Antifungal properties of lecithin- and terbinafine-loaded electrospun poly(ε-caprolactone) nanofibres†
Sriram Harini‡
a,
Mayandi Venkatesh‡a,
Sridhar Radhakrishnancd,
Mobashar Hussain Urf Turabe Fazile,
Eunice Tze Leng Goha,
Sun Ruic,
Chetna Dhanda,
Seow Theng Onge,
Veluchamy Amutha Barathiab,
Roger W. Beuermanab,
Seeram Ramakrishnacdf,
Navin Kumar Verma*ae and
Rajamani Lakshminarayanan*ab
aSingapore Eye Research Institute, The Academia, 20 College Road, Discovery Tower Level 6, Singapore 169856. E-mail: lakshminarayanan.rajamani@seri.com.sg
bOphthalmology and Visual Sciences Academic Clinical Program, Duke-NUS Graduate Medical School, Singapore 169857
cDepartment of Mechanical Engineering, National University of Singapore, Singapore 117584
dCenter for Nanofibres and Nanotechnology, National University of Singapore, Singapore 117584
eLee Kong Chian School of Medicine, Nanyang Technological University, Experimental Medicine Building, Singapore 636921. E-mail: nkverma@ntu.edu.sg
fGuangdong-Hongkong-Macau Institute of CNS Regeneration (GHMICR), Jinan University, Guangzhou 510632, China
First published on 20th April 2016
Abstract
The purpose of this article is to investigate the efficacy of electrospun polycaprolactone (PCL) fibres loaded with egg lecithin and terbinafine hydrochloride (terbinafine) against moulds and dermatophytic fungi which are responsible for superficial nail and skin mycoses. Scanning electron microscopy studies indicated that addition of lecithin and terbinafine decreased the average diameter of the PCL nanofibres, improved the wettability of the fibre mats, increased the mechanical properties and displayed pronounced blue fluorescence. Cellular adhesion studies and confocal microscopy images revealed that the PCL mats containing lecithin and terbinafine did not affect cell adhesion and maintained a normal phenotypic shape and viability of the primary human dermal fibroblasts. In vitro antifungal assays demonstrated that terbinafine-loaded mats maintained antifungal efficacy against moulds as well as dermatophytic fungus. Using an ex vivo porcine skin infection model, we showed that the drug-eluting mats resulted in >5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log reduction in the viability of T. mentagrophytes.
log reduction in the viability of T. mentagrophytes.
Introduction
Superficial fungal infections are one of the most frequent infections that affect the human population. Skin and nail fungal infections affect ∼1.7 billion populations worldwide.1 Dermatophytic fungi such as those belonging to the genera Trichophyton, Microsporum and Epidermophyton are some of the key etiological agents responsible for conditions such as athlete's foot, ringworm and infection of the nails.2,3 These fungi often find their susceptible host among immunocompromised individuals who eventually suffer from opportunistic infection of skin mycoses. The frequency of opportunistic fungal infections has increased over the past few decades. The use of anti-neoplastic and immunosuppressive agents, broad spectrum antibiotics, prosthetic devices and grafts and aggressive surgery are the major risk factors for the increasing incidence of fungal skin infections. Patients with burns, neutropenia, HIV infection and pancreatitis are highly prone to opportunistic infections due to their impaired cell immunity.4Current management of fungal infections includes both topical and systemic antifungal drugs. Topical application is preferred since a high concentration of the drugs can be achieved at the site of infections, enhancing the efficacy of the treatment and decreasing the possible side effects while increasing patient compliance.5 Topical allylamine antifungals show faster response against infection than the azoles, although their cure rates are very similar. Terbinafine hydrochloride is an allylamine antifungal agent which inhibits the squalene epoxidase, an enzyme involved in the early step of the ergosterol biosynthesis.6 Terbinafine is reported to be superior to griseofulvin and itraconazole formulations in many aspects such as gastrointestinal absorption, protein binding ability, half-life, strains against which they are effective, faster response, better cure rate and toxicity.7 Terbinafine is highly lipophilic in nature, diffuse rapidly through the dermis and gets concentrated in the lipophilic stratum corneum.8 The therapeutic efficacy of topical formulations is governed in part by the vehicle characteristics. Various strategies have been explored to improve the drug penetration for treating deep subcutaneous infections. Barot et al., reported that a microemulsion formulations containing oleic acid, Labrasol and Transcutol P in water had about three times superior penetration into the human cadaver skin compared to commercial formulations.9 Topical terbinafine formulations containing phospholipids, polymers, fatty acids, fatty alcohols, surfactants, terpenes have been shown to improve the transdermal delivery without affecting the antifungal properties.10–12
Electrospun nanofibres serve as an ideal matrix for the delivery of antibiotics to the infected sites. The method has a number of advantages over the other methods of drug delivery such as high surface area, ease of incorporation of the drug and limited time for drug recrystallization due to faster evaporation of the solvent. The drug loaded nanofibres helps to overcome the frequent use of the drugs formulations thereby reducing the chances of adverse reactions, side effects and evolution of drug resistance caused by excess active drug components.13–17 The nanofibre based drug delivery system for treatment of wounds and topical infections serves as a functional topical or local system for the purpose. By controlling the degradation of the nanofibre we can achieve a uniform, burst or controlled release of drug at the site of wound or infection through the high surface area to volume ratio of the nanofibres.18 Nanofibres prepared from formulations containing fatty acids/alcohols can enhance the bioavailability and drug permeability without compromising its pharmacological properties.19,20 Such type of formulations has been used by exploiting electrospinning for the release of eugenol, hexadecane, lysozyme, (R)-(+)-limonene and for improving the mucoadhesivity of α-tacopherol.21–24
Among the various synthetic polymer mats, electrospun polycaprolactone (PCL) has been widely used for drug delivery applications owing to its compatibility with numerous drugs, biocompatibility, low hydrolytic degradability and low cost.25–27 Blending of PCL with gelatin, plasma treatment or lecithin has been shown to improve the wettability and cell adhesion. In this work, we prepared the PCL membrane loaded with lecithin and terbinafine and investigated their effect on mechanical properties, swellability, cell adhesion, biocompatibility and photoluminescence properties. We assessed the antifungal properties against filamentous and dermatophytic fungi and established their efficacy in an ex vivo efficacy of a porcine skin fungal infection model.
Materials and methods
PCL (Mw = 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), chloroform and methanol were purchased from Sigma-Aldrich (Singapore). The phospholipids from eggs were obtained from VAV Life sciences PVT Limited, Mumbai (India). Rhodamine-phalloidin fluorescent dye was purchased from Molecular Probes®, Antibody anti-α-tubulin, Hoechst staining solution and Flouromount™ Aqueous mounting medium were purchased from Sigma-Aldrich. DMEM cell growth medium was purchased from Gibco®. All the cell culture reagents were obtained from Life Technologies Corporation (Singapore). All other chemicals and reagents were obtained from Sigma Aldrich (S) Pvt. Ltd. All chemicals were of analytical grade and used without further purifications.
000), chloroform and methanol were purchased from Sigma-Aldrich (Singapore). The phospholipids from eggs were obtained from VAV Life sciences PVT Limited, Mumbai (India). Rhodamine-phalloidin fluorescent dye was purchased from Molecular Probes®, Antibody anti-α-tubulin, Hoechst staining solution and Flouromount™ Aqueous mounting medium were purchased from Sigma-Aldrich. DMEM cell growth medium was purchased from Gibco®. All the cell culture reagents were obtained from Life Technologies Corporation (Singapore). All other chemicals and reagents were obtained from Sigma Aldrich (S) Pvt. Ltd. All chemicals were of analytical grade and used without further purifications.
Preparation of drug loaded PCL nanofibres
PCL nanofibres were fabricated by the electrospinning method. PCL was dissolved in methanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) chloroform mixture (3
chloroform mixture (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 ratio) at a concentration of 10% w/v. Egg lecithin was loaded at the concentration of 5% (w/w of PCL) into the polymer solution based on previous report that 5% lecithin provided optimum mechanical, hemocompatibility and cell adhesion properties.24 For the drug-loaded fibre mats, terbinafine hydrochloride powder was mixed with PCL/lecithin solution and the final drug concentration in the dope solution was varied from 0.5 to 2 wt% with respect to PCL. The mixture was stirred overnight at room temperature to achieve a homogenous solution. The polymer solution was poured into a standard 10 mL syringe that was attached to a steel needle. Electrospinning was carried out by introducing a high voltage (DC high voltage power supply from Gamma High Voltage Research, Florida, U.S.) to the needle as a positive electrode and a fixed sheet of grounded aluminum collector. The distance between needle tip and collector was set at 13 cm. Once positive electrode reached a critical voltage of 18 kV, spinning head extruded polymer solution and the electrospun fibers were deposited on the aluminum foil collector. The emitting rate of the polymer solution was controlled at 1 mL h−1 by means of a syringe pump (KD Scientific Inc., M.A., USA). For simplicity the mats were labeled as follows: pristine PCL mats – PCL; PCL mats containing 2% terbinafine – PCL_2%TERB; PCL mats containing lecithin – PCL_Lt and PCL mats containing lecithin and 2% terbinafine – PCL_Lt_2%TERB.
7 ratio) at a concentration of 10% w/v. Egg lecithin was loaded at the concentration of 5% (w/w of PCL) into the polymer solution based on previous report that 5% lecithin provided optimum mechanical, hemocompatibility and cell adhesion properties.24 For the drug-loaded fibre mats, terbinafine hydrochloride powder was mixed with PCL/lecithin solution and the final drug concentration in the dope solution was varied from 0.5 to 2 wt% with respect to PCL. The mixture was stirred overnight at room temperature to achieve a homogenous solution. The polymer solution was poured into a standard 10 mL syringe that was attached to a steel needle. Electrospinning was carried out by introducing a high voltage (DC high voltage power supply from Gamma High Voltage Research, Florida, U.S.) to the needle as a positive electrode and a fixed sheet of grounded aluminum collector. The distance between needle tip and collector was set at 13 cm. Once positive electrode reached a critical voltage of 18 kV, spinning head extruded polymer solution and the electrospun fibers were deposited on the aluminum foil collector. The emitting rate of the polymer solution was controlled at 1 mL h−1 by means of a syringe pump (KD Scientific Inc., M.A., USA). For simplicity the mats were labeled as follows: pristine PCL mats – PCL; PCL mats containing 2% terbinafine – PCL_2%TERB; PCL mats containing lecithin – PCL_Lt and PCL mats containing lecithin and 2% terbinafine – PCL_Lt_2%TERB.
Scanning electron microscopy
The morphology and diameter of the electrospun fibres were studied by scanning electron microscope (SEM) (Quanta 200F, FEI, Oregon, US). Prior to imaging with SEM, the mats were sputter coated with gold (JEOL JFC-1200 fine coater, Japan). Imaging was done at an accelerating voltage of 8 kV.Water contact angle measurements
The wettability of the mats was measured by sessile drop method using video contact angle instrument (VCA-optima, AST, Inc.). The mats were glued to the glass slide using double-sided carbon tapes and placed onto the samples stage. 1 μL of deionized water was placed onto the fibre mats by means of a syringe and the contact angle was measured at 5 different locations after 15 s at room temperature.Uniaxial tensile test
Mechanical properties of electrospun fibrous membranes were determined with a tabletop uniaxial testing machine (INSTRON 3345, USA) with the use of a 10 N load cell under a cross-head speed of 5 mm min−1 at ambient conditions (25 °C and 70% relative humidity). All samples were prepared in the form of rectangular strips with dimensions of 20 × 10 mm from the electrospun fibrous membranes. Sample thickness was measured with a digital Vernier caliper. A set of 7 samples for each group of electrospun fibrous membranes were tested and the average values were reported.Biocompatibility of the nanofibre mats
Human dermal fibroblast cells, which are a well-characterized and widely used model system for determining the skin biocompatibility, were cultured as described by Verma et al.28 Briefly, cells were cultured in DMEM medium (Gibco®) supplemented with 10% (v/v) fetal bovine serum, 50 U mL−1 penicillin and 50 μg mL−1 streptomycin in a humidified incubator at 37 °C and 5% CO2. All the cell culture reagents were obtained from Life Technologies Corporation (Singapore). For biocompatibility experiments, cells were seeded onto coverslips precoated with nanofibre mats and placed at the bottom of the 12-well plates (Nunc®) at a density of 10 × 104 cells per well and allowed to grow for 24 h before analysis. Cells were incubated at 37 °C in a 5% CO2 incubator to allow them to attach on the surface and grow for various time-points depending on particular experiments. At the end of the incubation/treatment, dermal fibroblasts were analyzed for their adherence and morphologies by molecular imaging and cell viability by a MTS-based assay.Cell adhesion of dermal fibroblast on the nanofibres
For determining cellular adhesion over time, human dermal fibroblasts cultured on nanofibre preparations were fixed at various time-points over a range of 1 h to 24 h, stained, imaged using a ×20 objective lens and subsequently quantified by ImageJ software. Fluorescent images of the electrospun nanofibres were acquired by the Zeiss LSM710-Meta laser scanning microscope at 490 nm excitation and BP520 emission filter using a ×40 objective lens. At least 20 different microscopic fields were analyzed for each sample.Cell viability of the dermal fibroblast after incubation with the nanofibres
Human dermal fibroblast cell viability was determined using CellTier 96® Aqueous One solution cell proliferation assay kit according to manufacturer's instructions (Promega Corporation, Madison, WI, USA) and as described previously by Verma et al., 2012.28 This assay evaluates mitochondrial functions by measuring the ability of viable cells to reduce MTS ((3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium)) into a quantifiable blue insoluble formazon product. Briefly, at the end of the treatment period, cells growing on the nanofibre-coated coverslips placed in a 12-well plate containing 500 μL of cell culture medium were incubated with 50 μL of MTS tetrazolium solution for 2 h at 37 °C. Subsequently, the absorbance was measured at 490 nm using a microplate reader (Infinite M200 Pro, Tecan, and Mannedorf, Switzerland) and then relative cell viability was calculated. Each treatment was performed in triplicate.Fluorescence microscopy
For confocal microscopy of human dermal fibroblasts, cells were cultured on nanofibre-coated cover-slips for 24 h and then fixed in 3% paraformaldehyde. After washing with PBS, cells were fluorescently labeled with FITC-conjugated anti-α-tubulin (Sigma-Aldrich) and rhodamine-phalloidin (Molecular Probes®) to visualize the cytoskeletal systems (as indicators of cell morphologies and health) and Hoechst (Sigma-Aldrich) to visualize the nuclear morphology. Coverslips were mounted on glass slides using Flouromount™ aqueous mounting medium (Sigma-Aldrich). Confocal imaging was carried out by a laser scanning microscope (Zeiss LSM710-Meta, Carl Zeiss Microimaging Inc., NY, USA) using a ×40 oil immersion objective lens. Excitation wavelengths used were 540 nm, 490 nm and 561 nm and emission filters were BP 565 nm, BP 520 nm and 572–754 nm respectively. At least 20 different microscopic fields were analyzed for each sample.Photoluminescent properties of drug-loaded PCL mats
Fluorescence spectra of the nanofibre preparations were obtained using a BioTek Synergy H1 Multi-Mode Reader equipped with Gen5™ 2.06 software. Briefly, emission was recorded from 400 nm to 600 nm with a step interval of 10 nm at fixed excitation of 360 nm. Xenon flash light source with bottom probe was used at a read height of 4.7 mm at normal speed. To obtain an averaged value of emission spectra for the entire region of the nanofibre mats, area scanning was performed by taking multiple measurements in a matrix format (excitation 360 nm, emission 400 nm).Radial diffusion assay
Cultures of Aspergillus and Fusarium strains (at a concentration of 0.5 McFarland standards) were spread onto the surface of sterile Sabouraud dextrose agar (SDA) plates using a cotton swab in a 9 cm diameter Petri dishes. The fibre mats (1 cm × 1 cm) were placed on top of the swabbed cultures and incubated at 37 °C. Antifungal activity of fibre mats was expressed as the zone of inhibition in diameter after 72 h. The assay was performed in two independent duplicates and the average values are reported.For dermatophytic fungi, Epidermophyton and Trichophyton strains were received from American Type Culture Collection (ATCC) and subcultured on a potato dextrose agar (PDA) plate at 30 °C for 4 to 15 days. Following growth, conidia were harvested in sterile saline and adjusted to 1.0 × 106 conidia per mL. Mueller-Hinton (MH) agar (Remel, KS) plates were streaked evenly with a swab dipped into the standardized inoculum suspension. Lids were left ajar for 3 min in a laminar flow cabinet to allow for any excess surface moisture to be absorbed into the agar and the drug loaded nanofibre mats (1 cm × 1 cm) were placed on the surfaces of inoculated plates. Plates were inverted and incubated at 30 °C for 4 to 7 days to allow for fungal growth. The zone of inhibition was measured in centimeters. The assay was performed in two independent duplicates and the average value was reported.
Ex vivo porcine skin infection model
Fresh porcine skin was obtained from the Singapore Experimental Medical Centre. The skin was sterilized by immersing in 70% ethanol for 5 min, then the excised skin was washed with 0.9% saline solution 3 times. After washing, skin was soaked in saline for 5 min in order to remove the visible excess subcutaneous fat, dermis tissues with scalpel blade. Under sterile conditions the skin was cut into 2 cm × 2 cm pieces. The dried skin was placed at the bottom of a Petri dish and the experimental groups were assigned. The control and treatment groups contained four skins per plate (n = 4) in duplicates. A moist atmosphere was created by placing a sterile wet tissue paper at the bottom of the Petri dish. Trichophyton mentagrophytes were subcultured as reported before. About 100 μL aliquot of the fungal inoculum (4.0 × 106 conidia per mL) was applied to the dry epidermal surface of the skin and spread uniformly over the entire surface region using a sterile pipette tip. The skin required incubation for 6 days at 30 °C to a create skin infection. After 6 days, the epidermis at the sites of skin infections was covered with electrospun mats (2 cm × 2 cm). The Petri dish was incubated at 30 °C for 24 h. After 24 h incubation, both the control and treated skin specimens were immersed into the inhibitor solution (1 mL saline solution containing 1% polyvinyl sulphonic acid) except control group. After preparing one-log (10-fold) serial dilutions of the above solution in 0.9% saline solution was prepared. 100 μL of each dilution was plated on PDA plates, incubated at 30 °C for 5 to 7 days and the colony forming units was counted.Statistical analysis
For statistical analysis, the data were analyzed by using GraphPad Prism 6.0 software. All the experimental data was expressed in mean ± standard deviations. The two-tailed Students t-test and one-way analysis of variance with Newman–Keuls post hoc analysis were used for determining the significance of differences. The data with P < 0.05 was considered significance.Results and discussion
Morphology of drug loaded PCL nanofibre mats
Electrospinning of 10% PCL solution in methanol:chloroform binary solvent was carried out at a flow rate of 1 mL h−1 and field strength of 18 kV/13 cm. Under these conditions, electrospun PCL nanofibres exhibited coiling, looping and bending morphologies (Fig. 1(a)), suggesting structural instability as the polymer jet extends under elongational flow.29 The average diameter of the randomly aligned PCL fibres was 205 ± 35 nm (Fig. 1(a)). Addition of a natural phospholipid egg lecithin to the PCL solution (PCL_Lt) resulted in the formation of homogeneous fibres without considerable looping and coiling with decreased diameter 127.7 ± 43.7 nm (Fig. 1(b)). To determine if the addition of terbinafine had any effect on fibre morphologies, SEM analysis was performed for the PCL_Lt mats loaded with varying concentrations of terbinafine ranging from 0.25% to 2% (w/w of PCL) (Fig. 1(c–f)). It was observed that addition of terbinafine did not alter the morphology of PCL_Lt nanofibres (Fig. 1(c–f) vs. (b); Fig. 1(g)). Increasing the concentration of terbinafine in the dope solution, however, resulted in fibre mats with narrow distribution of diameter without significant change in the average diameter (p > 0.05) (Fig. 1(g)). The average diameters of fibres containing 1% and 2% terbinafine in the PCL_Lt dope solution were 104 ± 25 nm and 105 ± 17 nm, respectively (Fig. 1(e) and (f)) indicating the presence of cationic drug imposes higher elongation and thinning forces by increasing the charged density of polymer jet.30 Scanning electron micrographs indicated that addition of lecithin together with terbinafine decreased the average diameter of nanofibres in pristine PCL mats, presumably caused by increased solution conductivity due to the presence of higher amounts of cationic terbinafine in the dope solution.31 It should be noted that all the electrospinning parameters such as the working voltage, flow rate, collector distance etc. were maintained constant for the fabrication of the nanofibres and the only factor altered was the concentration of terbinafine. It was also observed that with the increase in the terbinafine concentration the fibres exhibited a straighter morphology rather than a curly one which was visible in lower concentrations. Lecithin with a flexible hydrophobic tail might influence the curvature observed during electrospinning. The curvature observed in lower concentrations of the drug gradually showed straight fibres at higher drug concentration with the lecithin concentration maintained constant at 5% (w/w). The decreased diameter of the fibres obtained can be owed to the increased solution conductivity with the increased drug concentration.32Mechanical properties of PCL nanofibre mats
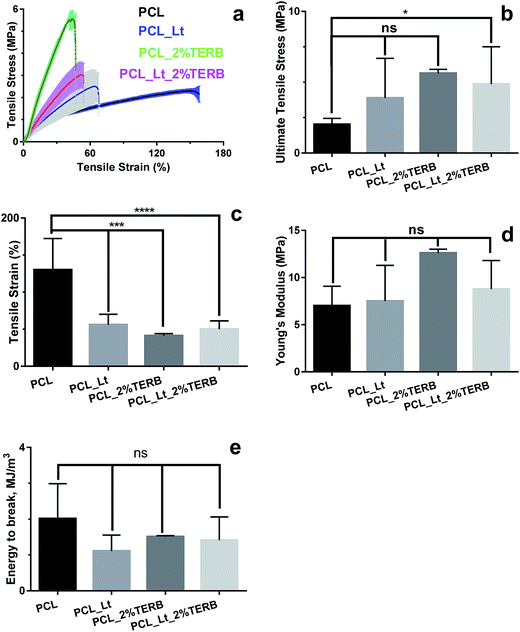
In order to evaluate this, the effect of lecithin and/or terbinafine on the tensile properties of the electrospun nanofibre mats was investigated. PCL mats displayed flexible behavior as indicated by low tensile stress (Fig. 2(a and b)) and high tensile strain (Fig. 2(c)). Incorporation of lecithin or terbinafine decreased the elastic properties of the mats, as indicated by significant decrease in the ultimate tensile strain for mats containing lecithin or terbinafine (p < 0.001). While no significant change in ultimate tensile strength was observed upon incorporation of lecithin (p > 0.05), remarkable increase was observed upon incorporation of terbinafine into PCL mats (p < 0.0001). However, we did not observe statistically significant (p > 0.05) changes in the Young's modulus and toughness in comparison to pristine PCL mats for both lecithin (PCL_Lt) and terbinafine (PCL_2%TERB) loaded mats (Fig. 2(d and e)). Incorporation of lecithin along with the drug decreased the elasticity of the nanofibre mats while increasing the ultimate tensile strength when compared to pristine PCL mats (Fig. 2(a)). It is likely that the decreased bending instability upon incorporation of lecithin or terbinafine and a decrease in the average diameter of the nanofibers could be responsible for the increase in ultimate tensile stress and decrease in elastic properties of pristine PCL mats.33 Apart from the durability of mats while handling and application, the dressings must withstand the applied load generated during cell attachment, migration and differentiation.34–36Wettability of nanofibre mats
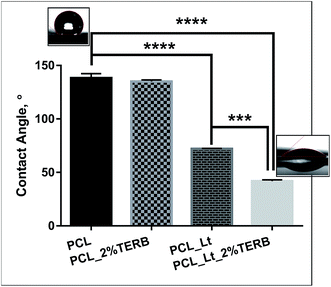
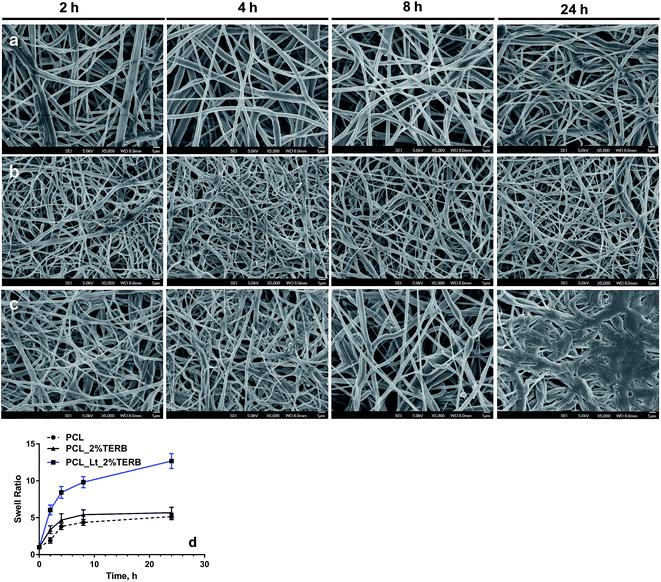
To confirm that lecithin loading altered the surface wettability of nanofibre mats, we determined the static water contact angle for various nanofibre mats. It was noted that the water contact angle of the pristine PCL nanofibre mats was 138.2 ± 4.2°, indicating their hydrophobic nature (Fig. 3). Incorporation of terbinafine did not alter the surface wettability of PCL as no change in contact angle was observed. However, addition of lecithin to PCL decreased the contact angle of the nanofibers to 72.1 ± 0.3° (Fig. 3) thus increasing the wettability of the PCL_Lt mats surfaces, consistent with the previous reports that lecithin improved the wettability and biocompatibility of synthetic polymers such as PCL, poly-L-lactide and polyamide-6.30,37 Notably, when 2% terbinafine was added to PCL_Lt, the water contact angle decreased significantly further to 41.9 ± 1.2° (Fig. 3), thus enhancing the wettability of electrospun mat surfaces further. These results suggest that, loading of PCL mats with terbinafine and lecithin reversed the hydrophobicity of PCL nanofibers. To infer the effect of increased wettability on the morphology of lecithin/terbinafine loaded PCL fibres, we examined the swelling characteristics of the fiber mats. Fig. 4(a–c) show the SEM images of PCL, PCL_2%TERB and PCL_Lt_2%TERB mats after immersing the mats in PBS buffer at various time interval. The diameter of fibres increased gradually for both PCL and PCL_2%TERB mats with increasing incubation time whereas for PCL_Lt_2%TERB mats, individual fibres coalesce to form film-like texture after 24 h. To obtain quantitative information, we determined average swelling ratio for various mats. For both PCL and PCL_TERB mats, the swelling ratio increased with immersion time and reached plateau after 24 h (Fig. 4(d)). For both the mats the average diameter increased by about 5-fold at 24 h after soaking in PBS. In contrast, PCL_Lt_TERB mats displayed faster swelling characteristics as >5-fold increase in swelling ratio was achieved within first 2 h of immersion in PBS. Consistent with the static contact angle measurements, these results indicate that addition of lecithin and terbinafine enhanced the rate of swelling of hydrophobic PCL mats (Fig. 4(d)). Water contact angle measurements showed an expected decrease in the contact angle from a value of 138.15° to 72.1° upon addition of lecithin. These results augment the earlier studies by others that incorporation of lecithin promotes the wettability of hydrophobic polymer fibres/films.24,29,30,38,39 A further decrease in the water contact angle to 41.9° was observed with the PCL_Lt_2% TERB nanofibre mats. Lecithin is a phospholipid and has an amphiphilic chemical structure consisting of a zwitterionic head groups (hydrophilic) and hydrocarbon chains with varying degree of unsaturation (hydrophobic). Due to the amphiphilic nature, lecithin self-assembles during electrospinning to form a hydrophilic layer on the PCL nanofibre surface, thus enhancing the wettability.32,38,39 As a result, the PCL_Lt_2%TERB mats displayed a greater increase in the rate of swelling than PCL or PCL_2%TERB mats. Human dermal fibroblasts seeded onto PCL mats with varying degree of hydrophilicity did not show significant differences in cell adhesion and maintained normal phenotypic shape, indicating that cell adhesion and biocompatibility was not altered by the presence of lecithin/terbinafine.Cell adhesion and biocompatibility of PCL mats
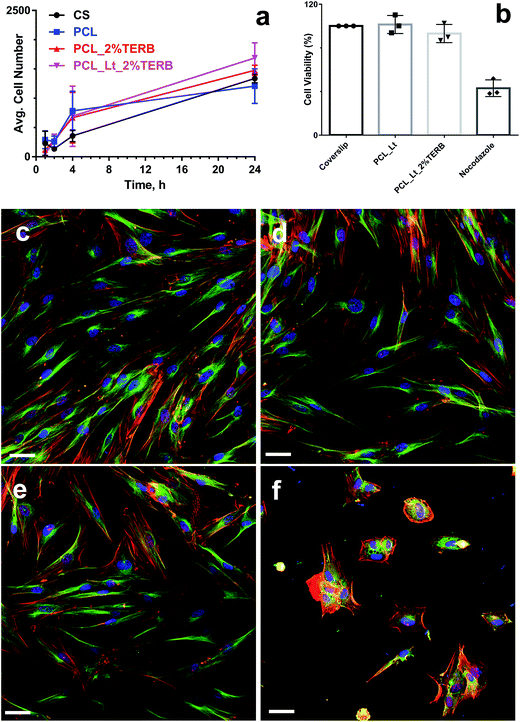
Next, we determined if the alterations in wettability of PCL upon addition of lecithin and terbinafine could alter the cell adhesion and biocompatibility for primary human dermal fibroblasts. The number of adhered cells seeded on various scaffolds was enumerated at predetermined time intervals. Fig. 5(a) showed the time-dependent increase in the number of adhered cells on various PCL mats and on microscopy coverslips, the latter served as a control. Though, the number of cells adhered to PCL_Lt_2%TERB scaffold was higher, the final count was not statistically significant (p > 0.5) among various groups. These results indicate that increased wettability of PCL did not alter the fibroblasts cell adhesion during the early stages. Human dermal fibroblasts seeded onto PCL mats with varying degree of hydrophilicity did not show significant differences in cell adhesion and maintained normal phenotypic shape, indicating that cell adhesion and biocompatibility was not altered by the presence of lecithin/terbinafine.Biocompatibility of electrospun mats were evaluated by monitoring their effects on the metabolic activity and by imaging the morphology of human dermal fibroblasts. The viability of the human dermal fibroblasts was analyzed by quantitative MTS reduction assay. No alterations in the metabolic activity of the cultures were observed as indicated by no significant decrease in the MTS values for all the electrospun mats (Fig. 5(b)). However, only 40% cell survival was observed for the nocodazole treated cultures, confirming the cytotoxicity of the compound. These results indicate that lecithin/terbinafine did not alter the biocompatibility of electrospun PCL mats. Morphological features of hDFs seeded on various mats were further visualized by confocal microscopy after immuno staining of the cytoskeletal components (α-tubilin and β-actin) and nuclei to confirm any alterations in cell morphology. Fluorescent confocal microscopic imaging and analysis showed no obvious abnormalities in the cellular or nuclear morphologies of dermal fibroblasts seeded on electrospun nanofibre mats in comparison to that observed in untreated cells seeded on coverslips (Fig. 5(c–e)). In contrast, cells treated with a toxic drug nocodazole (5 μg mL−1) as a control showed denatured cytoskeleton and reduced cell spreading (Fig. 5(f)). Together with the cell adhesion and MTS assay, these results demonstrated excellent biocompatibility of both PCL and lecithin/terbinafine loaded PCL mats.
Photoluminescent properties of drug-loaded PCL mats
As naphthalene-based dyes display fluorescence properties depending on the polarity and viscosity of the environment, we examined the photoluminescence properties of pristine PCL, PCL and lecithin/lecithin-loaded mats.40 Fig. 6(a–d) show the fluorescence images of nanofiber mats prepared under various conditions. Pristine PCL mats displayed weak blue fluorescence properties at an excitation wavelength of 360 nm. Incorporation of terbinafine or lecithin/terbinafine resulted in significant enhancement in the fluorescence intensity compared to pristine PCL mats. To confirm the results, we scanned the various areas of the mats (Fig. 6(a–d) insets) and estimated the average fluorescence intensity. When compared to pristine PCL mats, the average fluorescence intensity increased by about 2 fold and 3 fold for PCL_TERB and PCL_Lt_TERB mats, respectively, indicating that the presence of terbinafine confer enhanced fluorescence properties to PCL (Fig. 6(e)). Photoluminescence of lecithin and terbinafine loaded PCL nanofibre mats was examined as terbinafine contains naphthalene group and naphthalene-based dyes display fluorescence properties depending on the polarity and viscosity of the environment. Interestingly it was observed that pristine PCL mats displayed weak blue fluorescent properties when excited at 360 nm. A moderate increase in fluorescence intensity was observed for PCL_2%TERB mats. It was observed that addition of lecithin resulted in 3-fold increase in fluorescence intensity of the PCL_Lt_2%TERB mats when compared to pristine PCL mats. The changes in intrinsic fluorescence intensity of PCL_Lt_2%TERB mats could be used to track drug release at the infected site and also provide information on scaffold degradation.Antifungal susceptibility of the drug loaded nanofibre mats
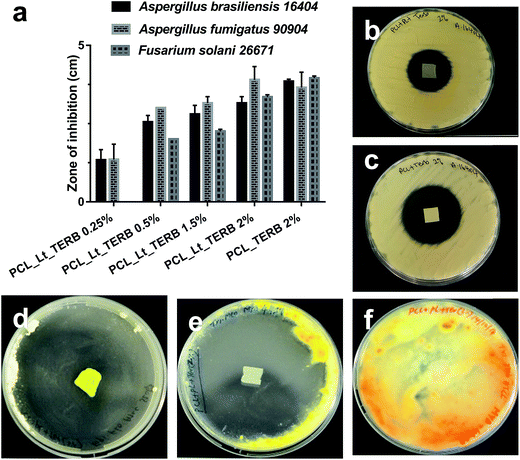
The antifungal activity of the drug-loaded nanofibre mats was evaluated against clinically relevant moulds by disc diffusion assay. An increase in the clear zone of inhibition was observed for the fungal species as the concentration of terbinafine-loaded mats was increased and maximum inhibitory activity was achieved when the final terbinafine concentration was 2% (w/w) (Fig. 7(a)). Table 1 summarizes the antifungal activity of electrospun PCL mats containing lecithin and terbinafine against various moulds. In addition, the PCL_Lt mats did not show any noticeable zone of inhibition which were prominent for the mats loaded with various concentrations of terbinafine (ESI Fig. S1†). However, against Aspergillus brasiliensis 16404 and Fusarium solani 26671 strains, statistically significant difference (p < 0.005) in the zone of inhibition was observed between PCL_Lt_2%TERB and PCL_2%TERB whereas no difference was observed against Aspergillus fumigatus 90904 strains (Fig. 7(b and c) and ESI Fig. S1†). It is likely that the interaction between terbinafine and lecithin may weaken the antifungal properties of the drug, thus decreasing the diameter of the zone of inhibition.| Zone of inhibition (cm) against | |||||
|---|---|---|---|---|---|
| Aspergillus brasiliensis ATCC 16404 | Aspergillus fumigatus ATCC 90904 | Fusarium solani 26671 | Epidermophyton flocossum ATCC 52063 | Trichophyton mentagrophytes ATCC MYA-4439 | |
| PCL_Lt | 0 | 0 | 0 | 0 | 0 |
| PCL_Lt_1%TERB | 2.8 ± 0.3 | 3.2 ± 0.2 | 2.3 ± 0.1 | 8 | 7.5 |
| PCL_Lt_2%TERB | 3.9 ± 0.1 | 3.7 ± 0.5 | 4 ± 0.1 | 8 | 7.4 ± 0.14 |
The antifungal properties of lecithin/terbinafine loaded mats were further examined against dermatophytic fungal pathogens, Epidermophyton floccosum ATCC 52063 and Trichophyton mentagrophytes ATCC MYA 4439 and Trichophyton rubrum MYA 4438 strains as well. The dermatophytic T. rubrum MYA 4438 strain was included as it showed resistant to terbinafine and served as the CLSI recommended quality control standard.41 The results indicated clear zone of inhibition against terbinafine-susceptible dermatophytes whereas no inhibitory zone was observed against the terbinafine-resistant T. rubrum strains (Fig. 7(d–f)). As was observed before for the filamentous fungus, PCL_Lt_2% TERB mats displayed clearer zone of inhibition than PCL_Lt_1%TERB (ESI Fig. S2†). The antifungal activity of terbinafine was very pronounced as shown by the radial disc diffusion assay against the moulds and the dermatophytic fungi. The lecithin did not play any role in the fungal growth inhibition as PCL_Lt mats did not display a clear zone of inhibition. The antifungal effect of 1 cm2 PCL_Lt_2%TERB mats was remarkable against the dermatophytic fungi that there was no detectable growth in the entire Petri dish, augmenting the superior activity of terbinafine against dermatophytic pathogens than against Aspergillus or Fusarium spp. In addition, to test the storage stability, we measured the antifungal activity of mats that were kept in a sealed aluminium foil at room temperature for 4 months. The mats displayed a clear zone of inhibition against E. flocossum and T. mentagrophytes, suggesting excellent storage stability and longer shelf life (data not shown).
Ex vivo porcine skin colonisation model of dermatophytes
To investigate the antifungal efficacy of terbinafine loaded PCL_Lt mats we have developed an ex vivo porcine skin colonisation model. In both the untreated control skin and PCL_Lt treated skins showed significant growth of T. mentagrophytes. However, no colonies could be detected from the skin that were exposed to PCL_Lt mats containing 1% or 2% terbinafine, confirming >5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log reduction in the fungal viability (Fig. 8). These results further support the complete inhibitory activity of the mats, thus reproducing the antifungal activity of the drug.
log reduction in the fungal viability (Fig. 8). These results further support the complete inhibitory activity of the mats, thus reproducing the antifungal activity of the drug.
Conclusion
In summary, electrospun PCL nanofibre mats were prepared containing terbinafine antifungal and lecithin as a surface modifier. Electrospun PCL mats containing lecithin/antifungal increased mechanical properties, wettability and photoluminescence without altering the cell adhesion and biocompatibility for primary human dermal fibroblasts. Antifungal studies demonstrated complete retention of activity against filamentous and dermatophytic fungi. We further demonstrated the efficacy of the drug-loaded fibre mats in an ex vivo porcine skin infection model. Together with the biocompatibility, these results indicate their potential applications in the treatment of superficial skin and nail fungal infections.Acknowledgements
The authors thank VAV Life sciences PVT Limited, Mumbai (India) for providing Lecithin. This research is supported by the Singapore National Research Foundation under its Translational and Clinical Research Flagship Programme (NMRC/TCR/008-SERI/2013) and administered by the Singapore Ministry of Health's National Medical Research Council and RL thanks the funding support from National Medical Research Council's Co-operative Basic Research Grant (NMRC/CBRG/0048/2013). NKV acknowledges funding support from the NTU Lee Kong Chian School of Medicine (L0412130 and L0412290) and the Ministry of Education Singapore AcRF-Tier I (2014-T1-001-141) grants.References
- G. D. Brown, D. W. Denning, N. A. Gow, S. M. Levitz, M. G. Netea and T. C. White, Sci. Transl. Med., 2012, 4, 165rv13 Search PubMed.
- B. Havlickova, V. A. Czaika and M. Friedrich, Mycoses, 2008, 51, 2–15 CrossRef PubMed.
- J. Thomas, G. A. Jacobson, C. K. Narkowicz, G. M. Peterson, H. Burnet and C. Sharpe, J. Clin. Pharm. Ther., 2010, 35, 497–519 CrossRef CAS PubMed.
- N. Singh, Clin. Infect. Dis., 2001, 33, 1692–1696 CrossRef CAS PubMed.
- I. P. Kaur and S. Kakkar, Expert Opin. Drug Delivery, 2010, 7, 1303–1327 CrossRef CAS PubMed.
- L. Ostrosky-Zeichner, A. Casadevall, J. N. Galgiani, F. C. Odds and J. H. Re, Nat. Rev. Drug Discovery, 2010, 9, 719–727 CrossRef CAS PubMed.
- R. Hart, E. M. Bell-Syer, F. Crawford, D. J. Torgerson, P. Young and I. Russell, Br. Med. J., 1999, 319, 79–82 CrossRef CAS PubMed.
- M. S. Erdal, A. Y. Peköz, B. Aksu and A. Araman, Pharm. Dev. Technol., 2014, 19, 565–570 CrossRef CAS PubMed.
- B. S. Barot, P. B. Parejiya, H. K. Patel, M. C. Gohel and P. K. Shelat, AAPS PharmSciTech, 2012, 13, 184–192 CrossRef CAS PubMed.
- N. Zalas, P. Suvanprakorn and P. Vayumhasuwan, US Pat., 0017234 A1, 2015.
- J. R. Baker, M. R. Flack, M. Ciotti and J. A. Sutcliff, WO Pat., 132342, 2009.
- G. Cevc and U. Vierl, US Pat., 7820720 B2, 2010.
- Y. F. Goh, I. Shakir and R. Hussain, J. Mater. Sci., 2013, 48, 3027–3054 CrossRef CAS.
- S. Y. Chew, J. Wen, E. Yim and K. Leong, Biomacromolecules, 2005, 6, 2017–2024 CrossRef CAS PubMed.
- Z. M. Huang, C. L. He, A. Yang, X. J. Han, J. Yin and Q. Wu, J. Biomed. Mater. Res., Part A, 2006, 77, 169–179 CrossRef PubMed.
- K. Kim, Y. K. Luu, C. Chang, D. Fang, B. S. Hsiao, B. Chu and M. Hadjiargyrou, J. Controlled Release, 2004, 98, 47–56 CrossRef CAS PubMed.
- J. Zeng, X. Xu, X. Chen, Q. Leng, X. Bian, L. Yang and X. Jing, J. Controlled Release, 2003, 92, 227–231 CrossRef CAS PubMed.
- A. J. Meinel, O. Germershaus, T. Luhmann, H. P. Merkle and L. Meinel, Eur. J. Pharm. Biopharm., 2012, 81, 1–13 CrossRef CAS PubMed.
- M. R. Singh, K. Pradhan and D. Singh, Curr. Drug Delivery, 2012, 9, 243–254 CrossRef CAS.
- C. Kriegel, K. M. Kit, D. J. McClements and J. Weiss, Langmuir, 2009, 25, 1154–1161 CrossRef CAS PubMed.
- A. Arecchi, S. Mannino and J. Weiss, J. Food Sci., 2010, 75, N80–N88 CrossRef CAS PubMed.
- T. Briggs and T. L. Arinzeh, J. Biomed. Mater. Res., Part A, 2014, 102, 674–684 CrossRef PubMed.
- A. Camerlo, C. Vebert-Nardin, R. M. Rossi and A. M. Popa, Eur. Polym. J., 2013, 49, 3806–3813 CrossRef CAS.
- M. Zhang, K. Wang, Z. Wang, B. Xing, Q. Zhao and D. Kong, J. Mater. Sci.: Mater. Med., 2012, 23, 2639–2648 CrossRef CAS PubMed.
- J. Venugopal, Y. Z. Zhang and S. Ramakrishna, Nanotechnology, 2005, 16, 2138–2142 CrossRef CAS PubMed.
- R. Goyal, L. K. Macri, H. M. Kaplan and J. Kohn, J. Controlled Release, 2016 DOI:10.1016/j.jconrel.2015.10.049.
- T. K. Dash and V. B. Konkimalla, J. Controlled Release, 2012, 158, 15–33 CrossRef CAS PubMed.
- N. K. Verma, J. Conroy, P. E. Lyons, J. Coleman, M. P. O'Sullivan, H. Kornfeld, D. Kelleher and Y. Volkov, Toxicol. Appl. Pharmacol., 2012, 264, 451–461 CrossRef CAS PubMed.
- D. H. Reneker, W. Kataphinan, A. Theron, E. Zussman and A. L. Yarin, Polymer, 2002, 43, 6785–6794 CrossRef CAS.
- R. Nirmala, H. M. Park, R. Navamathavan, H. S. Kang, M. El-Newehy and H. Y. Kim, Mater. Sci. Eng., C, 2011, 31, 486–493 CrossRef CAS.
- Z. Li and C. Wang, One-Dimensional nanostructures: Electrospinning Technique and Unique Nanofibers, SpringerBriefs in Materials, Berlin, Heidelberg, 2013, Effects of working parameters on electrospinning, 15–29.
- X. Fu, L. Ou, M. Zhang, Y. Guan, K. Wang, Y. Che, D. Kong, G. Steinhof, W. Li, Y. Yu and N. Ma, Int. J. Artif. Organs, 2010, 33, 161–170 CAS.
- S. C. Wong, A. Baji and S. Leng, Polymer, 2008, 49, 4713–4722 CrossRef CAS.
- M. Eastwood, R. Porter, U. Khan, G. McGrouther and R. Brown, J. Cell. Physiol., 1996, 166, 33–42 CrossRef CAS PubMed.
- M. Eastwood, D. McGrouther and R. Brown, Proc. Inst. Mech. Eng., Part H, 1998, 212, 85–92 CrossRef CAS.
- J. M. Zaleskas, B. Kinner, T. M. Freyman, I. V. Yannas, L. J. Gibson and M. Spector, Biomaterials, 2004, 25, 1299–1308 CrossRef CAS PubMed.
- Y. Wang, F. Z. Cui, Y. P. Jiao, K. Hu and D. D. Fan, Biomed. Mater. (Bristol, U. K.), 2008, 3, 15012 CrossRef CAS PubMed.
- N. Zhu, F. Z. Cui, K. Hu and L. Zhu, J. Biomed. Mater. Res., Part A, 2007, 82, 455–461 CrossRef CAS PubMed.
- D. Yu, C. B. White, G. R. Williams, S. W. Annie Bligh, K. White, L. Zhu and N. P. Chatterton, Soft Matter, 2011, 7, 8239–8247 RSC.
- L. A. Munishkina and A. L. Fink, Biochim. Biophys. Acta, 2007, 1768, 1862–1885 CrossRef CAS PubMed.
- E. I. Nweze, P. K. Mukherjee and M. A. Ghannoum, J. Clin. Microbiol., 2010, 48, 3750–3752 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra04755f |
| ‡ Contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2016 |