Efficiently enhancing the photocatalytic activity of faceted TiO2 nanocrystals by selectively loading α-Fe2O3 and Pt co-catalysts†
Chang Liu,
Ruifeng Tong,
Zhenkai Xu,
Qin Kuang*,
Zhaoxiong Xie and
Lansun Zheng
State Key Laboratory of Physical Chemistry of Solid Surfaces, Department of Chemistry, College of Chemistry and Chemical Engineering, Xiamen University, Xiamen 361005, China. E-mail: qkuang@xmu.edu.cn
First published on 17th March 2016
Abstract
The loading of oxidation and/or reduction co-catalysts onto the surface of semiconductor nanomaterials is one of the most efficient methods of improving the performance of semiconductor-based photocatalysts. However, in most of cases, the enhancing effect can be weakened by a random co-catalyst loading method because the different roles of photocatalyst facets in the photocatalytic process are not simultaneously considered. In this paper, a ternary composite photocatalyst, Fe2O3–TiO2–Pt, with α-Fe2O3 and Pt nanoparticles selectively deposited onto the {001} and {101} facets of TiO2, respectively, was successfully constructed using the facet-induced photogenerated electrons and holes of well-faceted anatase TiO2 nanocrystals as natural redox agents. The overall photocatalytic activity of this well-designed composite photocatalyst in H2 production has been enhanced greatly by as much as 2.2 times and 30 times compared to the photocatalysts loaded randomly and without a co-catalyst, respectively. The enhanced photocatalytic activity of Fe2O3–TiO2–Pt was attributed to the remarkably enhanced separation of photogenerated charge carriers with the excitation of UV light.
1. Introduction
Semiconductor-based photocatalysis has attracted increasing attention recently not only for the generation of solar fuels (e.g., by splitting water for H2 production and reducing CO2 for CH4 production) but also for the treatment of gas and water contaminants.1–6 The photocatalytic activity of a semiconductor catalyst primarily depends on its ability to create electron–hole pairs. The most effective strategy for promoting the photocatalytic activity of semiconductors is, therefore, to improve the separation efficiency of photogenerated electron–hole pairs.7,8 Noble metal (such as Pt) and/or metal oxide nanoparticles (such as NiO) have been loaded onto semiconductor nanomaterials as co-catalysts to successfully improve the performance of semiconductor photocatalysts.9,10 For these binary and ternary composite photocatalysts, the separation between electrons and holes can be considerably enhanced under the driving force of a built-in electric field at the heterojunction interface.11–14 Unfortunately, the performance of the composite photocatalysts have been far lower than anticipated, despite much higher than that of unloaded photocatalysts. This lower-than-expected performance might be the result of random co-catalyst loading. The exploration of rationally engineered co-catalysts at desired locations on photocatalyst surfaces is needed to optimize the photocatalytic activities of these composite photocatalysts.Recently, photogenerated electrons and holes were found to be spatially separated onto different facets of well-faceted semiconductor nanocrystals (NCs), leading to different redox reactivities towards photo-activated chemical reactions.15–18 These findings suggest a route towards loading co-catalysts at specific locations on photocatalyst surfaces. In other words, noble metal or metal oxide co-catalysts can be selectively loaded onto the specific reductive or oxidative facets of semiconductor nanocrystals (NCs) using the facet-induced photogenerated electrons and holes as natural redox agents. Recently, this synthetic strategy has been demonstrated with many semiconductors such as TiO2, BiVO4, BiOCl, and AgI.19–22 Despite great success in rationally engineering locations of co-catalyst, we are still facing a problem how to design band position matched heterostructure semiconductors, in which the synergy between the heterojunction-induced and facet-induced effects for the separation of photogenerated electrons and holes can be achieved.
Hematite (α-Fe2O3) is a narrow bandgap semiconductor (Eg: 2.0–2.2 eV) and thought to be a promising photocatalytic and photovoltaic material, because of its natural abundance and excellent stability in aqueous media. Limited by its high recombination and the very short lifetime of its photogenerated charge carriers, α-Fe2O3 is often employed as a oxidation co-catalyst for the wide bandgap semiconductor photocatalyst (such as TiO2) in photocatalytic reaction, instead of main photocatalyst.23–27 In previous studies, the deposition of α-Fe2O3 onto the surface of semiconductor photocatalysts was typically achieved using either impregnation or hydrothermal methods, which usually result in randomly loading of α-Fe2O3 nanoparticles.28,29 In this paper, a ternary composite photocatalyst was designedly constructed in which α-Fe2O3 and Pt co-catalysts were loaded on the photo-oxidative {001} facets and photo-reductive {101} facets, respectively, of tetragonal bipyramidal anatase TiO2 NCs. Our experimental results demonstrated that this well-designed composite photocatalyst was significantly more active in H2 evolution than the photocatalyst loaded randomly or with only one co-catalyst, due to its more efficient electron–hole separation, as we expected.
2. Experimental section
2.1 Chemicals
Potassium hydroxide (KOH, 90%), hexamethylenetetramine (HMTA, 99%), methanol (CH3OH, 99.5%), ethanol (C2H5OH, 99.9%), potassium iodate (KIO3, 99%), iron nitrate nonahydrate (Fe(NO3)3·9H2O, 99%) and ferrous sulfate (FeSO4·7H2O, 99%) were purchased from Sinopharm Chemical Reagent Co., Ltd. Potassium hexachloroplatinate (K2PtCl6·6H2O, 99.95%) was purchased from Alfar Aesar. The commercial photocatalyst Degussa P25 was purchased from Shanghai Haiyi Co. All reagents were used as received without further purification.2.2 Synthesis of samples
2.3 Characterization of samples
The morphology and structure of all samples were observed by transmission electron microscopy (TEM, JEOL JEM-2100) at a 200 kV accelerating voltage and scanning electron microscopy (SEM, Hitachi S-4800) at a 10 kV accelerating voltage. The UV-vis diffuse reflectance spectra (DRS) of samples were measured by UV-vis spectrophotometry (Varian Cary-5000). The specific surface areas of samples were measured by the Brunauer–Emmett–Teller (BET) method using nitrogen adsorption and desorption isotherms on a Micromeritics Instrument Corporation sorption analyzer (TriStar II 3020). The high resolution X-ray photoelectron spectroscopy (XPS) study was performed using a Thermo Scientific K-alpha photoelectron spectrometer (PHI Quantum-2000) using monochromatic Al Kα radiation; all peak positions were calibrated with the C 1s peak (284.5 eV) as an internal standard. The effective loading amounts of Pt and Fe2O3 on TiO2 NCs were determined by inductively coupled plasma-atomic emission spectrometry (ICP-AES, Baird PS-4).2.4 Photocatalytic activity measurement in H2 evolution
The photocatalytic H2 production experiments were conducted in a Labsolar-III (AG) photocatalytic H2 production system (Perfectlight Co. Ltd). Typically, 25 mg TiO2 sample was well dispersed in a mixed solution of deionized water (90 mL) and methanol (10 mL) under magnetically stirring. The reaction temperature was maintained at 10 °C by a cooling water circulation system. A 300 W Xe lamp (PLS-SXE-300UV, Beijing Trusttech Co. Ltd) with the wavelength of 300–2500 nm was employed as light source, and the power density was measured to be 1200 and 560 mW cm−2 at the light outlet and actual location of reactor, respectively. Before irradiation, the system was pumped for 1 h to remove the air. The amount of H2 evolved was determined by using a gas chromatography (GC 2060) with a TCD detector, and high purity Ar gas (99.999%) was used as carrier gas.3. Results and discussion
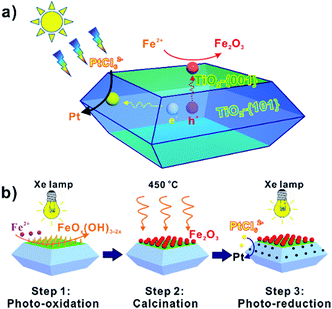
For anatase TiO2, the {001} and {101} facets become hole- and electron-rich, respectively, upon irradiation, thereby providing oxidation and reduction sites for chemical reaction, as depicted in Scheme 1a. Because of this unique photochemical behavior of anatase TiO2, a step-by-step photo-activated chemical deposition process was developed to construct a special ternary composite photocatalyst with co-catalysts at desired locations of TiO2 NCs. As illustrated in Scheme 1b, α-Fe2O3 nanoparticles can be selectively deposited on the hole-rich {001} facets of TiO2 through the photooxidation of an Fe(II) salt (FeSO4) (step 1) and a subsequent calcination process at 450 °C (step 2). And then Pt nanoparticles were deposited on the electron-rich {101} facets of TiO2 via the photoreduction of K2PtCl6 (step 3). In this ternary composite photocatalyst, Pt nanoparticles deposited on the photo-reductive {101} facets of TiO2 NCs not only act as sinks for photogenerated electrons, but also provide effective proton reduction sites for H2 production. In the meanwhile, α-Fe2O3 nanoparticles deposited on the photo-oxidative {001} facets of TiO2 NCs could induce a transfer of photogenerated holes from TiO2 to Fe2O3, because the valence band of α-Fe2O3 is higher than that of TiO2. Since the heterojunction-induced and facet-induced effects for the separation of photogenerated electrons and holes work cooperatively, this well-designed ternary composite photocatalyst is anticipated to exhibit a photocatalytic activity superior to that of a photocatalyst loaded randomly or with only one co-catalyst.In this study, anatase TiO2 NCs with a truncated tetragonal bipyramidal morphology were used as a model photocatalyst, in which the top/bottom surfaces were {001} facets and the side surfaces were {101} facets. For the truncated tetragonal bipyramidal TiO2 NCs used, the average lengths of the maximum and minimum sides of the square {001} faces in every particle were 250 nm and 200 nm, respectively, corresponding to 27.9% {001} facets exposed on the NCs' surfaces (Fig. S1†). Fig. 1 shows typical morphologies of TiO2 NCs after the selective deposition of α-Fe2O3 and Pt co-catalysts, i.e. Fe2O3–TiO2 and Fe2O3–TiO2–Pt. In Fe2O3–TiO2, the {001} facets of TiO2 NCs were decorated with a large number of spherical nanoparticles with an average diameter of 4–8 nm, while the {101} facets remained smooth (Fig. 1a and b). These spherical nanoparticles were actually α-Fe2O3 and they resulted from the dehydration of amorphous ferric oxyhydroxide (FeOx(OH)3−2x, x = 0–1) nanoparticles that were formed on the {001} facets of TiO2 NCs via the hydrolysis of Fe3+ after the photooxidation of Fe2+ (Fig. S2†).32 After the photoreduction process, the originally smooth {101} facets of TiO2 NCs became very coarse, indicating the presence of nanoparticles (Fig. 1c and d). Unfortunately, these nanoparticles on the {101} facets were outside the resolution limit of the SEM due to their small size.
 | ||
| Fig. 1 Low magnification and high magnification SEM images of (a and b) Fe2O3–TiO2, and (c and d) Fe2O3–TiO2–Pt. Insets in (b) and (d) are corresponding models to show the sample morphologies. | ||
In order to determine the composition of the deposits, XRD analysis was performed. However, the XRD patterns of these samples were the same as that of pure anatase TiO2 (PDF no. 84-1286), and the characteristic peaks of Fe2O3 and Pt were not detected, as shown in Fig. 2a. This may be attributed to the low loading content and small size of the deposited nanoparticles.25 ICP-AES results confirmed that the loading amounts of Fe and Pt were only ca. 3.17 wt% and 0.64 wt%, respectively, lower than their raw material ratios (ca. 5 wt% and 1 wt% for Fe and Pt, respectively).
More information about the composition of the deposits on each facet was provided by TEM. As shown in Fig. 2b, in Fe2O3–TiO2, 8–15 nm nanoparticles with a higher contrast appeared on the {001} facets of the TiO2 NCs. The lattice fringes of 0.27 nm spacing were observed on the nanoparticles, which corresponded to the {104} plane of α-Fe2O3 (Fig. 2b). These α-Fe2O3 nanoparticles presented good crystallinity and close contact with the TiO2 surface, which would facilitate charge transfer between α-Fe2O3 and TiO2. In Fe2O3–TiO2–Pt, the lattice fringes taken from the nanoparticles deposited on the {001} facets likewise corresponded to α-Fe2O3. Besides that, some very small nanoparticles of 2–3 nm in size were observed on the {101} facets of the TiO2 NCs. The lattice spacing of these small nanoparticles was measured to be 0.224 nm, which corresponded to the {111} planes of metallic Pt (Fig. 2b). Therefore, the deposits on the {101} facets of the TiO2 NCs were Pt nanoparticles.
To confirm the exact chemical states of Fe and Pt species deposited on the surface of the TiO2 NCs, high resolution XPS were further measured, and the results taken from Fe2O3–TiO2–Pt are shown in Fig. 3. The Ti 2p XPS spectrum displayed doublet peaks at 458.7 eV and 464.3 eV, which corresponded to Ti 2p3/2 and Ti 2p1/2, respectively (Fig. 3a). The splitting width between the two peaks was 5.7 eV, indicating a normal state of Ti4+. In the Fe 2p XPS spectrum (Fig. 3b), two peaks were observed near 710.4 eV (Fe 2p3/2) and 724.2 eV (Fe 2p1/2) with a weak shake-up satellite peak, which represented the presence of Fe3+–O.33,34 The O 1s XPS spectrum displayed a slightly asymmetric single peak centered at 530.0 eV, which was deconvoluted by Gaussian fitting into three components (Fig. 3c). The dominant component (OL) centered at 529.8 eV was attributed to the lattice oxygen in the oxides. The weak peak located at 530.7 eV was attributed to the component (OV) associated with O2− ions in oxygen-deficient regions (oxygen vacancies), and the weaker component (OC) at approximately 532.5 eV was attributed to chemisorbed oxygen species (e.g., OH−).35 It should be noted that Fe2O3–TiO2–Pt contained a much lower proportion of both OV and OC compared to the FeOx(OH)3−2x–TiO2 sample without calcination processing (Fig. S3†). This indicated that the crystallinity of ferric oxide nanoparticles deposited on the {001} facets of the TiO2 NCs was improved by calcination. The Pt 4f XPS spectrum showed two peaks centered at 74.9 eV and 71.8 eV, which corresponded to the 4f5/2 and 4f7/2 signals of metallic Pt, respectively (Fig. 3d).
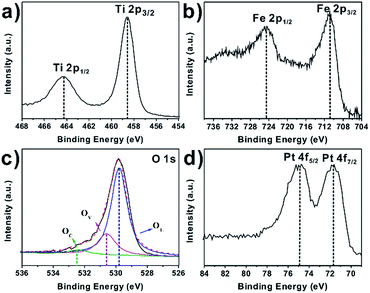 | ||
| Fig. 3 High resolution XPS survey of (a) Ti 2p, (b) Fe 2p, (c) O 1s, and (d) Pt 4f for Fe2O3–TiO2–Pt. | ||
According to the above characterizations, there was no doubt that the nanoparticles selectively deposited on the {001} and {101} facets of TiO2 in Fe2O3–TiO2–Pt were α-Fe2O3 and Pt, respectively, as was intended. According to our design, a synergy was expected between the facet-induced and heterojunction-induced effects for the separation of photogenerated carriers in Fe2O3–TiO2–Pt, which would efficiently enhance the photocatalytic activity of photocatalysts.
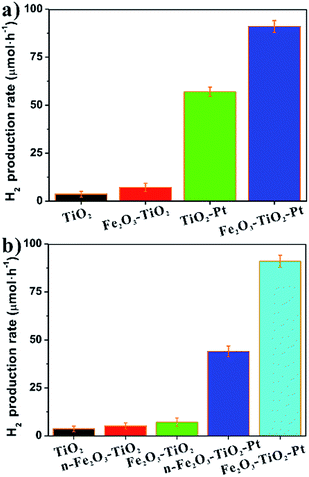
To validate the effectiveness of the fabrication strategy used, the photocatalytic performances of bare TiO2, TiO2 loaded only with Fe2O3 on {001} facets (Fe2O3–TiO2), and TiO2 loaded with dual co-catalysts (Fe2O3–TiO2–Pt) for H2 production were systematically evaluated. For comparison, TiO2 loaded only with Pt on {101} facets (TiO2–Pt), which has been previously proven to exhibit an excellent photocatalytic activity in H2 evolution, were specifically prepared and measured as reference (Fig. S4†).19 Fig. 4a shows the H2 production rate (μmol h−1) in the presence of four samples under irradiation by a 300 W Xe lamp for 4 h, which followed the order of Fe2O3–TiO2–Pt > TiO2–Pt ≫ Fe2O3–TiO2 > TiO2. In the absence of co-catalysts, TiO2 NCs exhibited very poor photocatalytic activity, and the hydrogen production rate was only 3.6 μmol h−1. When loaded with α-Fe2O3, the photocatalyst displayed an enhanced photocatalytic activity, and the H2 production rate increased to 7.1 μmol h−1, near twice that of bare TiO2. The hydrogen production rate of TiO2–Pt was 20 times that of the bare TiO2 NCs and 8 times that of Fe2O3–TiO2. Excitingly, the hydrogen production rate of the ternary photocatalyst Fe2O3–TiO2–Pt increased up to 90.0 μmol h−1, which was 150% that of TiO2–Pt. This result was entirely consistent with our expectation.
In fact, the importance of rationally engineering the locations of co-catalysts to improve photocatalytic activity can be further highlighted by comparing the photocatalytic performances between the composite photocatalysts with co-catalysts selectively deposited and their counterparts with randomly deposited. We have systematically studied the effect of Pt nanoparticles selective and random deposition on the surface of TiO2 NCs in our previous study.19 The results indicated that the selective Pt deposition greatly enhanced the photocatalytic performance of TiO2 NCs whereas this enhancement was reduced by randomly depositing Pt nanoparticles on the surface of TiO2 NCs. In order to explore whether the deposition location of Fe2O3 nanoparticles have similar effect, two photocatalysts with α-Fe2O3 randomly deposited, n-Fe2O3–TiO2 (Fig. S5†) and n-Fe2O3–TiO2–Pt (Fig. S6†) was specifically synthesized, in which α-Fe2O3 were deposited on the TiO2 NCs via a traditional impregnation–calcination method.31 It should be noted that the photocatalytic activity of TiO2 NCs relates to the size of Fe2O3 nanoparticles. Therefore, different precursors and calcination temperatures were optimized in our study to ensure that the size distribution range of Fe2O3 selectively and randomly deposited on the surface of TiO2 NCs were similar. As shown in Fig. S8,† the size distribution range of Fe2O3 in Fe2O3–TiO2 and n-Fe2O3–TiO2 sample are 6.4 ± 1.0 nm and 6.0 ± 1.0 nm, respectively. ICP-AES result confirmed the loading amounts of Fe (ca. 3.62 wt%) and Pt (ca. 0.69 wt%) in n-Fe2O3–TiO2–Pt were similar as Fe2O3–TiO2–Pt. As shown in Fig. 4b, the H2 production rate of n-Fe2O3–TiO2 was 5.1 μmol h−1, which was slightly higher than that of bare TiO2 (3.6 μmol h−1) but lower than that of Fe2O3–TiO2 (7.2 μmol h−1). Even after the deposition of Pt nanoparticles, the H2 production rate of n-Fe2O3–TiO2–Pt was only 44.0 μmol h−1, which was approximately half that of Fe2O3–TiO2–Pt. Obviously, the photocatalysts with co-catalyst selectively deposited are much superior to their counterparts with co-catalysts deposited randomly in the photocatalytic activity. This indicated that the enhanced photocatalytic activity of Fe2O3–TiO2–Pt indeed originated from the rational locations of dual co-catalysts on the TiO2 NCs. In other words, the composite photocatalysts could not hit peak photocatalytic efficiency, if co-catalysts are randomly located on the surface of photocatalysts.
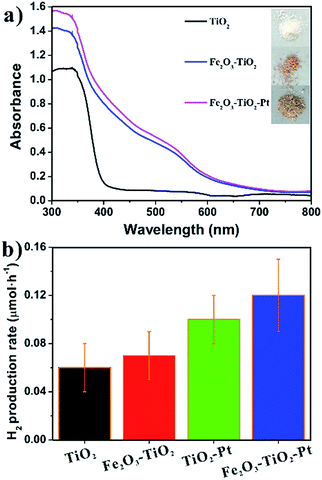
It should be pointed out that the photocatalytic activity of photocatalysts is actually influenced by many factors, such as the specific surface area of photocatalysts and the ability to absorb light. However, there was very few difference in their specific surface areas between the measured samples (10.35, 12.03, 11.80, 12.64 m2 g−1 for TiO2, Fe2O3–TiO2, TiO2–Pt, and Fe2O3–TiO2–Pt, respectively), and the specific surface area of Fe2O3–TiO2–Pt was just slightly higher than Fe2O3–TiO2 and TiO2–Pt. Therefore, the significant enhancement of Fe2O3–TiO2–Pt in photocatalytic activity compared to Fe2O3–TiO2 and TiO2–Pt has no relationship with the differences in their specific surface areas. On the other hand, a significant change in the color of the TiO2 NCs occurred after the loading of the co-catalysts (insets to Fig. 5a). The UV-vis DRS of the samples revealed that the bare TiO2 NCs absorbed only UV light below 400 nm, while Fe2O3–TiO2 and Fe2O3–TiO2–Pt not only absorbed light more strongly in the UV region but also had a considerable absorption in the visible region. The absorption edges of two samples were significantly red-shifted to approximately 620 nm, which is consistent with the bandgap of Fe2O3 (ca. 2.00 eV). Therefore, the extension of the light absorption range in the visible light region for Fe2O3–TiO2 and Fe2O3–TiO2–Pt can be attributed to α-Fe2O3 deposited on the TiO2 NCs. Previous studies have demonstrated that TiO2–Fe2O3 nanocomposites exhibited a pretty good photocatalytic activity under visible light, which was usually attributed to the visible light driven electron transfer.23,26,27,29 That is, the electrons in the valence band of α-Fe2O3 could be excited to higher energy levels of the conduction band with the excitation of high-energy visible light (400–550 nm), and these high energy electrons, in which energy level was even higher than the conduction band position of TiO2, could thermodynamically transfer to the conduction band of TiO2, thereby enhancing photocatalytic activity of photocatalysts.23 To determine the contribution from the visible-light driven photocatalytic activity of α-Fe2O3, the H2 production rates of TiO2, Fe2O3–TiO2, TiO2–Pt, and Fe2O3–TiO2–Pt were measured under irradiation by visible light (λ ≥ 400 nm) using a UV400 cut-off filter. As shown in Fig. 5b, these photocatalysts performed badly under visible-light irradiation, and the photocatalytic H2 evolution rates were orders of magnitude lower than those illuminated by an unfiltered Xe lamp. Such low visible light photocatalytic activities revealed that the contribution from the visible light driven electron transfer from α-Fe2O3 to TiO2 were minimal. However, the photocatalytic activities of the different photocatalysts followed the same order under visible and UV-visible irradiation. Therefore, the rational locations of co-catalysts was exactly the underlying cause for the enhanced photocatalytic activities of Fe2O3–TiO2–Pt.
It should be noted Fe2O3–TiO2–Pt possessed not only remarkably enhanced photocatalytic ability but also an excellent stability for H2 production. As shown in Fig. 6, the photocatalytic activity of Fe2O3–TiO2–Pt decreased by less than 10% after four H2 production cycles. Previous studies revealed that the photogenerated electrons in the conduction band of α-Fe2O3 could reduce Fe3+ to Fe2+, resulting in the dissolution of α-Fe2O3 in a liquid photocatalytic environment, which greatly restricts stability and recyclability of Fe2O3-based photocatalyst.36–38 However, XPS analysis of the photocatalyst after the photocatalytic H2 production reaction confirmed that Fe did not undergo a change of valence state (Fig. S7†). Therefore, the slight decrease in photocatalytic activity might have been caused by the incomplete recovery of the photocatalyst in these experiments.
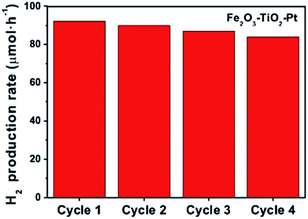 | ||
| Fig. 6 Multiple cycles of photocatalytic H2 production in the presence of Fe2O3–TiO2–Pt under irradiation by a 300 W Xe lamp. The amount of photocatalyst was 25 mg and the irradiation time was 4 h. | ||
Given the above results, Fe2O3–TiO2–Pt exhibited the best photocatalytic activity among all the photocatalysts and good stability for the H2 evolution reaction as was expected. The rational engineering of the locations at which co-catalysts are deposited on a photocatalyst, i.e. the selective deposition of reduction and oxidation co-catalysts onto the relevant reduction and oxidation facets of TiO2 NCs, was indeed able to effectively achieve an optimal photocatalytic activity. To interpret the remarkably enhanced photocatalytic performance of Fe2O3–TiO2–Pt, the transfer of photogenerated electrons and holes in Fe2O3–TiO2–Pt is illustrated in Scheme 2, where both the facet- and heterojunction-induced effects are fully considered. Note that the electrons and holes in the conduction bands and valence bands of TiO2 and α-Fe2O3 are dominantly generated by the excitation with UV light of Xe lamp, since the contribution from the visible light has proven to be negligible. Previous studies have revealed that the positions of both the conduction and valence band of the {101} facets of anatase TiO2 are slightly lower than those of the {001} facets.17,39,40 Photogenerated electrons prefer to transfer toward the {101} facets, while photogenerated holes transfer toward the {001} facets (the facet-induced effect). As a result, the {101} and {001} facets provide reduction and oxidation sites, respectively, in photochemical reactions. When Pt nanoparticles are selectively deposited on the {101} electron-rich facets, the photogenerated electrons transfer to the Pt nanoparticles from the {101} facets of TiO2, causing the Pt nanoparticles to become the catalytic centers for H2 production (the semiconductor/metal heterojunction-induced effect). When α-Fe2O3 nanoparticles are deposited on the {001} facets of TiO2 NCs, the holes concentrated on the {001} facets are likewise easily transferred to α-Fe2O3 because of higher valence band position of α-Fe2O3 (the semiconductor/semiconductor heterojunction-induced effect). In contrast, there is no pathway for the electrons, which are concentrated on the {101} facets of TiO2 NCs, to transfer to α-Fe2O3 located on the {001} facets of TiO2 NCs due to spatial separation, although α-Fe2O3 is lower than TiO2 in conduction band positions. That is, if only co-catalysts locate on the right facets, the facet-induced effect acts in cooperation with the heterojunction-induced effect, thereby leading to an optimized separation efficiency of photogenerated electrons and holes. For the randomly loaded photocatalysts, in which some co-catalyst nanoparticles locate on the facets with an opposite effect, the recombination of photogenerated electrons and holes would become serious, because the heterojunction-induced effect is in conflict with the facet-induced effect.
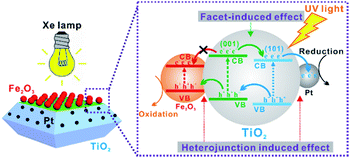 | ||
| Scheme 2 Schematic diagram of the proposed mechanism for the enhanced photocatalytic activity of Fe2O3–TiO2–Pt under UV light. | ||
4. Conclusions
A specific ternary heterojunction photocatalyst was designedly synthesized, in which two co-catalysts, α-Fe2O3 and Pt, were selectively loaded onto the photo-oxidative {001} and photo-reductive {101} facets, respectively, of TiO2 NCs by utilizing the facet-dependent photochemical reactivity of TiO2 NCs. Compared to the photocatalysts loaded randomly and without co-catalyst, this well-designed ternary photocatalyst, Fe2O3–TiO2–Pt, exhibited superior photocatalytic activity (as high as 2.2 times and 30 times, respectively) and excellent recycling stability for H2 production. The fundamental cause was that the separation of photogenerated charge carriers with the excitation of UV light were remarkably improved due to the synergy between the facet-induced and heterojunction induced effects. This photocatalyst engineering strategy, which was based on the selective deposition of co-catalysts, allows the intrinsic effects of specific facets and heterojunctions to be probed for their role in the enhanced photocatalytic performance of the composite photocatalysts. In addition, since the proposed enhancement mechanism is universal, the synthesized photocatalysts are expected to perform best in all photocatalytic processes.Acknowledgements
This work was supported by the National Basic Research Program of China (2015CB932301), the National Natural Science Foundation of China (21473146 and 21333008) and the Fundamental Research Funds for the Central Universities (20720160026).Notes and references
- T. Hisatomi, J. Kubota and K. Domen, Chem. Soc. Rev., 2014, 43, 7520–7535 RSC.
- H. Tong, S. Ouyang, Y. Bi, N. Umezawa, M. Oshikiri and J. Ye, Adv. Mater., 2012, 24, 229–251 CrossRef CAS PubMed.
- B. Oregan and M. Gratzel, Nature, 1991, 353, 737–740 CrossRef CAS.
- M. S. Dresselhaus, G. Chen, M. Y. Tang, R. Yang, H. Lee, D. Wang, Z. Ren, J.-P. Fleurial and P. Gogna, Adv. Mater., 2007, 19, 1043–1053 CrossRef CAS.
- C. K. Chan, H. Peng, G. Liu, K. McIlwrath, X. F. Zhang, R. A. Huggins and Y. Cui, Nat. Nanotechnol., 2008, 3, 31–35 CrossRef CAS PubMed.
- X. Chen, L. Liu, P. Y. Yu and S. S. Mao, Science, 2011, 331, 746–750 CrossRef CAS PubMed.
- C. Chen, W. Ma and J. Zhao, Chem. Soc. Rev., 2010, 39, 4206–4219 RSC.
- R. Li, H. Han, F. Zhang, D. Wang and C. Li, Energy Environ. Sci., 2014, 7, 1369–1376 CAS.
- R. Marschall, Adv. Funct. Mater., 2014, 24, 2421–2440 CrossRef CAS.
- W. Fan, Q. Zhang and Y. Wang, Phys. Chem. Chem. Phys., 2013, 15, 2632–2649 RSC.
- K. Woan, G. Pyrgiotakis and W. Sigmund, Adv. Mater., 2009, 21, 2233–2239 CrossRef CAS.
- M. Murdoch, G. I. N. Waterhouse, M. A. Nadeem, J. B. Metson, M. A. Keane, R. F. Howe, J. Llorca and H. Idriss, Nat. Chem., 2011, 3, 489–492 CAS.
- Y. Ohko, T. Tatsuma, T. Fujii, K. Naoi, C. Niwa, Y. Kubota and A. Fujishima, Nat. Mater., 2003, 2, 29–31 CrossRef CAS PubMed.
- S. Sakthivel, M. V. Shankar, M. Palanichamy, B. Arabindoo, D. W. Bahnemann and V. Murugesan, Water Res., 2004, 38, 3001–3008 CrossRef CAS PubMed.
- T. Ohno, K. Sarukawa and M. Matsumura, New J. Chem., 2002, 26, 1167–1170 RSC.
- T. Tachikawa, S. Yamashita and T. Majima, J. Am. Chem. Soc., 2011, 133, 7197–7204 CrossRef CAS PubMed.
- J. Yu, J. Low, W. Xiao, P. Zhou and M. Jaroniec, J. Am. Chem. Soc., 2014, 136, 8839–8842 CrossRef CAS PubMed.
- M. D'Arienzo, M. V. Dozzi, M. Redaelli, B. D. Credico, F. Morazzoni, R. Scotti and S. Polizzi, J. Phys. Chem. C, 2015, 119, 12385–12393 Search PubMed.
- C. Liu, X. Han, S. Xie, Q. Kuang, X. Wang, M. Jin, Z. Xie and L. Zheng, Chem.–Asian J., 2013, 8, 282–289 CrossRef CAS PubMed.
- R. Li, F. Zhang, D. Wang, J. Yang, M. Li, J. Zhu, X. Zhou, H. Han and C. Li, Nat. Commun., 2013, 4, 1432 CrossRef PubMed.
- L. Zhang, W. Wang, S. Sun, D. Jiang and E. Gao, Appl. Catal., B, 2015, 162, 470–474 CrossRef CAS.
- Q. Kuang, X. Zheng and S. Yang, Chem.–Eur. J., 2014, 20, 2637–2645 CrossRef CAS PubMed.
- P. Luan, M. Xie, X. Fu, Y. Qu, X. Sun and L. Jing, Phys. Chem. Chem. Phys., 2015, 17, 5043–5050 RSC.
- K. E. deKrafft, C. Wang and W. Lin, Adv. Mater., 2012, 24, 2014–2018 CrossRef CAS PubMed.
- S. J. A. Moniz, S. A. Shevlin, X. An, Z.-X. Guo and J. Tang, Chem.–Eur. J., 2014, 20, 15571–15579 CrossRef CAS PubMed.
- T. Madrakian, A. Afkhami, R. Haryani and M. Ahmadi, Rsc Adv., 2014, 4, 44841–44847 RSC.
- P. Luan, M. Xie, D. Liu, X. Fu and L. Jing, Sci. Rep., 2014, 4, 6180 CrossRef CAS PubMed.
- G. H. Du, Q. Chen, P. D. Han, Y. Yu and L. M. Peng, Phys. Rev. B: Condens. Matter Mater. Phys., 2003, 67, 035323 CrossRef.
- L. Peng, T. Xie, Y. Lu, H. Fan and D. Wang, Phys. Chem. Chem. Phys., 2010, 12, 8033–8041 RSC.
- G. H. Du, Q. Chen, P. D. Han, Y. Yu and L. M. Peng, Phys. Rev. B: Condens. Matter Mater. Phys., 2003, 67, 035323 CrossRef.
- W. Zhou, H. Fu, K. Pan, C. Tian, Y. Qu, P. Lu and C.-C. Sun, J. Phys. Chem. C, 2008, 112, 19584–19589 CAS.
- I. Jang, K.-E. You, Y. C. Kim and S.-G. Oh, Appl. Surf. Sci., 2014, 316, 187–193 CrossRef CAS.
- T. Yamashita and P. Hayes, Appl. Surf. Sci., 2008, 254, 2441–2449 CrossRef CAS.
- Y. Luo, J. Luo, J. Jiang, W. Zhou, H. Yang, X. Qi, H. Zhang, H. J. Fan, D. Y. W. Yu, C. M. Li and T. Yu, Energy Environ. Sci., 2012, 5, 6559–6566 CAS.
- P. T. Hsieh, Y. C. Chen, K. S. Kao and C. M. Wang, Appl. Phys. A, 2008, 90, 317–321 CrossRef CAS.
- B. Palanisamy, C. M. Babu, B. Sundaravel, S. Anandan and V. Murugesan, J. Hazard. Mater., 2013, 252, 233–242 CrossRef PubMed.
- Y. Wang, W. Du and Y. Xu, Langmuir, 2009, 25, 2895–2899 CrossRef CAS PubMed.
- Z. Li, J. Sheng, Y. Wang and Y. Xu, J. Hazard. Mater., 2013, 254, 18–25 CrossRef PubMed.
- J. Pan, G. Liu, G. Q. Lu and H.-M. Cheng, Angew. Chem., Int. Ed., 2011, 50, 2133–2137 CrossRef CAS PubMed.
- Y.-F. Li, Z.-P. Liu, L. Liu and W. Gao, J. Am. Chem. Soc., 2010, 132, 13008–13015 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: XRD pattern and SEM images of TiO2 NCs; SEM, TEM images and XPS spectra of FeOx(OH)3−2x–TiO2; SEM and TEM images of TiO2–Pt; SEM and EDX analysis of n-Fe2O3–TiO2 and n-Fe2O3–TiO2–Pt NCs; XPS spectra of Fe2O3–TiO2–Pt. See DOI: 10.1039/c6ra04552a |
| This journal is © The Royal Society of Chemistry 2016 |