Corrosion management of carbon steel material: operational modes influence corrosion rate – an in vitro study
P. Balamurugana,
P. Chandramohanb and
T. S. Rao*b
aDepartment of Biotechnology, Pondicherry University, Puducherry-605 014, India
bWater and Steam Chemistry Division, BARC, Kalpakkam-603 102, India. E-mail: subbarao@igcar.gov.in
First published on 20th April 2016
Abstract
This work describes the corrosion management of carbon steel structures of a firewater distribution system under laboratory conditions. The influence of operational modes i.e. regular replacement and extended stagnant condition of firewater were tested. Corrosion rate at different experimental conditions was determined and the corrosive bacteria were enumerated using conventional microbial culture methods. Denaturing gradient gel electrophoresis showed a significant variation in bacterial diversity under the different test conditions. Raman spectroscopy identified iron oxide phases like lepidocrocite, goethite, hematite, magnetite and the underlying corrosion mechanism was described. Based on the results obtained a plausible carbon steel corrosion management was explained.
1. Introduction
Industrial water distribution pipeline networks are usually made of carbon steel (CS). Among the various industrial distribution systems, firewater system (FWS) which is a dead end system is vital for industrial safety. A proper FWS in an industry should supply the required water in critical times without any failure. Nevertheless, CS being the structural material of FWS, it is more likely to get corroded. Corrosion of CS by microorganisms in various systems have been investigated by many research groups.1–4 However, there are only a couple of published reports on the microbially induced corrosion (MIC) failures of CS in FWS. A study report published in 2007 described the deterioration of firewater pipelines due to MIC, wherein the authors have detected sulphate reducing bacteria (SRB) and iron bacteria in the firewater that were responsible for the biogenic corrosion.5 Likewise, another case study on the FWS failure was reported from a Danish stainless steel production plant and Finnish power plant, where MIC of iron pipes was noticed in both the units.6Corrosion of CS in stagnant water condition, as the case seen in FWS, is a major concern in industrial safety. Firewater distribution systems are pipeline networks with hydrants at necessary places that ensure the safety of industrial units, big hospitals, large academic and research institutions. FWS is generally maintained in stagnant condition with pressures of 1–1.5 kg cm−2. In case of fire fighting, high pressures of 10–12 kg cm−2 are employed to spray water at optimal force. However, in case of unanticipated failures due to corrosion of the pipelines, the vital pressure required to pump the water would not be available in the system and the outcome could be disastrous in critical fire scenarios. Some of the common failures could be due to mechanical faults, chocking by biogenic growth and corrosion products in the valves of pipelines and pipe punctures due to localized corrosion. Hence, proper maintenance of the FWS is essential to avoid such unanticipated failures during serious fire accidents that could cause damage to industrial equipments and loss of life.
Microorganisms are omnipresent and are reported in a variety of iron corrosion failures.7–10 In FWS, since the water is stagnant most of the time it encourages anaerobic bacteria growth resulting in localized corrosion that leads to the deposition of organic debris along with corrosion products. Microbes adopt a biofilm mode of growth on these metal surfaces and this sessile growth with exopolymer matrix, assist in the transport of nutrients from the bulk water that supports the microbes in deriving their energy. Microorganisms that promote corrosion generally include not only bacteria but also algae and fungi. However, the damage caused by bacteria is more pronounced and more commonly six categories of corrosion causing bacteria are well recognized; iron oxidizing bacteria (IOB), iron reducing bacteria (IRB), sulphate reducing bacteria (SRB), sulphur oxidizing bacteria (SOB), nitrate reducing bacteria (NRB) and the slime producing bacteria. Of these, IOB and SRB are typical examples of the aerobic and anaerobic synergistic interaction which is more common in microbial corrosion of iron and steel alloys.2 The primary effect by these microbes would be the formation of differential ion concentration cells on the metal surfaces. Thus, a respiring microbial colony removes the oxidized metal (at anodic sites) and the processes which utilize hydrogen (at cathodic sites) will cause electrochemical corrosion reactions resulting in metal dissolution.
The present study was conceived and carried out after observing a few failures of FWS at Kalpakkam nuclear centre site, India. A systematic study was conducted to investigate the underlying problem in controlled laboratory conditions. In the methodological study, the corrosion pattern of CS in the actually practiced condition of FWS (regular replacement i.e. firewater replacement at every 30 days) was recorded in the light of microbial corrosion. Additionally, an experimental condition (extended stagnant i.e. firewater unchanged for a period of 120 days) was also evaluated as a step towards corrosion management and thus extending the life of FWS pipelines.
2. Experimental
2.1 Water analysis
The firewater system includes the open reservoir, sump and pipelines. Water samples were collected from all the locations of FWS and analysed for pH, temperature, hardness, micro-nutrients and a few metal ions. The physicochemical parameters were analysed according to the standard procedures as described in “Standard methods for the examination of water and wastewater”.112.2 Experimental conditions and samples
Carbon steel coupons (length × width × thickness = 76 mm × 12 mm × 1 mm) were polished to 600 grit silicon carbide metallurgical paper. The pre-weighed and pre-sterilized (with alcohol) coupons were immersed in raw firewater collected from the open reservoir of FWS. Two experimental conditions were chosen for the investigation. In the first condition, coupons were exposed to firewater with regular changing at every 30 days (actually practiced procedure in the FWS of nuclear centre site) for a period of 120 days. In the second condition, water was not replaced but stored up to 120 days (experimental condition). The experimental setup is a closed unit and maintained airtight in dark condition at room temperature (25 °C) and hence no significant evaporation was noticed. The exposed coupons were retrieved after 7, 15, 30, 60, 90 and 120 days. One set of coupons was used for corrosion deposit analysis and another set of coupons for microbiological analysis. Filter sterilized (membrane size 0.22 μm, Millipore) firewater was used as a control set, to compare with that of microbial corrosion. Throughout the sampling period, the changes in pH and dissolved oxygen were measured. To have a diversity profile of bacterial biofilm development at the initial stages, coupon samples of 1, 2, 3, 4, 5, and 6 days were taken from an experimental setup. The biofilm developed on these coupons was extracted for whole genomic DNA and used for denaturing gradient gel electrophoresis (DGGE) analysis.2.3 Weight loss and corrosion rate determination
The weight loss and corrosion rate of the coupons was measured by standard ASTM procedures for corrosion studies. The metal coupon preparation, cleaning, and corrosion test specimen weight loss measurement were made as per the standard methods described in ASTM G1-03 and corrosion rate using ASTM D2688-15.12,132.4 Corrosion product analysis by confocal Raman spectroscopy
Raman spectra were recorded with the powdered corrosion product samples using LAB HR-800 spectrometer (JobinYvon, France). The spectrometer was operated with 50× long working objective lens with 1800 grooves per mm grating. Ar+ ion laser with 514.54 nm wavelength was used for the analysis. Laser power for Raman signal was optimized based on S/N ratio and sample degradation. A Peltier cooled CCD was used for the Raman signal detection. A multiple point Raman spectra were acquired to see the nature of homogeneity in the samples. Raman spectra covering a spectral range of 100–2000 cm−1 with a resolution of <1 cm−1 were recorded to interpret the iron oxide phases or the corrosion products.2.5 Microbiological analysis
Retrieved coupons for microbiological analysis were handled in aseptic conditions and the biofilm was scrapped with the help of a sterile nylon brush in 50 ml of 1× PBS buffer. The samples were serially diluted and plated on Luria Bertani agar, iron bacteria agar, and modified iron sulphite agar media to enumerate the total viable counts of the heterotrophic bacteria, iron bacteria and sulphidogenic bacteria respectively.2.6 Biofilm diversity by DGGE
The biofilm samples collected in 50 ml of 1× PBS were filtered on to 0.22 μm filter paper and the filter discs were cut into pieces by a sterile scalpel. The pieces were suspended in 200 μl of the lysozyme (lysis buffer) solution. DNA extraction was carried out using the QIAamp DNA mini kit (Invitrogen) according to the manufacturer's protocol. The extracted DNA was analysed electrophoretically on a 0.8% agarose gel. DGGE specific PCR and DGGE analysis were performed as described earlier.142.7 Statistical analysis
All the experiments were carried out in triplicates and the values were expressed as mean ± SD. SPSS 20.0 statistics package (IBM SPSS Statistics software is a comprehensive, predictive analytical tool for analysts and statistical programmers) was used for the analysis. The effect of different experimental conditions on the corrosion rate was analysed statistically by the independent t-test and was considered significant only if the P value was ≤0.05.3. Results and discussion
3.1 Water analysis
The results of various physicochemical parameters such as pH, temperature, hardness, micronutrient levels, metal ion analysis of the firewater was presented in Table 1. Significant to moderate fluctuation in the values of the analyzed parameters were observed. The dissolved solids, chloride and sulphate content of the water were high in all the samples of open reservoir, sump, and pipelines. The pH ranged from 6.8 to 7.6 in the samples collected from different locations. The dissolved oxygen levels were higher in the open reservoir and sump, while it was low in the pipeline samples.| Parameters | Unit | Open reservoir (Min–Max) | Sump (Min–Max) | Pipelines (Min–Max) |
|---|---|---|---|---|
| a (Min–Max = minimum–maximum). | ||||
| Temperature | °C | 24.3–34 | 25–28.2 | 23–31 |
| Conductivity | μS cm−1 | 216–326 | 266–306 | 265–317 |
| Total dissolved solids | mg l−1 | 170–324 | 178–254 | 160–301 |
| Total suspended solids | mg l−1 | 4–14 | 5–9 | 3–10 |
| Dissolved oxygen | mg l−1 | 6.2–7.6 | 5.7–7 | 3–4.7 |
| pH | — | 7.1–7.4 | 7–7.8 | 6.8–7.5 |
| Total organic carbon | mg l−1 | 14–90 | 8–102 | 5–86 |
| Chloride | mg l−1 | 55–75 | 55–75 | 40–70 |
| Sulphate | mg l−1 | 19–109 | 10–98 | 22–89 |
| Calcium | mg l−1 | 40–70 | 40–80 | 20–80 |
| Magnesium | mg l−1 | 30–60 | 36–60 | 0–60 |
| Carbonate | mg l−1 | 40–60 | 40–50 | 20–50 |
| Bicarbonate | mg l−1 | 20–40 | 20–40 | 20–60 |
| Alkalinity | mg l−1 | 60–100 | 60–80 | 40–110 |
| Hardness | mg l−1 | 76–118 | 76–128 | 40–130 |
| Nitrate | μg l−1 | 8.2–28.2 | 9.2–14.8 | 4.1–17 |
| Nitrite | μg l−1 | 0.06–0.71 | 0.02–0.6 | 0.004–0.43 |
| Iron, Fe2+ | μg l−1 | 12.2–33.5 | 16.2–45.3 | 58.4–97.6 |
| Phosphate | μg l−1 | 39.8–136 | 29.3–125 | 25–112 |
| Ammonia | μg l−1 | 4.4–21.6 | 7.2–14.5 | 4–91.5 |
Quality of water is a major concern as it is directly linked to human health15 as well as the performance of distribution system material.3,5,10 Thus, the quality of the water used for an industrial unit is an important criterion when fouling and corrosion free operation are the primary goals. Normally, the water used in an industrial system can be either corrosive to the structural material or scale forming. This mostly depends on the physicochemical parameters like temperature, pH, total dissolved solids, calcium hardness and alkalinity. In addition, corrosion of materials is enhanced by the interactions of biotic and abiotic components.16 The abiotic components are the physicochemical parameters present in the surrounding milieu that normally leads to generalized corrosion, which is also referred to as uniform corrosion that occurs as a result of the aggressive ions in water. The biotic components are the living microbes that can lead to localized corrosion, which usually occurs with the development of ion concentrated corrosion cells due to microbial growth and its metabolism.2,14
The results of water analysis revealed that the water used in FWS is highly conducive for microbial growth and metabolism. The near neutral pH range i.e. 6.8 to 7.8 and water temperature of 24 to 30 °C were optimal for most of the mesophilic microbes. In addition, the firewater had an adequate supply of nutrients like nitrate, phosphate and iron that are very essential for microbial life. Levels of dissolved oxygen determine the type of bacteria in the system. In the present study, the dissolved oxygen levels were high in the open reservoir and sump, whereas in the pipelines it was less. The reduced dissolved oxygen levels observed could be due to the limitation of air diffusion as the system is a closed pipeline network and also due to respiration of the bacterial population. It has been previously reported that within 2 h of water stagnation microbial activity can deplete oxygen up to 90%.17 This reduced dissolved oxygen level would provide a favourable condition for anaerobic bacterial growth and reduction reactions. The microbiological analysis also confirmed the presence of significant anaerobic bacteria counts in the pipelines such as SRB and iron reducing bacteria that reduces the sulphate and ferric ions respectively.
The dissolved solids in the firewater were found to be high which can increase the electrical conductivity of water. Indirectly, it can also increase the corrosion current at the metal/water interface resulting in significant material damage. High levels of calcium carbonate in water tend to form, scale and decrease the corrosion rate. However, the presence of chlorate and sulphate ions in the water tends to keep the calcium in solution and prevent scale formation. In this study, the levels of chloride and sulphate present in the firewater were relatively high. Fairly high chloride content in water can induce pitting corrosion of CS5 and sulphates are directly related to MIC problems where it acts as an important electron acceptor for SRB.
3.2 Weight loss and corrosion rate determination
In the first experimental condition, where firewater was changed every 30 days, a metal loss of 0.7–21 mg cm−2 was observed and in the second condition, where water was not changed the metal loss was 0.5–13 mg cm−2. Hence, the metal loss was more in case of water changed set than the water unchanged set (Fig. 1A). The corrosion rate calculated by the weight loss method showed a comparative reduction in the water unchanged condition (∼1.6 mpy in sterile firewater; ∼2.7 mpy in raw firewater) than the water changed condition (∼2.8 mpy in sterile firewater; ∼3.7 mpy in raw firewater). This reduction in corrosion rate (Fig. 1B) was statistically significant by independent t-test (P ≤ 0.001). | ||
| Fig. 1 Weight loss (A) and corrosion rate (B) of carbon steel exposed to firewater changed and unchanged conditions. | ||
Pure cultures were often used to elucidate the mechanism of bacterial adhesion, biofilm formation and corrosion phenomena. However, this will not exactly reflect the complex and constantly changing environment of feed water in industrial systems.18 Simulation studies of large distribution systems had been carried out previously to elucidate the effect of biofilm on cast iron pipe of the drinking water distribution system10 and in situ corrosion control studies on industrial water systems.19
In this study, corrosion of FWS material, CS by raw firewater and the role of microbes in corrosion enhancement was elucidated by simulation of the FWS conditions in the laboratory. In addition, an experimental condition was tested as a step towards corrosion management as detailed in the methodology. In the actual FWS, the water will be maintained in a stagnant condition for a period of 30 days. After 30 days, the water will be drained in storm drains and replenished with new water whereas in the second set of experimental test condition (this study) the water was made to stagnate for 120 days without changing. Irrespective of the presence or absence of bacteria, the weight loss and corrosion rate of CS was more in the actual practiced condition wherein water had been changed while the corrosion was significantly less (P ≤ 0.001) in water unchanged condition. Total weight loss of the metal is an indicator of generalized corrosion and the results of this study showed that corrosion was more in the actual practiced condition.
The relatively high corrosion rate in the water changed condition could be due to the oxidative corrosion factors like high dissolved oxygen, fresh unsaturated water with aggressive chemical ions, oxidation of reduced chemical species such as sulphide to thiosulphate, increased metabolism of heterotrophic aerobic bacteria, revival of the dormant microbes present in the biofilm, disruption of the protective oxide layer on the metal surface and so on. On the other hand, the reduced corrosion rate in the water unchanged experimental condition is an advantage. However, making the untreated raw firewater to stagnate for a long time also moderately increased the corrosion rate (from 1.7 to 2.0 mpy after 60 days) [Fig. 1B]. This could be due to the metabolism of the anaerobic bacteria, as we observed increased sulphidogenic bacteria counts in the water unchanged experimental condition (Table 3). The reduced corrosion rate observed in sterile unchanged firewater (Fig. 1B) supports the above fact, where the corrosion was due to the presence of bacteria. This reduced corrosion rates observation in sterile unchanged firewater is noteworthy for recommendation to the FWS where water can be stored for longer periods provided the water used is free from bacteria.
3.3 Dissolved oxygen and pH of the bulk water
The dissolved oxygen measured in the bulk phase of the water at the start of the experiment was 4.5 ppm. Subsequently, it reduced to 1.4 ppm and remained stable. In the case of water unchanged sets, the dissolved oxygen after 60th and 120th day was ∼1 ppm. A separate set of samples were analyzed for day one to six that showed dissolved oxygen level of 1.8 ppm. The pH at the start of the experiment was 7.6 which showed a marginal reduction over time, which was 0.3–0.5 units and the final pH at the end of 120 days was near neutral (Table 2).| Days | Dissolved oxygen (ppm) | pH | ||
|---|---|---|---|---|
| Water | Water | |||
| Changed | Not changed | Changed | Not changed | |
| a Dissolved oxygen and pH levels at the time of water replacement. | ||||
| 0 | 4.5 | 4.2 | 7.6 | 7.5 |
| 7 | 1.6 | 1.7 | 7.1 | 7.2 |
| 15 | 1.7 | 1.6 | 7.2 | 7.1 |
| 30 | 1.8 (4.9a) | 1.6 | 7.2 (7.4a) | 7.0 |
| 60 | 1.6 (5.1a) | 0.9 | 7.1 (7.7a) | 6.7 |
| 90 | 1.8 (3.9a) | 1 | 7.1 (7.4a) | 7.1 |
| 120 | 1.5 | 0.9 | 7.0 | 7.0 |
3.4 Corrosion product analysis by confocal Raman spectroscopy
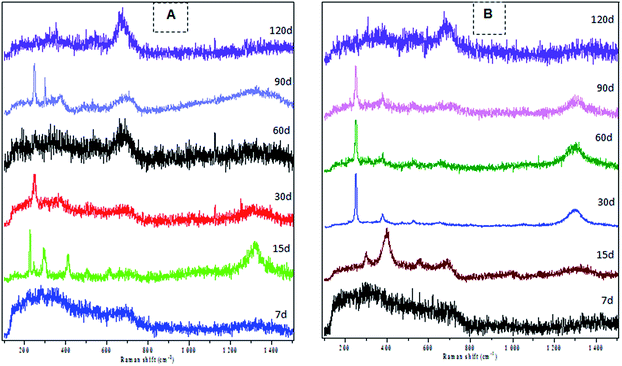
Raman spectroscopic analysis of the deposits on the carbon steel revealed, major phases corresponding to lepidocrocite, goethite, hematite and magnetite type iron oxide. Different types of iron oxide phases presence on the CS material was observed, when exposed to FWS water as a function of exposure time. A very thin oxide film was visually observed on the surface of CS after 7 days of exposure whereas in Raman spectroscopic analysis we could not detect any iron oxide phase on CS surface. However, on long exposure upto 15 days, corrosion products from the sample surface showed Raman peaks (vibrational modes) at 308 and 407 cm−1 suggesting lepidocrocite and goethite type of iron oxides. On further exposure of carbon steel specimens, hematite iron phase was observed as indicated by the Raman shifts at 250.2, 377.4, 526, 651, 1317 cm−1. This oxide was further transformed to magnetite phase, marked by its Raman shift at ∼676.6 cm−1 when exposed further. Generally, dissolved oxygen presence in the water plays an important role during the conversion of iron hydroxides to oxide phase or oxide formation. A transformation from hematite to magnetite phase occurred after 30 days of exposure, however, when the water was replaced, formation of the hematite phase is facilitated (Fig. 2A). Whereas, in the case of water unchanged condition magnetite phase domination was recorded with time (Fig. 2B).The chemical characterization of the oxide layer formed on metals and its mechanism of formation are important factors for the development and improvement in existing methodologies for materials resistance against corrosion.20 Raman spectroscopy was used as a tool to determine the type of iron oxides formed on the CS surface, which in turn reveals the type of corrosion process. All specimens analyzed by Raman spectroscopy showed regions of three different colours yellow, reddish-brown and black. The main phases reported in iron corrosion are lepidocrocite (γ-FeO(OH)), goethite (α-FeO(OH)), hematite (α-Fe2O3) and magnetite (Fe3O4).21 Lepidocrocite was usually formed in the early stages of corrosion phenomena, but as the exposure time increased it was transformed into goethite. Generally, when iron is exposed to water or atmospheric oxygen it forms lepidocrocite, siderite, goethite, and hematite type of iron oxides. These oxides further transform to maghemite and magnetite under the low dissolved oxygen environment which are stable and offer a protective oxide layer to the alloy surface.22 In the present study, the coupons that were exposed to water changed condition, showed a transition of hematite (red oxide) to magnetite phase (black oxide). However, the magnetite phase formed was disturbed by the replacement of new water at 30th day that contained increased dissolved oxygen level (Table 2). The higher dissolved oxygen level has favoured oxidative corrosion as reflected in the dominant presence of the red oxide once again (Fig. 2A). Unsaturated water tends to dissolve metal from the surface to establish saturation with respect to iron and microbes. This could be either accelerating or inhibiting the localized corrosion of metals through alteration of the protective layer formed on the surface.23 In our present study, coupons that were exposed in water unchanged conditions had a dominant magnetite phase (Fig. 2B). This result suggests that minimizing the frequent change of water creates low oxygen environment and result in the formation of a more protective iron oxide (magnetite) layer.
3.5 Microbiological analysis and biofilm diversity by DGGE
Total viable count enumerated in biofilm scrapings showed heterotrophic bacteria in the order of 102 to 106 cfu cm−2, iron bacteria were 102 to 105 cfu cm−2 and the maximum sulphidogenic bacteria count was 3.0 × 103 cfu cm−2. The heterotrophic bacterial count was relatively less in the case of water unchanged samples than the water changed samples (Table 3).| Days | Heterotrophic bacteria (cfu cm−2) | Iron bacteria (cfu cm−2) | Sulphidogenic bacteria (cfu cm−2) | |||
|---|---|---|---|---|---|---|
| A | B | A | B | A | B | |
| a A – water changed; B – water unchanged. | ||||||
| 7 | 8.7 × 105 | 2.7 × 105 | 9.0 × 105 | 4.4 × 105 | 2.2 × 102 | 1.2 × 102 |
| 15 | 1.7 × 106 | 1.4 × 105 | 1.4 × 105 | 2.4 × 105 | 2.8 × 102 | 2.1 × 102 |
| 30 | 5.0 × 105 | 5.2 × 104 | 3.0 × 103 | 1.8 × 103 | 1.2 × 102 | 1.5 × 102 |
| 60 | 3.3 × 106 | 2.1 × 104 | 1.0 × 104 | 1.1 × 102 | 3.0 × 102 | 1.0 × 102 |
| 90 | 1.2 × 105 | 1.1 × 103 | 2.0 × 102 | 1.2 × 102 | 6.2 × 101 | 2.2 × 103 |
| 120 | 2.6 × 105 | 1.1 × 102 | 3.7 × 103 | 2.2 × 102 | 3.8 × 102 | 3.0 × 103 |
Denaturing gradient gel electrophoresis of the scrapped biofilm samples showed a diversified profile of bacterial community (Fig. 3). In the initial development of the biofilm community, the banding pattern was identical in day 1 and day 2 samples. Similarly, banding patterns of day 3, 4 and 5 samples were relatively identical (Fig. 3A). Species richness was more in 7, 15 and 30 days as noticed by the number of bands (Fig. 3B). On the water unchanged set of experiments (Fig. 3C) i.e., after 30 days, there was a decrease in the species diversity and subsequently, it was observed that there was domination by one or two types of bacteria in the 60th, 90th and 120th day samples. In the case of biofilm samples wherein firewater was replaced every 30 days, the number of bands observed were more in the case of 60, 90 and 120 days indicating the richness in bacterial diversity (Fig. 3D).
Heterotrophic aerobic bacteria utilize organic carbon and fresh nutrients available in water for the growth and establishment of bacterial communities. This was apparent from the DGGE fingerprints (Fig. 3A and B), where significant establishment of a bacterial community was observed. Presence of reductive bacterial species such as iron reducing and sulphidogenic bacteria in bulk water explains the tolerance and adaptation of these bacteria to the system condition where high dissolved oxygen conditions transiently exist. Moreover, the dominant presence of a few bands (Fig. 3C) could be from anaerobic bacteria species (Table 3). The increase in the bacterial count in biofilm samples and the diverse profiles of DGGE shows that the microbial community is replenished with the addition of new bacterial load and fresh nutrients (Table 3 and Fig. 3D).
3.6 Corrosion mechanism and its management
The results of our experimental test condition i.e. water unchanged for an extended period at an average temperature of ∼27 °C, demonstrated the transformation of hematite phase to magnetite. This transformation occurred without significant involvement of molecular H2 or any hydrothermal conditions as suggested in the literature.24 Generally, hematite (α-Fe2O3) and magnetite (Fe3O4) are the two most common iron oxides in near-surface environments. Magnetite is stable under more reducing conditions than hematite. Thus, in water unchanged experimental condition, magnetite phase was recorded after 30 days, while in sterile water we have not seen such phase formation. Thus, we attribute a plausible role of bacteria mediated reductive conditions in the formation of magnetite. According to Otake et al.24 transformation between magnetite and hematite has been considered as a redox reaction involving an oxidant (e.g., O2, SO42−) or a reductant (e.g., H2, organic compounds). In the present study, Raman spectroscopy results provide proof for an independent mechanism for iron oxide transformation in industrial systems, where the bacterial population has a significant role in the transformation process. However, whether this mechanism is common or dominant in such systems has to be determined. Yet, we attribute to the consortium of microbes present in the FWS for the inter-conversion of the oxides with a prominent role of oxygen in the experimental set where water was replaced sequentially. Whenever water was changed the magnetite film that has formed with time was transformed to hematite due to dissolved oxygen. There was also a view among investigators that transformation of magnetite to hematite requires molecular O2. In a similar context the presence of magnetite and/or hematite in rocks has been linked to a specific oxidation environment. However, when the availability of reductants or oxidants was low to account in many geologic and industrial environments, the transformations of iron oxides through redox reactions can be impaired.25Bacterial cells usually have a net negative charge due to the presence of surface associated acidic groups; this makes them react with positively charged metal species.26,27 An earlier study has reported that cells can act as templates for the nucleation of metals and there is evidence that minerals in natural environments nucleate on cells.28 Microbial Fe(III) reduction is commonly referred to as dissimilatory iron reduction and in this process microorganisms transfer electrons to external Fe(III), reducing it to Fe(II) for a purpose other than assimilation of iron into the biomass.29 The known Fe(III) reducing micro-organisms belong to different phylogenetic groups, suggesting that the ability to reduce Fe(III) is spread throughout the microbial domain.30 One of the strategy that bacteria adapt is by solubilisation of the solid phase iron by high-affinity Fe(III) chelators, the so-called siderophores. The siderophores are water-soluble, low molecular weight, Fe(III)-specific ligands which are produced at low iron concentrations.31 Siderophores facilitate solubilisation of Fe(III) making it more accessible to the microorganisms and enabling transport into or close to the cell. Pseudomonas group of bacteria are well known for the production of siderophores which is also called iron reducing bacteria. In the various bacterial types characterized in the FWS, Pseudomonas sp. was one of the dominant bacteria in the water distribution systems.32 It can also be assumed that the FWS biofilm bacteria had an effective electron shuttle that can accept electrons from iron reducing bacteria and then transfer the electrons to the Fe(III) oxide surface. This mode can regenerate the electron shuttle in the oxidized form. In this manner, small amounts of an electron shuttle can act as catalyst and undergo multiple reduction–oxidation cycles.33 Most recently, it has been proposed that some microorganisms may transfer electrons from the cell surface to the surface of Fe(III) oxides through biological nanowires.34 Based on the observations of the present study it can be surmised that: (1) the bacterial consortium of the FWS has the ability to reduce hydrous ferric oxides, goethite, and hematite to magnetite under slow growth conditions i.e., it took more than 30 days to observe the magnetite peak in the Raman analysis. On the contrary, in the water changed condition the magnetite phase was disturbed on new water addition. (2) The water changed condition could have added more dissolved oxygen to the system, thereby resulting in the oxidation of magnetite phase to hematite phase. It may be noted that dissolved oxygen levels were less than 1.6 ppm within 7 days of stagnation and remained so till 30 days, thereafter it was ∼1.0 ppm. This attributes that dissolved oxygen is playing a role in the transformation of the iron oxides.
A schematic illustration, Fig. 4 was prepared based on the observations of CS biofouling and corrosion. The illustration shows the microbial fouling (i.e., biofilm formation) and the formation of iron oxide phases with time scale.
It can be interpreted that under an extended period of firewater stagnation/no frequent change of firewater, the protective iron oxide magnetite formation is microbially mediated. However, the biologically generated magnetite film tenacity needs to be tested. Based on further experimental data the magnetite formation was confirmed due to bacterial action, since magnetite film was not perceived on the CS coupons exposed in sterilized firewater. Nevertheless, further research is needed (1) to identify the specific signatures of the biofilm bacteria in initiating the iron oxide conversion. (2) Iron isotope ratios can be used to distinguish the iron oxides. (3) Further characterization of the corrosion deposits using surface analytical techniques can aid in elucidating the precise mechanism of iron oxide formation and transformation.
In other chemical processes magnetite is transformed into hematite through progressive oxidation of ferrous iron to ferric state. In a mineralogenesis process, markedly different from that observed in other systems, where the transformation of the Fe3O4 into Fe2O3 occurs via maghemite formation. Hematite is the most stable compound among the iron oxides and all the other iron oxides get converted to Fe2O3 easily. As for our case, it is quite possible that the mixture contains iron oxide with +3 state, and by heating it gets converted to Fe3O4. It would be premature to confirm it. Thus conversion from Fe3O4 to Fe2O3 is possible without reducing agents. The process is called oxygen dissociation and can occur at temperatures > 800 °C, however, the exact temperature varies due to composition and oxygen partial pressure. A quick hysteresis loop and back field curve analysis should give a quick indication of magnetite/maghemite (soft) versus hematite (hard) or a mixture of both.35
4. Conclusions
Physicochemical analysis of the freshwater used for the firewater system showed the presence of aggressive ions as well as corrosive bacteria in the system. The mode of operational conditions altered the corrosion rate of carbon steel corrosion, which has been well illustrated by the results obtained in the study. Significantly, less corrosion rate observed in the experimental condition (without changing the water) suggests that the firewater can be stored for longer periods for better corrosion management. In addition, usage of corrosive bacteria free fire water would be ideal to store water for longer periods as well to control corrosion. The MIC in the system can be minimized by controlling the fouling bacteria in the system by the addition of biocides.Acknowledgements
P. B. acknowledges the support of the Atomic Energy Regulatory Board, Mumbai, to carry out this study. P. B. was a recipient of Senior Research Fellowship from the Council of Scientific and Industrial Research, New Delhi and the financial assistance is duly acknowledged.References
- T. S. Rao, T. N. Sairam, B. Viswanathan and K. V. K. Nair, Corros. Sci., 2000, 42, 1417 CrossRef CAS.
- T. S. Rao, J. K. Aruna, P. Chandramohan, B. S. Panigrahi and S. V. Narasimhan, Biofouling, 2009, 25, 581 CrossRef CAS PubMed.
- M. T. S. Lutterbach and F. P. Franca, World J. Microbiol. Biotechnol., 1996, 12, 391 CrossRef CAS PubMed.
- S. Rajagopal, N. Sasikumar, J. Azariah and K. V. K. Nair, Biofouling, 1991, 3, 311 CrossRef.
- J. Starosvetsky, D. Starosvetsky and R. Armon, Eng. Failure Anal., 2007, 14, 1500 CrossRef CAS.
- L. Carpén and T. Ohligschläger, A case study on corrosion of stainless steel in firewater mains, 6th European stainless steel conference, Finland, 2008 Search PubMed.
- P. Angell and K. Urbanic, Corros. Sci., 2000, 42, 897 CrossRef CAS.
- I. Beech, V. Zinkevich, R. Tapper and R. Gubner, Geomicrobiol. J., 1997, 15, 121 CrossRef.
- E. Miranda, M. Bethencourt, F. J. Botana, M. J. Cano, J. M. Sanchez-Amaya, A. Corzo, J. Garcıa de Lomas, M. L. Fardeau and B. Ollivier, Corros. Sci., 2006, 48, 2417 CrossRef CAS.
- F. Teng, Y. T. Guan and W. P. Zhu, Corros. Sci., 2008, 50, 2816 CrossRef CAS.
- W. E. Federation and A. P. H. Association, APHA, Standard methods for the examination of water and waste water, American Public Health Association (APHA), Washington, DC, USA, 21st edn, 2005 Search PubMed.
- ASTM G1-03, ASTM International, West Conshohocken, PA, USA, 2011 Search PubMed.
- ASTM D2688-15, ASTM International, West Conshohocken. PA, USA, 2015 Search PubMed.
- P. Balamurugan, M. Hiren Joshi and T. S. Rao, Biofouling, 2011, 27, 967 CrossRef CAS PubMed.
- A. M. Kalwale and P. A. Savale, Adv. Appl. Sci. Res., 2012, 3, 273 CAS.
- Y. T. Puyate and A. Rim-Rukeh, J. Appl. Sci. Environ. Manage., 2008, 12, 135 Search PubMed.
- P. Sarin, J. Bebee, K. K. Jim, M. A. Beckett, W. M. Kriven and J. A. Clemen, Water Res., 2004, 38, 1259 CrossRef CAS PubMed.
- J. Kielemoes, I. Bultinck, H. Storms, N. Boon and W. Verstraete, FEMS Microbiol. Ecol., 2002, 39, 41 CrossRef CAS PubMed.
- J. F. Batista, R. F. C. Pereira, J. M. Lopes, M. F. M. Carvalho, M. J. Feio and M. A. M. Reis, Biodegradation, 2000, 11, 441 CrossRef CAS PubMed.
- R. A. Antunes, I. Costa and D. L. A. de Faria, Mater. Res., 2003, 6, 403 CrossRef CAS.
- T. Misawa, K. Hashimoto and S. Shimodaira, Corros. Sci., 1974, 14, 131 CrossRef CAS.
- X. Nie, X. Li, C. Du, Y. Huangb and H. Du, J. Raman Spectrosc., 2009, 40, 76 CrossRef CAS.
- H. A. Videla and L. K. Herrera, Int. Microbiol., 2005, 8, 169 CAS.
- T. Otake, D. J. Wesolowski, L. M. Anovitz, L. F. Allard and H. Ohmoto, Geochim. Cosmochim. Acta, 2010, 74, 6141 CrossRef CAS.
- H. Ohmoto, Econ. Geol., 2003, 98, 157 CrossRef CAS.
- J. B. Fein, C. J. Daughne, N. Yee and T. A. Davis, Geochim. Cosmochim. Acta, 1997, 61, 3319 CrossRef CAS.
- C. J. Daughney and J. B. Fein, J. Colloid Interface Sci., 1998, 198, 53 CrossRef CAS.
- S. Langley and T. J. Beveridge, Appl. Environ. Microbiol., 1999, 65, 489 CAS.
- D. R. Lovley, FEMS Microbiol. Ecol., 1997, 20, 305 CrossRef CAS.
- D. J. Lonergan, H. L. Jenter, D. J. Coates, E. J. Phillips, T. M. Schmidt and D. R. Lovley, J. Bacteriol., 1996, 178, 2402 CAS.
- Y. Luu and J. A. Ramsay, World J. Microbiol. Biotechnol., 2003, 19, 215 CrossRef CAS.
- B. Palaniappan and S. R. Toleti, J. Biosci. Bioeng., 2016, 121, 435 CrossRef CAS PubMed.
- K. P. Nevin and D. R. Lovley, Geomicrobiol. J., 2002, 19, 141 CrossRef CAS.
- G. Reguera, K. D. McCarthy, T. Mehta, J. S. Nicoll, M. T. Tuominen and D. R. Lovley, Nature, 2005, 435, 1098 CrossRef CAS PubMed.
- F. Genuzio, A. Sala, T. Schmidt, D. Menzel and H. Freund, J. Phys. Chem. C, 2014, 118, 29068 CAS.
| This journal is © The Royal Society of Chemistry 2016 |