Polyelectrolyte Brønsted acid catalyzed three-component Mannich reactions accelerated by emulsion†
Abstract
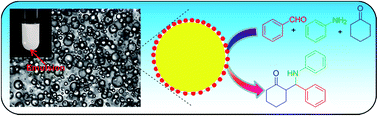
An effective polyelectrolyte Brønsted acid (polyacrylic acid) catalyzed three-component Mannich reaction accelerated by emulsion has been developed. The results demonstrated that the polyacrylic acid (PAA) provided the best catalytic activity in water because of the formation of emulsions during the reaction. This newly developed simple catalyst could be recycled at least five times without any loss of activity.

 Please wait while we load your content...
Please wait while we load your content...