Design, synthesis and mesomorphic behaviour of a four-ring achiral bent-core liquid crystal in the nematic phase†
Amina Nafeesa,
Aloka Sinha*a,
Nandiraju V. S. Raob,
Gayatri Kalitab,
Golam Mohiuddinb and
Manoj Kumar Paulb
aDepartment of Physics, Indian Institute of Technology Delhi, Hauz Khas, New Delhi-110016, India. E-mail: aloka@physics.iitd.ac.in; Tel: +91-11-26596003
bChemistry Department, Assam University, Silchar-788011, Assam, India. E-mail: drnvsrao@gmail.com; Tel: +91-94-93222541
First published on 25th April 2016
Abstract
Bent-core nematics have attracted growing interest because of the unconventional properties and extraordinary effects exhibited by these liquid crystalline phases. In this report we design, synthesize and characterize four different four-ring achiral bent-core liquid crystals containing a methyl group in the central phenyl ring. The mesomorphic behaviour of compounds has been investigated by optical polarizing microscopy, differential scanning calorimetry, electro-optical investigation, and dielectric spectroscopy. Calculations based on density functional theory of the molecules were also carried out. The addition of a methyl group in the central phenyl ring not only induces the lowering of the clearing point but also enhances the nematic phase range. Dielectric spectroscopy reveals a low frequency relaxation peak and a very large dielectric permittivity; which is the characteristic of cybotactic clusters. In electro-optic studies, spontaneous polarization peaks are not observed. However, a periodic pattern is observed under the application of an ac electric field. Finally, the compounds show a weak tendency to crystallize; which makes it possible to supercool the cybotactic nematic phase down to room temperature.
1. Introduction
After the discovery of bent-core liquid crystals (LCs) that exhibit nematic phases, bent-core nematics (BCNs) have been the subject of intense experimental and theoretical investigation due to their unique properties, extraordinary effects and potential application in the area of fast switching displays.1–5 The nematic phases of bent-core compounds possess unusual physical properties, like a giant flexoelectric response,2 low frequency dielectric relaxation,6 unusual electro-convection scenarios,7 magnetic field induced birefringence;8 which may be related to the presence of polar smectic clusters in the nematic phase. The majority of the bent-core molecules are based on a five- six- or seven-ring rigid core in which the central ring represents the bend unit, usually a 1,3 substituted benzene with an angle between the two attached arms of 120°. Recently new bent-core compounds, such as a five-membered heterocycle, either a 2,5-disubstituted 1,3,4-oxadiazole or a 3,5-disubstituted 1,2,4-oxadiazole possessing a larger bent angle of 134–140°, are also reported.6 The nematic phases are rare in bent core systems9,10 because of the constraints imposed by the shape of the molecules contributing to steric hindrance for free rotation, core–core interactions promoting the layer formation due to the segregation between extended aromatic moieties and end alkyl chains rather than interdigitation of molecules along the orientational direction of the long axes. The incompatibility between the bent aromatic cores and aliphatic moieties is another limitation for the formation of nematic phases. Additionally, BCNs have been regarded as promising candidates for indefinable biaxial nematic11,12 and polar ferroelectric nematic13,14 phases. The fluid ferroelectric nematic phase is expected to exhibit a much faster and easier response to an external electric field in comparison to the conventional ferroelectric smectic LCs and explore the possibility for the new applications in the area of electro-optic device technology. On the other hand, biaxiality was first reported in a 1, 3, 4-oxadiazole-based bent-core mesogen,11,12 but its very nature (spontaneous vs. induced; local vs. macroscopic) is still controversial.15Although, there is an ongoing scientific debate on these topics, there is now a general consensus that the unique properties of BCNs (Scheme 1a) result from the presence of nanometre size clusters of molecules (cybotactic groups) characterized by short range smectic (Sm) positional order and biaxial orientational order.9,13,16–19 These clusters persist all over the nematic temperature range and they are now widely recognized as an inherent feature of the nematic phase of bent-core mesogens, and accordingly, the phase is generally called the cybotactic nematic (Ncyb, Scheme 1b) phase.9 The four-spot small angle X-ray diffraction (SAXRD) pattern typical of BCNs provides the clear evidence of tilted smectic-C-like order within clusters.9,10,13,15–17 The same pattern was initially interpreted as a signature of long-range biaxiality. This was recently confirmed by direct observation of cybotactic groups in cryo-transmission electron microscope (cryo-TEM) images.18 Another important property of the bent-core LC which has become a major focal point for the liquid crystal research is the twist-bend nematic phase.19 Recently, the existence of a new condensed phase of matter with unique properties designated as twist-bend nematic (NTB or twist-bend, Scheme 1c) phase, a fundamentally new type of nematic ground state of achiral molecules below the nematic phase, is confirmed in n-meric (n = dimer, n = 3, trimer, n = 4, tetramer) compounds20–23 containing flexible methylene spacers20–23 as well as rigid bent-core19,24,25 molecular materials. This was theoretically conjectured earlier.26 These compounds exhibit layer-free, helical liquid crystal ordering of nano-scale pitch made up of three or four molecules (10–15 nm) and have been structurally identified and characterized. The NTB phase was also observed in the ether and mixed ether/ester-linked bimesogens and it is concluded that the gross topologies dictate the incidence of this fascinating phase and not molecular properties such as dipole moment and bend angle.27,28
BCNs are relatively uncommon and it is generally more difficult and challenging to design and synthesise four-ring bent-core compounds exhibiting only nematic phase. At present, little is known about the structural parameters that control the induction of nematic phase for these compounds. Detailed studies and characterizations of different type of bent-core LCs are needed to further understand the induction of the nematic phase and the physical properties such as biaxiality and ferroelectric switching. Currently, there are dozens of bent-core LC molecules synthesized and explored as mesomorphic material, but a majority of them are five-ring bent-shaped molecules based on oxadiazole as central core6 or 4-substituted phenyl ring10 with the presence of the lower temperature smectic phase in addition to nematic phase. Recently few three-ring bent-core compounds29 exhibiting monotropic nematic phases (∼40 ± 10 °C) with a low temperature smectic phase are reported. However, for practical applications, enantiotropic nematic phase with a wider temperature range, which must be around room temperature and without the presence of the low temperature smectic or columnar phases, would be required. A few bent-core compounds exhibiting only nematic phases have been also reported.6,9,14,30
Hence any attempt to synthesize bent-core compounds consisting of few rings say three or four with a combination of different linking groups and a combination of substituents to exhibit enantiotropic nematic phase at ambient temperatures is still a challenging goal for chemists that can lead to detailed understanding of structure–property relationship as well as its functional characteristics. It cannot be taken for granted a priori that all the new bent-core molecular structures would still be able to form nematic phase, and it is also conceivable that there would be a complete loss of mesophase behaviour in some cases. In this paper, the design, synthesis, dielectric properties, electro-optical and energy optimised molecular structure with density functional theory calculation of unsymmetrical achiral four-ring bent-core LCs with a broad nematic phase range are presented. The salient features of these bent-core liquid crystalline compounds are (i) wide temperature nematic phases which can also be cooled down to room temperature without the presence of the lower temperature smectic phase and (ii) the observation of unique 2D periodic pattern with the application of the electric field which can be obtained and controlled by the applied electric field.
2. Design of the molecule
The recent experimental support in favour of a twist-bend nematic phase (NTB), biaxial nematic phase (Nb) that is exhibited by rigid as well as dimer bent-core mesogens composed of two rod-like mesogenic wings coupled to a central linking moiety has been debated very well. The central linking moiety possesses a transverse dipole with an obtuse bent angle between the two arms. This represents a banana or V-shaped molecule composed of two uniaxial arms with a central transverse dipole. The study of the influence of dipole–dipole correlations on the stability of the biaxial nematic phase, in the two-particle-cluster approximation,31 revealed that (a) the polar-molecular-shape correlations between neighboring molecules substantially favour the stabilization of biaxial nematic phases, and (b) the electrostatic interactions between permanent transverse dipoles of bent-core molecules also significantly stabilize the biaxial nematic phases. The introduction of a 2-methyl group in the 1,3-disubstituted phenyl ring of a bent-core molecular architecture can generate an obtuse bond angle of ∼145°, which gives rise to an increase in bend angle as well as a dipole in the bending direction. The reduced bend from 120 to ∼145° of the 1,3-phenyl moiety by the introduction of a methyl group in the 2-position and with the decrease in the number of rings from five or more to four in the molecular unit, places these compounds at the borderline between classical rod-like LCs and bent-core mesogens. The four-ring molecules can also be recognized as true hockey-stick model molecules. Further the use of linking groups viz., azobenzene or phenyl benzoate or salicylideneimine units in one of the arms of the bent molecule promote mesomorphism. The introduction of methyl moiety in the central phenyl ring with an additional methyl moiety in adjacent ring not only changes the electrostatic interaction between adjacent molecules but also enhances the intermolecular distance between molecules. In addition, the influence of the bent shape to negate the nematic phase formation can be decreased by these two methyl moieties in the lateral direction in adjacent rings. The realization of such a molecular architecture leads to a reduction in rotational disorder as well as a dipole in the bent direction. If molecular interactions are strong enough then the molecular structure can promote polar biaxial nematic phases. In this study, we aimed to combine the lateral dipole in the form of a methoxy moiety at the terminal position in one of the arms of the bent-core molecule, while the central bent core possesses methyl substituents projected at a location inside the molecular core to represent the transverse dipole. The other end of the molecule is linked to 4-n-alkoxysalicylaldehyde through an imine moiety, which actually seems superior to the benzylidene aniline core and is more stable towards hydrolysis due to intermolecular hydrogen bonding.3. Experimental
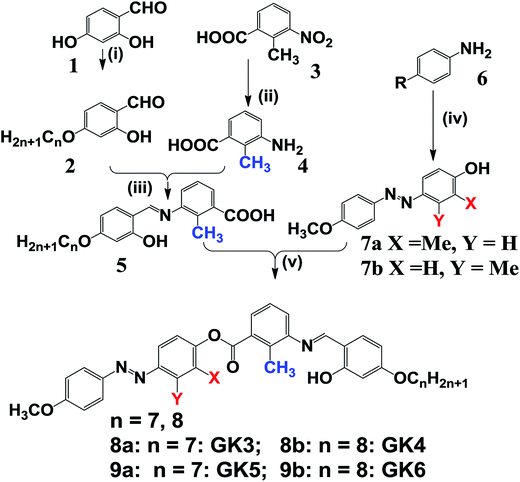
3.1 Synthesis
The starting material in the present study 4-n-alkyloxy-2-hydroxybenzaldehyde was prepared by Williamson etherification of 2,4-dihydroxybenzaldehyde with appropriate n-alkyl bromide. 2-Methyl-3-nitrobenzoic acid was converted into corresponding amine by reduction using 5% Pd–C, which was condensed with 4-n-alkoxy salicylaldehyde in presence of few drops of glacial acetic acid to get 2-methyl-3-N-(4-n-alkyloxysalicylidene) amino benzoic acid in good yields. 2-Methyl-3-N-(4-n-alkyloxysalicylidene) amino benzoic acid was further esterified with 3-methyl-4-hydroxy-4′-methoxyazobenzene/2-methyl-4-hydroxy-4′-methoxy-azobenzene using DCC-DMAP reaction to yield the designed products GK3, GK4, GK5 and GK6 (Scheme 2). The compounds were further recrystallized repeatedly to get the pure samples. All the compounds were characterized by elemental analysis, FTIR, UV-Vis and 1H NMR studies. The synthetic procedures for all the homologous series of compounds are followed as detailed for unsubstituted compounds in an earlier publication.32The assigned νO–H–N stretching band confirmed the presence of intermolecular H-bonding in all the compounds. The introduction of ortho hydroxyl group in benzylidene moiety not only enhances the stability of the imines through intramolecular H-bonding to overcome the hydrolytic instability of the molecules towards moisture but also enhances the transverse dipole-moment. The infrared spectra of all the compounds exhibited characteristic bands in region 1460 ± 15 cm−1 (νN![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, azo), 1602–1620 cm−1 (νCH
N, azo), 1602–1620 cm−1 (νCH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, imine), 1740 ± 15 cm−1 (νC
N, imine), 1740 ± 15 cm−1 (νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, ester), 3200 ± 50 cm−1 (νO–H, H-bonding). The 13C NMR (Fig. S2†) data of a representative example GK5 is furnished below. 13C NMR: δC (100 MHz, CDCl3) 165.9, 163.9, 163.7, 162.7, 161.9, 152.2, 149.6, 148.5, 147.4, 139.2, 134.2, 133.6, 130.5, 128.4, 126.6, 124.8, 124.1, 123.9, 122.6, 119.7, 116.7, 114.2, 113.8, 113.0, 107.8, 101.5, 68.3, 55.6, 31.7, 30.9, 29.0, 25.9, 22.6, 17.6, 15.4, 14.1.
O, ester), 3200 ± 50 cm−1 (νO–H, H-bonding). The 13C NMR (Fig. S2†) data of a representative example GK5 is furnished below. 13C NMR: δC (100 MHz, CDCl3) 165.9, 163.9, 163.7, 162.7, 161.9, 152.2, 149.6, 148.5, 147.4, 139.2, 134.2, 133.6, 130.5, 128.4, 126.6, 124.8, 124.1, 123.9, 122.6, 119.7, 116.7, 114.2, 113.8, 113.0, 107.8, 101.5, 68.3, 55.6, 31.7, 30.9, 29.0, 25.9, 22.6, 17.6, 15.4, 14.1.
3.2 Investigation
Indigenous cells with a thickness of 3.2 μm were used for the investigation of the electro-optical and dielectric behaviour. The LC cells were made with indium tin oxide (ITO) – coated glass substrates. The inner surfaces of the cells were spin coated with Nylon 6/6 and subsequently rubbed in an anti-parallel direction for planar alignment. The cells were assembled with the glass plates having their coated sides facing one over another and separated by high precision spacers. The cells were filled with LC via capillary action in the isotropic phase. The temperature of the cell was controlled by an Instec (HCS302) hot stage attached to the STC200 temperature controller. The spontaneous polarization was measured using polarization reversal triangular wave method. The electric field induced patterns were observed simultaneously under the polarizing microscope. A Tektronix AFG 3021 function generator, TPS 2024 oscilloscope, and homemade amplifier were used for the electro-optical measurement. Dielectric measurements were carried out using an Agilent (E4980A) precision LCR meter with a probing voltage of 100 mV in a wide frequency range of 20 Hz to 2 MHz in the nematic phase. The polarizing optical microscope (Olympus BX51P) was used to record the texture. The textures were captured at 50× magnification in transmission mode under crossed polarizer. The melting and clearing transition temperature of the samples were obtained using differential scanning calorimetry (DSC) and further confirmed by polarizing optical microscopy (POM) studies. The bent-core materials under investigation are denoted by GK3, GK4, GK5, and GK6. The molecular structures of the compounds are given in Scheme 2 and phase transition temperatures in °C (ΔH, ΔS) are summarised in Table 1, where the enthalpies ΔH (kJ mol−1) and the entropies ΔS are given in parentheses respectively. Representative DSC thermogram for GK4 is displayed in Fig. 1. The inset shows the nematic–isotropic phase transitions.| Compound | Phase transition temperatures °C (ΔH, ΔS) | ΔN (°C) |
|---|---|---|
| GK3 | Cr 114.6 (58.4, 18.1) N 144.4 (0.619, 0.18) Iso | 29.8 |
| Cr 65.6 (44.7, 15.8) N 143.8 (0.655, 0.19) Iso | 78.2 | |
| GK4 | Cr 107.7 (49.5, 15.6) N 139.8 (0.454, 0.13) Iso | 32.1 |
| No crystallization N 139.2 (0.517, 0.15) Iso | >120 | |
| GK5 | Cr 110.6 (41.7, 13.0) N 135.6 (0.495, 0.14) Iso | 25 |
| No crystallization N 134.9 (0.512, 0.15) Iso | >120 | |
| GK6 | Cr 104.3 (48.2, 15.4) N 132.2 (0.438, 0.13) Iso | 27.9 |
| No crystallization N 131.4 (0.548, 0.16) Iso | >120 |
 | ||
| Fig. 1 DSC trace of GK4 obtained during second heating and cooling cycles scanned at a rate 5 K min−1. | ||
4. Results and discussion
4.1 Density functional theory (DFT) calculations
DFT computational studies based on quantum mechanical calculations were performed to obtain the information related to molecular conformation, bend angle, dipole moment, molecular polarizability and electrostatic potential distribution of the selected molecules GK3–GK6. Full geometry optimizations have been carried out without imposing any constraints using the Gaussian 09 program package.33 Spin restricted DFT calculations were carried out in the framework of the generalized gradient approximation (GGA) using Becke3 – Lee–Yang–Parr hybrid functional (B3LYP) exchange–correlation functional and the 6-311G(d,p) basis set.34,35 B3LYP functional with the standard basis set 6-311G have been used due to its successful application for larger organic molecules, as well as hydrogen bonded systems in the past,36–38 and bent-core molecules39–43 recently. The molecules with planar conformation possessing methyl substituent either downward (hindered position) or upwards (open position) were submitted for optimization. The difference in optimized energy between the pairs of GK3–GK3up and GK4–GK4up is ∼0.1 kJ mol−1. GK3up and GK4up have the lower energy in comparison to GK3 and GK4. Therefore, the most preferred geometry will be GK3up and GK4up. Similarly for GK5–GK5up and GK6–GK6up the difference in energy is ∼9.6 kJ mol−1. Hence, the GK5 and GK6 with lower energy will be the preferred optimized geometry. The position of the methyl group in GK3 and GK3up contribute to a small change in dihedral angle between the two neighbouring rings. The methyl group in GK3 is more hindered whereas in GK3up the hindered environment is relaxed with a noticeable increase in the dihedral angle thus, facilitating it to be the most preferred optimized geometry [similarly for the GK4 and GK4up molecules]. In case of GK5 and GK5up, the methyl moiety in GK5up is hindered by the interaction between the lone pair of N atom of azo group. However, the interaction of methyl moiety in GK5 with the azo linkage is comparatively smaller. Thus, GK5 will be the preferred optimized geometry [same for GK6 and GK6up]. The difference in bend angles 142° for GK3up and GK4up and 148° for GK5 and GK6 is noticeable (Table 1 in ESI†). The variation in dipole moment is not significant but the major contribution of the dipole moment component comes from the Y and Z components for GK5 and GK6, whereas in the case of GK3up/GK4up it comes from X and Z components. Polarizabilities are crucial for an understanding of the molecular properties in molecular optics and spectroscopy. Electrostatic intermolecular interaction energy is related to this quantity, in particular for systems without a permanent dipole moment.44DFT calculated principal polarizability components (αXX, αYY and αZZ), isotropic polarizability component αiso, polarizability anisotropy Δα = [αXX − (αYY + αZZ)/2] and asymmetry parameter, η = [(αYY − αZZ)/(αXX − αiso)], parameters relative to the molecular polarizability tensor, α, in the Cartesian reference frame are displayed in Table 2 in ESI.† The molecular polarizability component αXX is comparatively larger along the molecular longitudinal X-axis (Table 2 in ESI†) for all the molecules than the other two directions. The asymmetry parameter η = [(αYY − αZZ)/(αXX − αiso)] (0.04–0.05) is rather very small for GK3up/GK4up and is dependent on the bending angle, which is found to be 142°. However, η value is 0.070–0.078 for meta methyl substituted compounds (GK5 and GK6) with a bend angle of 148°. The principle components of the polarizability tensor (αXX, αYY and αZZ) and αiso = (αXX + αYY + αZZ)/3 is rather small. Although the molecular formula and number of electrons of the two sets of studied molecules are the same, the main component of static polarizability along the molecular long X-axis differs significantly due to the different position of the methyl moieties in the phenyl ring adjacent to the central ring which has manifested the bend in the molecule.
The dihedral angle between the central phenyl ring and phenyl ring with benzylidene linkage is found to be 136–138° in both sets of compounds (Fig. 2). The dihedral angle between the central phenyl ring and phenyl ring of azo moiety is 104° in GK3up/GK4up as well as 135° in GK5/GK6. Hence, these three phenyl rings are out of plane and can be visualized to promote the twist in the molecule. Further a significant deviation in the dihedral angles between the central phenyl ring and azo phenyl ring between two sets of molecules ∼135° for GK5/GK6, ∼104° for GK3up/GK4up reflects the influence of methyl moiety substitution in meta-position and ortho position to ester linkage.
4.2 Liquid crystalline phase
4.3 Dielectric spectroscopy
The dielectric permittivity and the electrical conductivity are the key electric properties determining the behaviour of a compound in an electric field. The dielectric spectroscopy yields information on the frequency dependence of the complex dielectric permittivity, ε*(f) = ε′(f) − iε′′(f) where the real part ε′(f) corresponds to the dielectric permittivity, and ε′′(f) is its imaginary parts (the dielectric loss). The dielectric studies on all the four compounds were carried out in the planar aligned cell of thickness 3.2 μm in the frequency range 20 Hz to 2 MHz. The real part of the dielectric permittivity was calculated as ε′ = C/Co, where C is the capacitance of the filled cell and Co is the capacitance of the empty cell. The frequency dependence of loss factor (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) and dielectric permittivity at different temperature for sample GK4 and GK6 is shown in Fig. 5 and 6, respectively.
δ) and dielectric permittivity at different temperature for sample GK4 and GK6 is shown in Fig. 5 and 6, respectively.
 | ||
| Fig. 6 Dielectric permittivity versus frequency at different temperature of GK4 and GK6 respectively. | ||
The loss factor (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) curve shows one relaxation peak between 100 and 1500 Hz in the measured frequency range. The loss peak appears in the low frequency region and shifted towards the higher frequency side as the temperature is increased. The reduction in relaxation frequency with decreasing temperature suggests an increase in cluster size as well as decrease in viscosity with decreasing temperature.50 The low frequency process is attributed to the collective motion of the molecules characteristic of nematic phase possessing cybotactic molecular arrangement due to the following reasons. First, the temperature dependence of the dielectric permittivity and relaxation frequency of the process shows very large value of dielectric permittivity (Fig. 6). The large dielectric permittivity is a characteristic of a cybotactic cluster as seen previously for other bent-core liquid materials.6,14 The second reason is that the dielectric loss peak becomes completely suppressed under the applied bias voltage (Fig. 7) reflecting that it is due to the collective process of the molecules. The same phenomenon is observed in all the other samples.
δ) curve shows one relaxation peak between 100 and 1500 Hz in the measured frequency range. The loss peak appears in the low frequency region and shifted towards the higher frequency side as the temperature is increased. The reduction in relaxation frequency with decreasing temperature suggests an increase in cluster size as well as decrease in viscosity with decreasing temperature.50 The low frequency process is attributed to the collective motion of the molecules characteristic of nematic phase possessing cybotactic molecular arrangement due to the following reasons. First, the temperature dependence of the dielectric permittivity and relaxation frequency of the process shows very large value of dielectric permittivity (Fig. 6). The large dielectric permittivity is a characteristic of a cybotactic cluster as seen previously for other bent-core liquid materials.6,14 The second reason is that the dielectric loss peak becomes completely suppressed under the applied bias voltage (Fig. 7) reflecting that it is due to the collective process of the molecules. The same phenomenon is observed in all the other samples.
4.4 Electro-optical studies
In order to know the electro-optical property of the compounds an ac electric field is applied across the sample. All the four compounds exhibited parallel stripe pattern along the rubbing direction (Fig. 8b, f and h) initially. However, these stripes transformed to chocolate-grid like pattern (Fig. 8c and g) followed by the appearance of a turbulent state when the amplitude of the applied voltage is increased (Fig. 8d). When the frequency is increased, the formation of parallel stripe pattern appeared as shown in the Fig. 8e (GK3) and Fig. 8i (GK6). The parallel stripe pattern at low frequency was previously observed by the Wiant et al. in a bent-core LC.7 The possible origin of parallel stripe pattern was thought to be a flexo-electric effect. The flexo-electric effect has been reported to produce two kinds of patterns composed of parallel stripes in calamitic liquid crystal-one at low frequencies and another at high frequencies.51 A similar stripe pattern is also observed in achiral rigid bent-core mesogen,19 in achiral bimesogen, which has a molecular structure composed of differing mesogenic core units linked together by a methylene dioxy chain28 and in a mixture of monomer and methylene units linked mesogenic dimer.52 The periodic stripe pattern parallel to rubbing direction is observed in the presence as well as in the absence of the electric field. Another pattern observed is the chocolate-grid like pattern which is observed when the frequency and the voltage of the applied field are increased (Fig. 8a–i). This type of wavy pattern is observed in the strongly twisted planar aligned cholesteric LC compounds under the influence of ac electric field.53 Both parallel stripe pattern and chocolate-grid like pattern are observed in the chiral compounds depending on the pitch of the cholesteric liquid crystal. However, for the first time we have observed a 2D periodic pattern in bent-core LC. The induction of periodic pattern may be due to the replacement of H-atom by the bulky methyl group in the second benzene ring which enhances the intermolecular separation. This result is also corroborated by DFT calculation. The DFT calculations shows a significant deviation from the planar shape to a twisted molecular shape (in particular GK3 and GK4) originating from the dihedral angles imposed by the methyl substituent replacing H-atom. This may have resulted in a periodic structure and is characterized by the presence of periodic pattern in the presence of electric field. The periodic pattern may have resulted because of the induced twisted in molecular shape. This is also confirmed by our previous study of similar compounds, in which the only difference is the absence of methyl group in the second benzene ring.32 No change in texture in the presence of the ac electric field was observed in our previous studied compounds without a methyl substituent, even though the dihedral angles and polarizabilities do not deviate significantly from GK5 and GK6 (the molecular structures, (S1) and related data (Tables 1 and 2) are provided in ESI†). However, to know the origin and properties of the periodic pattern, detailed study of the material using other characterization techniques are under investigation.The measurement of the spontaneous polarization using the repolarization current technique was also undertaken.54 A triangular wave voltage of frequency from 1 Hz to 50 Hz and voltages up to 80 Vpp is applied but no peak corresponding to spontaneous polarization is observed in all the four samples. When the electric current is measured through the samples under the triangular electric fields, the current peaks are found to be absent generally in bent-core materials exhibiting nematic phase polarization. This is due to the symmetry-allowed twist and splay instability which often destroy the polar structure and cancel the macroscopic polarization.9 However, if an applied electric field is strong enough to cooperatively orient the dipoles and increase the correlation length of polar order in the cybotactic domains, ferroelectric-like switching may occur. The current response of GK4 is shown in Fig. 9 as a representative example.
5. Conclusions
Bent-core compounds containing methyl group in the central phenyl ring have been successfully designed and synthesized. The addition of methyl group enhances the nematic phase range and also lowers the clearing temperature. All the studied compounds exhibit a wide range of the nematic phase which can be cooled down to room temperature. The dielectric spectrum shows the presence of only a lower frequency mode in the entire nematic range during cooling conditions. The low frequency mode indicates the formation of temporary polarized clusters even in fluid phases. The large permittivity at low frequency is unusual for these bent-core LCs and could be seen as another proof of the presence of cybotactic clusters. No current peak corresponding to spontaneous polarization is observed. This may be due to large threshold field required for these materials. Electro-optical studies revealed the existence of 1D and 2D periodic pattern under the influence of applied ac electric field. The two types of periodic pattern that are observed is the periodic stripe pattern and another is the chocolate grid like pattern. For the first time, we report here the unusual chocolate pattern, a 2D periodic pattern in the presence of the electric field in the bent-core LC. Such a periodic pattern has potential application in variable grating mode device, in structured nano-composite material and in nano-patterning required for advanced micro-chip fabrication.Acknowledgements
Department of Science and Technology (SR/S2/CMP-007/2010) and University Grant Commission, New Delhi, India is gratefully acknowledged by A. Sinha and A. Nafees, respectively. The authors acknowledge Dr P. Tandon, Lucknow University for useful discussions related to DFT studies. Prof. Narayanan Kurur, IIT-Delhi are also acknowledged for 13C NMR experiment.References
- A. Jakli, Liq. Cryst. Rev., 2013, 1, 65 CrossRef CAS.
- J. Harden, B. Mbanga, N. Eber, K. Fodor-Csorba, S. Sprunt, J. T. Gleeson and A. Jakli, Phys. Rev. Lett., 2006, 97, 157802 CrossRef CAS PubMed.
- C. Bailey, K. Fodor-Csorba, R. Verduzco, J. T. Gleeson, S. Sprunt and A. Jákli, Phys. Rev. Lett., 2009, 103, 237803 CrossRef CAS PubMed.
- O. Francescangeli, F. Vita, F. Fauth and E. T. Samulski, Phys. Rev. Lett., 2011, 107, 207801 CrossRef PubMed.
- F. Vita, I. F. Placentino, C. Ferrero, G. Singh, E. T. Samulski and O. Francescangeli, Soft Matter, 2013, 9, 6475 RSC.
- G. Shanker, M. Nagaraj, A. Kocot, J. K. Vij, M. Prehm and C. Tschierske, Adv. Funct. Mater., 2012, 22, 1671 CrossRef CAS.
- D. Wiant, J. T. Gleeson, N. Eber, K. Fodor-Csorba, A. Jakli and T. Toth-Katona, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys., 2005, 72, 041712 CrossRef CAS PubMed.
- D. Wiant, S. Stojadinovic, K. Neupane, S. Sharma, K. Fodor-Csorba, A. Jakli, J. T. Gleeson and S. Sprunt, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys., 2006, 73, 030703 CrossRef CAS PubMed.
- C. Keith, A. Lehmann, U. Baumeister, M. Prehm and C. Tschierske, Soft Matter, 2010, 6, 1704 RSC.
- M. Alaasar, M. Prehm, K. May, A. Eremin and C. Tschierske, Adv. Funct. Mater., 2014, 24, 1703 CrossRef CAS.
- B. R. Acharya, A. Primak and S. Kumar, Phys. Rev. Lett., 2004, 92, 145506 CrossRef PubMed.
- L. A. Madsen, T. J. Dingemans, M. Nakata and E. T. Samulski, Phys. Rev. Lett., 2004, 92, 145505 CrossRef CAS PubMed.
- O. Francescangeli, V. Stanic, S. I. Torgova, A. Strigazzi, N. Scaramuzza, C. Ferrero, I. P. Dolbnya, T. M. Weiss, R. Berardi, L. Muccioli, S. Orlandi and C. Zannoni, Adv. Funct. Mater., 2009, 19, 2592 CrossRef CAS.
- S. Ghosh, N. Begum, S. Turlapati, S. K. Roy, A. K. Das and N. V. S. Rao, J. Mater. Chem. C, 2014, 2, 425 RSC.
- C. Tschierske and D. J. Photinos, J. Mater. Chem., 2010, 20, 4263 RSC.
- O. Francescangeli and E. T. Samulski, Soft Matter, 2010, 6, 2413 RSC.
- S. Chakraborty, J. T. Gleeson, A. Jákli and S. Sprunt, Soft Matter, 2013, 9, 1817 RSC.
- C. Zhang, M. Gao, N. Diorio, W. Weissflog, U. Baumeister, S. Sprunt, J. T. Gleeson and A. Jákli, Phys. Rev. Lett., 2012, 109, 107802 CrossRef CAS PubMed.
- D. Chen, M. Nakata, R. Shao, M. R. Tuchband, M. Shuai, U. Baumeister, W. Weissflog, D. M. Walba, M. A. Glaser, J. E. Maclennan and N. A. Clark, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys., 2014, 89, 022506 CrossRef PubMed.
- (a) V. Borshch, Y.-K. Kim, J. Xiang, M. Gao, A. Jakli, V. P. Panov, J. K. Vij, C. T. Imrie, M. G. Tamba, G. H. Mehl and O. D. Lavrentovich, Nat. Commun., 2013, 4, 2635 CAS; (b) M. G. Tamba, S. M. Salili, C. Zhang, A. Jakli, G. H. Mehl, R. Stannarius and A. Eremin, RSC Adv., 2015, 5, 11207 RSC.
- D. Chen, J. H. Porada, J. B. Hooper, A. Klittnick, Y. Shen, M. R. Tuchband, E. Korblova, D. Bedrov, D. M. Walba, M. A. Glaser, J. E. Maclennan and N. A. Clark, Proc. Natl. Acad. Sci. U. S. A., 2013, 110, 15931 CrossRef CAS PubMed.
- (a) R. J. Mandle and J. W. Goodby, ChemPhysChem, 2016, 17, 967 CrossRef CAS PubMed; (b) R. J. Mandle, E. J. Davis, C. T. Archbold, C. C. A. Voll, J. L. Andrews, S. J. Cowling and J. W. Goodby, Chem.–Eur. J., 2015, 21, 8158–8167 CrossRef CAS PubMed.
- Y. Wang, G. Singh, D. M. Agra-Kooijman, M. Gao, H. K. Bisoyi, C. Xue, M. R. Fisch, S. Kumar and Q. Li, CrystEngComm, 2015, 17, 2778 RSC.
- M. W. Schroder, S. Diele, G. Pelzl, U. Dunemann, H. Kresse and W. Weissflog, J. Mater. Chem., 2003, 13, 1877 RSC.
- V. Gortz, C. Southern, N. W. Roberts, H. F. Gleeson and J. W. Goodby, Soft Matter, 2009, 5, 463 RSC.
- R. B. Meyer, Phys. Rev. Lett., 1969, 22, 918 CrossRef CAS.
- R. J. Mandle, C. C. A. Vol, D. J. Lewis and J. W. Goodby, Liq. Cryst., 2016, 43, 13 CrossRef CAS.
- R. J. Mandle, E. J. Davis, S. A. Lobato, C. C. A. Vol, S. J. Cowling and J. W. Goodby, Phys. Chem. Chem. Phys., 2014, 16, 6907 RSC.
- G. Mohiuddin, V. Punjani and S. K. Pal, ChemPhysChem, 2015, 16, 2015 CrossRef PubMed.
- H. F. Gleeson, S. Kaur, V. Gçrtz, A. Belaissaoui, S. Cowling and J. W. Goodby, ChemPhysChem, 2014, 15, 1251 CrossRef CAS PubMed.
- (a) M. A. Osipov and G. Pajak, J. Phys.: Condens. Matter, 2012, 24, 142201 CrossRef PubMed; (b) M. A. Bates, Chem. Phys. Lett., 2007, 437, 189 CrossRef CAS.
- A. Nafees, G. Kalita, M. K. Paul, A. Sinha and N. V. S. Rao, RSC Adv., 2015, 5, 7001 RSC.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochters, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox, Gaussian 09, Revision B.01, Gaussian, Inc., Wallingford CT, 2010 Search PubMed.
- K. Kim and K. D. Jordan, J. Phys. Chem., 1994, 98, 10089 CrossRef CAS.
- P. J. Stephens, F. J. Devlin, C. F. Chabalowski and M. J. Frisch, J. Phys. Chem., 1994, 98, 11623 CrossRef CAS.
- A. R. Leach, Molecular Modelling – Principles and Applications, Pearson education limited, England, 2nd edn, 2001 Search PubMed.
- N. H. March, Electron Density Theory of Atoms and Molecules, Academic, London, 1992 Search PubMed.
- E. S. Kryachko and E. V. Ludena, Energy Density Functional Theory of Many-Electron System, Kluwer, Dordrecht, 1990 Search PubMed.
- J. Seltmann, A. Marini, B. Mennucci, S. Dey, S. Kumar and M. Lehmann, Chem. Mater., 2011, 23, 2630 CrossRef CAS.
- A. R. K. Selvaraj, W. Weissflog and R. Friedemann, J. Mol. Model., 2007, 13, 907 CrossRef CAS PubMed.
- A. R. K. Selvaraj, W. Weissflog, G. Pelzl, S. Diele, H. Kresse, Z. Vakhovskaya and R. J. Friedemann, Phys. Chem. Chem. Phys., 2006, 8, 1170 RSC.
- W. Weissflog, G. Naumann, B. Kosata, M. W. Schroeder, A. Eremin, S. Diele, H. Kresse, R. Friedemann, A. R. K. Selvaraj and G. Pelzl, J. Mater. Chem., 2005, 15, 4328 RSC.
- W. Koch and M. C. Holthausen, A Chemist's Guide to Density Functional Theory, Wiley-VCH Verlag GmbH, 2nd edn, 2001 Search PubMed.
- R. K. Nath, R. Deb, N. Chakraborty, G. Mohiuddin, D. S. S. Rao and N. V. S. Rao, J. Mater. Chem. C, 2013, 1, 663 RSC.
- R. Deb, R. K. Nath, M. K. Paul, N. V. S. Rao, F. Tuluri, Y. Shen, R. Shao, D. Chen, C. Zhu, I. I. Smalyukh and N. A. Clark, J. Mater. Chem., 2010, 20, 7332 RSC.
- R. K. Nath, D. D. Sarkar, D. S. S. Rao and N. V. S. Rao, Liq. Cryst., 2012, 39, 889 CrossRef CAS.
- D. D. Sarkar, R. Deb, N. Chakraborty and N. V. S. Rao, Liq. Cryst., 2012, 39, 1003 CrossRef CAS.
- M. K. Paul, G. Kalita, A. R. Laskar, S. Debnath, V. Gude, D. D. Sarkar, G. Mohiuddin, S. K. Varshney and N. V. S. Rao, J. Mol. Struct., 2013, 78, 1049 Search PubMed.
- L. Chakraborty, N. Chakraborty, D. D. Sarkar, N. V. S. Rao, S. Aya, K. V. Le, F. Araoka, K. Ishikawa, D. Pociecha, E. Gorecka and H. Takezoe, J. Mater. Chem. C, 2013, 1, 1562 RSC.
- G. Shanker, M. Prehm, M. Nagaraj, J. K. Vij, M. Weyland, A. Eremin and C. Tschierske, ChemPhysChem, 2014, 15, 1323 CrossRef CAS PubMed.
- N. V. Madhusudana and V. A. Raghunathan, Liq. Cryst., 1989, 5, 1789 CrossRef CAS.
- V. P. Panov, R. Balachandran, J. K. Vij, M. G. Tamba, A. Kohlmeier and G. H. Mehl, Appl. Phys. Lett., 2012, 101, 234106 CrossRef.
- B. I. Senyuk, I. I. Smalyukh and O. D. Lavrentovich, Opt. Lett., 2005, 30, 349 CrossRef CAS PubMed.
- M. F. Achard, J. P. Bedel, J. P. Marcerou, H. T. Nguyen and J. C. Rouillon, Eur. Phys. J. E, 2003, 10, 129 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra05125a |
| This journal is © The Royal Society of Chemistry 2016 |