One-step hydrothermal synthesis of magnetic responsive TiO2 nanotubes/Fe3O4/graphene composites with desirable photocatalytic properties and reusability†
Ping Zhangab,
Zunli Mo*a,
Yawen Wanga,
Lijuan Hanc,
Chun Zhanga,
Guoping Zhaoa and
Zheng Lia
aKey Laboratory of Eco-Environment-Related Polymer Materials, Ministry of Education of China, College of Chemistry and Chemical Engineering, Northwest Normal University, Lanzhou, Gansu 730070, People's Republic of China. E-mail: mozlnwnu2011@163.com; Tel: +86-931-7971829
bGansu Science and Technology Museum, Lanzhou, Gansu 730030, People's Republic of China
cNatural Energy Institute, Gansu Academy of Sciences, Lanzhou, Gansu 730046, People's Republic of China
First published on 1st April 2016
Abstract
Here, we report a facile approach to preparing a magnetic responsive TiO2 nanotubes/Fe3O4/graphene composite (TNTs/Fe3O4/GN), in which the reduction of graphene oxide as well as the formation of Fe3O4 nanoparticles and TNTs occurred simultaneously. The preparation time and procedure were effectively simplified and shortened. A series of techniques including XRD, TEM, BET, VSM and EDX were employed to characterize the composite. Also, the photocatalytic activity of the composite was evaluated via the removal of methylene blue. The results showed that TNTs and Fe3O4 nanoparticles were deposited on layers of GN, and a synergistic effect was observed in the composite. Owing to interfacial contact between TNTs and GN, the recombination of photogenerated electrons/holes was suppressed and the photocatalytic performance was enhanced. The composite not only displayed higher photocatalytic activity than TNTs and P25, but also exhibited desirable reusability and rapid separation performance, which make it a potential candidate for applications in wastewater treatment.
1. Introduction
In recent years, the treatment of refractory organic contaminants in wastewater by photocatalytic reactions has attracted considerable attention from scholars. Photocatalytic reactions have been widely used for the degradation of organic compounds in wastewater owing to their non-polluting nature and rapid response.1,2 TiO2 is considered to be the most promising photocatalyst because of its chemical stability, resistance to acid and alkali, non-toxic nature, low cost, strong oxidative capacity and good catalytic performance.3,4 To further improve the photocatalytic properties of TiO2, scientists have made many meaningful investigations and achieved good effects.5–7 However, how to reduce the loss of photocatalysts and retain their maximum performance during the separation and recycling process has become a bottleneck in the practical application of photocatalysts.8,9 Magnetic separation is considered to be a quicker and more effective technique than traditional separation technology including centrifugation and filtration. When magnetic media are introduced into a photocatalyst, they can not only enable the solid–liquid separation and reuse of the photocatalyst but also avoid the loss of the catalyst and reduce the cost invested in equipment for separation.10 Therefore, more and more attention has been paid to developing magnetically separated photocatalysts with high stability and desirable photocatalytic performance.The advent of GN has earned much attention in the field of environmental rehabilitation materials owing to its flexible two-dimensional structure and unique properties.11,12 On the one hand, it can be employed as a high-quality adsorbent for environmental restoration owing to its large surface area, unique π–π conjugation system and two-dimensional planar structure. Organic pollutants can be adsorbed strongly by the surface of GN via interaction forces between dyes and the aromatic regions of GN.13–15 On the other hand, GN has better electron transport capacity and can serve as an ideal partner for improving the photocatalytic activity of TiO2. Because GN can accept and quickly transfer photogenerated electrons and suppress the recombination of electrons and holes, the photocatalytic performance of TiO2 can be enhanced.16,17 In general, GN has been prepared by exfoliating graphite to form individual graphene oxide (GO) layers using strong oxidizing agents, which introduce oxygen functional groups. These functional groups make the GO sheets readily soluble in many solvents and facilitate surface modifications.18,19 However, GO exhibits poor electronic conductivity, owing to the interruption of the π system by substitution with oxygen functional groups. Therefore, various reducing agents20,21 and heating in alkaline conditions18,22,23 have been used to reduce GO to restore its sp2 hybridized network and increase its electronic conductivity.
Fe3O4 is one of the most widely used soft magnetic materials. However, pure iron tetroxide is easily aggregated during preparation and degradation processes owing to its large magnetic dipole interaction, which can reduce the catalytic activity and stability of Fe3O4.24,25 Currently, loading this magnetic metal oxide on GN sheets is believed to be a good strategy for solving this problem owing to its large specific surface area.26,27 Lin et al.28 synthesized ternary GN–TiO2–Fe3O4 composites by a two-step procedure, in which a GN–TiO2 precursor was first synthesized and then Fe3O4 was deposited by a co-precipitation method, which exhibited higher removal efficiency for rhodamine B and durability under UV light irradiation. Compared with TiO2 nanoparticles, TiO2 nanotubes (TNTs), which have a tubular structure and two open ends, have a much larger specific surface area and numerous active sites, which exhibit good adsorption behavior and photocatalytic activity.29,30 Moreover, a greater degree of interfacial contact between TNTs and graphene should favor charge separation to enhance the photocatalytic activity.31 However, until now, reports about the assembly of Fe3O4 and TiO2 nanotubes compounded with GN have been limited.
In this paper, we report a facile route for the growth of TNTs and Fe3O4 nanoparticles on graphene oxide sheets via a one-step hydrothermal method in alkaline conditions. In this method, graphene oxide (GO), TiO2 and water-soluble divalent and ferric iron salts were used as raw materials. In alkaline hydrothermal conditions, TNTs and an aggregate of Fe3O4 nanoparticles were grown on the surface of GO while reduced graphene was formed simultaneously by the deoxygenation of GO. The reduction of GO as well as the formation of Fe3O4 nanoparticles and TNTs was completed in one step, which can effectively simplify the procedure and shorten the preparation time of the new magnetic responsive TNTs/Fe3O4/GN photocatalyst. Importantly, we tested the photocatalytic properties and reusability of the composites. The results showed that the TNTs/Fe3O4/GN composites exhibited desirable photocatalytic activity and durability for the degradation of organic dyes. The enhancement of the photocatalytic activity of the TNTs/Fe3O4/GN composites could be ascribed to the favorable charge transfer kinetics of the graphene structure and the higher photocatalytic activity of TNTs.
2. Experimental section
2.1. Materials and methods
Graphite was purchased from the Chengdu Institute of Organic Chemistry. FeCl3·6H2O and acetic acid were bought from Shanghai Chemical Reagents Company. FeSO4·7H2O was obtained from the Beijing Chemical Reagent Factory. Tetra-n-butyl titanate, anhydrous ethanol, NaOH and methylene blue (MB) were supplied by China National Pharmaceutical Group. Potassium permanganate (KMnO4) was purchased from Dan Dong No. 1 Chemical Plant. Hydrochloric acid (HCl) was supplied by Baiyin Liangyou Chemical Reagents Co., Ltd. All the reagents were of analytical grade and were used without further purification. Deionized water was used for the preparation of all aqueous solutions. TiO2 (P25) was obtained from Degussa (Germany). GO was prepared by a modified Hummers method.322.2. Preparation of magnetic responsive TNTs/Fe3O4/GN composites
Initially, 0.05 g GO was dispersed in 50 mL deionized water and treated by ultrasound for 1 h to obtain a uniform dispersion. Then, 0.45 g P25 was added slowly to the dispersion with continuous ultrasound treatment and stirring for 0.5 h, then 0.135 g FeCl3·6H2O and 0.070 g FeSO4·7H2O were added and mixed completely with further ultrasound treatment and stirring for 15 min. Next, 1 mol L−1 NaOH was dropped into the above mixed solution to provide a pH value of 10. After that, 20.0 g NaOH was added. Then, the mixture was transferred to a Teflon-lined stainless-steel autoclave, which was kept at 120 °C for 24 h. After cooling to room temperature, the mixture was washed with deionized water to neutral pH and soaked for 1 h in 0.1 M HCl solution. Finally, the final product was washed with deionized water and ethanol and separated magnetically by employing a magnet. The resulting powders were dried in a vacuum oven at 60 °C for 12 h, and calcined for 60 min at 300 °C in a N2 protective atmosphere. Then, the magnetic responsive TNTs/Fe3O4/GN composite was obtained.2.3. Characterization
The morphology and structure of a sample were determined on a Hitachi S-4800 scanning electron microscope (SEM) operating at 30 kV, with Au sprayed prior to examination, and a JEOL JEM-2100 transmission electron microscope (TEM). X-ray diffraction (XRD) patterns of samples were collected at room temperature with Cu Kα X-ray sources using a D/Max-2400 powder diffraction meter (Japanese Physical Company). Nitrogen adsorption–desorption isotherms were obtained at 77 K using an ASAP-2020 physical adsorption analyzer (Micromeritics Instruments, USA) after samples were vacuum-degassed at 150 °C. The surface area and pore volume of samples were determined by the Brunauer–Emmett–Teller (BET) model using adsorption data and the Barrett–Joyner–Halenda (BJH) model based on desorption isotherms, respectively. Isothermal magnetization curves were recorded at several different points with an external magnetic field of 0–80 kOe.2.4. Adsorption and photocatalytic performance measurements
The adsorptive and photocatalytic performances of the TNTs/Fe3O4/GN composite were evaluated by measuring the adsorption and photocatalytic degradation of MB in aqueous solution in the dark and then under UV light irradiation, respectively. The photocatalytic activity of samples was measured using an XPA-7 photocatalytic reactor (Xujiang Machine Plant, Nanjing). The temperature of the reaction was maintained at room temperature by circulating water. Firstly, a 0.05 g sample was dispersed in a 50 mL aqueous solution of MB with an initial concentration of 40 mg L−1. The suspension was stirred in the dark for 30 min to reach adsorption–desorption equilibrium. Then, the solution was irradiated by a 100 W medium-pressure mercury lamp and samples were taken from the mixture at intervals of 20 min for testing the photocatalytic activity. The concentration of MB was determined at a wavelength of 666 nm by a UV-vis spectrophotometer.3. Results and discussion
The morphology of the as-prepared samples was characterized by TEM. Fig. 1a shows an image of pure graphene. From Fig. 1a, we can clearly see the morphology of GN sheets, of which the roughness obviously exists in monolayer and multilayer form. Fig. 1b–c show images of the TNTs/Fe3O4/GN composite at different magnifications. As shown in Fig. 1b, Fe3O4 particles aggregate with a diameter of 100–130 nm dispersed homogeneously in the graphene layer, and the boundary of the graphene layer is clearly visible. Moreover, tiny nanotubes can be seen in the graphene layer. These nanotubes, which have a diameter of about 10 nm and a length of about 100 nm, are uniformly loaded on the lamellar structure of GN. When the image is magnified (Fig. 1c), it can be seen that the aggregate of Fe3O4 particles is composed of many small nanoparticles with a size of 10–20 nm. The inset of Fig. 1c shows the characteristic diffraction patterns of graphene, anatase TiO2 and Fe3O4. Therefore, we can conclude that the formation of magnetic Fe3O4 particles and TNTs, as well as the reduction of GO, occurred simultaneously, which indicates that the one-step hydrothermal co-precipitation method is an efficient method for the preparation of multi-component composites. Fig. 1d shows the EDS spectrum of the TNTs/Fe3O4/GN composite. The composite consists of Fe, Ti and O, as well as C and Cu from a C-coated Cu grid, which confirms the formation of Fe oxides and Ti oxides on GN during the hydrothermal process.The XRD patterns of GO, GN, Fe3O4, TNTs and the TNTs/Fe3O4/GN composite are shown in Fig. 2. It can be observed that the characteristic peak of GO at 2θ = 10.6° has disappeared and a new peak of GN at 23.3° has appeared in the XRD pattern of the TNTs/Fe3O4/GN composite, which indicates that GO has been successfully reduced via the hydrothermal process.33 Fig. 2e shows the XRD pattern of the TNTs/Fe3O4/GN composite. The strong diffraction peaks that appear at 25.4°, 37.9°, 48.0°, 55.1° and 62.9° are attributed to the (101), (004), (200), (211) and (204) crystal planes of the anatase phase, respectively.34 The diffraction peaks at 30.3° and 57.6° correspond to the characteristic peaks of Fe3O4.35 All the observations confirm that the TNTs/Fe3O4/GN composite was successfully prepared by the one-step hydrothermal co-precipitation method. The grain size of Fe3O4 was calculated by the Scherrer formula (D = Kλ/β![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ), where D is the estimated grain size, K refers to a constant value of 0.89, β is the integral of the width at half-height, θ is the diffraction angle, and λ is the wavelength of the X-rays, which was equal to 0.15418 nm.36 The results show that the average size of the Fe3O4 particles is about 12.1 nm, which is in agreement with the result of TEM analysis.
θ), where D is the estimated grain size, K refers to a constant value of 0.89, β is the integral of the width at half-height, θ is the diffraction angle, and λ is the wavelength of the X-rays, which was equal to 0.15418 nm.36 The results show that the average size of the Fe3O4 particles is about 12.1 nm, which is in agreement with the result of TEM analysis.
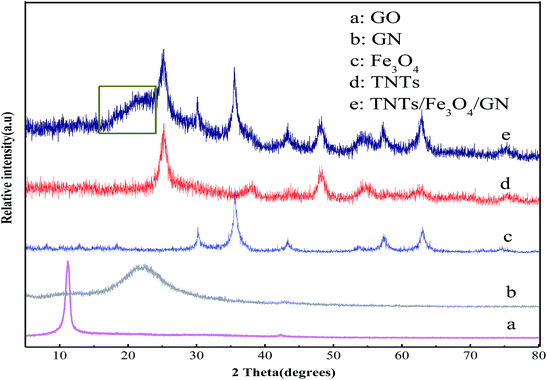 | ||
| Fig. 2 XRD patterns of obtained samples: GO (a), GN (b), Fe3O4 (c), TNTs (d), and TNTs/Fe3O4/GN composite (e). | ||
Fig. 3 depicts the preparation scheme of the magnetic responsive TNTs/Fe3O4/GN composite. The huge surface area of GO was taken as a template, P25 powder (80% anatase and 20% rutile) was dispersed in a dispersion of GO in deionized water with continuous ultrasound treatment to enable complete mixing, and then a certain proportion of FeCl3·6H2O and FeSO4·7H2O was added with ultrasound treatment to form a stable system. Because the surface of GO contains many functional groups such as epoxides and alcohols and the edges of GO possess carboxylic acids and other keto groups, TiO2 nanoparticles and divalent and ferric iron bind to functional groups on the surface of GO. Then, a certain amount of NaOH was added and the mixture was transferred to a Teflon-lined stainless-steel autoclave to conduct the alkaline hydrothermal reaction. In the alkaline hydrothermal reaction, TNTs were formed by the rolling up of exfoliated titanate sheets while Fe3O4 nanoparticles formed an aggregate on the surface of GN layers. It is remarkable that GO was transformed into GN in the alkaline hydrothermal reaction. It is thought that GO actually consists of two distinct components that are non-covalently complexed together, the majority by mass being large functionalized graphene-like sheets together with small, more highly oxidized fragments or debris.18 The highly oxidized debris was deprotonated, and the charge of the debris was negative. The interaction between the oxidized debris and the functionalized graphene sheet became repulsive under basic conditions. The debris was easily separated from the functionalized graphene sheet, and was hard to reattach. Meanwhile, GN formed owing to the loss of the highly oxidized debris. In the hydrothermal reaction, GO was reduced to GN, the magnetic Fe3O4 particles aggregated, and TNTs were deposited on the layer of GN. Then, the magnetic responsive TNTs/Fe3O4/GN composite was prepared in one step. The formation of magnetic Fe3O4 particles and TNTs, as well as the reduction of GO, occurred simultaneously, which effectively omitted the steps of modification of the inorganic nanoparticles and redispersion in the precursor.
To study the textural properties and nature of porosity of the composite, analysis of the nitrogen adsorption/desorption isotherm was carried out. As shown in Fig. 4a, the nitrogen adsorption/desorption isotherm of the TNTs/Fe3O4/GN composite exhibited a typical IUPAC type IV form with a distinct type H3 hysteresis loop in the relative pressure range of 0.7–0.95, which suggests the characteristics of mesoporous materials.37 The BET specific surface area was thus calculated from the adsorption data to be 341.83 m2 g−1. The pore size distribution of the TNTs/Fe3O4/GN composite determined by the BJH method shows a large range from 2 to 80 nm, as shown in Fig. 4b. The peaks in the pore size distribution of the sample were found to be around 3 nm and 19.2 nm, which can be ascribed to the pores of TNTs and the gap between Fe3O4 particles, and the curvature of GN, respectively. The large surface area and porous structure are beneficial for applications as a catalyst for the adsorption and degradation of dyes.
 | ||
| Fig. 4 Nitrogen adsorption–desorption isotherm curve (a) and pore diameter distribution (b) of TNTs/Fe3O4/GN composite. | ||
Fig. 5 shows the saturation magnetization (Ms) values of the TNTs/Fe3O4/GN nanocomposite and pure Fe3O4 particles. All the samples exhibit typical superparamagnetic features with negligible coercivity and remanence. It can be seen that the saturation magnetization values of pure Fe3O4 particles and the magnetic responsive TNTs/Fe3O4/GN composite are 60.02 emu g−1 and 22.32 emu g−1, respectively, which indicates that the magnetic property of the composite is weaker than that of pure Fe3O4. This may be due to the fact that Fe3O4 particles in the composite were coated by GN sheets and TNTs, which reduced the saturation magnetization and the magnetic response.38 Although the saturation magnetization of TNTs/Fe3O4/GN was weakened compared with that of pure Fe3O4 particles, the magnetically responsive TNTs/Fe3O4/GN composite can still be successfully extracted under the influence of an external magnetic field. As shown in the inset of Fig. 5, when the photocatalyst was dispersed in an aqueous solution of MB, the dispersion turned dark blue. The color of the suspension faded after irradiation by UV light. Thus, it can be observed that the composite is rapidly attracted toward a magnet under an external magnetic field, which indicates excellent magnetic response.
In this study, MB was used as a model organic pollutant to evaluate the adsorption ability and photocatalytic performance of different catalysts. In a test of photocatalytic performance, an adsorption equilibrium experiment was conducted by stirring for 30 min firstly under dark conditions. Secondly, a photocatalytic performance test was performed after turning on a UV light source (100 W mercury lamp). The dependence of the relative concentration on the reaction time is plotted in Fig. 6. As displayed in Fig. 6a, during the adsorption process the concentration of the dye in different samples declined to different extents. P25 exhibited negligible adsorption activity for the adsorption of MB after stirring for 30 min. In contrast, about 37.5% and 69% of MB was adsorbed by GN and TNTs, respectively. With the TNTs/Fe3O4/GN composite, almost 90.0% of MB can be adsorbed under identical conditions, which can be ascribed to the combined synergistic effect of GN and TNTs. After irradiation for 20 minutes, the removal rate of MB by the magnetic responsive TNTs/Fe3O4/GN composite was 99.8%, which is higher than those of pure TNTs and P25. Its huge specific area and porous structure may be responsible for the better adsorption property of the TNTs/Fe3O4/GN composite. In order to further study the optimal properties of the TNTs/Fe3O4/GN composite, we also evaluated the photocatalytic properties of the TNTs/Fe3O4/GN composite with various TNTs contents (Fig. 6b). Interestingly, the photocatalytic activity of pure TNTs was still relatively low. In contrast, considerably higher removal efficiency was recorded for the TNTs/Fe3O4/GN composite. In particular, 70% TNTs/Fe3O4/GN exhibited the best performance and could remove almost all of the MB after UV irradiation for 20 min, which implies that it has great potential in applications for removing dyes from water.
 | ||
| Fig. 6 Adsorption and photodegradation of MB under UV light irradiation by different photocatalysts: (a) TNTs/Fe3O4/GN, TNTs, P25 and GN. (b) TNTs/Fe3O4/GN composite with different TiO2 contents. | ||
Besides the activity of photocatalysts, their reusability is significant for their practical application.39 Therefore, six successive recycling tests for the degradation of MB by the TNTs/Fe3O4/GN composite were performed. In these tests, the initial concentration of MB solution was 40 mg L−1 in each run, and the catalyst was recovered by a magnet at the end of the experiment. The results are shown in Fig. 7. The removal rate of MB was still greater than 90% after six repeated uses, which reveals that this photocatalyst possesses high stability and good reusability. Under the same measurement conditions as for MB, the adsorption ability and photocatalytic performance of TNTs/Fe3O4/GN composites for rhodamine B were tested, as shown in Fig. S1.† The result showed that TNTs/Fe3O4/GN composites also exhibited good adsorption ability and photocatalytic performance for rhodamine B. About 85.2% of rhodamine B was adsorbed after stirring for 30 min. After irradiation for 20 minutes, the removal rate of magnetic responsive TNTs/Fe3O4/GN composite to MB was 95.6%, i.e., the TNTs/Fe3O4/GN composites exhibited good adsorption ability and photocatalytic performance for different dyes.
Based on the characterization and experimental data discussed above, a simple photocatalytic mechanism of the TNTs/Fe3O4/GN composite is depicted in Fig. 8. This hybrid is of ternary architecture and intimate interfacial contact exists between the components. During the adsorption process, the large surface area of TNTs and GN endow them with excellent adsorption ability, which can increase the chance of contact between contaminant molecules and materials. Under UV light illumination, electrons (e−) can be excited from the valence band (VB) to the conduction band (CB) of TNTs. Owing to the interfacial contact between TNTs and GN, these electrons can migrate subsequently into GN, which is an excellent conductor. Thus, the recombination of photogenerated electrons/holes can be suppressed. In this process, GN acts as an electron acceptor and carrier to improve the separation efficiency of photogenerated electrons/holes, which further enhances the photocatalytic performance. Then, the transferred electrons are captured by adsorbed O2 on the surface of GN and finally yield superoxide radicals, which can oxidize dyes adsorbed on the surface of TNTs/Fe3O4/GN. Moreover, photogenerated holes that accumulate in the VB of TNTs could react with surface-adsorbed H2O or OH− to produce hydroxyl radicals. Both superoxide radicals and hydroxyl radicals have strong oxidative abilities and can degrade dyes into CO2, H2O and other intermediates.40,41 Moreover, dye molecules may also be oxidized by holes directly. In conclusion, a synergistic effect exists in TNTs and GN, which makes the photocatalytic performance enhanced compared with that of simple TNTs or GN. Owing to the addition of magnetic materials, the composites can be separated easily and exhibit good reusability.
4. Conclusions
In summary, a new type of magnetic responsive TNTs/Fe3O4/GN composite was prepared by a facile one-step hydrothermal process. In this process, the reduction of GO, as well as the formation of Fe3O4 nanoparticles and TNTs, is completed in one step. The method not only effectively simplifies the procedure of synthesizing a multi-component magnetic responsive photocatalyst, but also maintains the uniformly dispersed state of the different components. Compared with TNTs and P25, the TNTs/Fe3O4/GN composite possesses higher adsorption capacity and photocatalytic activity for the removal of MB under UV light irradiation. Importantly, the TNTs/Fe3O4/GN composite can be rapidly separated from aqueous solutions under an external magnetic field, and exhibits remarkable reusability owing to the incorporation of magnetic particles.Acknowledgements
The authors are grateful for financial aid from the National Natural Science Foundation of China (51262027), the financial support of the Natural Science Foundation of Gansu Province (1104GKCA019; 1010RJZA023), Science and Technology Tackle Key Problem Item of Gansu Province (2GS064-A52-036-08) and the fund of the State Key Laboratory of Solidification Processing in NWPU (SKLSP201011), Gansu Province Major Project of Science and Technology (143GKDA013), and the Youth Science and Technology Fund of Gansu Province (1308RJYA051).References
- Q. C. Liu, D. K. Ma, Y. Y. Hu, Y. W. Zeng and S. M. Huang, ACS Appl. Mater. Interfaces, 2013, 5, 11927–11934 CAS.
- S. Rasalingam, R. Peng and R. T. Koodali, Appl. Catal., B, 2015, 174, 49–59 CrossRef.
- J. R. Chen, F. X. Qiu, W. Z. Xu, S. S. Cao and H. J. Zhu, Appl. Catal., A, 2015, 495, 131–140 CrossRef CAS.
- H. Y. Liu, J. B. Joo, M. Dahl, L. S. Fu, Z. Z. Zeng and Y. D. Yin, Energy Environ. Sci., 2015, 8, 286–296 CAS.
- D. Dolat, S. Mozia, R. J. Wróbel, D. Moszynski, B. Ohtani, N. Guskos and A. W. Morawski, Appl. Catal., B, 2015, 162, 310–318 CrossRef CAS.
- X. Pan, Y. Zhao, S. Liu, C. L. Korzeniewski, S. Wang and Z. Y. Fan, ACS Appl. Mater. Interfaces, 2012, 4(8), 3944–3950 CAS.
- D. Sarkar, C. K. Ghosh, S. Mukherjee and K. K. Chattopadhyay, ACS Appl. Mater. Interfaces, 2013, 5(2), 331–337 CAS.
- T. J. Xin, M. L. Ma, H. P. Zhang, J. Gu, S. Wang, M. Liu and Q. Zhang, Appl. Surf. Sci., 2014, 288, 51–59 CrossRef CAS.
- K. Mandel, F. Hutter, C. Gellermann and G. Sextl, Sep. Purif. Technol., 2013, 109, 144–147 CrossRef CAS.
- J. Y. Jing, J. Li, J. Feng, W. Li and W. Y. William, Chem. Eng. J., 2013, 219, 355–360 CrossRef CAS.
- W. J. Liu, J. Y. Cai and Z. H. Li, ACS Sustainable Chem. Eng., 2015, 3, 277–282 CrossRef CAS.
- D. Kalel and P. Thakur, J. Porous Mater., 2015, 22, 797–806 CrossRef.
- B. Weng, M. Q. Yang, N. Zhang and Y. Xu, J. Mater. Chem. A, 2014, 2, 9380–9389 CAS.
- L. Zhang, H. Z. Li, Y. Liu, Z. Tian, B. Yang, Z. B. Sun and S. Q. Yan, RSC Adv., 2014, 4, 48703–48711 RSC.
- W. Yan, F. He, S. L. Gai, P. Gao, Y. J. Chen and P. P. Yang, J. Mater. Chem. A, 2014, 2, 3605–3612 CAS.
- L. X. Zhang, J. Zhang, H. F. Jiu, C. H. Ni, X. Zhang and M. L. Xu, J. Phys. Chem. Solids, 2015, 86, 82–89 CrossRef CAS.
- Z. X. Gan, X. L. Wu, M. Meng, X. B. Zhu, L. Yang and P. K. Chu, ACS Nano, 2014, 8, 9304–9310 CrossRef CAS PubMed.
- J. P. Rourke, P. A. Pandey, J. J. Moore, M. Bates, I. A. Kinloch, R. J. Young and N. R. Wilson, Angew. Chem., 2011, 123, 3231–3235 CrossRef.
- Y. Chen, X. Zhang, D. Zhang, P. Yu and Y. Ma, Carbon, 2011, 49, 573–580 CrossRef CAS.
- H. J. Shin, K. K. Kim, A. Benayad, S. M. Yoon, H. K. Park, I. S. Jung, M. H. Jin, H. K. Jeong, J. M. Kim, J. Y. Choi and Y. H. Lee, Adv. Funct. Mater., 2009, 19, 1987–1992 CrossRef CAS.
- S. Park, J. An, J. R. Potts, A. Velamakanni, S. Murali and R. S. Ruoff, Carbon, 2011, 49, 3019–3023 CrossRef CAS.
- T. Bayer, B. V. Cunning, R. Selyanchyn, T. Daio, M. Nishihara, S. Fujikawa, K. Sasakia and S. M. Lyth, J. Membr. Sci., 2016, 508, 51–61 CrossRef CAS.
- S. Sun, M. Sun, Y. Fang, Y. Wang and H. Wang, RSC Adv., 2016, 6, 13063–13071 RSC.
- H. C. Cui, D. C. Lin and Z. L. Zhang, Mater. Lett., 2015, 143, 38–40 CrossRef CAS.
- M. Abboud, S. Youssef, J. Podlecki, R. Habchi, G. Germanos and A. Foucaran, Mater. Sci. Semicond. Process., 2015, 39, 641–648 CrossRef CAS.
- X. B. Zhu, Z. L. Mo, C. Zhang, B. Wang, G. P. Zhao and R. B. Guo, Mater. Res. Bull., 2014, 59, 223–226 CrossRef CAS.
- Y. Z. Wan, Z. W. Yang, G. Y. Xiong, R. S. Guo, Z. Liu and H. L. Luo, J. Power Sources, 2015, 294, 414–419 CrossRef CAS.
- Y. Lin, Z. G. Geng, H. B. Cai, L. Ma, J. Chen, J. Zeng, N. Pan and X. P. Wang, Eur. J. Inorg. Chem., 2012, 28, 4439–4444 CrossRef.
- D. Yang, Y. Sun, Z. Tong, Y. Tian, Y. Li and Z. Jiang, J. Phys. Chem. C, 2015, 119, 5827–5835 CAS.
- L. J. Han, Z. Ma, Z. H. Luo, G. Liu, J. T. Ma and X. C. An, RSC Adv., 2016, 6, 6643–6650 RSC.
- M. Sun, Y. Fang, Y. Wang, S. Sun, J. He and Z. Yan, J. Alloys Compd., 2015, 650, 520–527 CrossRef CAS.
- D. C. Marcano, D. V. Kosynkin, J. M. Berlin, A. Sinitskii, Z. Z. Sun, A. Slesarev, L. B. Alemany, W. Lu and J. M. Tour, ACS Nano, 2010, 4, 4806–4814 CrossRef CAS PubMed.
- W. H. Feng, Z. X. Pei, Z. B. Fang, M. L. Huang, M. L. Lu, S. X. Weng, Z. Y. Zheng, J. Hu and P. Liu, J. Mater. Chem. A, 2014, 2, 7802–7811 CAS.
- J. Lu, M. Wang, C. Deng and X. Zhang, Talanta, 2013, 105, 20–27 CrossRef CAS PubMed.
- Z. L. Mo, C. Zhang, R. B. Guo, S. Meng and J. Zhang, Ind. Eng. Chem. Res., 2011, 50, 3534–3539 CrossRef CAS.
- H. El-Deeb and M. Bron, J. Power Sources, 2015, 275, 893–900 CrossRef CAS.
- Y. Peng, M. Yan, Q. G. Chen, C. M. Fan, H. Y. Zhou and A. W. Xu, J. Mater. Chem. A, 2014, 2, 8517–8524 CAS.
- P. Zhang, Z. L. Mo, L. J. Han, Y. W. Wang, G. P. Zhao, C. ZhanG and Z. Li, J. Mol. Catal. A: Chem., 2015, 402, 17–22 CrossRef CAS.
- J. F. Wang, T. Tsuzuki, B. Tang, X. L. Hou, L. Sun and X. G. Wang, ACS Appl. Mater. Interfaces, 2012, 4, 3084–3090 CAS.
- L. Zhang, H. Z. Li, Y. Liu, Z. Tian, B. Yang, Z. B. Sun and S. Q. Yan, RSC Adv., 2014, 4, 48703–48711 RSC.
- Y. Q. Shi, K. Q. Zhou, B. B. Wang, S. H. Jiang, X. D. Qian, Z. Gui, R. K. K. Yuen and Y. Hu, J. Mater. Chem. A, 2014, 2, 535–544 CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra03389j |
| This journal is © The Royal Society of Chemistry 2016 |





