Experimental and theoretical studies of molecular complexes of theophylline with some phenylboronic acids†
Mayura TalwelkarShimpi*a,
Sven Öberga,
Lopamudra Giri*b and
V. R. Pedireddib
aMaterial Science, Luleå University of Technology, 971 87 Sweden. E-mail: mayura.talwelkarshimpi@ltu.se
bSolid State & Supramolecular Structural Chemistry Laboratory, School of Basic Sciences, Indian Institute of Technology Bhubaneswar, Bhubaneswar 751 007, India
First published on 25th April 2016
Abstract
Molecular complexes of the active pharmaceutical ingredient (API) theophylline, 1 with 4-halophenylboronic acids [4-chlorophenylboronic acid (a), 4-bromophenylboronic acid (b), 4-iodophenylboronic acid (c)], 4-hydroxyphenylboronic acid (d) and 1,4-phenylene-bis-boronic acid (e) have been reported. The complexes were characterized and analysed using the intensity data obtained by X-ray diffraction techniques. All the halo substituted boronic acid complexes are found to be isostructural (1.a, 1.b and 1.c) irrespective of the variations in size and electronegativity of halogen atoms while complexes with non-halogenated boronic acids, 1.d and 1.e, show distinctly different features between themselves as well as with that of 1.a–c, both in two and three-dimensional arrangements. Complexes 1.a–c are noted to be crystallized in the form of sheet structures, which are stacked in three dimensional arrangements, while channels and square grid networks are observed in 1.d and 1.e, respectively. Further the homomeric and heteromeric interactions which occur in the complexes have been analysed by a DFT-D3 method.
Introduction
In recent years boronic acids have received a lot of attention due to their wide range of applications – for example, as intermediates in various organic synthesis,1–5 as sensors due to their ability to form reversible diol formation with glucose,6–10 as reactants/substrates in the formation of boroxine networks,11–14 etc. Also, boronic acids are well employed in medicinal chemistry as antibiotics,15,16 for instance, in the treatment of tumors17 as well as inhibitors.18–20 In recent times, boronic acids also have been used as building blocks in non-covalent synthesis for the synthesis of exotic supramolecular assemblies, as has appeared in the literature in the domains of both experimental as well as theoretical studies.21–24A noteworthy examples being molecular complexes of 1,4-phenylene-bis-boronic acid, 4-bromophenylboronic acid, 4-hydroxyphenylboronic acid, etc. which are resulted through the formation of preferentially by hetero-dimers formed by boronic acid with various aza-donor compounds.25,26 Also, a recent report by some of us27 indeed demonstrates the competitive nature of boronic acid functionality even in the presence of a strong hydrogen bonding donor group like hydroxyl (–OH) in the formation of exotic supramolecular assemblies with various aza donor compounds.
Another notable example that highlights the significance of boronic acids in supramolecular synthesis is the crystallization experiments performed on 4-carboxyphenylboronic acid, in which the boronic acid forms heterodimers with carboxylic acid group.28–30 In fact, such experimental observations are corroborated with theoretical studies demonstrating that boronic acid heterodimers are energetically more favourable than their homodimers.31–35
Realization of such a distinct self-assembled assemblies are attributed to the ability of –B(OH)2 to yield syn–anti, syn–syn and anti–anti, and also ability to mimic strong hydrogen bond affinity functional groups like carboxylic acids (–COOH) and amides (–CONH2).26,28
Though boronic acids gained much interest as a co-crystal former in the development of myriad of supramolecular assemblies, theoretical studies performed on boronic acids mediated assemblies are very much limited.
In the parallel developments in the arena of supramolecular chemistry, in recent times, supramolecular synthesis is directed towards preparation of co-crystals with bio-active entities like Active Pharmaceutical Ingredients (APIs) as one of the components, to implant varied bioactivity for the APIs different from the native form. However, in such pharmaceutical co-crystals, boronic acids although possess effective bio properties have not been well explored except a few examples in recent years.35
Thus, in this article, we present the experimental as well as theoretical studies of supramolecular assemblies of a pharmaceutically active bio-compound, theophylline, with some boronic acid as illustrated below in Chart 1. The theophylline is chosen as it is an effective drug used in the treatment of acute asthma,36 apart from its use in tumour therapy37,38 and as diuretic, but its effectiveness is hindered by its low stability, mainly due to the interconversion between its anhydrous and hydrate forms as a function of relative humidity (RH).
Thus, co-crystals of theophylline, 1 with some boronic acids – 4-chlorophenylboronic acid (a), 4-bromophenylboronic acid (b), 4-iodophenylboronic acid (c), 4-hydroxyphenylboronic acid (d) and 1,4-phenylene-bis-boronic acid (e) – have been prepared by solvent evaporation method of crystallization and characterized through X-ray diffraction techniques, keeping in view the propensity of these boronic acids in the formation of molecular complexes as exemplified by us in the literature.26c,27 The energy of the heteromeric and homomeric interactions have been computed and compared using DFT calculations.
Experimental
All the chemicals have been purchased from Sigma-Aldrich with >99% purity and have been used without further purification. The solvents employed for the crystallization studies were of spectroscopy grade of highest available quality. All the molecular complexes 1.a–e were obtained by dissolving theophylline, 1 and the respective boronic acid, as the case may be, in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio, in either water or ethanol solvents depending upon the solubility of solutes and kept it aside by covering it with aluminium foil to attain supersaturation environment. In all the cases, good quality crystals suitable for X-ray diffraction studies were obtained within 48 hours.
1 ratio, in either water or ethanol solvents depending upon the solubility of solutes and kept it aside by covering it with aluminium foil to attain supersaturation environment. In all the cases, good quality crystals suitable for X-ray diffraction studies were obtained within 48 hours.
Molecular complex, 1.b, however, did not yield single crystals and the obtained microcrystalline solid was used for characterization by powder X-ray diffraction.
In a typical experiment, 90 mg of 1 (0.5 mmol) and 78 mg of a (0.5 mmol) were dissolved in about 15 ml ethanol by gently boiling the solution and kept aside. Good quality crystals are obtained within 24 hours which are utilized for single crystal diffraction studies.
Single crystal X-ray diffraction studies
The crystals of suitable dimension and good quality were chosen under a Leica microscope, equipped with a polarizer and a CCD camera. A crystal was glued to a glass fibre using an acrylate adhesive and placed on a goniometer of the Bruker single crystal X-ray diffractometer equipped with CCD area detector and a graphite-monochromated Mo-Kα radiation (λ = 0.71073 Å). The X-ray intensity data were collected at room temperature and the process was smooth in all the cases, and no additional precautions are necessitated, as the crystals were quite stable. The intensity data were processed using Bruker's suite of programs (SAINT),39 and absorption corrections were applied using SADABS.40 The structure solution of all the compounds have been carried out by direct methods and refinements were performed by full matrix least squares on F2 using the SHELXTL-PLUS41 suite of programs. All the non-hydrogen atoms were refined anisotropically, while the hydrogen atoms are placed in either calculated positions or obtained from Fourier maps, are refined isotropically. Complete structure solution and refinement parameters are listed in Table 1. All the intermolecular interactions as listed in Table 2 were computed using PLATON.42| Parameters | 1.a | 1.c | 1.d | 1.e |
|---|---|---|---|---|
| Formula | C7H8N4O2:C6H6BClO2 | C7H8N4O2:C6H6BIO2 | C7H8N4O2:C6H7BO3 | 2(C7H8N4O2):C6H8B2O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O H2O |
| Mr | 336.54 | 427.99 | 318.10 | 542.09 |
| Crystal shape | Plate | Plate | Block | Plate |
| Crystal colour | Colourless | Colourless | Colourless | Yellow |
| Crystal system | Triclinic | Triclinic | Monoclinic | Triclinic |
| Space group | P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
P21/c | P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
| T, K | 293(2) | 293(2) | 293(2) | 293(2) |
| λ(Mo-Kα)/Å | 0.71073 | 0.71073 | 0.71073 | 0.71073 |
| a/Å | 7.510(1) | 7.475(1) | 12.858(3) | 8.528(2) |
| b/Å | 9.471(2) | 9.623(2) | 8.215(2) | 8.711(2) |
| c/Å | 11.878(2) | 11.957(3) | 14.199(3) | 9.661(3) |
| α/° | 85.63(3) | 87.25(1) | 90 | 109.74(1) |
| β/° | 79.47(4) | 80.94(1) | 98.03(4) | 94.47(1) |
| γ/° | 68.96(3) | 71.63(1) | 90 | 104.78(1) |
| V/Å3 | 775.1(3) | 806.1(3) | 1485.1(5) | 642.5(3) |
| Z | 2 | 2 | 4 | 1 |
| Dc/g cm−3 | 1.442 | 1.763 | 1.423 | 1.401 |
| μ, mm−1 | 0.271 | 2.011 | 0.110 | 0.108 |
| 2θ range [°] | 50.06 | 50.08 | 50.52 | 50.06 |
| Limiting indices | −8 ≤ h ≤ 8 | −8 ≤ h ≤ 8 | −15 ≤ h ≤ 15 | −10 ≤ h ≤ 10 |
| −11 ≤ k ≤ 11 | −11 ≤ k ≤ 11 | −9 ≤ k ≤ 9 | −10 ≤ k ≤ 10 | |
| −14 ≤ l ≤ 14 | −14 ≤ l ≤ 14 | −17 ≤ l ≤ 17 | −11 ≤ l ≤ 11 | |
| F(000) | 348 | 420 | 664 | 282 |
| Total reflections | 7555 | 3923 | 7220 | 6198 |
| Unique reflections | 2723 | 2626 | 2698 | 2268 |
| Reflection at I > 2σ(I) | 2716 | 2379 | 2021 | 1815 |
| No. of parameters | 248 | 236 | 244 | 213 |
| R1, I > 2σ(I) | 0.075 | 0.059 | 0.046 | 0.080 |
| wR2, I > 2σ(I) | 0.184 | 0.152 | 0.139 | 0.183 |
| GoF on F2 | 1.161 | 1.116 | 1.048 | 1.220 |
| CCDC no. | 1440006 | 1440007 | 1440008 | 1440009 |
| D–H⋯A | 1.a | 1.c | 1.d | 1.e | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a Three columns for each structure represent H⋯A, D⋯A distances and ∠D–H⋯A angles, respectively, for a typical hydrogen bond, being represented as D–H⋯A. | ||||||||||||
| O–H⋯O | 2.11 | 2.81 | 150 | 2.11 | 2.79 | 167 | 1.85 | 2.74 | 153 | 2.06 | 2.86 | 162 |
| 1.91 | 2.71 | 172 | ||||||||||
| N–H⋯O | 1.94 | 2.78 | 170 | 2.02 | 2.80 | 156 | 1.90 | 2.79 | 174 | 1.86 | 2.74 | 166 |
| O–H⋯N | 1.92 | 2.85 | 167 | 2.15 | 2.90 | 173 | 2.02 | 2.80 | 152 | 2.06 | 2.81 | 169 |
| C–H⋯O | 2.47 | 3.32 | 156 | 2.35 | 3.28 | 159 | 2.41 | 3.19 | 139 | 2.63 | 3.27 | 125 |
| 2.53 | 3.33 | 149 | 2.54 | 3.33 | 151 | 2.57 | 3.52 | 170 | ||||
| C–H⋯X | 2.84 | 3.67 | 144 | 3.11 | 3.92 | 144 | ||||||
Powder diffraction studies
X-ray powder diffraction (XPRD) data were collected on a PANalytical diffractometer with Cu-Kα radiation (λ = 1.54060 Å). X-ray generator set up at 40 kV and 30 mA was used to collect intensity data with a step size of 0.017° (2θ) in a continuous scanning mode. Diffraction patterns were collected in the 2θ range of 5–50° at room temperature.DFT methodology
Theoretical calculations using DFT methods, implemented in NWChem,43 have been performed on all the complexes to compute the strength of the hydrogen bonding. The association energy of hydrogen bonded dimers have been calculated by subtracting energy of isolated molecules from the total energy of hydrogen bonded molecular motif, by using DFT-D3 method by Grimme44 with B3LYP45 functional. The initial coordinates are taken from the single crystal diffraction data. Observed conformations have been used without further optimization. We have used 6-31G(3df,3pd) basis set for complexes 1.a, 1.c and 1.d while 6-311G** for 1.b complex and results obtained with these basis sets have been presented in similar studies.46Results and discussion
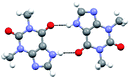
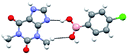
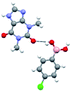
The co-crystals 1.a and 1.c–e, as specified in Chart 1, thus, obtained as described in preceding section, were characterized by single crystal X-ray diffraction method. All the complexes are found to be anhydrous, except 1.e, and the resultant asymmetric units are shown in Fig. 1 in the form of ORTEP. The structural features of each assembly is discussed in following section along with correlations and diversity among the structures. | ||
| Fig. 1 ORTEP Of molecular contents in the crystal structures of (a) 1.a, (b) 1.c, (c) 1.d and (d) 1.e. | ||
An important characteristic feature to be emphasized that in all the assemblies, respective boronic acids adopt syn–anti conformation, which was noted as stable amongst others, as reported in the literature.26,28 Further, structural analyses of all the complexes reveal that boronic acid functionality exclusively participate in heteromeric interaction with theophylline molecule, while theophylline forms both homomeric and heteromeric interactions, except in the complex 1.d where it forms only heteromeric interaction with the corresponding boronic acid moiety.
Structural description of complex, 1.a
Co-crystallisation of theophylline 1, with 4-chlorophenylboronic acid (a) from an ethanol solution gave plate-like colourless crystals 1.a, suitable for X-ray diffraction studies. Structural analysis reveals that in the molecular complex 1.a, the co-former are present in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molecular ratio, in a triclinic space group. Contents of an asymmetric unit are shown in Fig. 1(a), while pertinent crystallographic parameters are listed in Table 1.
1 molecular ratio, in a triclinic space group. Contents of an asymmetric unit are shown in Fig. 1(a), while pertinent crystallographic parameters are listed in Table 1.
From the molecular structure, it is apparent that –B(OH)2 moiety of boronic acid is in syn–anti conformation. Further, packing analysis of the molecules in the crystal lattice reveals that theophylline and molecules of a are held together through O–H⋯N (H⋯N, 1.92 Å) and N–H⋯O hydrogen bonds (H⋯O, 2.53 Å) yielding binary units. Such adjacent dimers are further self-assembled through the formation of O–H⋯O hydrogen bonds (H⋯O, 2.11 Å) yielding a tetrameric network, as shown in Fig. 2(a).
 | ||
| Fig. 2 (a) A two dimensional sheet structure in the complex 1.a (b) stacked layer arrangement observed in 1.a. | ||
Further, the juxtaposed tetrameric ensembles are arranged in the form of tapes by homomeric interactions formed between adjacent theophylline molecules with the aid of cyclic and centrosymmetric N–H⋯O hydrogen bonds (H⋯O, 1.94 Å). Within the two-dimensional arrangement, the tapes are connected to each other by heteromeric interactions formed between co-formers by C–H⋯Cl hydrogen bonds (H⋯Cl, 2.84 Å), as represented in Fig. 2(a). Complete characteristics of hydrogen bonds are given in Table 2.
In three-dimensional arrangement, such sheets are further stacked as is shown in Fig. 2(b). To continue the exploration with other halo derivatives, co-crystallization of 1 and 4-bromophenylboronic acid, b is carried out.
Structure evolution of molecular complex, 1.b
Co-crystallization of 1 and b from any of the solvents did not yield good quality single crystals suitable for X-ray diffraction studies to determine molecular structure of the complex and further to analyse recognition and self-assembly process. However, the microcrystalline solid obtained shows high crystallinity as the powder X-ray diffraction patterns show well resolved peaks, with a characteristic peak of high intense at 26.5°, as shown in Fig. 3(a). Further, the pattern has distinctly different peaks than in the patterns of the co-formers, as shown in Fig. 3(b) and (c), respectively, in their native forms. In particular, the characteristic peaks corresponding to either of the co-formers, for instance, at 2θ 7, 11.5, 14.5 and 8.5, 18, 24 in Fig. 3(b) and (c) are not present in Fig. 3(a), thus suggesting formation of a molecular complex, 1.b. | ||
| Fig. 3 Powder X-ray diffraction patterns of (a) complex, 1.b, (b) theophylline, 1, (c) 4-bromophenylboronic acid, b, (d) co-crystals of 1.a, (e) simulated patterns of 1.a and (f) co-crystals of 1.c. | ||
Interestingly, upon comparison of the patterns of the microcrystalline solid of 1.b with that of the X-ray powder patterns of the 1.a, as represented in Fig. 3(d), both of them are noted to be similar in all aspects, especially the coincided signature peaks at 26.5°. Such correlated patterns may be resultant of iso-structurality of the solids, hence it may be considered that replacement of –Cl by –Br showed no structural deviations between 1.a and 1.b. In fact, it is well known from the literature that in many instances, compounds of halogen series are iso-structural.26b,c Thus, 1.b may be regarded to have the same structural packing features as that of 1.a which were derived experimentally from single crystal X-ray diffraction studies. Furthermore, to ensure that crystalline phases are of the same from both single and microcrystalline solids, simulated patterns of 1.a (Fig. 3(e)) are compared with that of the experimental patterns and noted that both are identical.
Establishing structural correlation between 1.a and 1.b, further experimentation was followed up by preparing co-crystals of 1 with 4-iodophenylboronic acid, c.
Supramolecular assembly in molecular complex, 1.c
Structure determination of molecular complex, 1.c, reveals that the crystal lattice is similar to that of the molecular complex 1.a, with closely related unit cell parameters (1.a, a = 7.510, b = 9.471, c = 11.878 Å, α = 85.63, β = 79.47, γ = 68.96°; 1.c, a = 7.475, b = 9.623, c = 11.957 Å, α = 87.25, β = 80.94, γ = 71.63°). Furthermore, the space group (triclinic, P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) is also found to be as same as that of 1.a. Complete structure parameters are listed in Table 1. Thus, asymmetric unit of complex 1.c also contains one molecule of each reactant, as shown in Fig. 1(b). The packing of the molecules in the crystal lattice are shown in Fig. 4, which are very much similar to that were shown in Fig. 2 for the complex 1.a. The iso-structurality is also established by the matching of PXRD patterns of 1.c with that of 1.a and 1.b. Such observations further, support the literature survey on the iso-structural features among the halogenated structures, although halogens differ with each other by various factors like, electronegativity, size, etc.
) is also found to be as same as that of 1.a. Complete structure parameters are listed in Table 1. Thus, asymmetric unit of complex 1.c also contains one molecule of each reactant, as shown in Fig. 1(b). The packing of the molecules in the crystal lattice are shown in Fig. 4, which are very much similar to that were shown in Fig. 2 for the complex 1.a. The iso-structurality is also established by the matching of PXRD patterns of 1.c with that of 1.a and 1.b. Such observations further, support the literature survey on the iso-structural features among the halogenated structures, although halogens differ with each other by various factors like, electronegativity, size, etc.
 | ||
| Fig. 4 (a) Two dimensional sheet structure in complex 1.c; (b) layer structure of complex 1.c in three dimensions. | ||
It is apparent from the structural analysis of 1.a–c, that both –B(OH)2 and halo groups show structure directing features of the observed assemblies. Since only halogen groups have been used in conjunction to –B(OH)2 in 1.a–c, the study has also been extended to non-halogenated functional groups like –OH, to explore its impact on the process of molecular recognition of such boronic acids towards theophylline, 1. Thus, co-crystallisation experiments of 1, carried out with 4-hydroxyphenylboronic acid (d) from an ethanol solution, gave a molecular complex, 1.d.
Packing of molecules in molecular complex, 1.d
Structure determination of 1.d reveals that it is an anhydrous molecular complex, in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of co-formers, with the asymmetric unit is as shown Fig. 1(c). It is apparent that in 1.d also, the boronic acid moiety adopts the syn–anti conformation, as found in the complexes 1.a and 1.c. Analysis of recognition features discloses that the co-formers are held together in the form of binary units by holding each other through a pair-wise hydrogen bonds O–H⋯O and N–H⋯O, with the corresponding H⋯O distances being 1.85 and 1.90 Å, respectively. A typical interaction is shown in Fig. 5(a). Although in the molecular complexes 1.a and 1.c also, the co-formers comprise of binary units, it is interesting to note that in 1.d, the pattern of interaction between the co-formers is vividly different (O–H⋯N/N–H⋯O vs. O–H⋯O/N–H⋯O). The dimers are further connected to each other through O–H⋯O hydrogen bonds, that are being established by –OH group, with an H⋯O distance of 1.91 Å (see Fig. 5(a)) leading to the formation of tetrameric linear chains. The four-component ensembles, indeed, progress along a direction yielding infinite chains due to the periodicity. Thus, in the complex 1.d, unlike 1.a and 1.c, homomeric dimers are not observed even between the molecules of 1. The chains are arranged in the crystal lattice in the form of crossed chains, by establishing a O–H⋯N (H⋯N, 2.02 Å) hydrogen bond between the entangled chains. Such packing arrangement yields a square-grid network as is shown in Fig. 5(b).
1 ratio of co-formers, with the asymmetric unit is as shown Fig. 1(c). It is apparent that in 1.d also, the boronic acid moiety adopts the syn–anti conformation, as found in the complexes 1.a and 1.c. Analysis of recognition features discloses that the co-formers are held together in the form of binary units by holding each other through a pair-wise hydrogen bonds O–H⋯O and N–H⋯O, with the corresponding H⋯O distances being 1.85 and 1.90 Å, respectively. A typical interaction is shown in Fig. 5(a). Although in the molecular complexes 1.a and 1.c also, the co-formers comprise of binary units, it is interesting to note that in 1.d, the pattern of interaction between the co-formers is vividly different (O–H⋯N/N–H⋯O vs. O–H⋯O/N–H⋯O). The dimers are further connected to each other through O–H⋯O hydrogen bonds, that are being established by –OH group, with an H⋯O distance of 1.91 Å (see Fig. 5(a)) leading to the formation of tetrameric linear chains. The four-component ensembles, indeed, progress along a direction yielding infinite chains due to the periodicity. Thus, in the complex 1.d, unlike 1.a and 1.c, homomeric dimers are not observed even between the molecules of 1. The chains are arranged in the crystal lattice in the form of crossed chains, by establishing a O–H⋯N (H⋯N, 2.02 Å) hydrogen bond between the entangled chains. Such packing arrangement yields a square-grid network as is shown in Fig. 5(b).
 | ||
| Fig. 5 (a) Molecular recognition between 1 and d and arrangement of linear tetrameric units. (b) Square-grid network observed in the crystal lattice of 1.d. | ||
Structural description of complex, 1.e
Structure determination avowed that the molecular complex, 1.e, crystallises in a triclinic P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group. Unlike the complexes 1.a–d, discussed above, complex 1.e is a hydrate structure comprises of theophylline and molecules of e in a 2
space group. Unlike the complexes 1.a–d, discussed above, complex 1.e is a hydrate structure comprises of theophylline and molecules of e in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio along with a water molecule. The asymmetric unit is delineated in Fig. 1(e) and the crystal data is presented in Table 1. The three-dimensional packing of molecules is quite alluring and different from other complex. The molecules are packed in the crystal lattice, in the form of stacked layers, as shown in Fig. 6(a).
1 ratio along with a water molecule. The asymmetric unit is delineated in Fig. 1(e) and the crystal data is presented in Table 1. The three-dimensional packing of molecules is quite alluring and different from other complex. The molecules are packed in the crystal lattice, in the form of stacked layers, as shown in Fig. 6(a).
 | ||
| Fig. 6 (a) Stacking of layers observed in the molecular complex 1.e. (b) Two dimensional structural arrangement in 1.e. (c) Channel structure of complex 1.e in three dimensions. | ||
In a typical sheet, however the adjacent molecules of 1 and e establish a recognition through the formation of a pair-wise hydrogen bonds, O–H⋯N/C–H⋯O, with the corresponding H⋯N and H⋯O distances being 2.06 and 2.63 Å, respectively. It is interesting to note that the recognition pattern is entirely different than observed in 1.a–d. The dimers, thus, obtained are held together by a O–H⋯O (H⋯O, 2.06 Å) hydrogen bonding, realizing a quartet ensemble, somewhat similar to such units observed in complex, 1.a–c. However, in 1.e, the quartet ensembles are evolved with a void space of dimension ∼7 × 6 Å2, suitable to fit water molecules as shown Fig. 6(b). Such quartet ensembles embedded with water molecules are further connected to each other through homomeric interactions, in the form of centrosymmetric N–H⋯O (H⋯O, 1.86 Å) hydrogen bonds formed between theophylline molecules, (see Fig. 6(b)), yielding a planar layer structure. Ultimately, in the crystal lattice, the arrangement is stabilized by stacking of the layers (Fig. 6(a)). Since the stacking is through translational symmetry, the channels within the sheets are positioned such that voids are aligned channels. Within the channels, the water molecules are held each other as well as with boronic acids in the host network, by different types of O–H⋯O hydrogen bonds.
Density functional modeling
The formation of a co-crystal depends upon the lattice energy which is partly directed by favourable energy minimization through intermolecular interactions, for example, hydrogen bonds, present between the adjacent molecules, in the form of homomers and heteromers. Hence the estimation of binding energy for all such feasible and possible recognition patterns is appropriate even to emphasize the formation of a specific type of pattern over the other. Such an exercise has been carried out using DFT-D3 method with B3LYP functional. Energies have been estimated and their values are presented in Table 3.On comparing the results obtained for binding energy calculations, it shows that the homodimer formed between the theophylline molecules which is present in complexes 1.a, 1.c and 1.e shows lowest energy among all the motifs present in the crystal structures of 1.a, 1.c–e. This can be attributed to centrosymmetric cyclic dimer present between strong hydrogen bond donor and acceptor atoms, resulted into the formation of strong hydrogen bonded dimer.
Heterodimers formed by boronic acids with theophylline shows topological difference hence binding energy required to form these motifs also differs significantly. Heterodimer formed between d and theophylline involves strong N–H⋯O and O–H⋯O hydrogen bonds hence it shows lowest energy among all heterodimers formed by boronic acid with theophylline which involves weak C–H⋯O hydrogen bond in their dimer formation. Heteromer formed between hydroxyl group and theophylline in complex 1.d shows lowest energy among heteromers formed by other functional groups such as boronic acid and halo groups. Though the heteromer formed by hydroxy group is more stable compared to heteromer formed by boronic acid, the difference in energy is very less (0.627 kcal mol−1).
Haloboronic acid complexes are iso-structural and form similar structural motifs in their crystal structures, but these motifs differ significantly in their binding energy. Such variations may be accounted for the difference in the electronic factors between –Cl and –I atoms. Thus, heteromers with pair-wise hydrogen bonds formed by a are lower in energy (−9.413 kcal mol−1) compared to the corresponding ones formed by c (−6.903 kcal mol−1) and also a similar trend is observed in case of C–H⋯X (X–, Cl, I). In contrast, single hydrogen bonded heteromers appear to be favourable in 1.c (−8.158 kcal mol−1) than in 1.a (−7.530 kcal mol−1). However, in overall, 1.a shows more stability compared to 1.c by an amount of −2.509 kcal mol−1, taken into account energy of all the interactions.
Conclusion
This study supports the use of boronic acid functionality as a good chromophore for the synthesis of co-crystals of pharmaceutically important ingredients (APIs) and will help to study further physico-chemical properties of such complexes. It is important to note that despite the affinity of 1 for hydration under humid conditions, the complexes are anhydrous except 1.e. Further, from the structural aspects, the molecular recognition patterns in the form of homomeric and heteromeric interactions play an important role in the formation of co-crystal, except in case of complex 1.d wherein only heteromeric interactions are present in the crystal structure.Theoretical calculations indicate that binding energy of structural motifs depends upon type of functional groups involved in the motif formation. Haloboronic acids though form iso-structural motifs, differs significantly in their binding energies. The energy difference between the heteromers formed by boronic acid and hydroxyl group is very less which supports our previous study indicating boronic acid is as competitive as hydroxyl group in the formation of hydrogen bonded complexes.
Acknowledgements
We are thankful to the computational resources provided by the Swedish National Infrastructure for Computing (SNIC) at HPC2N and PDC. LG and VRP thank IIT Bhubaneswar for Infrastructure facilities and LG thanks MHRD for Senior Research Fellowship.References
- D. G. Hall, Boronic Acids: Prep. and Appl. in Organic Synth. and Med., 2006, p. 1 Search PubMed.
- (a) X. Lu and S. Lin, J. Org. Chem., 2005, 70, 9651 CrossRef CAS PubMed; (b) D. J. Paymode and C. V. Ramana, J. Org. Chem., 2015, 80, 11551 CrossRef CAS PubMed.
- N. Miyaura, Bull. Chem. Soc. Jpn., 2008, 81, 1535 CrossRef CAS.
- H. Rao, H. Fu, Y. Jiang and Y. Zhao, Angew. Chem., Int. Ed., 2009, 48, 1114 CrossRef CAS PubMed.
- Z. Tang and Q. Hu, J. Am. Chem. Soc., 2004, 126, 3058 CrossRef CAS PubMed.
- M. Li, W. Zhu, F. Marken and T. D. James, Chem. Commun., 2015, 51, 14562 RSC.
- T. D. James, K. R. A. S. Sandanayake and S. Shinkal, Nature, 1995, 374, 345 CrossRef CAS.
- E. W. Miller, A. E. Albers, A. Pralle, E. Y. Isacoff and C. J. Chang, J. Am. Chem. Soc., 2005, 127, 16652 CrossRef CAS PubMed.
- A. Schiller, R. A. Wessling and B. A Singaram, Angew. Chem., Int. Ed., 2007, 46, 6457 CrossRef CAS PubMed.
- J. C. Pickup, F. Hussain, N. D. Evans, O. J. Rolinski and D. J. S. Birch, Biosens. Bioelectron., 2005, 20, 2555 CrossRef CAS PubMed.
- J. R. Hunt, C. J. Doonan, J. D. LeVangie, A. P. Côté and O. M. Yaghi, J. Am. Chem. Soc., 2008, 130, 11872 CrossRef CAS PubMed.
- M. A. Mehta, T. Fujinami, S. Inoue, K. Matsushita, T. Miwa and T. Inoue, Electrochim. Acta, 2000, 45, 1175 CrossRef CAS.
- D. Salazar-Mendoza, J. Guerrero-Alvarez and H. Höpfl, Chem. Commun., 2008, 6543 RSC.
- G. Zhang, O. Presly, F. White, I. M. Oppel and M. A Mastalerz, Angew. Chem., Int. Ed., 2014, 53, 1516 CrossRef CAS PubMed.
- J. N. Cambre and B. S. Sumerlin, Polymer, 2011, 52, 4631 CrossRef CAS.
- P. C. Trippler and C. McGuigan, MedChemComm, 2010, 1, 183 RSC.
- M. Sung, L. Bagain, Z. Chen, T. Karpova, X. Yang, C. Silvin, T. C. Voss, J. G. McNally, C. Van Waes and G. L. Hager, Mol. Pharmacol., 2008, 74, 1215 CrossRef CAS PubMed.
- D. B. Carter, D. A. Ross, K. S. Ishaq, G. M. Suarez and C. Chae, Biochim. Biophys. Acta, 1977, 484, 103 CrossRef CAS.
- J. Liu, H. Zhang, Z. Xiao, F. Wang, X. Wang and Y. Wang, Int. J. Mol. Sci., 2011, 12, 1807 CrossRef CAS PubMed.
- Y. Zhu, S. Yao, B. Xu, Z. Ge, J. Cui, T. Cheng and R. Li, Bioorg. Med. Chem., 2009, 17, 6851 CrossRef CAS PubMed.
- M. K. Cyrański, A. Jezierska, P. Klimentowska, J. J. Panek, G. Z. Zukowska and A. Sporzyński, J. Chem. Phys., 2008, 128 Search PubMed.
- M. Kurt, T. R. Sertbakan and M. Özduran, Spectrochim. Acta, Part A, 2008, 70, 664 CrossRef CAS PubMed.
- R. Nishiyabu, Y. Kubo, T. D. James and J. S. Fossey, Chem. Commun., 2011, 47, 1124 RSC.
- R. Nishiyabu, Y. Kubo, T. D. James and J. S. Fossey, Chem. Commun., 2011, 47, 1106 RSC.
- (a) P. De, S. R. Gondi, D. Roy and B. S. Sumerlin, Macromolecules, 2009, 42, 5614 CrossRef CAS; (b) J. Fournier, T. Maris, J. D. Wuest, W. Guo and E. Galoppini, Molecular tectonics, J. Am. Chem. Soc., 2003, 125, 1002 CrossRef CAS PubMed; (c) P. M. Mitrasinovic, Curr. Org. Synth., 2012, 9, 233 CrossRef CAS; (d) Y. Zhang, M. Li, S. Chandrasekaran, X. Gao, X. Fang, H. Lee, K. Hardcastle, J. Yang and B. A. Wang, Tetrahedron, 2007, 63, 3287 CrossRef CAS PubMed; (e) P. Rogowska, M. K. Cyranski, A. Sporzynski and A. Ciesielski, Tetrahedron Lett., 2006, 47, 1389 CrossRef CAS.
- (a) V. R. Pedireddi and N. SeethaLekshmi, Tetrahedron Lett., 2004, 45, 1903 CrossRef CAS; (b) S. Seethalekshmi, S. Varughese, L. Giri and V. R. Pedireddi, Cryst. Growth Des., 2014, 14, 4143 CrossRef CAS; (c) M. R. Shimpi, N. Seethalekshmi and V. R. Pedireddi, Cryst. Growth Des., 2007, 7, 1958 CrossRef CAS.
- M. Talwelkar and V. R. Pedireddi, Tetrahedron Lett., 2010, 51, 6901 CrossRef CAS.
- N. SeethaLekshmi and V. R. Pedireddi, Cryst. Growth Des., 2007, 7, 944 CAS.
- N. Seethalekshmi and V. R. Pedireddi, Inorg. Chem., 2006, 45, 2400 CrossRef CAS PubMed.
- C. B. Aakeröy, J. Desper and B. Levin, CrystEngComm, 2005, 7, 102 RSC.
- P. Rodríguez-Cuamatzi, O. I. Arillo-Flores, M. I. Bernal-Uruchurtu and H. Höpfl, Cryst. Growth Des., 2005, 5, 167 Search PubMed.
- D. Tzeli, G. Theodorakopoulos, I. D. Petsalakis, D. Ajami and J. Rebek, J. Am. Chem. Soc., 2011, 133, 16977 CrossRef CAS PubMed.
- J. D. Larkin, K. L. Bhat, G. D. Markham, B. R. Brooks, H. F. Schaefer III and C. W. Bock, J. Phys. Chem. A, 2006, 110, 10633 CrossRef CAS PubMed.
- S. Varughese, S. B. Sinha and G. R. Desiraju, Sci. China: Chem., 2011, 54, 1909 CrossRef CAS.
- (a) S. Varughese, Y. Azim and G. R. Desiraju, J. Pharm. Sci., 2010, 99, 3743 CrossRef CAS PubMed; (b) V. Dyulgerov, R. P. Nikolova, L. T. Dimova and B. L. Shivachev, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2012, 68, o2320 CAS; (c) J. H-Paredes, A. L. Olvera-Tapia, J. I. Arenas-García, H. Höpfl, H. Morales-Rojas, D. Herrera-Ruiz, A. I. Gonzaga-Morales and L. Rodríguez-Fragoso, CrystEngComm, 2015, 17, 5166 RSC.
- (a) C. T. Furukawa, G. G. Shapiro, C. W. Bierman, M. J. Kraemer, D. J Ward and W. E. Pierson, Pediatrics, 1984, 74, 453 CAS; (b) J. W. Jenne, Clin. Chest Med., 1984, 5, 645 CAS; (c) Y. Ono, T. Kondo, T. Tanigaki, Y. Syouyama, Y. Moue, U. Kamiya, G. Tazaki and Y. Ohta, Nihon Kokyuki Gakkai Zasshi, 2001, 39, 75 CAS; (d) M. Spears, I. Donnelly, L. Jolly, M. Brannigan, K. Ito, C. McSharry, J. Lafferty, R. Chaudhuri, G. Braganza, I. M. Adcock, P. J. Barnes, S. Wood and N. C. Thomson, Eur. Respir. J., 2009, 33, 1010 CrossRef CAS PubMed; (e) A. E. B. Yassin, A. H. Aodah, S. Al-Suwayeh and E. I. Taha, Pharm. Dev. Technol., 2012, 17, 712 CrossRef CAS PubMed.
- M. Sung, L. Bagain, Z. Chen, T. Karpova, X. Yang, C. Silvin, T. C. Voss, J. G. McNally, C. Van Waes and G. L. Hager, Mol. Pharmacol., 2008, 74, 1215 CrossRef CAS PubMed.
- A. Paramore and S. Frantz, Drug Discov., 2003, 2, 611 CrossRef CAS PubMed.
- SAINT, Version 6.02, Bruker AXS, Inc., Analytical X-ray Systems, 5465 East Cheryl Parkway, Madison, WI, 2000, pp. 53711–55373 Search PubMed.
- SADABS [Area-Detector Absorption Correction], Siemens Industrial Automation, Inc., Madison, WI, 1996 Search PubMed.
- G. M. Sheldrick, SHELXTL, University of Gottingen, Gottingen, Germany, 1997 Search PubMed.
- (a) A. L. Spek, Acta Crystallogr., 2009, 65, 148 CAS; (b) A. L. Spek, PLATON, A Multipurpose Crystallographic Tool, Utrecht University, Utrecht, Netherland, 2002 Search PubMed.
- M. Valiev, E. J. Bylaska, N. Govind, K. Kowalski, T. P. Straatsma, H. J. J. Van Dam, D. Wang, J. Nieplocha, E. Apra, T. L. Windus and W. A. De Jong, Comput. Phys. Commun., 2010, 181, 1477 CrossRef CAS.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132 Search PubMed.
- C. Lee, W. Yang and R. G. Parr, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 37(2), 785 CrossRef CAS.
- W. J. Hehre, K. Ditchfield and J. A. Pople, J. Chem. Phys., 1972, 56, 2257 CrossRef CAS.
Footnote |
| † CCDC reference numbers for complexes 1.a, 1.c, 1.d and 1.e are 1440006–1440009, respectively. For crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra04100k |
| This journal is © The Royal Society of Chemistry 2016 |