Synthesis of decahydropyrrolo[1,2-a]tetrazolo[1,5-d]pyrazines via Strecker reaction and intramolecular [3+2] cycloaddition†
Shakil N. Afraja,
Chinpiao Chen*ab and
Gene-Hsian Leec
aDepartment of Chemistry, National Dong Hwa University, Soufeng, Hualien, 974, Taiwan. E-mail: chinpiao@mail.ndhu.edu.tw; Fax: +886-3-863-0475
bDepartment of Nursing, Tzu Chi University of Science and Technology, Hualien, 970, Taiwan
cInstrumentation Center, National Taiwan University, Taipei 10617, Taiwan
First published on 11th March 2016
Abstract
This paper reports an efficient two-step method for synthesizing a series of new (5aS,10R)-10-aryl-5,5a,6,7,8,10-hexahydropyrrolo[1,2-a]tetrazolo[1,5-d]pyrazines by a catalyst-free three-component Strecker reaction followed by an intramolecular [3+2] cycloaddition. The first step involved three-component coupling of aldehydes, (S)-2-(azidomethyl)pyrrolidine and potassium cyanide in presence of water to form (R)-2-((S)-2-(azidomethyl)pyrrolidin-1-yl)-2-phenylacetonitriles (3a–3v). In the next step products (3a–3v) underwent intramolecular [3+2] cycloaddition on heating to form (5aS,10R)-10-aryl-5,5a,6,7,8,10-hexahydropyrrolo[1,2-a]tetrazolo[1,5-d]pyrazines (4a–4v). The present methodology is cost-effective, operationally simple, and applicable for various aromatic, aliphatic and heterocyclic aldehydes, and gives desired products in satisfactory yields and diastereoselectivities. A proposed mechanism for synthesis of 3a and 4a is reported.
Introduction
Multicomponent Strecker reactions1 are excellent reactions for the synthesis of α-amino nitriles.2,3 These α-aminonitriles have gained considerable attention over the past few years, in particular because the α-aminonitrile functionality acts as an intermediate in the synthesis of pharmaceutically crucial chiral amino acids and their derivatives,4 ligands, peptides, and natural products.5 Developing a simple, efficient, and cost-effective method for synthesizing α-aminonitriles has become increasingly crucial. Numerous efforts have resulted in the development of highly efficient methods for synthesizing α-aminonitriles.6–8 Dipolar cycloaddition reactions have been the cornerstone of heterocyclic chemistry for the previous decades.9 The Huisgen 1,3-dipolar cycloaddition10 between an azide and a nitrile is the most favourable and powerful mechanism for constructing a tetrazole moiety.11a Many strategies have been developed for synthesizing tetrazoles; the conventional synthesis of 5-substituted 1H-tetrazoles through the [3+2] cycloaddition of an azide with the corresponding nitrile was first reported by Hantzsch and Vagt.11b Sharpless et al. later reported the innovative formation of fused tetrazoles and 1H-tetrazoles, created by using ZnII salt.11c,d Pizzo et al. reported a TBAF catalyzed [3+2] cycloaddition,11e and Yamamoto et al. studied a Cu2O-catalyzed azide–nitrile cycloaddition.11f A highly efficient iron-catalyzed synthesis of 5-substituted 1H-tetrazole was demonstrated by Bolm et al.11g Pitchumani et al. studied a CoY zeolite-catalyzed 5-substituted 1H-tetrazole;11h Stephen et al. reported a facile Lewis acid-mediated synthesis of bicyclic tetrazoles;11i and Smith et al. investigated a tricyclic fused tetrazole created by thermal cyclization.11j Mohapatra and co-workers described synthesis of tricyclic fused tetrazoles from α-amino acid derivatives following ‘click’ chemistry.11k Finally, Frejd et al. developed tricyclic fused tetrazoles from allylic bromides generated by the DiazAll reaction.11l Tetrazoles have gained considerable attention from organic chemists because they can act as metabolically stable surrogates for the carboxylic acid group12 and as precursors in various nitrogen-containing heterocycles,13 and they are useful synthons in synthetic organic chemistry (Fig. 1).14aVarious biologically active compounds incorporating the tetrazole moiety have been developed. For example, compound (A), an angiotensin II antagonist, has been used to treat hypertension;14b,c compound (B) has been extensively used in models for anxiety;14d,e and compound (C) has shown affinity toward benzodiazepine receptors.14f Moreover, tetrazoles have antibacterial15 and anticancer16 activities and exert protective effects against heart disease17 and neurodegenerative disorder.18
Results and discussion
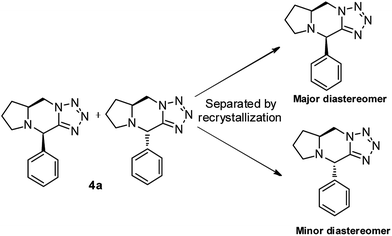
To design new fused heterocycles,19 we developed a simple, efficient, and cost-effective two-step strategy. The first step involved a three-component reaction of benzaldehyde, (S)-2-(azidomethyl)pyrrolidine, and KCN in the presence of water to form (R)-2-((S)-2-(azidomethyl)pyrrolidin-1-yl)-2-phenylacetonitrile (3a). The second step involved the catalyst-free intramolecular [3+2] cycloaddition of 3a to form (5aS,10R)-10-phenyl-5,5a,6,7,8,10-hexahydropyrrolo[1,2-a]tetrazolo[1,5-d]pyrazine (4a). The first step involved using a readily available cyanide source20 and water as the solvent to form aminonitrile (3a); this is an alternative method for the catalyst-free Strecker reaction. The second step enabled the fused tetrazole moiety, created through the catalyst-free intramolecular [3+2] cycloaddition. First, we prepared (S)-2-(azidomethyl)pyrrolidine (2a) by using the commercially available L-proline and following a previously reported procedure.21To establish the optimal experimental conditions for the synthesis of 3a, we use benzaldehyde, (S)-2-(azidomethyl)pyrrolidine (2a) and KCN as a model substrate. Table 1 summarizes the results, which indicated that the reaction temperature, solvents, and the loading of potassium cyanide influenced the product yields of the Strecker reaction. The most satisfactory condition was heating the mixture of benzaldehyde (1 eq.), (S)-2-(azidomethyl)pyrrolidine (1.1 eq.), and potassium cyanide (1.2 eq.) at 70 °C in the presence of water for 8 h (Table 1, entry 18). Initially, several organic solvents were screened for the three-component Strecker reaction of a model substrate by using 1.5 eq. of potassium cyanide: toluene at 111 °C afforded 3a and 4a in 28% and 1% yields, respectively; (Table 1, entry 1), dichloromethane at 40 °C afforded 3a and 4a in 24% and 1.5% yields, respectively; (Table 1, entry 2), acetonitrile at 80 °C afforded 3a and 4a in 17% and 1% yields, respectively; (Table 1, entry 3), and methanol at 65 °C afforded only product 3a in 23% yield (Table 1, entry 4). In addition, we screened solvents such as dioxane, dimethylformamide, and dimethylsulfoxide at 101, 140 and 145 °C, respectively; however, no product was formed (Table 1, entries 5–7). We then used water as the solvent at 100 °C, which afforded 3a and 4a in 69% and 1.3% yields, respectively (Table 1, entry 8). Therefore, we heated the reaction mixture at 100, 90, and 80 °C, which produced 3a in 76%, 81%, and 86% yields and 4a in 1.5%, 2%, and 1.5% yields, respectively (Table 1, entries 9–11). According to these results, as the temperature of the reaction decreased, the yields of the corresponding products increased. Therefore, we heated the reaction mixture of benzaldehyde (1 eq.), (S)-2-(azidomethyl)pyrrolidine (1.1 eq.), and potassium cyanide (1.5 eq.) at 70 °C for 8 h, which produced 3a and 4a in 93% and 1.8% yields, respectively (Table 1, entry 12). In addition, we heated the reaction mixture at 60 °C, 50 °C, 40 °C, and room temperature; however, increase in the reaction time and decrease in the product yields were observed (Table 1, entries 13–16). To further investigate the effect of the amount of potassium cyanide during the reaction, we conducted the same reaction by using 1.0 and 1.2 eq. of potassium cyanide, which afford 3a in 89% and 93% yields and 4a in 1.8% and 2.0% yields, respectively (Table 1, entries 17, 18). Therefore, heating 1.0 eq. of benzaldehyde, 1.1 eq. of (S)-2-(azidomethyl)pyrrolidine, and 1.2 eq. of potassium cyanide at 70 °C in the presence of water was the optimal condition for the Strecker reaction.
| Entry | Solvent | KCN (eq.) | Temp. (°C) | Time (h) | 3ab (%) | 4ab (%) |
|---|---|---|---|---|---|---|
a Reaction conditions: benzaldehyde (1.0 eq.), (S)-2-(azidomethyl)pyrrolidine (1.1 eq.) and potassium cyanide was heated in different solvent at different temperature in screw caped vial.b Isolated yields after flash chromatography.c dr: 86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 determined by 1H NMR spectroscopy of crude reaction mixture. 14 determined by 1H NMR spectroscopy of crude reaction mixture. |
||||||
| 1 | Toluene | 1.5 | 111 | 30 | 28 | 1 |
| 2 | DCM | 1.5 | 40 | 36 | 24 | 1.5 |
| 3 | CH3CN | 1.5 | 80 | 40 | 17 | 1 |
| 4 | CH3OH | 1.5 | 65 | 22 | 23 | 0 |
| 5 | Dioxane | 1.5 | 101 | 32 | Traces | 0 |
| 6 | DMF | 1.5 | 140 | 20 | Traces | 0 |
| 7 | DMSO | 1.5 | 145 | 25 | Traces | 0 |
| 8 | H2O | 1.5 | 110 | 10 | 69 | 1.3 |
| 9 | H2O | 1.5 | 100 | 8 | 76 | 1.5 |
| 10 | H2O | 1.5 | 90 | 8 | 81 | 2 |
| 11 | H2O | 1.5 | 80 | 8 | 86 | 1.5 |
| 12c | H2O | 1.5 | 70 | 8 | 93 | 1.8 |
| 13 | H2O | 1.5 | 60 | 10 | 87 | 1 |
| 14 | H2O | 1.5 | 50 | 13 | 76 | 0 |
| 15 | H2O | 1.5 | 40 | 18 | 69 | 0 |
| 16 | H2O | 1.5 | RT | 34 | 64 | 0 |
| 17 | H2O | 1.0 | 70 | 8 | 89 | 1.8 |
| 18c | H2O | 1.2 | 70 | 8 | 93 | 2 |
After determining the optimized conditions for the synthesis of 3a, we developed the intramolecular [3+2] azide–nitrile cycloaddition to obtain 4a. The optimization studies for the synthesis of 3a, in some reactions, we obtained some amounts of 4a. Therefore, we screened various solvents for the intramolecular [3+2] azide–nitrile cycloaddition to obtain the tricyclic product. First, we heated 3a in water at 100 °C, which produced 4a in a 20% yield and 88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 diastereoselectivity (Table 2, entry 1). We then screened toluene, acetonitrile, dimethyl sulfoxide, and dimethylformamide at 111, 80, 160, and 150 °C, which afforded 4a in 58%, 63%, 87%, and 94% yields and 86
12 diastereoselectivity (Table 2, entry 1). We then screened toluene, acetonitrile, dimethyl sulfoxide, and dimethylformamide at 111, 80, 160, and 150 °C, which afforded 4a in 58%, 63%, 87%, and 94% yields and 86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14, 89
14, 89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11, 90
11, 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, 91
10, 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 diastereoselectivities respectively (Table 2, entries 2–5). Therefore, heating 3a in the presence of dimethylformamide without any catalyst was the optimal condition for the intramolecular [3+2] azide–nitrile cycloaddition.
9 diastereoselectivities respectively (Table 2, entries 2–5). Therefore, heating 3a in the presence of dimethylformamide without any catalyst was the optimal condition for the intramolecular [3+2] azide–nitrile cycloaddition.
| Entry | Solvent | T (°C) | Time (h) | 4ab (%) | drc |
|---|---|---|---|---|---|
| a Reaction conditions: (0.2 mmol) of (R)-2-((S)-2-(azidomethyl)pyrrolidin-1-yl)-2-phenylacetonitriles (3a) was heated in various solvent at different temperature in oven dried, argon flushed, screw caped sealed tube.b Isolated yields after flash chromatography.c Diastereomeric ratio determined by 1H NMR spectroscopy of crude reaction mixture. | |||||
| 1 | H2O | 100 | 12 | 20 | 88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 12 |
| 2 | Toluene | 111 | 24 | 58 | 86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 14 |
| 3 | CH3CN | 80 | 72 | 63 | 89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 11 |
| 4 | DMSO | 160 | 16 | 87 | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
| 5 | DMF | 150 | 14 | 94 | 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 9 |
Under the optimized reaction conditions, the Strecker reaction and the intramolecular [3+2] azide–nitrile cycloaddition was investigated to illustrate the efficiency of this strategy; Table 3 outlines the obtained results. Various aromatic, aliphatic, and heterocyclic aldehydes were explored. Substituted benzaldehydes bearing electron-donating and electron-withdrawing groups at o-, m-, and p-positions, such as methyl, methoxy, fluoro, chloro, bromo, and cyano groups, generated the corresponding products 3a–3p in satisfactory to excellent yields and diastereoselectivities. Products 3a–3p underwent the intramolecular [3+2] cycloaddition to form the corresponding 4a–4p in satisfactory to excellent yields and diastereoselectivities (Table 3, entries 1–16). 2,4,6-Trimethoxybenzaldehyde reacted smoothly and produced an excellent yield and satisfactory diastereoselectivity of 3q; 3q then underwent the intramolecular [3+2] cycloaddition to form tricyclic 4q in an excellent yield and satisfactory distereoselectivity (Table 3, entry 17). Although 1-napthaldehyde is bulky, it reacted well under optimized conditions and produced 3r in an excellent yield and satisfactory diastereoselectivity; 3r then underwent the intramolecular [3+2] cycloaddition to form 4r in an excellent yield and satisfactory diastereoselectivity (Table 3, entry 18). Aliphatic aldehydes such as isovaleraldehyde and dihydrocenamaldehyde exhibited high reactivity under the optimized reaction conditions and generated 3s and 3t in satisfactory yields and diastereoselectivities; 3s and 3t then underwent the intramolecular [3+2] cycloaddition to form 4s and 4t in satisfactory yields and diastereoselectivities (Table 3, entries 19–20). Heterocyclic aldehydes such as 3-thienylcarbaldehyde and bulky 3-thionapthaldehyde showed satisfactory reactivities under the optimized reaction conditions and generated 3u and 3v, in satisfactory diastereoselectivities and excellent yields; 3u and 3v then underwent the intramolecular [3+2] cycloaddition to form 4u and 4v in excellent yields and satisfactory distereoselectivities (Table 3, entry 21–22). The electronic properties of the substituents on the phenyl ring did not affect the yields of the corresponding products. Due to inseparable diastereomeric mixture of products 3a–3v by flash chromatography we carry forward these products to next step [3+2] cycloaddition to form products 4a–4v. The product 3a was confirmed using 1H NMR, 13C NMR, IR, and mass spectrometry. In the 1H NMR (CDCl3) spectrum of 3a, a methine proton (N–CH–) at δ 5.31 ppm was observed as a characteristic singlet; the 13C NMR spectrum peak at δ 116.4 ppm and the IR spectrum band at 2100 cm−1 corresponded to the nitrile functionality of 3a and the characteristic ion at m/z = 241 was attributed to the [M]+ ion peak in the mass spectrum (EI). The product 4a was recrystallized and confirmed using X-ray diffraction analysis of monocrystal, which is clarified that newly formed stereocenter exhibited an (R) configuration. From X-ray study (Fig. 2), we assigned the absolute configuration of major and minor diastereomers as shown in Scheme 1.
| Entry | R | Productsa (dr)d | Yieldc (%) | Productsb (dr)d | Yieldc (%) |
|---|---|---|---|---|---|
| a Reaction conditions: aldehyde (1.0 eq.), (S)-2-(azidomethyl)pyrrolidine (1.1 eq.), KCN (1.2 eq.) and 0.1 mL water heated at 70 °C in screw caped vial.b Aminonitriles (1 eq.) in 2 mL dimethylformamide were heated at140–150 °C in screw caped sealed tube.c Isolated yields after flash chromatography.d Diastereomeric ratio determined by 1H NMR spectroscopy of crude reaction mixture. | |||||
| 1 | C6H5 | 3a (86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14) 14) |
93 | 4a (91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) 9) |
94 |
| 2 | o-CH3C6H4 | 3b (72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28) 28) |
88 | 4b (96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) 4) |
81 |
| 3 | m-CH3C6H4 | 3c (70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30) 30) |
90 | 4c (91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) 9) |
80 |
| 4 | p-CH3C6H4 | 3d (79![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 21) 21) |
91 | 4d (89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11) 11) |
88 |
| 5 | m-CH3OC6H4 | 3e (86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14) 14) |
89 | 4e (91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) 9) |
89 |
| 6 | p-CH3OC6H4 | 3f (76![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 24) 24) |
87 | 4f (92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8) 8) |
86 |
| 7 | o-FC6H4 | 3g (72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28) 28) |
90 | 4g (77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23) 23) |
90 |
| 8 | m-FC6H4 | 3h (82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18) 18) |
89 | 4h (91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) 9) |
81 |
| 9 | p-FC6H4 | 3i (86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14) 14) |
90 | 4i (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) 5) |
90 |
| 10 | o-ClC6H4 | 3j (67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33) 33) |
87 | 4j (87![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13) 13) |
77 |
| 11 | m-ClC6H4 | 3k (92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8) 8) |
84 | 4k (90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) 10) |
91 |
| 12 | p-ClC6H4 | 3l (94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6) 6) |
89 | 4l (96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) 4) |
93 |
| 13 | o-BrC6H4 | 3m (60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40) 40) |
90 | 4m (88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12) 12) |
71 |
| 14 | m-BrC6H4 | 3n (93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7) 7) |
91 | 4n (94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6) 6) |
92 |
| 15 | m-CNC6H4 | 3o (90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) 10) |
91 | 4o (98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
91 |
| 16 | p-CNC6H4 | 3p (97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 03) 03) |
92 | 4p (99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
93 |
| 17 | 2,4,6-(CH3O)3C6H2 | 3q (70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30) 30) |
93 | 4q (71![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 29) 29) |
88 |
| 18 | 1-Naphthyl | 3r (70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30) 30) |
90 | 4r (71![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 29) 29) |
91 |
| 19 | (CH3)2CHCH2 | 3s (93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 07) 07) |
91 | 4s (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) 5) |
81 |
| 20 | C6H5(CH2)2 | 3t (75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25) 25) |
88 | 4t (78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22) 22) |
87 |
| 21 | 3-Thienyl | 3u (82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18) 18) |
91 | 4u (82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18) 18) |
88 |
| 22 | Benzothiophen-3-yl | 3v (74![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 26) 26) |
89 | 4v (80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20) 20) |
89 |
Further structure of major diastereomer characterized using 1H NMR, 13C NMR, IR, and mass spectrometry. In the 1H NMR (CDCl3) spectrum of major diastereomer, a methine proton (N–CH–) at δ 4.60 ppm was observed as a characteristic singlet, in the 13C NMR spectrum, peak at δ 154.32 ppm corresponded to the carbon atom of the tetrazole ring, and the characteristic ion at m/z = 241 was attributed to the [M]+ ion peak in its mass spectrum (EI). Structure of minor diastereomer identified using 1H NMR, 13C NMR and IR. In the 1H NMR (CDCl3) spectrum of minor diastereomer a methine proton (N–CH–) at δ 5.00 ppm was observed as a characteristic singlet, and in the 13C NMR spectrum, peak at δ 153.06 ppm corresponded to the carbon atom of the tetrazole ring.
A plausible reaction mechanism for the synthesis of 3a and 4a is outlined in Scheme 2. Benzaldehyde (1a) reacted with (S)-2-(azidomethyl)pyrrolidine (2a) to form iminium ion, ‘re-face’ attack of cyanide on electron deficient carbon atom of iminium ion to form 3a, which is underwent intramolecular [3+2] cycloaddition on heating to form 4a.
To check safety, we studied Strecker reaction using 2-azidoethanamine (molecular weight: 86.09). It is safe to work with low molecular weight azide starting material, no any explosion was observed during course of reaction
 | ||
| Fig. 2 Crystal structure of (5aS,10R)-10-phenyl-5,5a,6,7,8,10-hexahydropyrrolo[1,2-a]tetrazolo[1,5-d]pyrazine (CCDC: 1041454) (major diastereomer, as structure shown in Scheme 1). | ||
Conclusion
In summary, we developed a simple, efficient and catalyst-free synthesis of (5aS,10R)-10-phenyl-5,5a,6,7,8,10-hexahydropyrrolo[1,2-a]tetrazolo[1,5-d]pyrazines through a three-component Strecker reaction, followed by an intramolecular [3+2] cycloaddition. The process is applicable for a wide range of aromatic, aliphatic, and heterocyclic aldehydes. Because the methodology does not involve using expensive starting materials, it is attractive for industrial and pharmaceutical applications.Experimental section
General experimental procedure for KCN/H2O mediated Strecker reaction
A mixture of aldehyde (1.0 mmol), (S)-2-(azidomethyl)pyrrolidine (1.1 mmol), KCN (1.2 mmol) and H2O (100 μL) in screw caped vial was stirred for 6–8 h at 70 °C. After completion of the reaction (monitored by TLC), the mixture was diluted with dichloromethane and washed with water, and the aqueous phase was extracted with dichloromethane, the combined organic extracts were dried over MgSO4, concentrated under reduced pressure and purified by column chromatography using hexane/EtOAc as eluent to give the desired product. (Note: aqueous phase was combined with (aq.) potassium permanganate solution and discarded in ventilated hood!!!). The products were further identified by FT-IR, 1H and 13C NMR spectroscopy, LRMS, HRMS, and all data were all in good agreement with assigned structures the characterization data of aminonitriles (3a–3v) are as follows:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −51.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.58–7.53 (m, 3H), 7.40–7.37 (m, 2H), 5.31 (s, 1H), 3.54–3.49 (dd, J1 = 4.3 Hz, J2 = 4.2 Hz, 1H), 3.34–3.28 (m, 1H), 3.19–3.16 (m, 1H), 2.69–2.67 (m, 1H), 2.59–2.51 (m, 1H), 2.10–2.01 (m, 1H), 1.81–1.74 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 133.9, 128.7, 128.0, 127.4, 116.4, 60.9, 58.6, 55.0, 49.3, 28.7, 33.0; IR (KBr, thin film cm−1): 2100, 1629, 1450, 1271, 1123, 910, 703; LRMS-EI (m/z): 241 (0.3), 219 (1), 199 (3), 185 (82), 174 (6), 149 (6), 116 (100), 105 (14), 89 (10), 77 (7), 55 (2); HRMS-EI (m/z): M+ calcd for C13H15N5, 241.1327; found: 241.1330.
9), pale yellow oil, [α]25D = −51.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.58–7.53 (m, 3H), 7.40–7.37 (m, 2H), 5.31 (s, 1H), 3.54–3.49 (dd, J1 = 4.3 Hz, J2 = 4.2 Hz, 1H), 3.34–3.28 (m, 1H), 3.19–3.16 (m, 1H), 2.69–2.67 (m, 1H), 2.59–2.51 (m, 1H), 2.10–2.01 (m, 1H), 1.81–1.74 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 133.9, 128.7, 128.0, 127.4, 116.4, 60.9, 58.6, 55.0, 49.3, 28.7, 33.0; IR (KBr, thin film cm−1): 2100, 1629, 1450, 1271, 1123, 910, 703; LRMS-EI (m/z): 241 (0.3), 219 (1), 199 (3), 185 (82), 174 (6), 149 (6), 116 (100), 105 (14), 89 (10), 77 (7), 55 (2); HRMS-EI (m/z): M+ calcd for C13H15N5, 241.1327; found: 241.1330.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9); pale yellow oil, [α]25D = −42.8° (c 1.0, CHCl3); 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.64–7.56 (m, 1H), 7.35–7.25 (m, 1H), 7.22–7.19 (m, 2H), 5.40 (s, 1H), 3.50–3.44 (dd, J1 = 5.0 Hz, J2 = 6.0 Hz, 1H), 3.24–3.21 (m 1H), 3.14–3.09 (m, 1H), 2.62–2.51 (m, 2H), 2.42 (s, 3H), 2.08–2.02 (m, 1H), 1.90–1.87 (m, 1H), 1.76–1.73 (m, 2H); 13C NMR (75 MHz, CDCl3, ppm) δ: 136.7, 131.8, 131.0, 128.1, 126.0, 116.6, 60.7, 56.5, 55.2, 49.4, 28.9, 22.6, 18.8; IR (KBr, thin film, cm−1): 2966, 2100, 1461, 1272, 1127, 918, 732; LRMS-EI (m/z): 259 (0.07), 258 (0.1), 255 (0.2), 252 (0.1), 251 (0.7), 249 (0.08), 248 (0.1), 246 (0.07), 213 (0.6), 199 (73), 185 (1), 173 (2), 144 (2), 116 (2), 105 (5), 99 (1), 77 (8), 55 (2); HRMS-EI (m/z): M+ calcd for C14H17N5, 255.1484; found: 255.1480.
9); pale yellow oil, [α]25D = −42.8° (c 1.0, CHCl3); 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.64–7.56 (m, 1H), 7.35–7.25 (m, 1H), 7.22–7.19 (m, 2H), 5.40 (s, 1H), 3.50–3.44 (dd, J1 = 5.0 Hz, J2 = 6.0 Hz, 1H), 3.24–3.21 (m 1H), 3.14–3.09 (m, 1H), 2.62–2.51 (m, 2H), 2.42 (s, 3H), 2.08–2.02 (m, 1H), 1.90–1.87 (m, 1H), 1.76–1.73 (m, 2H); 13C NMR (75 MHz, CDCl3, ppm) δ: 136.7, 131.8, 131.0, 128.1, 126.0, 116.6, 60.7, 56.5, 55.2, 49.4, 28.9, 22.6, 18.8; IR (KBr, thin film, cm−1): 2966, 2100, 1461, 1272, 1127, 918, 732; LRMS-EI (m/z): 259 (0.07), 258 (0.1), 255 (0.2), 252 (0.1), 251 (0.7), 249 (0.08), 248 (0.1), 246 (0.07), 213 (0.6), 199 (73), 185 (1), 173 (2), 144 (2), 116 (2), 105 (5), 99 (1), 77 (8), 55 (2); HRMS-EI (m/z): M+ calcd for C14H17N5, 255.1484; found: 255.1480.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −41.6° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.64–7.56 (m, 1H), 7.32–7.30 (m, 1H), 7.22–7.19 (m, 2H), 5.40 (s, 1H), 3.50–3.44 (dd, J1 = 5.0 Hz, J2 = 6.0 Hz, 1H), 3.23–3.21 (m, 1H), 3.14–3.09 (m, 1H), 2.67–2.51 (m, 2H), 2.42 (s, 3H), 2.07–2.02 (m, 1H), 1.90–1.87 (m, 1H), 1.76–1.64 (m, 2H); 13C NMR (75 MHz, CDCl3, ppm) δ: 138.4, 135.0, 133.7, 129.3, 128.4, 128.9, 116.4, 60.9, 58.4, 54.7, 49.1, 28.6, 22.9, 21.1; IR (KBr, thin film, cm−1): 2967, 2100, 1459, 1259, 1124, 916, 729; LRMS-EI (m/z): 255 (0.1), 229 (0.2), 213 (0.6), 199 (72), 185 (0.8), 172 (2), 130 (100), 103 (27), 91 (4), 77 (9), 70 (15), 55 (2); HRMS-EI (m/z): M+ calcd for C14H17N5, 255.1484; found: 255.1478.
9), pale yellow oil, [α]25D = −41.6° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.64–7.56 (m, 1H), 7.32–7.30 (m, 1H), 7.22–7.19 (m, 2H), 5.40 (s, 1H), 3.50–3.44 (dd, J1 = 5.0 Hz, J2 = 6.0 Hz, 1H), 3.23–3.21 (m, 1H), 3.14–3.09 (m, 1H), 2.67–2.51 (m, 2H), 2.42 (s, 3H), 2.07–2.02 (m, 1H), 1.90–1.87 (m, 1H), 1.76–1.64 (m, 2H); 13C NMR (75 MHz, CDCl3, ppm) δ: 138.4, 135.0, 133.7, 129.3, 128.4, 128.9, 116.4, 60.9, 58.4, 54.7, 49.1, 28.6, 22.9, 21.1; IR (KBr, thin film, cm−1): 2967, 2100, 1459, 1259, 1124, 916, 729; LRMS-EI (m/z): 255 (0.1), 229 (0.2), 213 (0.6), 199 (72), 185 (0.8), 172 (2), 130 (100), 103 (27), 91 (4), 77 (9), 70 (15), 55 (2); HRMS-EI (m/z): M+ calcd for C14H17N5, 255.1484; found: 255.1478.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −47.1° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.42–7.40 (d, J = 6.0 Hz, 2H), 7.28–7.18 (m, 2H), 5.26 (s, 1H), 3.52–3.47 (dd, J1 = 4.3 Hz, J2 = 4.2 Hz, 1H), 3.32–3.26 (m, 1H), 3.18–3.14 (m, 1H), 2.72–2.66 (m, 1H), 2.58–2.46 (m, 1H), 2.44 (s, 3H), 2.09–2.03 (m, 1H), 1.82–1.69 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 138.4, 130.9, 129.4, 129.2, 127.6, 127.2, 116.5, 60.8, 58.2, 54.7, 49.1, 28.6, 22.9, 20.9; IR (KBr, thin film, cm−1): 2961, 2100, 1684, 1511, 1448, 1270, 1201, 1122, 908, 839, 768; LRMS-EI (m/z): 255 (0.1), 239 (0.06), 229 (0.2), 199 (57), 173 (0.9), 149 (1), 130 (100), 103 (7), 91 (2), 77 (5), 70 (12), 55 (0.8); HRMS-EI (m/z): M+ calcd for C14H17N5, 255.1484; found 255.1480.
9), pale yellow oil, [α]25D = −47.1° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.42–7.40 (d, J = 6.0 Hz, 2H), 7.28–7.18 (m, 2H), 5.26 (s, 1H), 3.52–3.47 (dd, J1 = 4.3 Hz, J2 = 4.2 Hz, 1H), 3.32–3.26 (m, 1H), 3.18–3.14 (m, 1H), 2.72–2.66 (m, 1H), 2.58–2.46 (m, 1H), 2.44 (s, 3H), 2.09–2.03 (m, 1H), 1.82–1.69 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 138.4, 130.9, 129.4, 129.2, 127.6, 127.2, 116.5, 60.8, 58.2, 54.7, 49.1, 28.6, 22.9, 20.9; IR (KBr, thin film, cm−1): 2961, 2100, 1684, 1511, 1448, 1270, 1201, 1122, 908, 839, 768; LRMS-EI (m/z): 255 (0.1), 239 (0.06), 229 (0.2), 199 (57), 173 (0.9), 149 (1), 130 (100), 103 (7), 91 (2), 77 (5), 70 (12), 55 (0.8); HRMS-EI (m/z): M+ calcd for C14H17N5, 255.1484; found 255.1480.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −51.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.33–7.28 (m, 1H), 7.15–7.09 (m, 2H), 6.91–6.88 (d, J = 9.0 Hz, 1H), 5.29 (s, 1H), 3.82 (s, 3H), 3.54–3.49 (dd, J1 = 3.7 Hz, J2 = 4.2 Hz, 1H), 3.32–3.26 (m, 1H), 3.20–3.17 (m, 1H), 2.74–2.71 (m, 1H), 2.60–2.51 (m, 1H), 2.10–2.01 (m, 1H), 1.80–1.71 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 159.8, 135.3, 129.7, 119.5, 116.4, 114.3, 112.8, 61.0, 58.5, 55.2, 54.8, 49.3, 28.6, 23.1; IR (KBr, thin film, cm−1): 2967, 2100, 1445, 1267, 1122, 910, 699; LRMS-EI (m/z): 271 (0.1), 245 (0.2), 229 (0.4), 215 (84), 171 (0.3), 146 (100), 116 (7), 103 (5), 89 (4), 77 (2), 55 (3), 50 (0.04); HRMS-EI (m/z): M+ calcd for C14H17N5O, 271.1433; found: 271.1435.
9), pale yellow oil, [α]25D = −51.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.33–7.28 (m, 1H), 7.15–7.09 (m, 2H), 6.91–6.88 (d, J = 9.0 Hz, 1H), 5.29 (s, 1H), 3.82 (s, 3H), 3.54–3.49 (dd, J1 = 3.7 Hz, J2 = 4.2 Hz, 1H), 3.32–3.26 (m, 1H), 3.20–3.17 (m, 1H), 2.74–2.71 (m, 1H), 2.60–2.51 (m, 1H), 2.10–2.01 (m, 1H), 1.80–1.71 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 159.8, 135.3, 129.7, 119.5, 116.4, 114.3, 112.8, 61.0, 58.5, 55.2, 54.8, 49.3, 28.6, 23.1; IR (KBr, thin film, cm−1): 2967, 2100, 1445, 1267, 1122, 910, 699; LRMS-EI (m/z): 271 (0.1), 245 (0.2), 229 (0.4), 215 (84), 171 (0.3), 146 (100), 116 (7), 103 (5), 89 (4), 77 (2), 55 (3), 50 (0.04); HRMS-EI (m/z): M+ calcd for C14H17N5O, 271.1433; found: 271.1435.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −45.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.45–7.42 (d, J = 9.0 Hz, 2H), 6.94–6.89 (m, 2H), 5.23 (s, 1H), 3.81 (s, 3H), 3.52–3.46 (dd, J1 = 4.3 Hz, J2 = 4.2 Hz, 1H), 3.31–3.25 (m, 1H), 3.17–3.13 (m, 1H), 2.71–2.66 (m, 1H), 2.57–2.47 (m, 1H), 2.09–2.00 (m, 1H), 1.81–1.75 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 159.6, 131.8, 128.5, 125.6, 116.6, 114.1, 113.9, 60.8, 57.9, 55.4, 55.1, 54.8, 49.8, 28.6, 22.9; IR (KBr, thin film, cm−1): 2967, 2100, 1695, 1506, 1248, 1028, 908, 834; LRMS-EI (m/z): 271 (0.01), 265 (0.02), 245 (0.2), 217 (0.4), 215 (29), 189 (0.4), 146 (100), 135 (11), 103 (4), 91 (3), 77 (5), 65 (2), 51 (1); HRMS-EI (m/z): M+ calcd for C14H17N5O, 271.1433, found: 271.1437.
9), pale yellow oil, [α]25D = −45.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.45–7.42 (d, J = 9.0 Hz, 2H), 6.94–6.89 (m, 2H), 5.23 (s, 1H), 3.81 (s, 3H), 3.52–3.46 (dd, J1 = 4.3 Hz, J2 = 4.2 Hz, 1H), 3.31–3.25 (m, 1H), 3.17–3.13 (m, 1H), 2.71–2.66 (m, 1H), 2.57–2.47 (m, 1H), 2.09–2.00 (m, 1H), 1.81–1.75 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 159.6, 131.8, 128.5, 125.6, 116.6, 114.1, 113.9, 60.8, 57.9, 55.4, 55.1, 54.8, 49.8, 28.6, 22.9; IR (KBr, thin film, cm−1): 2967, 2100, 1695, 1506, 1248, 1028, 908, 834; LRMS-EI (m/z): 271 (0.01), 265 (0.02), 245 (0.2), 217 (0.4), 215 (29), 189 (0.4), 146 (100), 135 (11), 103 (4), 91 (3), 77 (5), 65 (2), 51 (1); HRMS-EI (m/z): M+ calcd for C14H17N5O, 271.1433, found: 271.1437.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −42.8° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.57–7.54 (m, 1H), 7.39–7.36 (m, 1H), 7.20–7.10 (m, 2H), 5.51 (s, 1H), 3.52–3.46 (dd, J1 = 4.7 Hz, J2 = 4.7 Hz, 1H), 3.35–3.29 (m, 1H), 3.13–3.10 (m, 1H), 2.72–2.68 (m, 2H), 2.68–2.02 (m, 1H), 1.87–1.72 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 162.8 (d, J = 245.0 Hz), 136.4 (d, J = 7.2 Hz), 130.2 (d, J = 8.1 Hz), 122.1 (d, J = 2.8 Hz), 121.6 (d, J = 13.0 Hz), 116.0, 115.5, 114.6, 114.3, 60.9, 58.1, 55.0, 49.2, 28.6, 23.0; IR (KBr, thin film, cm−1): 2967, 2100, 1492, 1270, 1097, 921, 806, 757; LRMS-EI (m/z): 259 (0.02), 258 (0.02), 233 (0.3), 215 (0.7), 203 (93), 177 (0.6), 163 (0.3), 146 (2), 134 (100), 107 (8), 83 (1), 68 (2), 57 (1); HRMS-EI (m/z): M+ calcd for C13H14FN5, 259.1233; found 259.1237.
9), pale yellow oil, [α]25D = −42.8° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.57–7.54 (m, 1H), 7.39–7.36 (m, 1H), 7.20–7.10 (m, 2H), 5.51 (s, 1H), 3.52–3.46 (dd, J1 = 4.7 Hz, J2 = 4.7 Hz, 1H), 3.35–3.29 (m, 1H), 3.13–3.10 (m, 1H), 2.72–2.68 (m, 2H), 2.68–2.02 (m, 1H), 1.87–1.72 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 162.8 (d, J = 245.0 Hz), 136.4 (d, J = 7.2 Hz), 130.2 (d, J = 8.1 Hz), 122.1 (d, J = 2.8 Hz), 121.6 (d, J = 13.0 Hz), 116.0, 115.5, 114.6, 114.3, 60.9, 58.1, 55.0, 49.2, 28.6, 23.0; IR (KBr, thin film, cm−1): 2967, 2100, 1492, 1270, 1097, 921, 806, 757; LRMS-EI (m/z): 259 (0.02), 258 (0.02), 233 (0.3), 215 (0.7), 203 (93), 177 (0.6), 163 (0.3), 146 (2), 134 (100), 107 (8), 83 (1), 68 (2), 57 (1); HRMS-EI (m/z): M+ calcd for C13H14FN5, 259.1233; found 259.1237.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −53.3° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.35–7.29 (m, 3H), 7.07–7.05 (m, 1H), 5.32 (s, 1H), 3.55–3.49 (dd, J1 = 3.9 Hz, J2 = 4.0 Hz, 1H), 3.34–3.29 (m, 1H), 3.20–3.16 (m, 1H), 2.71–2.69 (m, 1H), 2.56–2.50 (m, 1H), 2.10–2.04 (m, 1H), 1.81–1.72 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 160.1 (d, J = 249.0 Hz), 131.0 (d, J = 8.3 Hz), 129.2 (d, J = 2.6 Hz), 124.1 (d, J = 3.8 Hz), 121.7 (d, J = 12.9 Hz), 61.1, 54.7, 52.4, 52.3, 49.2, 28.7, 23.0; IR (KBr, thin film, cm−1): 2923, 2100, 1591, 1443, 1267, 1023, 798; LRMS-EI (m/z): 259 (0.03), 257 (0.2), 239 (0.1), 205 (1), 203 (100), 178 (6), 134 (94), 109 (11), 107 (7), 83 (3), 70 (4), 55 (3); HRMS-EI (m/z): M+ calcd for C13H14FN5, 259.1233; found: 259.1231.
9), pale yellow oil, [α]25D = −53.3° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.35–7.29 (m, 3H), 7.07–7.05 (m, 1H), 5.32 (s, 1H), 3.55–3.49 (dd, J1 = 3.9 Hz, J2 = 4.0 Hz, 1H), 3.34–3.29 (m, 1H), 3.20–3.16 (m, 1H), 2.71–2.69 (m, 1H), 2.56–2.50 (m, 1H), 2.10–2.04 (m, 1H), 1.81–1.72 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 160.1 (d, J = 249.0 Hz), 131.0 (d, J = 8.3 Hz), 129.2 (d, J = 2.6 Hz), 124.1 (d, J = 3.8 Hz), 121.7 (d, J = 12.9 Hz), 61.1, 54.7, 52.4, 52.3, 49.2, 28.7, 23.0; IR (KBr, thin film, cm−1): 2923, 2100, 1591, 1443, 1267, 1023, 798; LRMS-EI (m/z): 259 (0.03), 257 (0.2), 239 (0.1), 205 (1), 203 (100), 178 (6), 134 (94), 109 (11), 107 (7), 83 (3), 70 (4), 55 (3); HRMS-EI (m/z): M+ calcd for C13H14FN5, 259.1233; found: 259.1231.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −50.1° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.50–7.47 (d, J = 9.0 Hz, 2H), 7.42–7.35 (m, 2H), 5.29 (s, 1H), 3.54–3.49 (dd, J = 3.1 Hz, J = 4.0 Hz, 1H), 3.27–3.34 (m, 1H), 3.20–3.08 (m, 1H), 2.70–2.64 (m, 1H), 2.56–2.48 (m, 1H), 2.00–2.01 (m, 1H), 1.97–1.67 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 162.7 (d, J = 246.0 Hz), 130.0, 129.2, 129.1, 116.4, 115.8, 115.6, 60.9, 58.0, 55.1, 49.2, 28.7, 23.1; IR (KBr, thin film, cm−1): 2961, 2100, 1604, 1508, 1223, 834, 791; LRMS-EI (m/z): 259 (0.02), 257 (0.02), 241 (0.02), 229 (0.3), 205 (0.5), 203 (52), 177 (0.6), 148 (1), 134 (100), 107 (5), 83 (1), 68 (2), 57 (0.8); HRMS-EI (m/z): M+ calcd for C13H14FN5, 259.1233; found: 259.1239.
9), pale yellow oil, [α]25D = −50.1° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.50–7.47 (d, J = 9.0 Hz, 2H), 7.42–7.35 (m, 2H), 5.29 (s, 1H), 3.54–3.49 (dd, J = 3.1 Hz, J = 4.0 Hz, 1H), 3.27–3.34 (m, 1H), 3.20–3.08 (m, 1H), 2.70–2.64 (m, 1H), 2.56–2.48 (m, 1H), 2.00–2.01 (m, 1H), 1.97–1.67 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 162.7 (d, J = 246.0 Hz), 130.0, 129.2, 129.1, 116.4, 115.8, 115.6, 60.9, 58.0, 55.1, 49.2, 28.7, 23.1; IR (KBr, thin film, cm−1): 2961, 2100, 1604, 1508, 1223, 834, 791; LRMS-EI (m/z): 259 (0.02), 257 (0.02), 241 (0.02), 229 (0.3), 205 (0.5), 203 (52), 177 (0.6), 148 (1), 134 (100), 107 (5), 83 (1), 68 (2), 57 (0.8); HRMS-EI (m/z): M+ calcd for C13H14FN5, 259.1233; found: 259.1239.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −39.8° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.79–7.61 (d, J = 5.4 Hz, 1H), 7.64–7.61 (m, 1H), 7.36–7.31 (m, 2H), 5.59 (s, 1H), 3.53–3.47 (dd, J1 = 7.0 Hz, J2 = 5.5 Hz, 1H), 3.29–3.21 (m, 1H), 3.14–3.08 (m, 1H), 2.70–2.62 (m, 2H), 2.09–2.01 (m, 1H), 1.98–1.78 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 133.7, 131.4, 130.3, 130.2, 129.6, 126.8, 115.8, 61.1, 55.6, 54.3, 49.0, 28.7, 22.7; IR (KBr, thin film, cm−1): 2967, 2100, 1437, 1264, 1127, 924, 754; LRMS-EI (m/z): 278 (0.01), 275 (0.05), 249 (0.3), 219 (97), 192 (0.6), 150 (100), 123 (6), 89 (3), 68 (3), 50 (0.6); HRMS-EI (m/z): M+ calcd for C13H14ClN5, 275.0938; found: 275.0942.
9), pale yellow oil, [α]25D = −39.8° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.79–7.61 (d, J = 5.4 Hz, 1H), 7.64–7.61 (m, 1H), 7.36–7.31 (m, 2H), 5.59 (s, 1H), 3.53–3.47 (dd, J1 = 7.0 Hz, J2 = 5.5 Hz, 1H), 3.29–3.21 (m, 1H), 3.14–3.08 (m, 1H), 2.70–2.62 (m, 2H), 2.09–2.01 (m, 1H), 1.98–1.78 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 133.7, 131.4, 130.3, 130.2, 129.6, 126.8, 115.8, 61.1, 55.6, 54.3, 49.0, 28.7, 22.7; IR (KBr, thin film, cm−1): 2967, 2100, 1437, 1264, 1127, 924, 754; LRMS-EI (m/z): 278 (0.01), 275 (0.05), 249 (0.3), 219 (97), 192 (0.6), 150 (100), 123 (6), 89 (3), 68 (3), 50 (0.6); HRMS-EI (m/z): M+ calcd for C13H14ClN5, 275.0938; found: 275.0942.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −54.7° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.56–7.54 (d, J = 6.0 Hz, 1H), 7.44–7.43 (m, 1H), 7.34–7.32 (m, 2H), 5.31 (s, 1H), 3.55–3.49 (dd, J1 = 4.0 Hz, J2 = 4.2 Hz, 1H), 3.35–3.29 (m, 1H), 3.20–3.14 (m, 1H), 2.72–2.67 (m, 1H), 2.60–2.50 (m, 1H), 2.11–2.01 (m, 1H), 1.85–1.76 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 136.0, 134.8, 130.0, 129.0, 127.6, 125.5, 116.0, 61.0, 58.3, 55.3, 49.4, 28.8, 23.1; IR (KBr, thin film, cm−1): 2967, 2100, 1596, 1267, 1078, 888, 770; LRMS-EI (m/z): 275 (0.02), 249 (0.2), 233 (1), 219 (100), 184 (1), 150 (82), 123 (4), 89 (2), 70 (3), 55 (1); HRMS-EI (m/z): M+ calcd for C13H14ClN5, 275.0938; found: 275.0930.
9), pale yellow oil, [α]25D = −54.7° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.56–7.54 (d, J = 6.0 Hz, 1H), 7.44–7.43 (m, 1H), 7.34–7.32 (m, 2H), 5.31 (s, 1H), 3.55–3.49 (dd, J1 = 4.0 Hz, J2 = 4.2 Hz, 1H), 3.35–3.29 (m, 1H), 3.20–3.14 (m, 1H), 2.72–2.67 (m, 1H), 2.60–2.50 (m, 1H), 2.11–2.01 (m, 1H), 1.85–1.76 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 136.0, 134.8, 130.0, 129.0, 127.6, 125.5, 116.0, 61.0, 58.3, 55.3, 49.4, 28.8, 23.1; IR (KBr, thin film, cm−1): 2967, 2100, 1596, 1267, 1078, 888, 770; LRMS-EI (m/z): 275 (0.02), 249 (0.2), 233 (1), 219 (100), 184 (1), 150 (82), 123 (4), 89 (2), 70 (3), 55 (1); HRMS-EI (m/z): M+ calcd for C13H14ClN5, 275.0938; found: 275.0930.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −55.9° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.50–7.43 (d, J = 9.0 Hz, 2H), 7.38–7.35 (m, 2H), 5.29 (s, 1H), 3.54–3.49 (dd, J1 = 3.1 Hz, J2 = 4.0 Hz, 1H), 3.34–3.27 (m, 1H), 3.19–3.15 (m, 1H), 2.69–2.65 (m, 1H), 2.57–2.51 (m, 1H), 2.10–2.04 (m, 1H), 1.84–1.70 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 134.5, 132.5, 129.3, 129.0, 128.8, 128.7, 116.0, 60.9, 58.0, 55.0, 49.2, 28.6, 23.0; IR (KBr, thin film, cm−1): 2967, 2100, 1489, 1267, 1091, 1015, 908, 842; LRMS-EI (m/z): 275 (0.07), 249 (0.2), 233 (0.4), 219 (63), 193 (0.7), 152 (36), 150 (100), 114 (4), 89 (2), 68 (2), 55 (1); HRMS-EI (m/z): M+ calcd for C13H14ClN5, 275.0938; found: 275.0932.
9), pale yellow oil, [α]25D = −55.9° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.50–7.43 (d, J = 9.0 Hz, 2H), 7.38–7.35 (m, 2H), 5.29 (s, 1H), 3.54–3.49 (dd, J1 = 3.1 Hz, J2 = 4.0 Hz, 1H), 3.34–3.27 (m, 1H), 3.19–3.15 (m, 1H), 2.69–2.65 (m, 1H), 2.57–2.51 (m, 1H), 2.10–2.04 (m, 1H), 1.84–1.70 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 134.5, 132.5, 129.3, 129.0, 128.8, 128.7, 116.0, 60.9, 58.0, 55.0, 49.2, 28.6, 23.0; IR (KBr, thin film, cm−1): 2967, 2100, 1489, 1267, 1091, 1015, 908, 842; LRMS-EI (m/z): 275 (0.07), 249 (0.2), 233 (0.4), 219 (63), 193 (0.7), 152 (36), 150 (100), 114 (4), 89 (2), 68 (2), 55 (1); HRMS-EI (m/z): M+ calcd for C13H14ClN5, 275.0938; found: 275.0932.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −35.7° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.65–7.40 (m, 2H), 7.39–7.36 (m, 1H), 7.34–7.27 (m, 1H), 5.56 (s, 1H), 3.54–3.52 (d, J = 6.0 Hz, 1H), 3.14–3.11 (m, 1H), 3.09–3.08 (m, 1H), 2.68–2.59 (m, 2H), 2.09–2.01 (m, 1H), 1.99–1.75 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 135.0, 133.7, 133.0, 130.5, 129.9, 127.8, 124.0, 116.0, 61.22, 58.0, 54.3, 49.0, 28.8, 22.6; IR (KBr, thin film, cm−1): 2961, 2100, 1692, 1470, 1264, 1198, 924, 754; LRMS-ESI (m/z): 323 (15), 322 (95), 320 (100), 309 (0.4), 295 (4), 289 (0.6); HRMS-ESI (m/z): M+ calcd for C13H15BrN5, 320.0433, found: 320.0507.
9), pale yellow oil, [α]25D = −35.7° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.65–7.40 (m, 2H), 7.39–7.36 (m, 1H), 7.34–7.27 (m, 1H), 5.56 (s, 1H), 3.54–3.52 (d, J = 6.0 Hz, 1H), 3.14–3.11 (m, 1H), 3.09–3.08 (m, 1H), 2.68–2.59 (m, 2H), 2.09–2.01 (m, 1H), 1.99–1.75 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 135.0, 133.7, 133.0, 130.5, 129.9, 127.8, 124.0, 116.0, 61.22, 58.0, 54.3, 49.0, 28.8, 22.6; IR (KBr, thin film, cm−1): 2961, 2100, 1692, 1470, 1264, 1198, 924, 754; LRMS-ESI (m/z): 323 (15), 322 (95), 320 (100), 309 (0.4), 295 (4), 289 (0.6); HRMS-ESI (m/z): M+ calcd for C13H15BrN5, 320.0433, found: 320.0507.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), [α]25D = −55.3° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.69 (m, 1H), 7.50–7.48 (d, J = 6.0 Hz, 2H), 7.29–7.24 (m, 1H), 5.31 (s, 1H), 3.55–3.49 (dd, J1 = 4.1 Hz, J2 = 4.2 Hz, 1H), 3.35–3.29 (m, 1H), 3.18–3.13 (m, 1H), 2.72–2.67 (m, 1H), 2.60–2.53 (m, 1H), 2.11–2.04 (m, 1H), 1.85–1.70 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm): δ = 136.2, 131.8, 130.3, 130.2, 125.9, 122.8, 115.9, 60.9, 58.0, 55.1, 49.3, 28.7, 23.1; IR (KBr, thin film, cm−1): 2961, 2100, 1559, 1470, 1270, 1070, 930, 770; LRMS-ESI (m/z): 325 (2), 323 (16), 322 (93), 320 (100), 309 (2), 278 (5), 276 (12); HRMS-ESI (m/z): M+ calcd for C13H15BrN5, 320.0433; found: 320.0508.
9), [α]25D = −55.3° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.69 (m, 1H), 7.50–7.48 (d, J = 6.0 Hz, 2H), 7.29–7.24 (m, 1H), 5.31 (s, 1H), 3.55–3.49 (dd, J1 = 4.1 Hz, J2 = 4.2 Hz, 1H), 3.35–3.29 (m, 1H), 3.18–3.13 (m, 1H), 2.72–2.67 (m, 1H), 2.60–2.53 (m, 1H), 2.11–2.04 (m, 1H), 1.85–1.70 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm): δ = 136.2, 131.8, 130.3, 130.2, 125.9, 122.8, 115.9, 60.9, 58.0, 55.1, 49.3, 28.7, 23.1; IR (KBr, thin film, cm−1): 2961, 2100, 1559, 1470, 1270, 1070, 930, 770; LRMS-ESI (m/z): 325 (2), 323 (16), 322 (93), 320 (100), 309 (2), 278 (5), 276 (12); HRMS-ESI (m/z): M+ calcd for C13H15BrN5, 320.0433; found: 320.0508.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −53.5° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.87–7.79 (m, 2H), 7.68–7.65 (d, J = 9.0 Hz, 1H), 7.56–7.50 (m, 1H), 5.39 (s, 1H), 3.58–3.53 (dd, J1 = 3.9 Hz, J2 = 4.0 Hz, 1H), 3.38–3.32 (m, 1H), 3.21–3.17 (m, 1H), 2.67–2.62 (m, 1H), 2.58–2.53 (m, 1H), 2.12–2.06 (m, 1H), 1.87–1.68 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 139.1, 132.6, 128.2, 118.1, 115.6, 112.9, 61.0, 58.6, 55.4, 49.4, 28.7, 23.2; IR (KBr, thin film, cm−1): 2967, 2225, 2100, 1602, 1267, 910, 853; LRMS-ESI (m/z): 279 (0.5), 269 (1.5), 268 (16), 267 (100), 240 (4), 224 (0.7), 244 (5), 238 (3), 222 (2.5); HRMS-ESI (m/z): M+ calcd for C14H15N6, 267.1280; found: 267.1349.
9), pale yellow oil, [α]25D = −53.5° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.87–7.79 (m, 2H), 7.68–7.65 (d, J = 9.0 Hz, 1H), 7.56–7.50 (m, 1H), 5.39 (s, 1H), 3.58–3.53 (dd, J1 = 3.9 Hz, J2 = 4.0 Hz, 1H), 3.38–3.32 (m, 1H), 3.21–3.17 (m, 1H), 2.67–2.62 (m, 1H), 2.58–2.53 (m, 1H), 2.12–2.06 (m, 1H), 1.87–1.68 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 139.1, 132.6, 128.2, 118.1, 115.6, 112.9, 61.0, 58.6, 55.4, 49.4, 28.7, 23.2; IR (KBr, thin film, cm−1): 2967, 2225, 2100, 1602, 1267, 910, 853; LRMS-ESI (m/z): 279 (0.5), 269 (1.5), 268 (16), 267 (100), 240 (4), 224 (0.7), 244 (5), 238 (3), 222 (2.5); HRMS-ESI (m/z): M+ calcd for C14H15N6, 267.1280; found: 267.1349.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −57.7° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.69 (s, 4H), 5.39 (s, 1H), 3.57–3.52 (dd, J1 = 3.8 Hz, J2 = 3.7 Hz, 1H), 3.36–3.29 (m, 1H), 3.22–3.17 (m, 1H), 2.66–2.61 (m, 1H), 2.56–2.48 (m, 1H), 2.11–2.01 (m, 1H), 1.86–1.70 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 135.6, 132.2, 131.5, 130.8, 129.5, 117.9, 115.4, 112.8, 60.8, 57.9, 55.0, 49.1, 28.5, 22.9; IR (KBr, thin film, cm−1): 2972, 2225, 2100, 1478, 1275, 1124, 899, 680; LRMS-ESI (m/z): 285 (4), 269 (2), 268 (17), 267 (100), 256 (7); HRMS-ESI (m/z): M+ calcd for C14H15N6; 267.1280, found: 267.1351.
9), pale yellow oil, [α]25D = −57.7° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.69 (s, 4H), 5.39 (s, 1H), 3.57–3.52 (dd, J1 = 3.8 Hz, J2 = 3.7 Hz, 1H), 3.36–3.29 (m, 1H), 3.22–3.17 (m, 1H), 2.66–2.61 (m, 1H), 2.56–2.48 (m, 1H), 2.11–2.01 (m, 1H), 1.86–1.70 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 135.6, 132.2, 131.5, 130.8, 129.5, 117.9, 115.4, 112.8, 60.8, 57.9, 55.0, 49.1, 28.5, 22.9; IR (KBr, thin film, cm−1): 2972, 2225, 2100, 1478, 1275, 1124, 899, 680; LRMS-ESI (m/z): 285 (4), 269 (2), 268 (17), 267 (100), 256 (7); HRMS-ESI (m/z): M+ calcd for C14H15N6; 267.1280, found: 267.1351.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −41.6° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 6.12 (s, 2H), 5.45 (s, 1H), 3.85 (s, 9H), 3.46–3.40 (m, 2H), 3.25–3.10 (m, 1H), 2.86–3.79 (dd, J = 9.9 Hz, J = 8.1 Hz, 2H), 2.00–1.94 (m, 1H), 1.79–1.74 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 161.9, 161.8, 158.96, 158.8, 117.4, 103.5, 90.9, 60.4, 58.6, 55.7, 55.2, 53.8, 50.4, 45.9, 28.4, 23.3; IR (KBr, thin film, cm−1): 2961, 2100, 1459, 1369, 1116, 924; LRMS-ESI (m/z): 334 (2), 333 (17), 332 (100), 330 (3.3), 305 (3), 289 (0.3); HRMS-ESI (m/z): M+ calcd for C16H21N5O3, 332.1644; found: 332.1712.
9), pale yellow oil, [α]25D = −41.6° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 6.12 (s, 2H), 5.45 (s, 1H), 3.85 (s, 9H), 3.46–3.40 (m, 2H), 3.25–3.10 (m, 1H), 2.86–3.79 (dd, J = 9.9 Hz, J = 8.1 Hz, 2H), 2.00–1.94 (m, 1H), 1.79–1.74 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 161.9, 161.8, 158.96, 158.8, 117.4, 103.5, 90.9, 60.4, 58.6, 55.7, 55.2, 53.8, 50.4, 45.9, 28.4, 23.3; IR (KBr, thin film, cm−1): 2961, 2100, 1459, 1369, 1116, 924; LRMS-ESI (m/z): 334 (2), 333 (17), 332 (100), 330 (3.3), 305 (3), 289 (0.3); HRMS-ESI (m/z): M+ calcd for C16H21N5O3, 332.1644; found: 332.1712.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −41.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 8.28–8.25 (d, J = 9.0 Hz, 1H), 7.90–7.83 (m, 3H), 7.61–7.45 (m, 3H), 6.01 (s, 1H), 3.67–3.65 (m, 1H), 3.28–3.27 (m, 1H), 2.66–2.64 (m, 1H), 2.43–2.42 (m, 1H), 2.11–2.08 (m, 1H), 1.75–1.71 (m, 1H), 1.71–1.63 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 135.2, 133.8, 130.5, 129.9, 129.3, 128.61, 126.7, 126.2, 125.7, 124.8, 123.4, 116.7, 60.6, 57.0, 55.8, 49.6, 28.9, 22.6; IR (KBr, thin film, cm−1): 2956, 2100, 1690, 1448, 1267, 1122, 913, 781; LRMS-EI (m/z): 291 (0.03), 265 (0.2), 237 (0.6), 235 (40), 208 (1), 180 (1), 166 (100), 139 (10), 115 (2), 89 (0.7), 70 (1), 50 (0.1); HRMS-EI (m/z): M+ calcd for C17H17N5, 291.1484, found: 291.1486.
9), pale yellow oil, [α]25D = −41.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 8.28–8.25 (d, J = 9.0 Hz, 1H), 7.90–7.83 (m, 3H), 7.61–7.45 (m, 3H), 6.01 (s, 1H), 3.67–3.65 (m, 1H), 3.28–3.27 (m, 1H), 2.66–2.64 (m, 1H), 2.43–2.42 (m, 1H), 2.11–2.08 (m, 1H), 1.75–1.71 (m, 1H), 1.71–1.63 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 135.2, 133.8, 130.5, 129.9, 129.3, 128.61, 126.7, 126.2, 125.7, 124.8, 123.4, 116.7, 60.6, 57.0, 55.8, 49.6, 28.9, 22.6; IR (KBr, thin film, cm−1): 2956, 2100, 1690, 1448, 1267, 1122, 913, 781; LRMS-EI (m/z): 291 (0.03), 265 (0.2), 237 (0.6), 235 (40), 208 (1), 180 (1), 166 (100), 139 (10), 115 (2), 89 (0.7), 70 (1), 50 (0.1); HRMS-EI (m/z): M+ calcd for C17H17N5, 291.1484, found: 291.1486.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −54.8° (c 1.0, CHCl3); 1H NMR (CDCl3, 300 MHz, ppm) δ: 3.98–3.93 (t, J = 9.0 Hz, 1H), 3.36–3.31 (dd, J1 = 4.5 Hz, J2 = 4.4 Hz, 1H), 3.12–3.18 (m, 1H), 2.95–3.03 (m, 2H), 2.54–2.63 (m, 1H), 1.82–1.87 (m, 4H); 1.82–1.60 (m, 3H), 0.96 (d, J = 1.8 Hz, 3H), 0.94 (d, J = 1.8 Hz, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 118.3, 61.3, 54.7, 52.8, 48.7, 41.3, 28.4, 24.5, 23.2, 22.1, 22.0; IR (KBr, thin film, cm−1): 2956, 2100, 1467, 1270, 1141, 1039, 817, 655; LRMS-EI (m/z): 221 (0.7), 206 (0.5), 191 (2), 179 (2), 165 (100), 138 (8), 109 (18), 97 (1), 80 (3), 70 (21), 55 (5), 52 (0.4); HRMS-EI (m/z): M+ calcd for C11H19N5, 221.1640; found: 221.1648.
9), pale yellow oil, [α]25D = −54.8° (c 1.0, CHCl3); 1H NMR (CDCl3, 300 MHz, ppm) δ: 3.98–3.93 (t, J = 9.0 Hz, 1H), 3.36–3.31 (dd, J1 = 4.5 Hz, J2 = 4.4 Hz, 1H), 3.12–3.18 (m, 1H), 2.95–3.03 (m, 2H), 2.54–2.63 (m, 1H), 1.82–1.87 (m, 4H); 1.82–1.60 (m, 3H), 0.96 (d, J = 1.8 Hz, 3H), 0.94 (d, J = 1.8 Hz, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 118.3, 61.3, 54.7, 52.8, 48.7, 41.3, 28.4, 24.5, 23.2, 22.1, 22.0; IR (KBr, thin film, cm−1): 2956, 2100, 1467, 1270, 1141, 1039, 817, 655; LRMS-EI (m/z): 221 (0.7), 206 (0.5), 191 (2), 179 (2), 165 (100), 138 (8), 109 (18), 97 (1), 80 (3), 70 (21), 55 (5), 52 (0.4); HRMS-EI (m/z): M+ calcd for C11H19N5, 221.1640; found: 221.1648.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −42.3° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.34–7.31 (m, 2H), 7.29–7.19 (m, 3H), 3.85–3.79 (t, J = 7.8 Hz, 1H), 3.28–3.23 (dd, J1 = 4.6 Hz, J2 = 4.7 Hz, 1H), 3.12–3.06 (m, 2H), 2.98–2.90 (m, 1H), 2.83–2.78 (t, J = 7.2 Hz, 2H), 2.67–2.58 (m, 1H), 2.13–2.10 (m, 2H), 2.10–1.82 (m, 3H), 1.79–1.70 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 139.8, 128.5, 128.3, 126.4, 117.9, 61.4, 54.5, 53.6, 48.7, 33.9, 31.7, 28.4, 23.2; IR (KBr, thin film, cm−1): 2956, 2100, 1451, 1273, 1119, 908, 699; LRMS-EI (m/z): 269 (0.05), 263 (0.2), 241 (3), 213 (100), 172 (13), 145 (1), 109 (15), 91 (29), 70 (5), 54 (2); HRMS-EI (m/z): M+ calcd for C15H19N5, 269.1640; found: 269.1648.
9), pale yellow oil, [α]25D = −42.3° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.34–7.31 (m, 2H), 7.29–7.19 (m, 3H), 3.85–3.79 (t, J = 7.8 Hz, 1H), 3.28–3.23 (dd, J1 = 4.6 Hz, J2 = 4.7 Hz, 1H), 3.12–3.06 (m, 2H), 2.98–2.90 (m, 1H), 2.83–2.78 (t, J = 7.2 Hz, 2H), 2.67–2.58 (m, 1H), 2.13–2.10 (m, 2H), 2.10–1.82 (m, 3H), 1.79–1.70 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 139.8, 128.5, 128.3, 126.4, 117.9, 61.4, 54.5, 53.6, 48.7, 33.9, 31.7, 28.4, 23.2; IR (KBr, thin film, cm−1): 2956, 2100, 1451, 1273, 1119, 908, 699; LRMS-EI (m/z): 269 (0.05), 263 (0.2), 241 (3), 213 (100), 172 (13), 145 (1), 109 (15), 91 (29), 70 (5), 54 (2); HRMS-EI (m/z): M+ calcd for C15H19N5, 269.1640; found: 269.1648.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −54.1° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.44–7.36 (m, 1H), 7.35–7.33 (m, 1H), 7.18–7.16 (m, 1H), 5.26 (s, 1H), 3.51–3.45 (dd, J1 = 4.1 Hz, J2 = 4.0 Hz, 1H), 3.28–3.22 (m, 1H), 3.16–3.13 (m, 1H), 2.76–2.75 (m, 1H), 2.59–2.56 (m, 1H), 2.09–2.03 (m, 1H), 1.88–1.80 (m, 1H), 1.79–1.71 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 135.2, 126.9, 126.6, 123.6, 116.7, 61.0, 54.9, 54.6, 49.6, 28.7, 23.1; IR (KBr, thin film, cm−1): 2956, 2100, 1445, 1275, 888, 792; LRMS-ESI (m/z): 253 (1), 250 (6), 249 (15), 248 (100), 237 (3), 221 (2), 211 (1), 206 (3); HRMS-ESI (m/z): M+ calcd for C11H14N5S, 248.0964, found: 248.0966.
9), pale yellow oil, [α]25D = −54.1° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.44–7.36 (m, 1H), 7.35–7.33 (m, 1H), 7.18–7.16 (m, 1H), 5.26 (s, 1H), 3.51–3.45 (dd, J1 = 4.1 Hz, J2 = 4.0 Hz, 1H), 3.28–3.22 (m, 1H), 3.16–3.13 (m, 1H), 2.76–2.75 (m, 1H), 2.59–2.56 (m, 1H), 2.09–2.03 (m, 1H), 1.88–1.80 (m, 1H), 1.79–1.71 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 135.2, 126.9, 126.6, 123.6, 116.7, 61.0, 54.9, 54.6, 49.6, 28.7, 23.1; IR (KBr, thin film, cm−1): 2956, 2100, 1445, 1275, 888, 792; LRMS-ESI (m/z): 253 (1), 250 (6), 249 (15), 248 (100), 237 (3), 221 (2), 211 (1), 206 (3); HRMS-ESI (m/z): M+ calcd for C11H14N5S, 248.0964, found: 248.0966.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), pale yellow oil, [α]25D = −42.1° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 8.06–8.03 (m, 1H), 7.87–7.84 (m, 1H), 7.68–7.43 (m, 1H), 7.42–739 (m, 2H), 5.60 (s, 1H), 3.62–3.53 (dd, J1 = 4.5 Hz, J2 = 6.7 Hz, 1H), 3.24–3.19 (m, 1H), 2.65–2.59 (m, 2H), 2.12–2.07 (m, 1H), 1.97–1.93 (m, 1H), 1.78–1.66 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 140.7, 136.5, 128.5, 125.8, 125.0, 124.4, 122.7, 122.4, 116.2, 60.6, 55.7, 54.2, 49.6, 29.0, 22.82; IR (KBr, thin film, cm−1): 2961, 2100, 1679, 1426, 1270, 1135, 1045, 883, 757; LRMS-EI (m/z): 297 (0.03), 271 (0.1), 241 (25), 214 (0.5), 172 (100), 145 (5), 101 (0.9), 89 (0.8), 68 (1), 55 (0.3); HRMS-EI (m/z): M+ calcd for C15H15N5S, 297.1048, found: 297.1044.
9), pale yellow oil, [α]25D = −42.1° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 8.06–8.03 (m, 1H), 7.87–7.84 (m, 1H), 7.68–7.43 (m, 1H), 7.42–739 (m, 2H), 5.60 (s, 1H), 3.62–3.53 (dd, J1 = 4.5 Hz, J2 = 6.7 Hz, 1H), 3.24–3.19 (m, 1H), 2.65–2.59 (m, 2H), 2.12–2.07 (m, 1H), 1.97–1.93 (m, 1H), 1.78–1.66 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 140.7, 136.5, 128.5, 125.8, 125.0, 124.4, 122.7, 122.4, 116.2, 60.6, 55.7, 54.2, 49.6, 29.0, 22.82; IR (KBr, thin film, cm−1): 2961, 2100, 1679, 1426, 1270, 1135, 1045, 883, 757; LRMS-EI (m/z): 297 (0.03), 271 (0.1), 241 (25), 214 (0.5), 172 (100), 145 (5), 101 (0.9), 89 (0.8), 68 (1), 55 (0.3); HRMS-EI (m/z): M+ calcd for C15H15N5S, 297.1048, found: 297.1044.General experimental procedure for intramolecular [3+2] cycloaddition
A solution of aminonitrile (1 eq.) in DMF (2 mL) was heated at 140–150 °C in screw caped sealed tube for 14–16 h. After completion of the reaction (monitored by TLC), the mixture was diluted with diethyl ether and washed with water, and the aqueous phase was extracted with diethyl ether. The combined organic extracts were dried over MgSO4 and concentrated under reduced pressure and purified by column chromatography using n-hexane/EtOAc as eluent to give the desired product. The products were identified by FT-IR, 1H and 13C NMR spectroscopy, and LRMS, HRMS, and all data were all in good agreement with assigned structures. The characterization data of tricyclic fused tetrazoles given as below:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), pale yellow solid; mp: 189–190. [α]25D = −45.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm): δ = 7.36 (m, 5H), 4.77–4.72 (dd, J1 = 3.3 Hz, J2 = 3.4 Hz, 1H), 4.60 (s, 1H), 4.18–4.11 (t, J = 12.0 Hz, 1H), 3.02–2.98 (m, 2H), 2.34–2.25 (q, J = 9.0 Hz, 1H); 2.23–2.14 (m, 1H), 2.02–1.83 (m, 2H), 1.79–1.70 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.3, 137.0, 128.7, 128.4, 128.3, 64.0, 59.6, 51.7, 50.8, 27.6, 22.3; IR (KBr, thin film, cm−1): 2950, 2807, 1659, 1454, 1155, 1075, 1012, 751, 699; LRMS-EI (m/z): 241 (9), 229 (1.0), 187 (1.1), 185 (100), 159 (61), 143 (5), 131 (19), 116 (60), 104 (28), 89 (10), 77 (9.1), 55 (9.6); HRMS-EI (m/z): M+ calcd for C13H15N5, 241.1327; found: 241.1333.
7), pale yellow solid; mp: 189–190. [α]25D = −45.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm): δ = 7.36 (m, 5H), 4.77–4.72 (dd, J1 = 3.3 Hz, J2 = 3.4 Hz, 1H), 4.60 (s, 1H), 4.18–4.11 (t, J = 12.0 Hz, 1H), 3.02–2.98 (m, 2H), 2.34–2.25 (q, J = 9.0 Hz, 1H); 2.23–2.14 (m, 1H), 2.02–1.83 (m, 2H), 1.79–1.70 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.3, 137.0, 128.7, 128.4, 128.3, 64.0, 59.6, 51.7, 50.8, 27.6, 22.3; IR (KBr, thin film, cm−1): 2950, 2807, 1659, 1454, 1155, 1075, 1012, 751, 699; LRMS-EI (m/z): 241 (9), 229 (1.0), 187 (1.1), 185 (100), 159 (61), 143 (5), 131 (19), 116 (60), 104 (28), 89 (10), 77 (9.1), 55 (9.6); HRMS-EI (m/z): M+ calcd for C13H15N5, 241.1327; found: 241.1333.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7); yellow oil; [α]25D = −26.6° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.38–7.30 (m, 3H), 7.17–7.09 (m, 2H), 5.00 (s, 1H), 4.80–4.77 (dd, J1 = 3.5 Hz, J2 = 3.3 Hz, 1H), 4.13–4.20 (t, J = 11.7 Hz, 1H), 3.12–3.00 (m, 2H), 2.44–2.35 (q, J = 8.8 Hz, 1H), 2.28–2.17 (m, 1H), 2.02–1.96 (m, 2H), 1.82–1.75 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 153.0, 134.0, 127.7, 127.3, 125.5, 57.3, 50.4, 48.3, 29.5, 27.4, 22.1; IR (KBr, thin film, cm−1): 2950, 2805, 1659, 1450, 1155, 1075, 1012, 750.
7); yellow oil; [α]25D = −26.6° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.38–7.30 (m, 3H), 7.17–7.09 (m, 2H), 5.00 (s, 1H), 4.80–4.77 (dd, J1 = 3.5 Hz, J2 = 3.3 Hz, 1H), 4.13–4.20 (t, J = 11.7 Hz, 1H), 3.12–3.00 (m, 2H), 2.44–2.35 (q, J = 8.8 Hz, 1H), 2.28–2.17 (m, 1H), 2.02–1.96 (m, 2H), 1.82–1.75 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 153.0, 134.0, 127.7, 127.3, 125.5, 57.3, 50.4, 48.3, 29.5, 27.4, 22.1; IR (KBr, thin film, cm−1): 2950, 2805, 1659, 1450, 1155, 1075, 1012, 750.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −47.6° (c 1.0, CHCl3), 1NMR (CDCl3, 300 MHz, ppm) δ: 7.34–7.31 (d, J = 9.0 Hz, 2H), 7.23–7.07 (m, 2H), 4.92 (s, 1H), 4.78–4.73 (dd, J1 = 2.6 Hz, J2 = 2.6 Hz, 1H), 4.18–4.10 (t, J = 11.2 Hz, 1H), 3.09–2.97 (m, 2H), 2.58 (s, 3H), 2.26–2.19 (q, J = 7.4 Hz, 1H), 2.17–2.15 (m, 1H), 1.95–1.88 (m, 2H), 1.79–1.77 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.1, 130.7, 128.9, 128.1, 127.3, 126.4, 125.7, 59.6, 59.1, 51.5, 50.7, 27.6, 22.3, 19.7; IR (KBr, thin film, cm−1): 2923, 2807, 1603, 1464, 1156, 1077, 726, 700; LRMS-EI (m/z): 255 (18), 227 (14), 200 (8), 199 (74), 173 (54), 172 (65), 158 (12), 144 (29), 130 (100), 115 (17), 104 (19), 84 (15), 70 (11), 55 (8); HRMS-EI (m/z): M+ calcd for C14H17N5, 255.1484; found: 255.1480.
7), brown gum, [α]25D = −47.6° (c 1.0, CHCl3), 1NMR (CDCl3, 300 MHz, ppm) δ: 7.34–7.31 (d, J = 9.0 Hz, 2H), 7.23–7.07 (m, 2H), 4.92 (s, 1H), 4.78–4.73 (dd, J1 = 2.6 Hz, J2 = 2.6 Hz, 1H), 4.18–4.10 (t, J = 11.2 Hz, 1H), 3.09–2.97 (m, 2H), 2.58 (s, 3H), 2.26–2.19 (q, J = 7.4 Hz, 1H), 2.17–2.15 (m, 1H), 1.95–1.88 (m, 2H), 1.79–1.77 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.1, 130.7, 128.9, 128.1, 127.3, 126.4, 125.7, 59.6, 59.1, 51.5, 50.7, 27.6, 22.3, 19.7; IR (KBr, thin film, cm−1): 2923, 2807, 1603, 1464, 1156, 1077, 726, 700; LRMS-EI (m/z): 255 (18), 227 (14), 200 (8), 199 (74), 173 (54), 172 (65), 158 (12), 144 (29), 130 (100), 115 (17), 104 (19), 84 (15), 70 (11), 55 (8); HRMS-EI (m/z): M+ calcd for C14H17N5, 255.1484; found: 255.1480.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7); brown gum, [α]25D = −45.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.15 (s, 4H), 4.77–4.72 (dd, J1 = 3.4 Hz, J2 = 3.3 Hz, 1H); 4.55 (s, 1H), 4.19–4.11 (t, J = 11.3 Hz, 1H), 3.04–2.98 (m, 2H), 2.34 (s, 3H), 2.27–2.14 (m, 2H), 2.02–1.80 (m, 2H), 1.78–1.74 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.4, 138.4, 136.8, 129.5, 128.9, 128.5, 125.4, 64.15, 59.6, 51.7, 50.8, 27.6, 22.3, 21.3; IR (KBr, thin film, cm−1): LRMS-EI (m/z): 255 (25), 238 (4), 221 (9), 199 (100), 173 (65), 158 (12), 144 (13), 130 (0.01), 118 (28), 103 (10), 77 (8), 55 (6); HRMS-EI (m/z): M+ calcd for C14H17N5, 255.1483; found: 255.1478.
7); brown gum, [α]25D = −45.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.15 (s, 4H), 4.77–4.72 (dd, J1 = 3.4 Hz, J2 = 3.3 Hz, 1H); 4.55 (s, 1H), 4.19–4.11 (t, J = 11.3 Hz, 1H), 3.04–2.98 (m, 2H), 2.34 (s, 3H), 2.27–2.14 (m, 2H), 2.02–1.80 (m, 2H), 1.78–1.74 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.4, 138.4, 136.8, 129.5, 128.9, 128.5, 125.4, 64.15, 59.6, 51.7, 50.8, 27.6, 22.3, 21.3; IR (KBr, thin film, cm−1): LRMS-EI (m/z): 255 (25), 238 (4), 221 (9), 199 (100), 173 (65), 158 (12), 144 (13), 130 (0.01), 118 (28), 103 (10), 77 (8), 55 (6); HRMS-EI (m/z): M+ calcd for C14H17N5, 255.1483; found: 255.1478.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7); brown gum, [α]25D = −44.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.24 (s, 1H), 7.16–7.12 (m, 3H), 4.77–4.71 (dd, J1 = 3.6 Hz, J2 = 3.7 Hz, 1H), 4.56 (s, 1H), 4.18–4.10 (t, J = 11.3 Hz, 1H), 3.04–2.98 (m, 2H), 2.34 (s, 3H), 2.32–2.14 (m, 2H), 1.99–1.83 (m, 2H), 1.79–1.74 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.4, 138.4, 133.8, 129.3, 128.1, 63.7, 59.4, 51.6, 50.7, 27.6, 22.2, 21.0; IR (KBr, thin film, cm−1): 2949, 2800, 1604, 1464, 1317, 1156, 1077, 776, 700; LRMS-EI (m/z): 255 (54), 229 (74), 199 (83), 173 (82), 143 (29), 118 (38), 115 (27), 91 (32), 70 (20), 55 (22); HRMS-EI (m/z): M+ calcd for C14H17N5, 255.1484, found: 255.1488.
7); brown gum, [α]25D = −44.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.24 (s, 1H), 7.16–7.12 (m, 3H), 4.77–4.71 (dd, J1 = 3.6 Hz, J2 = 3.7 Hz, 1H), 4.56 (s, 1H), 4.18–4.10 (t, J = 11.3 Hz, 1H), 3.04–2.98 (m, 2H), 2.34 (s, 3H), 2.32–2.14 (m, 2H), 1.99–1.83 (m, 2H), 1.79–1.74 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.4, 138.4, 133.8, 129.3, 128.1, 63.7, 59.4, 51.6, 50.7, 27.6, 22.2, 21.0; IR (KBr, thin film, cm−1): 2949, 2800, 1604, 1464, 1317, 1156, 1077, 776, 700; LRMS-EI (m/z): 255 (54), 229 (74), 199 (83), 173 (82), 143 (29), 118 (38), 115 (27), 91 (32), 70 (20), 55 (22); HRMS-EI (m/z): M+ calcd for C14H17N5, 255.1484, found: 255.1488.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −45.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: = 7.30–7.28 (m, 1H), 6.98–6.96 (d, J = 7.4 Hz, 1H), 6.89–6.86 (m, 2H), 4.75–4.70 (dd, J1 = 3.3 Hz, J2 = 3.2 Hz, 1H), 4.56 (s, 1H), 4.17–4.10 (t, J = 11.0 Hz, 1H), 3.77 (s, 3H), 3.02–3.00 (m, 2H), 2.33–2.24 (q, J = 8.7 Hz, 1H), 2.20–2.14 (m, 1H), 2.02–1.94 (m, 2H), 1.82–1.69 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: = 159.8, 154.2, 138.4, 129.7, 120.7, 114.1, 63.9, 59.5, 55.1, 51.7, 50.8, 27.6, 22.4; IR (KBr, thin film, cm−1): 2945, 2840, 1599, 1485, 1316, 1154, 1042, 737, 693; LRMS-EI (m/z): 271 (83), 229 (29), 215 (95), 189 (73), 146 (100), 130 (25), 91 (20), 77 (15), 55 (16); HRMS-EI (m/z): M+ calcd for C14H17N5O, 271.1433, found: 271.1435.
7), brown gum, [α]25D = −45.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: = 7.30–7.28 (m, 1H), 6.98–6.96 (d, J = 7.4 Hz, 1H), 6.89–6.86 (m, 2H), 4.75–4.70 (dd, J1 = 3.3 Hz, J2 = 3.2 Hz, 1H), 4.56 (s, 1H), 4.17–4.10 (t, J = 11.0 Hz, 1H), 3.77 (s, 3H), 3.02–3.00 (m, 2H), 2.33–2.24 (q, J = 8.7 Hz, 1H), 2.20–2.14 (m, 1H), 2.02–1.94 (m, 2H), 1.82–1.69 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: = 159.8, 154.2, 138.4, 129.7, 120.7, 114.1, 63.9, 59.5, 55.1, 51.7, 50.8, 27.6, 22.4; IR (KBr, thin film, cm−1): 2945, 2840, 1599, 1485, 1316, 1154, 1042, 737, 693; LRMS-EI (m/z): 271 (83), 229 (29), 215 (95), 189 (73), 146 (100), 130 (25), 91 (20), 77 (15), 55 (16); HRMS-EI (m/z): M+ calcd for C14H17N5O, 271.1433, found: 271.1435.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −46.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: = 7.28 (s, 1H), 7.90–7.87 (d, J = 9.0 Hz, 3H), 4.75–4.70 (dd, J1 = 3.7 Hz, J2 = 3.8 Hz, 1H), 4.54 (s, 1H), 4.16–4.09 (t, J = 9.0 Hz, 1H), 3.79 (s, 3H), 3.02–2.96 (m, 2H), 2.32–2.26 (q, J = 8.9 Hz, 1H), 2.20–2.13 (m, 1H), 1.98–1.80 (m, 2H), 1.78–1.72 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: = 159.8, 154.6, 129.4, 129.1, 114.1, 63.5, 59.6, 55.2, 51.7, 50.8, 27.7, 22.3; IR (KBr, thin film, cm−1): 2956, 2791, 1610, 1511, 1465, 1511, 1256, 1163, 1020, 812; LRMS-EI (m/z): 271 (30), 215 (19), 190 (18), 189 (83), 146 (100), 134 (30), 103 (4), 77 (7), 55 (5); HRMS-EI (m/z): M+ calcd for C14H17N5O, 271.1433; found: 271.1438.
7), brown gum, [α]25D = −46.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: = 7.28 (s, 1H), 7.90–7.87 (d, J = 9.0 Hz, 3H), 4.75–4.70 (dd, J1 = 3.7 Hz, J2 = 3.8 Hz, 1H), 4.54 (s, 1H), 4.16–4.09 (t, J = 9.0 Hz, 1H), 3.79 (s, 3H), 3.02–2.96 (m, 2H), 2.32–2.26 (q, J = 8.9 Hz, 1H), 2.20–2.13 (m, 1H), 1.98–1.80 (m, 2H), 1.78–1.72 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: = 159.8, 154.6, 129.4, 129.1, 114.1, 63.5, 59.6, 55.2, 51.7, 50.8, 27.7, 22.3; IR (KBr, thin film, cm−1): 2956, 2791, 1610, 1511, 1465, 1511, 1256, 1163, 1020, 812; LRMS-EI (m/z): 271 (30), 215 (19), 190 (18), 189 (83), 146 (100), 134 (30), 103 (4), 77 (7), 55 (5); HRMS-EI (m/z): M+ calcd for C14H17N5O, 271.1433; found: 271.1438.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −38.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.36–7.28 (m, 2H), 7.15–7.07 (m, 2H), 4.98 (s, 1H), 4.78–4.73 (dd, J1 = 3.8 Hz, J2 = 3.6 Hz, 1H), 4.18–4.11 (t, J = 11.5 Hz, 1H), 2.05–2.98 (m, 2H), 2.42–2.33 (q, J = 9.0 Hz, 1H), 2.24–2.19 (m, 1H), 2.00–1.80 (m, 2H), 1.79–1.76 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 160.7 (d, J = 247.0 Hz), 153.6, 130.3 (d, J = 8.4 Hz), 124.3 (d, J = 3.0 Hz), 124.0, 115.6, 59.6, 51.4, 50.6, 27.5, 22.1; IR (KBr, thin film, cm−1): 2950, 2813, 1588, 1486, 1267, 1157, 1078, 883, 776, 691; LRMS-EI (m/z): 259 (5), 229 (2), 203 (100), 177 (34), 149 (18), 134 (43), 122 (16), 107 (7), 75 (1), 55 (4); HRMS-EI (m/z): M+ calcd for C13H14FN5, 259.1233; found: 259.1228.
7), brown gum, [α]25D = −38.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.36–7.28 (m, 2H), 7.15–7.07 (m, 2H), 4.98 (s, 1H), 4.78–4.73 (dd, J1 = 3.8 Hz, J2 = 3.6 Hz, 1H), 4.18–4.11 (t, J = 11.5 Hz, 1H), 2.05–2.98 (m, 2H), 2.42–2.33 (q, J = 9.0 Hz, 1H), 2.24–2.19 (m, 1H), 2.00–1.80 (m, 2H), 1.79–1.76 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 160.7 (d, J = 247.0 Hz), 153.6, 130.3 (d, J = 8.4 Hz), 124.3 (d, J = 3.0 Hz), 124.0, 115.6, 59.6, 51.4, 50.6, 27.5, 22.1; IR (KBr, thin film, cm−1): 2950, 2813, 1588, 1486, 1267, 1157, 1078, 883, 776, 691; LRMS-EI (m/z): 259 (5), 229 (2), 203 (100), 177 (34), 149 (18), 134 (43), 122 (16), 107 (7), 75 (1), 55 (4); HRMS-EI (m/z): M+ calcd for C13H14FN5, 259.1233; found: 259.1228.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −48.8° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.36–7.31 (m, 1H), 7.21–7.19 (m, 1H), 7.11–7.02 (m, 2H), 4.77–4.72 (dd, J1 = 3.6 Hz, J2 = 3.3 Hz, 1H), 4.61 (s, 1H), 4.18–4.10 (t, J = 11.1 Hz, 1H), 3.03 (m, 2H), 2.34–2.25 (q, J = 9.0 Hz, 1H), 2.22–2.15 (m, 1H), 2.04–1.99 (m, 2H), 1.83–1.74 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 162.9 (d, J = 123.0 Hz), 153.8, 139.5, 130.3 (d, J = 8.2 Hz), 124.2 (d, J = 2.9 Hz), 115.9, 115.7, 115.3, 115.0, 63.3, 59.5, 51.6, 50.8, 27.6, 22.3; IR (KBr, thin film, cm−1): 2950, 2802, 1580, 1448, 1242, 1155, 790, 688; LRMS-EI (m/z): 259 (7), 229 (2), 203 (100), 177 (46), 149 (18), 134 (47), 122 (23), 107 (9), 81 (3), 55 (9); HRMS-EI (m/z): M+ calcd for C13H14FN5, 259.1233, found: 259.1242.
7), brown gum, [α]25D = −48.8° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.36–7.31 (m, 1H), 7.21–7.19 (m, 1H), 7.11–7.02 (m, 2H), 4.77–4.72 (dd, J1 = 3.6 Hz, J2 = 3.3 Hz, 1H), 4.61 (s, 1H), 4.18–4.10 (t, J = 11.1 Hz, 1H), 3.03 (m, 2H), 2.34–2.25 (q, J = 9.0 Hz, 1H), 2.22–2.15 (m, 1H), 2.04–1.99 (m, 2H), 1.83–1.74 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 162.9 (d, J = 123.0 Hz), 153.8, 139.5, 130.3 (d, J = 8.2 Hz), 124.2 (d, J = 2.9 Hz), 115.9, 115.7, 115.3, 115.0, 63.3, 59.5, 51.6, 50.8, 27.6, 22.3; IR (KBr, thin film, cm−1): 2950, 2802, 1580, 1448, 1242, 1155, 790, 688; LRMS-EI (m/z): 259 (7), 229 (2), 203 (100), 177 (46), 149 (18), 134 (47), 122 (23), 107 (9), 81 (3), 55 (9); HRMS-EI (m/z): M+ calcd for C13H14FN5, 259.1233, found: 259.1242.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −46.9° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.33 (s, 4H), 4.77–4.71 (dd, J1 = 4.9 Hz, J2 = 3.8 Hz, 1H); 4.59 (s, 1H), 4.17–4.10 (t, J = 11.2 Hz, 1H), 3.00 (m, 2H), 2.32–2.23 (q, J = 7.8 Hz, 1H), 2.22–2.19 (m, 1H), 1.97–1.95 (m, 2H), 1.83–1.74 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.0, 135.1 (d, J = 64.0 Hz), 130.0, 129.7, 129.0, 128.7, 63.3, 59.6, 51.7, 50.8, 27.6, 22.3; IR (KBr, thin film, cm−1): 2950, 2813, 1489, 1160, 1083, 803; LRMS-EI (m/z): 259 (5), 229 (5), 219 (21), 203 (100), 193 (18), 177 (68), 149 (41), 134 (51), 122 (31), 105 (21), 77 (8), 55 (22); HRMS-EI (m/z): M+ calcd for C13H14FN5, 259.1233, found: 259.1236.
7), brown gum, [α]25D = −46.9° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.33 (s, 4H), 4.77–4.71 (dd, J1 = 4.9 Hz, J2 = 3.8 Hz, 1H); 4.59 (s, 1H), 4.17–4.10 (t, J = 11.2 Hz, 1H), 3.00 (m, 2H), 2.32–2.23 (q, J = 7.8 Hz, 1H), 2.22–2.19 (m, 1H), 1.97–1.95 (m, 2H), 1.83–1.74 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.0, 135.1 (d, J = 64.0 Hz), 130.0, 129.7, 129.0, 128.7, 63.3, 59.6, 51.7, 50.8, 27.6, 22.3; IR (KBr, thin film, cm−1): 2950, 2813, 1489, 1160, 1083, 803; LRMS-EI (m/z): 259 (5), 229 (5), 219 (21), 203 (100), 193 (18), 177 (68), 149 (41), 134 (51), 122 (31), 105 (21), 77 (8), 55 (22); HRMS-EI (m/z): M+ calcd for C13H14FN5, 259.1233, found: 259.1236.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −43.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.64–7.62 (d, J = 6.0 Hz, 1H), 7.28–7.23 (m, 2H), 7.21–7.16 (m, 1H), 5.33 (s, 1H), 4.79–4.73 (dd, J1 = 3.0 Hz, J2 = 3.2 Hz, 1H), 4.18–4.10 (t, J = 11.2 Hz, 1H), 3.16–3.06 (m, 1H), (m, 2.99–2.94, 1H), 2.46–2.37 (q, J = 8.9 Hz, 1H), 2.25–2.15 (m, 1H), 1.97–1.82 (m, 2H), 1.79–1.69 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 153.7, 136.4, 133.0, 130.1, 129.9, 128.0, 124.4, 61.9, 59.6, 51.2, 50.8, 27.6, 22.3; IR (KBr, thin film, cm−1): 2956, 2813, 1574, 1473, 1075, 883, 781, 691; LRMS-ESI (m/z): 276 (47), 270 (4), 267 (2), 256 (10), 243 (17), 242 (100), 240 (7), 238 (3), 227 (8); HRMS-ESI (m/z): M+ calcd for C13H14ClN5, 276.0938; found: 276.0998.
7), brown gum, [α]25D = −43.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.64–7.62 (d, J = 6.0 Hz, 1H), 7.28–7.23 (m, 2H), 7.21–7.16 (m, 1H), 5.33 (s, 1H), 4.79–4.73 (dd, J1 = 3.0 Hz, J2 = 3.2 Hz, 1H), 4.18–4.10 (t, J = 11.2 Hz, 1H), 3.16–3.06 (m, 1H), (m, 2.99–2.94, 1H), 2.46–2.37 (q, J = 8.9 Hz, 1H), 2.25–2.15 (m, 1H), 1.97–1.82 (m, 2H), 1.79–1.69 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 153.7, 136.4, 133.0, 130.1, 129.9, 128.0, 124.4, 61.9, 59.6, 51.2, 50.8, 27.6, 22.3; IR (KBr, thin film, cm−1): 2956, 2813, 1574, 1473, 1075, 883, 781, 691; LRMS-ESI (m/z): 276 (47), 270 (4), 267 (2), 256 (10), 243 (17), 242 (100), 240 (7), 238 (3), 227 (8); HRMS-ESI (m/z): M+ calcd for C13H14ClN5, 276.0938; found: 276.0998.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −44.6° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm): δ = 7.36 (s, 1H), 7.30 (s, 3H), 4.76–4.71 (dd, J1 = 2.7 Hz, J2 = 2.9 Hz, 1H), 4.58 (s, 1H), 4.17–4.10 (t, J = 11.2 Hz, 1H), 3.03–2.98 (m, 2H), 2.33–2.24 (q, J = 9.0 Hz, 1H), 2.20–2.15 (m, 1H), 2.02–1.95 (m, 2H), 1.83–1.73 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 153.8, 139.0, 134.5, 130.0, 128.9, 128.2, 126.7, 63.3, 59.4, 51.6, 50.7, 27.5, 22.3; IR (KBr, thin film, cm−1): 2950, 2807, 1596, 1470, 1316, 1078, 779, 691; LRMS-EI (m/z): 275 (7.5), 265 (3.8), 244 (1.2), 221 (33), 219 (100), 193 (48), 150 (46), 13 (30), 89 (13), 55 (14); HRMS-EI (m/z): M+ calcd for C13H14ClN5, 275.0938, found: 275.0930.
7), brown gum, [α]25D = −44.6° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm): δ = 7.36 (s, 1H), 7.30 (s, 3H), 4.76–4.71 (dd, J1 = 2.7 Hz, J2 = 2.9 Hz, 1H), 4.58 (s, 1H), 4.17–4.10 (t, J = 11.2 Hz, 1H), 3.03–2.98 (m, 2H), 2.33–2.24 (q, J = 9.0 Hz, 1H), 2.20–2.15 (m, 1H), 2.02–1.95 (m, 2H), 1.83–1.73 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 153.8, 139.0, 134.5, 130.0, 128.9, 128.2, 126.7, 63.3, 59.4, 51.6, 50.7, 27.5, 22.3; IR (KBr, thin film, cm−1): 2950, 2807, 1596, 1470, 1316, 1078, 779, 691; LRMS-EI (m/z): 275 (7.5), 265 (3.8), 244 (1.2), 221 (33), 219 (100), 193 (48), 150 (46), 13 (30), 89 (13), 55 (14); HRMS-EI (m/z): M+ calcd for C13H14ClN5, 275.0938, found: 275.0930.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −47.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: = 7.37–7.33 (t, J = 8.0 Hz, 2H), 7.08–7.02 (t, J = 8.4 Hz, 2H), 4.77–4.72 (dd, J1 = 2.9 Hz, J2 = 3.3 Hz, 1H), 4.59 (s, 1H), 4.17–4.10 (t, J = 11.0 Hz, 1H), 3.03–2.96 (m, 2H), 2.33–2.24 (q, J = 8.9 Hz, 1H), 2.22–2.12 (m, 1H), 1.97–1.83 (m, 2H), 1.79–1.70 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.2, 130.1, 123.0, 115.9, 115.6, 63.3, 59.6, 51.7, 50.8, 27.7, 22.3; IR (KBr, thin film, cm−1): 2856, 2813, 1602, 1508, 1219, 1150, 872, 551; LRMS-EI (m/z): 275 (0.1), 259 (3), 255 (5), 227 (5), 203 (39), 177 (35), 149 (17), 134 (37), 119 (20), 93 (26), 85 (100), 79 (27), 55 (31); HRMS-EI (m/z): M+ calcd for C13H14ClN5, 275.0938; found: 275.0931.
7), brown gum, [α]25D = −47.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: = 7.37–7.33 (t, J = 8.0 Hz, 2H), 7.08–7.02 (t, J = 8.4 Hz, 2H), 4.77–4.72 (dd, J1 = 2.9 Hz, J2 = 3.3 Hz, 1H), 4.59 (s, 1H), 4.17–4.10 (t, J = 11.0 Hz, 1H), 3.03–2.96 (m, 2H), 2.33–2.24 (q, J = 8.9 Hz, 1H), 2.22–2.12 (m, 1H), 1.97–1.83 (m, 2H), 1.79–1.70 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.2, 130.1, 123.0, 115.9, 115.6, 63.3, 59.6, 51.7, 50.8, 27.7, 22.3; IR (KBr, thin film, cm−1): 2856, 2813, 1602, 1508, 1219, 1150, 872, 551; LRMS-EI (m/z): 275 (0.1), 259 (3), 255 (5), 227 (5), 203 (39), 177 (35), 149 (17), 134 (37), 119 (20), 93 (26), 85 (100), 79 (27), 55 (31); HRMS-EI (m/z): M+ calcd for C13H14ClN5, 275.0938; found: 275.0931.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −43.5° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.44–7.41 (d, J = 9.0 Hz, 1H), 7.25–7.22 (m, 3H), 5.30 (s, 1H), 4.76–4.70 (dd, J1 = 3.2 Hz, J2 = 3.1 Hz, 1H), 4.15–4.07 (t, J = 11.0 Hz, 1H), 3.09–3.00 (m, 1H), 2.96 (m, 1H), 2.40–2.32 (q, J = 8.7 Hz, 1H), 2.24–2.19 (m, 1H), 1.95–1.90 (m, 2H), 1.79–1.70 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 153.6, 134.6, 133.9, 132.6, 129.6, 127.3, 126.7, 59.6, 59.0, 51.2, 50.7, 27.5, 22.2; IR (KBr, thin film, cm−1): 2956, 2813, 1470, 1157, 1048, 757; LRMS-EI (m/z): 319 (0.1), 307 (0.5), 275 (5), 255 (2), 219 (100), 193 (33), 165 (14), 150 (46), 130 (21), 89 (12), 55 (12); HRMS-EI (m/z): M+ calcd for C13H14BrN5, 319.0433; found: 319.0432.
7), brown gum, [α]25D = −43.5° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.44–7.41 (d, J = 9.0 Hz, 1H), 7.25–7.22 (m, 3H), 5.30 (s, 1H), 4.76–4.70 (dd, J1 = 3.2 Hz, J2 = 3.1 Hz, 1H), 4.15–4.07 (t, J = 11.0 Hz, 1H), 3.09–3.00 (m, 1H), 2.96 (m, 1H), 2.40–2.32 (q, J = 8.7 Hz, 1H), 2.24–2.19 (m, 1H), 1.95–1.90 (m, 2H), 1.79–1.70 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 153.6, 134.6, 133.9, 132.6, 129.6, 127.3, 126.7, 59.6, 59.0, 51.2, 50.7, 27.5, 22.2; IR (KBr, thin film, cm−1): 2956, 2813, 1470, 1157, 1048, 757; LRMS-EI (m/z): 319 (0.1), 307 (0.5), 275 (5), 255 (2), 219 (100), 193 (33), 165 (14), 150 (46), 130 (21), 89 (12), 55 (12); HRMS-EI (m/z): M+ calcd for C13H14BrN5, 319.0433; found: 319.0432.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −46.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.52–7.47 (t, J = 7.0 Hz, 2H), 7.35–7.27 (m, 2H), 4.77–4.72 (dd, J1 = 3.5 Hz, J2 = 3.4 Hz, 1H), 4.58 (s, 1H), 4.18–4.11 (t, J = 11.1 Hz, 1H), 3.04–3.01 (m, 2H), 2.34–2.28 (q, J = 8.9 Hz, 1H), 2.25 (m, 1H), 2.05–1.98 (m, 2H), 1.84–1.77 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 153.8, 139.2, 132.0, 131.2, 130.3, 127.2, 122.8, 63.4, 59.6, 51.7, 50.8, 27.6, 22.3; IR (KBr, thin film, cm−1): 2950, 2818, 1467, 1273, 1160, 1050, 858, 760; LRMS-ESI (m/z): 359 (33), 341 (8), 331 (6), 323 (16), 322 (89), 321 (22), 320 (100), 310 (4), 296 (3), 293 (7); HRMS-ESI (m/z): M+ calcd for C13H14BrN5, 320.0433; found: 320.0507.
7), brown gum, [α]25D = −46.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.52–7.47 (t, J = 7.0 Hz, 2H), 7.35–7.27 (m, 2H), 4.77–4.72 (dd, J1 = 3.5 Hz, J2 = 3.4 Hz, 1H), 4.58 (s, 1H), 4.18–4.11 (t, J = 11.1 Hz, 1H), 3.04–3.01 (m, 2H), 2.34–2.28 (q, J = 8.9 Hz, 1H), 2.25 (m, 1H), 2.05–1.98 (m, 2H), 1.84–1.77 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 153.8, 139.2, 132.0, 131.2, 130.3, 127.2, 122.8, 63.4, 59.6, 51.7, 50.8, 27.6, 22.3; IR (KBr, thin film, cm−1): 2950, 2818, 1467, 1273, 1160, 1050, 858, 760; LRMS-ESI (m/z): 359 (33), 341 (8), 331 (6), 323 (16), 322 (89), 321 (22), 320 (100), 310 (4), 296 (3), 293 (7); HRMS-ESI (m/z): M+ calcd for C13H14BrN5, 320.0433; found: 320.0507.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −48.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.71–7.67 (m, 1H), 7.66–7.64 (m, 2H), 7.53–7.48 (m, 1H), 4.80–4.75 (dd, J1 = 2.0 Hz, J2 = 3.7 Hz, 1H), 4.68 (s, 1H), 4.20–4.12 (t, J = 11.5 Hz, 1H), 3.05–2.94 (m, 2H), 2.32–2.29 (q, J = 9.0 Hz, 1H), 2.23 (m, 1H), 2.04–1.98 (m, 2H), 185–1.80 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 135.4, 138.6, 132.9, 132.5, 131.9, 129.7, 118.2, 113.0, 63.1, 59.6, 51.7, 50.8, 27.5, 22.3; IR (KBr, thin film, cm−1): 2993, 2950, 2230, 1676, 1445, 1163, 792, 688; LRMS-EI (m/z): 266 (5), 237 (0.9), 210 (100), 184 (39), 156 (15), 141 (25), 129 (17), 115 (6), 70 (2), 55 (5); HRMS-EI (m/z): M+ calcd for C14H14N6, 266.1280, found: 266.1279.
7), brown gum, [α]25D = −48.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.71–7.67 (m, 1H), 7.66–7.64 (m, 2H), 7.53–7.48 (m, 1H), 4.80–4.75 (dd, J1 = 2.0 Hz, J2 = 3.7 Hz, 1H), 4.68 (s, 1H), 4.20–4.12 (t, J = 11.5 Hz, 1H), 3.05–2.94 (m, 2H), 2.32–2.29 (q, J = 9.0 Hz, 1H), 2.23 (m, 1H), 2.04–1.98 (m, 2H), 185–1.80 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 135.4, 138.6, 132.9, 132.5, 131.9, 129.7, 118.2, 113.0, 63.1, 59.6, 51.7, 50.8, 27.5, 22.3; IR (KBr, thin film, cm−1): 2993, 2950, 2230, 1676, 1445, 1163, 792, 688; LRMS-EI (m/z): 266 (5), 237 (0.9), 210 (100), 184 (39), 156 (15), 141 (25), 129 (17), 115 (6), 70 (2), 55 (5); HRMS-EI (m/z): M+ calcd for C14H14N6, 266.1280, found: 266.1279.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −48.9° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.71–7.68 (d, J = 11.6 Hz, 1H), 7.67–7.53 (m, 2H), 7.50–7.25 (m, 1H), 4.80–4.79 (dd, J1 = 3.5 Hz, J2 = 3.7 Hz, 1H), 4.68 (s, 1H), 4.20–4.12 (t, J = 11.3 Hz, 1H), 3.08–2.95 (m, 2H), 2.35–2.26 (q, J = 8.8 Hz, 1H), 2.23–2.16 (m, 1H), 2.04–1.98 (m, 2H), 1.86–1.79 (1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.2, 130.1, 123.0, 115.9, 115.6, 63.3, 59.6, 51.7, 50.8, 27.7, 22.3; IR (KBr, thin film, cm−1): 2917, 2846, 2225, 1607, 1467, 1273, 1157, 809, 556; LRMS-EI (m/z): 266 (2), 237 (0.2), 210 (100), 184 (42), 155 (17), 141 (32), 129 (17), 115 (7), 73 (4), 55 (14); HRMS-EI (m/z): M+ calcd for C14H14N6, 266.1280, found: 266.1277.
7), brown gum, [α]25D = −48.9° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.71–7.68 (d, J = 11.6 Hz, 1H), 7.67–7.53 (m, 2H), 7.50–7.25 (m, 1H), 4.80–4.79 (dd, J1 = 3.5 Hz, J2 = 3.7 Hz, 1H), 4.68 (s, 1H), 4.20–4.12 (t, J = 11.3 Hz, 1H), 3.08–2.95 (m, 2H), 2.35–2.26 (q, J = 8.8 Hz, 1H), 2.23–2.16 (m, 1H), 2.04–1.98 (m, 2H), 1.86–1.79 (1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.2, 130.1, 123.0, 115.9, 115.6, 63.3, 59.6, 51.7, 50.8, 27.7, 22.3; IR (KBr, thin film, cm−1): 2917, 2846, 2225, 1607, 1467, 1273, 1157, 809, 556; LRMS-EI (m/z): 266 (2), 237 (0.2), 210 (100), 184 (42), 155 (17), 141 (32), 129 (17), 115 (7), 73 (4), 55 (14); HRMS-EI (m/z): M+ calcd for C14H14N6, 266.1280, found: 266.1277.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −35.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 6.16 (s, 1H), 6.08 (s, 1H), 5.29 (s, 1H), 4.71–4.66 (dd, J1 = 3.0 Hz, J2 = 3.1 Hz, 1H), 4.14–4.06 (t, J = 10.8 Hz, 1H), 3.83 (s, 3H), 3.79 (s, 3H), 3.36 (s, 3H), 3.04–2.98 (m, 1H), 2.89–2.86 (m, 1H), 2.54–2.45 (q, J = 9.0 Hz, 1H), 2.11–2.06 (m, 1H), 1.95–1.88 (m, 1H), 1.78–168 (m, 1H), 1.66–1.58 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.2, 130.1, 130.0, 115.9, 115.6, 63.3, 59.6, 51.7, 50.8, 27.7, 22.3; IR (KBr, thin film, cm−1): 2923, 2851, 1462, 1256, 1031, 809; LRMS-EI (m/z): 331 (18), 272 (36), 234 (20), 210 (53), 206 (100), 179 (37), 155 (10), 141 (17), 121 (7), 83 (5), 69 (8), 55 (9); HRMS-EI (m/z): M+ calcd for C16H21N5O3, 331.1644, found: 331.1640.
7), brown gum, [α]25D = −35.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 6.16 (s, 1H), 6.08 (s, 1H), 5.29 (s, 1H), 4.71–4.66 (dd, J1 = 3.0 Hz, J2 = 3.1 Hz, 1H), 4.14–4.06 (t, J = 10.8 Hz, 1H), 3.83 (s, 3H), 3.79 (s, 3H), 3.36 (s, 3H), 3.04–2.98 (m, 1H), 2.89–2.86 (m, 1H), 2.54–2.45 (q, J = 9.0 Hz, 1H), 2.11–2.06 (m, 1H), 1.95–1.88 (m, 1H), 1.78–168 (m, 1H), 1.66–1.58 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.2, 130.1, 130.0, 115.9, 115.6, 63.3, 59.6, 51.7, 50.8, 27.7, 22.3; IR (KBr, thin film, cm−1): 2923, 2851, 1462, 1256, 1031, 809; LRMS-EI (m/z): 331 (18), 272 (36), 234 (20), 210 (53), 206 (100), 179 (37), 155 (10), 141 (17), 121 (7), 83 (5), 69 (8), 55 (9); HRMS-EI (m/z): M+ calcd for C16H21N5O3, 331.1644, found: 331.1640.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −34.9° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.90–7.82 (m, 3H), 7.66–7.54 (m, 2H), 7.48–7.37 (m, 2H), 4.90 (s, 1H), 4.87–4.78 (dd, J1 = 3.5 Hz, J2 = 3.0 Hz, 1H), 4.32–4.40 (t, J = 11.0 Hz, 1H), 3.08–3.02 (m, 2H), 2.37–2.27 (m, 2H), 1.98–1.86 (m, 2H), 1.73–1.68 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.3, 134.4, 131.7, 130.2, 128.7, 126.5, 125.8, 125.6, 124.9, 124.5, 122.8, 66.0, 60.1, 59.7, 51.5, 51.0, 27.7, 22.3; IR (KBr, thin film, cm−1): 2961, 2807, 1508, 1377, 1155, 979, 795, 617; LRMS-EI (m/z): 291 (53), 281 (0.7), 262 (4), 235 (32), 210 (67), 208 (100), 180 (33), 166 (96), 141 (25), 127 (10), 102 (1), 68 (0.9), 55 (5); HRMS-EI (m/z): M+ calcd for C17H17N5, 291.1484; found: 291.1481.
7), brown gum, [α]25D = −34.9° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.90–7.82 (m, 3H), 7.66–7.54 (m, 2H), 7.48–7.37 (m, 2H), 4.90 (s, 1H), 4.87–4.78 (dd, J1 = 3.5 Hz, J2 = 3.0 Hz, 1H), 4.32–4.40 (t, J = 11.0 Hz, 1H), 3.08–3.02 (m, 2H), 2.37–2.27 (m, 2H), 1.98–1.86 (m, 2H), 1.73–1.68 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.3, 134.4, 131.7, 130.2, 128.7, 126.5, 125.8, 125.6, 124.9, 124.5, 122.8, 66.0, 60.1, 59.7, 51.5, 51.0, 27.7, 22.3; IR (KBr, thin film, cm−1): 2961, 2807, 1508, 1377, 1155, 979, 795, 617; LRMS-EI (m/z): 291 (53), 281 (0.7), 262 (4), 235 (32), 210 (67), 208 (100), 180 (33), 166 (96), 141 (25), 127 (10), 102 (1), 68 (0.9), 55 (5); HRMS-EI (m/z): M+ calcd for C17H17N5, 291.1484; found: 291.1481.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D −46.9° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 3.98–3.93 (t, J = 7.9 Hz, 1H), 3.36–3.30 (dd, J1 = 4.5 Hz, J2 = 4.4 Hz, 1H), 3.18–3.12 (m, 1H), 2.02–2.95 (m, 2H), 2.63–2.54 (q, J = 9.0 Hz, 1H), 1.87–1.82 (m, 4H), 1.65–1.60 (m, 3H), 0.96 (d, J = 1.6 Hz, 3H), 0.94–0.93 (d, J = 1.7 Hz, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 158.8, 60.4, 58.6, 55.8, 55.7, 55.2, 53.8, 50.4, 45.9, 28.4, 23.3; IR (KBr, thin film, cm−1): 2956, 2868, 1467, 1166, 982, 762; LRMS-EI (m/z): 221 (3), 210 (0.7), 190 (0.9), 179 (10), 164 (100), 151 (3), 124 (3), 109 (5), 96 (15), 84 (8), 70 (9), 55 (10); HRMS-EI (m/z): M+ calcd for C11H19N5, 221.1640, found: 221.1643.
7), brown gum, [α]25D −46.9° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 3.98–3.93 (t, J = 7.9 Hz, 1H), 3.36–3.30 (dd, J1 = 4.5 Hz, J2 = 4.4 Hz, 1H), 3.18–3.12 (m, 1H), 2.02–2.95 (m, 2H), 2.63–2.54 (q, J = 9.0 Hz, 1H), 1.87–1.82 (m, 4H), 1.65–1.60 (m, 3H), 0.96 (d, J = 1.6 Hz, 3H), 0.94–0.93 (d, J = 1.7 Hz, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 158.8, 60.4, 58.6, 55.8, 55.7, 55.2, 53.8, 50.4, 45.9, 28.4, 23.3; IR (KBr, thin film, cm−1): 2956, 2868, 1467, 1166, 982, 762; LRMS-EI (m/z): 221 (3), 210 (0.7), 190 (0.9), 179 (10), 164 (100), 151 (3), 124 (3), 109 (5), 96 (15), 84 (8), 70 (9), 55 (10); HRMS-EI (m/z): M+ calcd for C11H19N5, 221.1640, found: 221.1643.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D −36.3° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.34–7.29 (m, 2H), 7.22–7.19 (m, 3H), 3.85–3.80 (t, J = 9.0 Hz, 1H), 3.36–3.31 (dd, J1 = 4.5 Hz, J2 = 4.4 Hz, 1H), 3.29–3.23 (dd, J1 = 4.6 Hz, J2 = 4.7 Hz, 1H), 3.12–3.06 (m, 2H), 2.83–2.78 (t, J = 7.2 Hz, 2H), 2.67–2.64 (q, J = 9.0 Hz, 1H); 2.13–2.06 (m, 2H), 1.92–1.82 (m, 3H), 1.70–1.62 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.0, 141.5, 128.5, 128.40, 128.3, 125.9, 59.5, 58.5, 52.2, 50.8, 32.6, 30.0, 27.6, 22.5; IR (KBr, thin film, cm−1): 2950, 2858, 1690, 1454, 1155, 1075, 746, 696; LRMS-EI (m/z): 269 (5), 251 (2), 213 (12), 179 (50), 164 (100), 150 (25), 123 (14), 109 (27), 96 (26), 91 (55), 69 (16), 55 (19); HRMS-EI (m/z): M+ calcd for C15H19N5, 269.1640, found: 269.1648.
7), brown gum, [α]25D −36.3° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.34–7.29 (m, 2H), 7.22–7.19 (m, 3H), 3.85–3.80 (t, J = 9.0 Hz, 1H), 3.36–3.31 (dd, J1 = 4.5 Hz, J2 = 4.4 Hz, 1H), 3.29–3.23 (dd, J1 = 4.6 Hz, J2 = 4.7 Hz, 1H), 3.12–3.06 (m, 2H), 2.83–2.78 (t, J = 7.2 Hz, 2H), 2.67–2.64 (q, J = 9.0 Hz, 1H); 2.13–2.06 (m, 2H), 1.92–1.82 (m, 3H), 1.70–1.62 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 154.0, 141.5, 128.5, 128.40, 128.3, 125.9, 59.5, 58.5, 52.2, 50.8, 32.6, 30.0, 27.6, 22.5; IR (KBr, thin film, cm−1): 2950, 2858, 1690, 1454, 1155, 1075, 746, 696; LRMS-EI (m/z): 269 (5), 251 (2), 213 (12), 179 (50), 164 (100), 150 (25), 123 (14), 109 (27), 96 (26), 91 (55), 69 (16), 55 (19); HRMS-EI (m/z): M+ calcd for C15H19N5, 269.1640, found: 269.1648.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −40.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.39 (s, 1H), 7.32 (m, 1H), 7.25 (s, 1H), 7.02–7.01 (d, J = 3.0 Hz, 1H), 4.76 (s, 1H), 4.71 (m, 1H), 4.15–4.09 (t, J = 8.4 Hz, 1H), 3.10–3.06 (m, 1H), 2.98–2.96 (m, 1H), 2.35–2.28 (q, J = 6.9 Hz, 1H), 2.20–2.16 (m, 1H), 1.96–1.94 (m, 2H), 1.78–1.74 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 153.8, 137.4, 126.6, 124.4, 59.6, 59.2, 51.9, 50.8, 27.6, 22.2; IR (KBr, thin film, cm−1): 2961, 2807, 1467, 1150, 836, 776, 603; LRMS-EI (m/z): 247 (30), 235 (4), 218 (3), 205 (10), 191 (73), 166 (13), 165 (100), 136 (23), 122 (80), 110 (33), 97 (13), 70 (8), 55 (9); HRMS-EI (m/z): M+ calcd for C11H13N5S, 247.0892; found:247.0899.
7), brown gum, [α]25D = −40.2° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 7.39 (s, 1H), 7.32 (m, 1H), 7.25 (s, 1H), 7.02–7.01 (d, J = 3.0 Hz, 1H), 4.76 (s, 1H), 4.71 (m, 1H), 4.15–4.09 (t, J = 8.4 Hz, 1H), 3.10–3.06 (m, 1H), 2.98–2.96 (m, 1H), 2.35–2.28 (q, J = 6.9 Hz, 1H), 2.20–2.16 (m, 1H), 1.96–1.94 (m, 2H), 1.78–1.74 (m, 1H); 13C NMR (75 MHz, CDCl3, ppm) δ: 153.8, 137.4, 126.6, 124.4, 59.6, 59.2, 51.9, 50.8, 27.6, 22.2; IR (KBr, thin film, cm−1): 2961, 2807, 1467, 1150, 836, 776, 603; LRMS-EI (m/z): 247 (30), 235 (4), 218 (3), 205 (10), 191 (73), 166 (13), 165 (100), 136 (23), 122 (80), 110 (33), 97 (13), 70 (8), 55 (9); HRMS-EI (m/z): M+ calcd for C11H13N5S, 247.0892; found:247.0899.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), brown gum, [α]25D = −39.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 8.03–8.06 (m, 1H), 7.87–7.84 (m, 1H), 7.68–7.43 (m, 1H), 7.42–7.39 (m, 2H), 5.60 (s, 1H), 3.62–3.53 (dd, J1 = 4.5 Hz, J2 = 6.7 Hz, 1H), 3.24–3.19 (m, 1H), 2.65–2.59 (m, 2H), 2.12–2.07 (m, 1H), 1.97–1.93 (m, 1H), 1.78–1.66 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 140.7, 136.5, 128.5, 125.8, 125.0, 124.4, 122.7, 122.4, 116.2, 60.6, 55.7, 54.2, 49.6, 29.0, 22.8; IR (KBr, thin film, cm−1): 2956, 2850, 1637, 1442, 1155, 1075, 762, 732; LRMS-EI (m/z): 297 (68), 268 (3), 241 (23), 215 (80), 186 (13), 172 (100), 160 (36), 147 (19), 115 (7), 89 (5), 69 (5), 55 (6); HRMS-EI (m/z): M+ calcd for C15H15N5S, 297.1048; found: 297.1044.
7), brown gum, [α]25D = −39.4° (c 1.0, CHCl3), 1H NMR (CDCl3, 300 MHz, ppm) δ: 8.03–8.06 (m, 1H), 7.87–7.84 (m, 1H), 7.68–7.43 (m, 1H), 7.42–7.39 (m, 2H), 5.60 (s, 1H), 3.62–3.53 (dd, J1 = 4.5 Hz, J2 = 6.7 Hz, 1H), 3.24–3.19 (m, 1H), 2.65–2.59 (m, 2H), 2.12–2.07 (m, 1H), 1.97–1.93 (m, 1H), 1.78–1.66 (m, 3H); 13C NMR (75 MHz, CDCl3, ppm) δ: 140.7, 136.5, 128.5, 125.8, 125.0, 124.4, 122.7, 122.4, 116.2, 60.6, 55.7, 54.2, 49.6, 29.0, 22.8; IR (KBr, thin film, cm−1): 2956, 2850, 1637, 1442, 1155, 1075, 762, 732; LRMS-EI (m/z): 297 (68), 268 (3), 241 (23), 215 (80), 186 (13), 172 (100), 160 (36), 147 (19), 115 (7), 89 (5), 69 (5), 55 (6); HRMS-EI (m/z): M+ calcd for C15H15N5S, 297.1048; found: 297.1044.Acknowledgements
The author thanks to Ms L. M. Hsu, at the Instruments Center, National Chung Hsing University, for her help in obtaining mass spectral data, and the Ministry of Science and Technology of Taiwan, for financially supporting this research under the contract MOST 103-2113-M-259-009-MY3.Notes and references
- (a) A. Strecker and J. Liebigs, Ann. Chem., 1850, 75, 27–45 CrossRef; (b) A. Strecker and J. Liebigs, Ann. Chem., 1854, 91, 349–351 CrossRef.
- For reviews of the Strecker reaction please see: (a) C. Najera and J. M. Sansano, Chem. Rev., 2007, 107, 4584–4671 CrossRef CAS PubMed; (b) H. Gröger, Chem. Rev., 2003, 103, 2795–2827 CrossRef PubMed; (c) J. Wang, X. Liu and X. Feng, Chem. Rev., 2011, 111, 6947–6983 CrossRef CAS PubMed; (d) S. J. Zuend, M. P. Coughlin, M. P. Lalonde and E. N. Jacobsen, Nature, 2009, 461, 968–970 CrossRef CAS PubMed; (e) A. G. Doyle and E. N. Jacobsen, Chem. Rev., 2007, 107, 5713–5743 CrossRef CAS PubMed; (f) E. Sucas, M. Y. Ngai, V. Komanduri and M. J. Krische, Acc. Chem. Res., 2007, 40, 1394–1401 CrossRef PubMed; (g) P. Merino, E. Marques-Lopez, T. Tejero and R. Herrera, Tetrahedron, 2009, 65, 1219–1234 CrossRef CAS; (h) J. Martens, ChemCatChem, 2010, 2, 379–381 CrossRef CAS.
- (a) A. Perdih and M. S. Dolenc, Curr. Org. Chem., 2007, 11, 801–832 CrossRef CAS; (b) J. A. Ma, Angew. Chem., 2003, 115, 4426–4435 (Angew. Chem., Int. Ed., 2003, 42, 4290–4299) CrossRef.
- (a) V. A. Soloshonok and K. Izawa, Asymmetric Synthesis and Application of R Amino acid, American Chemical Society, Washington DC, 2009, vol. 1009 Search PubMed; (b) A. S. Bommarius, M. Schwarm and K. Drauz, Chimia, 2001, 55, 50–59 CAS.
- (a) D. Enders and J. P. Shilvock, Chem. Soc. Rev., 2000, 29, 359–373 RSC; (b) T. Opatz, Synthesis, 2009, 1941–1959 CrossRef CAS.
- (a) P. Fontaine, A. Chironi, G. Masson and J. Zhu, Org. Lett., 2008, 10, 1509–1512 CrossRef CAS PubMed; (b) H. Zali-Boeini, M. Mobin, K. Hajibabaei and M. Ghani, J. Org. Chem., 2012, 77, 5808–5812 CrossRef CAS PubMed; (c) M. Pori, P. Galleti, R. Soldati and D. Giacomini, Eur. J. Org. Chem., 2013, 1683–1695 CrossRef CAS; (d) F. Rajabi, S. Nourian, S. Ghiassian, A. M. Balu, M. R. Saidi, J. C. Serrano-Ruizc and R. Luquec, Green Chem., 2011, 13, 3282–3289 RSC.
- Examples for the Strecker reaction using chiral organocatalysts. (a) E. J. Corey and M. J. Grogan, Org. Lett., 1999, 1, 157–160 CrossRef CAS PubMed; (b) M. S. Sigman and E. N. Jacobsen, J. Am. Chem. Soc., 1998, 120, 4901–4902 CrossRef CAS; (c) M. S. Sigman, P. Vachal and E. N. Jacobsen, Angew. Chem., 2000, 112, 1336–1338 (Angew. Chem., Int. Ed., 2000, 39, 1279–1281) CrossRef; (d) M. Rueping, E. Sugiono and C. Azap, Angew. Chem., 2006, 118, 2679–2681 (Angew. Chem., Int. Ed., 2006, 45, 2617–2619) CrossRef; (e) M. Rueping, E. Sugiono and S. A. Moreth, Adv. Synth. Catal., 2007, 349, 759–764 CrossRef CAS; (f) Y. H. Wen, Y. Xiong, L. Chang, J. L. Huang, X. H. Liu and X. M. Feng, J. Org. Chem., 2007, 72, 7715–7719 CAS.
- (a) M. Takamura, Y. Hamashima, H. Usuda, M. Kanai and M. Shibasaki, Angew. Chem., 2000, 112, 1716–1178 (Angew. Chem., Int. Ed., 2000, 39, 1650–1652) CrossRef; (b) N. S. Josephsohn, K. W. Kuntz, M. L. Snapper and H. Hoveyda, J. Am. Chem. Soc., 2001, 123, 11594–11599 CrossRef CAS PubMed; (c) S. Masumoto, H. Usuda, M. Suzuki, M. Kanai and M. Shibasaki, J. Am. Chem. Soc., 2003, 125, 5634–5635 CrossRef CAS PubMed; (d) S. Lundgren, E. Wingstrand, M. Penhoat and C. Moberg, J. Am. Chem. Soc., 2005, 127, 11592–11593 CrossRef CAS PubMed.
- For reviews please see: (a) I. Coldham and R. Hufton, Chem. Rev., 2005, 105, 2765–2809 CrossRef CAS PubMed; (b) K. Harju and Y. J. Kauhaluoma, Recent Res. Dev. Org. Chem., 2004, 8, 111–157 CAS; (c) K. A. Jorgensen and K. V. Gothelf, Chem. Rev., 1998, 98, 863–909 CrossRef; (d) A. Padwa, Angew. Chem., Int. Ed., 1976, 15, 123–136 CrossRef.
- (a) R. Huisgen, Angew. Chem., 1963, 75, 742–754 (Angew. Chem., Int. Ed., 1963, 2, 633–645) CrossRef CAS; (b) R. Huisgen, in 1,3-Dipolar Cycloaddition Chemistry, ed. A. Pawda, Wiley, New York, 1984, pp. 1–176 Search PubMed; (c) R. Huisgen, Proc. Chem. Soc., 1961, 357–369 Search PubMed; (d) R. Huisgen, Pure Appl. Chem., 1989, 61, 613–628 CrossRef CAS.
- (a) R. N. Butler, A. R. Katritzky, C. W. Rees and E. F. V. Scriven, Comprehensive Heterocyclic Chemistry, Pergammon Press, Oxford, UK, 1996, vol. 4 Search PubMed; (b) A. Hantzsch, A. Vagt and J. Liebigs, Ann. Chem., 1901, 314, 339–369 CrossRef CAS; (c) P. D. Zachary and K. B. Sharpless, Org. Lett., 2001, 3, 4091–4094 CrossRef; (d) P. D. Zachary and K. B. Sharpless, Org. Lett., 2002, 4, 2525–2527 CrossRef; (e) D. Amantini, R. Beleggia, F. Fringuelli, F. Pizzo and L. J. Vaccoro, J. Org. Chem., 2004, 69, 2896–2898 CrossRef CAS PubMed; (f) T. Jin, F. Kitahara, S. Kamijo and Y. Yamamoto, Tetrahedron Lett., 2008, 49, 2824–2827 CrossRef CAS; (g) J. Bonnamour and C. Bolm, Chem.–Eur. J., 2009, 15, 4543–4545 CrossRef CAS PubMed; (h) V. Rama, K. Kanagraj and K. Pitchumani, J. Org. Chem., 2011, 76, 9090–9095 CrossRef CAS PubMed; (i) S. Hanessian, D. Simard, B. D. Simard, C. Chenel and E. Haak, Org. Lett., 2008, 10, 1381–1384 CrossRef CAS PubMed; (j) P. A. S. Smith, J. M. Clegg and J. H. Hall, J. Org. Chem., 1958, 23, 524–529 CrossRef CAS; (k) D. K. Mohapatra, P. K. Maity, R. V. Ghorpade and M. K. Gurjar, Heterocycles, 2009, 77, 865–872 CrossRef CAS; (l) F. Fredrik, S. Manner, L. Witstrand and T. Frejd, J. Org. Chem., 2004, 69, 1346–1352 CrossRef PubMed.
- H. Singh, A. S. Chawla, V. K. Kapoor, D. Paul and R. K. Malhotra, Prog. Med. Chem., 1980, 17, 151–183 CrossRef CAS PubMed.
- D. J. Moderhack, Prakt. Chem., 1998, 340, 687–709 CrossRef CAS.
- (a) T. Jin, S. Kamijo and Y. Yamamoto, Tetrahedron Lett., 2004, 45, 9435–9437 CrossRef CAS; (b) J. V. Duncia, D. J. Carini, A. T. Chiu, A. L. Johnson, W. A. Price, P. C. Wong, R. R. Wexler and P. B. Timmermans, Med. Res. Rev., 1992, 12, 149–191 CrossRef CAS PubMed; (c) R. D. Smith, J. V. Duncia, R. J. Lee, D. D. Christ, A. T. Chiu, D. J. Carini, W. F. Herblin, P. B. M. W. Timmermans, R. R. Wexler and P. C. Wong, Methods Neurosci., 1993, 13, 258–280 CAS; (d) R.-Q. Huang, C. L. Bell-Horner, M. I. Dibas, D. F. Covey, J. A. Drewe and G. H. Dillon, J. Pharmacol. Exp. Ther., 2001, 298, 986–995 CAS; (e) M. E. Jung, H. Lal and M. B. Gatch, Neurosci. Biobehav. Rev., 2002, 26, 429–439 CrossRef CAS PubMed; (f) S. Daya, P. T. Kaye and M. Mphahlele, J. Med. Sci. Res., 1996, 24, 137–141 CAS.
- E. E. Smissman, A. Terada and S. El-Antably, J. Med. Chem., 1976, 19, 165–167 CrossRef CAS PubMed.
- (a) J. J. McGuire, C. A. Russell, W. E. Bolanowska, C. M. Freitag, C. S. Jones and T. I. Kalman, Cancer Res., 1990, 50, 1726–1731 CAS; (b) F. Itoh, K. Yukishige, M. Wajima, K. Ootsu and H. Akimoto, Chem. Pharm. Bull., 1995, 43, 230–235 CrossRef CAS PubMed.
- J. A. Zablocky, M. Miyano, N. R. Sashidhar, S. Panzer-Knodle, N. Nicholson and L. Feigen, J. Med. Chem., 1992, 35, 4914–4917 CrossRef.
- (a) W. H. Lunn, D. D. Schoepp, D. O. Calligaro, R. T. Vasileff, L. J. Heinz, C. R. Salhoff and P. J. O'Malley, J. Med. Chem., 1992, 35, 4608–4612 CrossRef CAS PubMed; (b) P. L. Ornstein, M. B. Arnold, D. Evrard, J. D. Leander, D. Lodge and D. D. Shoepp, Bioorg. Med. Chem. Lett., 1993, 3, 43–48 CrossRef CAS.
- (a) A. P. Dhondge, S. N. Afraj, C. Nuzlia, C. Chen and G. H. Lee, Eur. J. Org. Chem., 2013, 19, 4119–4130 CrossRef; (b) S. N. Afraj, C. Chen and G. H. Lee, RSC Adv., 2014, 4, 26301–26308 RSC.
- (a) J. T. Kuethe, D. R. Gauthier, G. L. Beutner and N. A. Yasuda, J. Org. Chem., 2007, 72, 7469–7472 CrossRef CAS PubMed; (b) L. Shu and P. Wang, Org. Process Res. Dev., 2008, 12, 298–300 CrossRef CAS; (c) T. Ooi, Y. Uematsu and K. Maruoka, J. Am. Chem. Soc., 2006, 128, 2548–2549 CrossRef CAS PubMed.
- D. Nils, B. Anders and A. Hans, Adv. Synth. Catal., 2004, 346, 1101–1110 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 1041454. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra00063k |
| This journal is © The Royal Society of Chemistry 2016 |