diffusion coefficient data of 5.30, 6.14, 10.93, and 13.73 × 10−6 cm2 s−1 for water, and 3.70, 3.10, 11.90, and 22.90 × 10−7 cm2 s−1 for hydronium ion at 353 K, respectively, for λ values of 3.5, 6, 11, and 16. It is observed that calculated diffusion coefficients of water as well as hydronium ion are lower in swollen SPEEK–SPPO blend membranes than in hydrated Nafion PEM, which is in consistent with the experiments. The lower diffusion coefficient values found in the hydrated SPEEK–SPPO as compared to Nafion manifest less phase separation in the blend membrane, as molecules diffuse more rapidly through water-swollen PEMs of higher phase segregation level.69
4. Conclusion
In the present work, quantum mechanics and classical all-atom MD simulations techniques were utilized to comprehensively study various structural and dynamical features of acidic blend SPEEK–SPPO membranes under different water contents conditions. Additionally, MD simulations were conducted at different temperatures to probe temperature-dependency of the dynamical characteristics. Results derived from the QM computations indicated that water molecules localized near protogenic sulfonic acid groups of both SPEEK and SPPO models due to strong H-bond interactions, which caused the proton dissociation at higher water uptakes. According to the MD simulation results for structural analyses (RDF, coordination number, and intermolecular H-bond interactions), it was found that with increasing hydration degree, sulfonic acid moieties of SPEEK and SPPO chains were surrounded by more water molecules due to increased sulfonic acid–water H-bond interactions, which in turn decreased the hydronium ion interaction with the sulfonic acid fragments, and thereby allowed the hydronium ions to be away from the sulfonic acid groups. Furthermore, the analyses of structural characteristics for water and hydronium ion clarified their capability to form aqueous clusters across the membrane. From the quantitative evaluation of water clusters, it was understood that by enhancing the water content value, the degree of phase separation improved, and larger clusters containing almost all molecules formed. Diffusivities for the water and hydronium ion were observed to improve with increasing the membrane water uptake. Similarly, enhancing the operational temperature gave rise to an increase in molecular mobility. Finally, it was noted that the predicted water and hydronium ion diffusion coefficients in hydrated SPEEK–SPPO membrane were smaller than in Nafion under identical conditions, in agreement with the experimental results.Acknowledgements
This research was financially supported by the Renewable Energy Organization (SUNA). G. B. gratefully acknowledges the use of School of Computer Science, Institute for Research in Fundamental Science (IPM).References
- L. Carrette, K. A. Friedrich and U. Stimming, Fuel Cells, 2001, 1, 5–39 CrossRef CAS.
- S. K. Kamarudin, F. Achmad and W. R. W. Daud, Int. J. Hydrogen Energy, 2009, 34, 6902–6916 CrossRef CAS.
- K. A. Mauritz and R. B. Moore, Chem. Rev., 2004, 104, 4535–4586 CrossRef CAS PubMed.
- G. Alberti and M. Casciola, Solid State Ion, 2001, 145, 3–16 CrossRef CAS.
- M. M. Hasani-Sadrabadi, E. Dashtimoghadam, F. S. Majedi, S. Wu, A. Bertsch, H. Moaddel and P. Renaud, RSC Adv., 2013, 3, 7337–7346 RSC.
- M. M. Hasani-Sadrabadi, S. R. Ghaffarian and P. Renaud, RSC Adv., 2013, 3, 19357–19365 RSC.
- M. Rikukawa and K. Sanui, Prog. Polym. Sci., 2000, 25, 1463–1502 CrossRef CAS.
- M. Gil, X. Ji, X. Li, H. Na, J. E. Hampsey and Y. Lu, J. Membr. Sci., 2004, 234, 75–81 CrossRef CAS.
- T. Xu, D. Wu and L. Wu, Prog. Polym. Sci., 2008, 33, 894–915 CrossRef CAS.
- H. Ahmad, S. K. Kamarudin, U. A. Hasran and W. R. W. Daud, Int. J. Hydrogen Energy, 2010, 35, 2160–2175 CrossRef CAS.
- V. Saarinen, K. D. Kreuer, M. Schuster, R. Merkle and J. Maier, Solid State Ion, 2007, 178, 533–537 CrossRef CAS.
- J. C. Tsai, H. P. Cheng, J. F. Kuo, Y. H. Huang and C. Y. Chen, J. Power Sources, 2009, 189, 958–965 CrossRef CAS.
- J. C. Tsai, J. F. Kuo and C. Y. Chen, J. Power Sources, 2009, 194, 226–233 CrossRef CAS.
- S. Pasupathi, S. Ji, B. Jan Bladergroen and V. Linkov, Int. J. Hydrogen Energy, 2008, 33, 3132–3136 CrossRef CAS.
- B. Jung, B. Kim and J. M. Yang, J. Membr. Sci., 2004, 245, 61–69 CrossRef CAS.
- A. H. Haghighi, M. M. Hasani-Sadrabadi, E. Dashtimoghadam, G. Bahlakeh, S. E. Shakeri, F. S. Majedi, S. Hojjati Emami and H. Moaddel, Int. J. Hydrogen Energy, 2011, 36, 3688–3696 CrossRef CAS.
- M. M. Hasani-Sadrabadi, S. R. Ghaffarian, N. Mokarram-Dorri, E. Dashtimoghadam and F. S. Majedi, Solid State Ion, 2009, 180, 1497–1504 CrossRef CAS.
- E. Sengül, H. Erdener, R. G. Akay, H. Yücel, N. Baç and I. I. Eroglu, Int. J. Hydrogen Energy, 2009, 34, 4645–4652 CrossRef.
- A. Vishnyakov and A. V. Neimark, J. Phys. Chem. B, 2001, 105, 9586–9594 CrossRef CAS.
- S. S. Jang, V. Molinero, T. Cagin and W. A. Goddard, J. Phys. Chem. B, 2004, 108, 3149–3157 CrossRef CAS.
- S. Urata, J. Irisawa, A. Takada, W. Shinoda, S. Tsuzuki and M. Mikami, J. Phys. Chem. B, 2005, 109, 4269–4278 CrossRef CAS PubMed.
- S. Cui, J. Liu, M. E. Selvan, D. J. Keffer, B. J. Edwards and W. V. Steele, J. Phys. Chem. B, 2007, 111, 2208–2218 CrossRef CAS PubMed.
- R. Devanathan, A. Venkatnathan and M. Dupuis, J. Phys. Chem. B, 2007, 111, 8069–8079 CrossRef CAS PubMed.
- C. H. Cheng, P. Y. Chen and C. W. Hong, J. Electrochem. Soc., 2008, 155, B435–B442 CrossRef CAS.
- J. Karo, A. Aabloo, J. O. Thomas and D. Brandell, J. Phys. Chem. B, 2010, 114, 6056–6064 CrossRef CAS PubMed.
- H. Abroshan, H. Akbarzadeh, F. Taherkhani and G. Parsafar, Mol. Phys., 2011, 109, 709–724 CrossRef CAS.
- C. K. Knox and G. A. Voth, J. Phys. Chem. B, 2010, 114, 3205–3218 CrossRef CAS PubMed.
- G. Brunello, S. G. Lee, S. S. Jang and Y. Qi, J. Renewable Sustainable Energy, 2009, 1, 033101–033114 CrossRef.
- C. V. Mahajan and V. Ganesan, J. Phys. Chem. B, 2010, 114, 8357–8366 CrossRef CAS PubMed.
- R. D. Lins, R. Devanathan and M. Dupuis, J. Phys. Chem. B, 2011, 115, 1817–1824 CrossRef CAS PubMed.
- T. Ohkubo, K. Kidena, N. Takimoto and A. Ohira, J. Mol. Model., 2011, 17, 739–755 CrossRef CAS PubMed.
- S. Zhu, L. Yan, D. Zhang and Q. Feng, Polymer, 2011, 52, 881–892 CrossRef CAS.
- J. Pozuelo, E. Riande, E. Saiz and V. Compañ, Macromolecules, 2006, 39, 8862–8866 CrossRef CAS.
- P. Y. Chen, C. P. Chiu and C. W. Hong, J. Power Sources, 2009, 194, 746–752 CrossRef CAS.
- G. Bahlakeh, M. Nikazar, M.-J. Hafezi, E. Dashtimoghadam and M. M. Hasani-Sadrabadi, Int. J. Hydrogen Energy, 2012, 37, 10256–10264 CrossRef CAS.
- G. Bahlakeh and M. Nikazar, Int. J. Hydrogen Energy, 2012, 37, 12714–12724 CrossRef CAS.
- V.-A. Glezakou, M. Dupuis and C. J. Mundy, Phys. Chem. Chem. Phys., 2007, 9, 5752–5760 RSC.
- M. Koyama, K. Bada, K. Sasaki, H. Tsuboi, A. Endou, M. Kubo, C. A. Del Carpio, E. Broclawik and A. Miyamoto, J. Phys. Chem. B, 2006, 110, 17872–17877 CrossRef CAS PubMed.
- S. J. Paddison and J. A. Elliott, J. Phys. Chem. A, 2005, 109, 7583–7593 CrossRef CAS PubMed.
- C. Wang and S. J. Paddison, J. Phys. Chem. A, 2013, 117, 650–660 CrossRef CAS PubMed.
- Y.-K. Choe, E. Tsuchida, T. Ikeshoji, A. Ohira and K. Kidena, J. Phys. Chem. B, 2010, 114, 2411–2421 CrossRef CAS PubMed.
- Y.-Y. Zhao, E. Tsuchida, Y.-K. Choe, T. Ikeshoji, M. A. Barique and A. Ohira, Phys. Chem. Chem. Phys., 2014, 16, 1041–1049 RSC.
- N. Idupulapati, R. Devanathan and M. Dupuis, J. Phys. Chem. A, 2010, 114, 6904–6912 CrossRef CAS PubMed.
- S. Li and J. R. Fried, Macromo. Theory Simul.s, 2013, 22, 410–425 CrossRef CAS.
- S. J. Paddison, K.-D. Kreuer and J. Maier, Phys. Chem. Chem. Phys., 2006, 8, 4530–4542 RSC.
- P. C. Hariharan and J. A. Pople, Theor. Chim. Acta, 1973, 28, 213–222 CrossRef CAS.
- A. D. Becke, Chem. Phys., 1992, 96, 2155 CAS.
- C. Lee, W. Yang and R. G. Parr, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 37, 785–789 CrossRef CAS.
- A. D. McLean and G. S. Chandler, J. Chem. Phys., 1980, 72, 5639–5648 CrossRef CAS.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, N. J. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, GAUSSIAN 09, Gaussian Inc., Wallingford, CT, 2009 Search PubMed.
- Material Studio 4.3, Accelrys Inc., San Diego, CA.
- S. L. Mayo, B. D. Olafson and W. A. Goddard, J. Phys. Chem., 1990, 94, 8897–8909 CrossRef CAS.
- G. F. Brunello, W. R. Mateker, S. G. Lee, J. I. Choi and S. S. Jang, J. Renewable Sustainable Energy, 2011, 3, 043111–043116 CrossRef.
- A. K. Rappe and W. A. Goddard, J. Phys. Chem., 1991, 95, 3358–3363 CrossRef CAS.
- M. Levitt, M. Hirshberg, R. Sharon, K. E. Laidig and V. Daggett, J. Phys. Chem. B, 1997, 101, 5051–5061 CrossRef CAS.
- G. Bahlakeh, M. Nikazar and M. M. Hasani-Sadrabadi, J. Membr. Sci., 2013, 429, 384–395 CrossRef CAS.
- G. Bahlakeh, M. Nikazar, M.-J. Hafezi and M. M. Hasani-Sadrabadi, J. Power Sources, 2013, 243, 935–945 CrossRef CAS.
- N. F. A. V. Vegt, W. J. Briels, M. Wessling and H. Strathmann, J. Chem. Phys., 1996, 105, 8849–8857 CrossRef.
- R. M. Sok, H. J. C. Berendsen and W. F. Van Gunsteren, J. Chem. Phys., 1992, 96, 4699–4704 CrossRef CAS.
- S. J. Plimpton, J. Comput. Phys., 1995, 117, 1–19 CrossRef CAS.
- R. W. Hockney and J. W. Eastwood, Computer Simulation Using Particles, McGraw-Hill, New York, 1981 Search PubMed.
- W. C. Swope, H. C. Andersen, P. H. Berens and K. R. Wilson, J. Chem. Phys., 1982, 76, 637–649 CrossRef CAS.
- S. Nose, J. Chem. Phys., 1984, 81, 511–519 CrossRef CAS.
- S. Nose, Mol. Phys., 1984, 52, 255–268 CrossRef CAS.
- S. Nose, Mol. Phys., 1986, 57, 187–191 CrossRef CAS.
- R. D. Lins, R. Devanathan and M. Dupuis, J. Phys. Chem. B, 2011, 115, 1817–1824 CrossRef CAS PubMed.
- W. Humphrey, A. Dalke and K. Schulten, J. Mol. Graphics, 1996, 14, 33–38 CrossRef CAS PubMed.
- A. Venkatnathan, R. Devanathan and M. Dupuis, J. Phys. Chem. B, 2007, 111, 7234–7244 CrossRef CAS PubMed.
- K. D. Kreuer, J. Membr. Sci., 2001, 185, 29–39 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2016 |






