Electrocatalytic degradation of ibuprofen in aqueous solution by a cobalt-doped modified lead dioxide electrode: influencing factors and energy demand
Ying Wang*a,
Chanchan Shena,
Lifang Lib,
Haiyan Lic and
Manman Zhanga
aThe Key Laboratory of Water and Sediment Sciences, Ministry of Education, School of Environment, Beijing Normal University, Beijing 100875, P. R. China. E-mail: yingwang@bnu.edu.cn; Tel: +86-10-5880-2851
bCollege of Chemical Engineering, Beijing University of Chemical Technology, Beijing 100029, P. R. China
cEnvironment of Ministry of Education, Beijing University of Civil Engineering and Architecture, Beijing 100875, P. R. China
First published on 18th March 2016
Abstract
A Co-doped modified PbO2 electrode was prepared to electrocatalytically oxidize IBU in aqueous solution. The effects of initial IBU concentration (40–320 mg L−1), initial pH (4–10), current density (3–30 mA cm−2), natural organic matter and small molecular organic acid were investigated. The structure, morphology and electrochemical properties of the electrode were studied by X-ray diffraction, scanning electron microscopy, linear sweep voltammetry and cyclic voltammograms. The doping of Co may decrease the particle size and increase the lifetime of PbO2, which favors the electrocatalytic activity. The results indicated that the Co-PbO2 electrode exhibited a highly effective oxidation capacity for IBU. After 60 min of electrolysis, the removal of IBU and COD at a current density of 3 mA cm−2 for 80 mg L−1 of IBU reached 98.7% and 32.1%, respectively, and the degradation of COD was 53.6% after 180 min of reaction. The reaction apparently followed a first-order kinetics model. When the IBU initial concentration was 80 mg L−1, the highest reaction rate and energy efficiency were observed. Considering the energy demand and space efficiency, the applied current density of 3 mA cm−2 was the most suitable. Lower pH favored degradation because the oxygen evolution reaction was restrained. The addition of low concentrations (10 mg L−1) of humic acid and fulvic acid could promote the degradation of IBU, whereas high concentrations (20–40 mg L−1) inhibited the degradation of IBU. Moreover, the addition of oxalic acid and citric acid (0.1–0.5 mmol L−1) could inhibit IBU degradation. Finally, the possible reaction pathways were proposed.
1 Introduction
An increasing number of pharmaceuticals are being produced and used worldwide, and there is a resulting subsequent discharge into the aquatic environment annually. Pharmaceutically active compounds in the water environment have caused great concern owing to their potential hazard to the safety of the ecosystem and humans.1,2Ibuprofen (IBU), the third most widely used drug in the world, is a non-steroidal, anti-inflammatory analgesic, antipyretic drug used for the treatment of fever, migraine, muscle aches, rheumatoid arthritis and tooth aches.3 Owing to its widespread applications, several kilotons of IBU are synthesized globally.4 Recently, IBU has been frequently detected at concentrations ranging between nanograms and micrograms per liter in wastewater and surface water. For example, IBU has been found in effluents (up to 22 μg L−1) and influents (up to 84 μg L−1) from sewage treatment plants.5 The average concentration detected in major rivers of Korea was 0.03 μg L−1.6
The conventional sewage treatment process has limits in the elimination of IBU because of its non-volatilization, long period and low oxidation. At present, advanced oxidation processes (AOPs), including UV, O3, photodegradation, ultrasound, Fenton and electrochemical degradation7–12 et al. showed obvious oxidation effects as a result of the formation of hydroxyl radicals. Among these, the electrochemical degradation process has been paid the most attention owing to its high efficiency and low cost, easy operation and lack of secondary pollution. The development of an electrode with higher activity and the investigation of the influence of system effects have aroused great interest.
Previous studies have reported that several materials can be used as anodes for oxidation, such as PbO2, SnO2 and boron-doped diamond (BDD) electrodes.13–17 At present, the electrochemical degradation of IBU has been studied on Ti/Pt/PbO2 and BDD electrodes because of their high oxygen evolution potential and strong mechanical properties. However, BDD anode is expensive and has weak adsorption performance, limiting its application. Recently, PbO2 electrode has been widely used as anode material because of its high electric catalytic performance and low cost. To further improve the electrocatalytic activity of PbO2 electrodes, Cd, La carbon nanotubes, etc. were doped into the surface layer for modification.18,19 Among these, cobalt (Co), as a abundant transition metal, displayed exceptional catalytic properties.20–23
In this work, a Co-doped PbO2 electrode was prepared for the electrochemical degradation of IBU. The electrochemical properties of the electrode were investigated by linear scanning voltammogram (LSV) and cyclic voltammograms (CV). Systematical experiments were conducted to study the effect of experimental parameters, such as initial concentration of IBU, initial pH and applied current density. Humic acid (HA) and fulvic acid (FA) (primary fractions of natural organic matter) as well as oxalic acid (OA) and citric acid (CA) (main small molecular organic acid) are widespread in the aquatic environment. They certainly have an influence on the degradation of IBU. However, to the best of our knowledge, their effects on the electrochemical degradation of organic matter are less. Therefore, the influences of HA, FA, OA and CA were also evaluated. Finally, the intermediates were identified and the possible degradation pathways were proposed.
2 Materials and methods
2.1 Materials
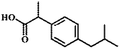
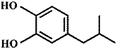
High-purity (99.5%) titanium mesh (Beijing Titanium Industrial and Trade Company) was selected as the metal matrix of PbO2 electrodes. Sodium 2-(4-isobutylphenyl) propanoate, known as ibuprofen (IBU), with a purity of 99.9%, was purchased from Sigma-Aldrich. Acetonitrile (HPLC grade) was procured from Merida. Other chemicals were of analytical grade and used without further purification. All solutions were prepared using deionized water.2.2 Electrode preparation
Before deposition, the Ti substrates underwent sandblasting, 30 min of ultrasonic cleaning in 40% NaOH, 10 min of ultrasonic cleaning in deionized water, 1 h of etching in boiling aqueous 10% oxalic acid, and rinsing with deionized water. The preparation of modified PbO2 electrode included three steps: thermal decomposition, electrodeposition of α-PbO2 and electrodeposition of β-PbO2-doped Co. The titanium mesh substrates were firstly brushed by the solution containing 8.5 g SnCl4·5H2O and 1.5 g SbCl3 in 50 mL of hydrochloric acid n-butanol mixture at room temperature. Upon drying, the substrates were calcined at 500 °C for 2 h. This procedure was repeated three times. A layer of mixed oxides of SnO2 and Sb2O3 on the surface of Ti mesh was obtained. The substrates were then electrodeposited with an intermediate layer in alkaline solution (0.1 M PbO, 3.5 M NaOH) at 40 °C, applying a current density of 3 mA cm−2. Finally, the top layer of β-PbO2-doped Co was electrodeposited on the substrates in acid solution at 65 °C, applying a current density of 40 mA cm−2. The acid solution composition consisted of 0.5 M Pb(NO3)2, 0.04 M KF and 5 mM Co(NO3)2·6H2O in 0.1 M HNO3.2.3 Electrochemical experiments
The electrochemical degradation of IBU was carried out in a batch reactor with an effective volume of 200 mL. The anode was Co-modified PbO2 electrode, and the cathode was titanium plate. The geometric surface areas of the anode and cathode were both 15 cm2 (3 cm × 5 cm). The inter electrode distance was 15 mm. A silicon Rectifier (Model DH 1718E-4, Beijing Dahua Rectifier Instruments Plant, Beijing, China) supplied direct current to the reactor. The solution was well mixed by a magnetic stirrer.The removal of IBU and COD was calculated according to the following equation:
| H = (C0 − Ct)/C0 × 100% | (1) |
Energy demand (Ed) was calculated according to the literature.24,25
 | (2) |
In addition to energy demand, one of the practical yardsticks in an industrial environment is the energy efficiency, which provides a description of the efficiency of a given process to determine its economic viability. The energy efficiency of degradation is defined as the ppm of organic contamination degraded in a given volume of solution per W h of electrical energy. The energy efficiency was calculated by the following equation:
| Energy efficiency (mg W−1 h−1) = m/E | (3) |
2.4 Analysis method
Scanning electron microscopy (SEM) was carried out on a Hitachi S-570 model instrument.X-ray diffraction (XRD) patterns of samples were obtained with an X'Pert Pro MPD Diffractometer (PANalytical, Netherlands) using Cu Kα radiation (40 kV, 40 mA). The X'Pert High Score Software was used for data collection and analysis. The standard patterns of the phase were taken from 2004 Release PDF database. The data were collected in step-scan mode with a step angle of 0.033° and a scan step time of 20 s.
Linear sweep voltammetry curves (LSVs) and cyclic voltammetry curves (CVs) were executed at the ALS/DY2323 (BAS, Japan) electrochemical workstation. The CV measurements were performed with a conventional three-electrode system, PbO2 and Co-PbO2 electrodes with an effective surface area of 1 cm2 were used as working electrodes, a platinum sheet with the same size was used as the counter electrode, and a saturated calomel electrode (SCE) was used as the reference electrode. All voltammetric curves were measured at room temperature.
The service lifetime tests of PbO2 and Co-PbO2 electrodes were investigated in 1 mol L−1 H2SO4 solution with the current density of 2 A cm−2. The temperature was kept at 60 °C. The anode potential was measured as a function of time, and it was considered that the electrode is deactivated when the potential increases 5 V from its initial value.
The concentration of IBU during reaction was measured by a high-performance liquid chromatograph (Dionex, USA). The injection volume was 20 μL of samples, and the mobile phase was acetonitrile (60%, volume content) and acetic acid (0.1%, volume content) in deionized water. The separation was performed using an ODS-18 reversed phase column (Varian, 5 μm, 150 mm × 4.6 mm) at the flow rate of 1.0 mL min−1 and column temperature of 30 ± 1 °C. A UV detector was used with the wavelength set at 254 nm.
The COD (chemical oxygen demand) was measured by the standard method.
The concentrations of metal ions in solution were determined with an inductively coupled plasma atomic emission spectrometer (ICP-AES, Perkin-Elmer Plasma 400; Norwalk, CT, USA).
The identification of the degradation intermediates of IBU was carried out by an ACQUITYUPLC system (Waters) coupled to a Xevo QTOF-MS (G2, Waters) equipped with an electrospray ionization (ESI) source operating in negative and positive ion mode. All the samples were separated on a Waters ACQUITY UPLC BEH C18 column (1.7 μm, 2.1 mm × 50 mm) at 40 °C. The mobile phase was composed of 0.1% formic acid aqueous solution (A) and acetonitrile (B) at a flow rate of 0.3 mL min−1. The injection volume was 5 μL. Initially, phase B was maintained at 10% for the first 1 min, followed by a linear increase to 100% during the next 9 min; then returning to initial conditions, which were equilibrated for 2 min before injection of the next sample.
3 Results and discussion
3.1 Electrode characterization
3.2 Electrochemical performance
Fig. 2 showed the results of LSV and CV experiments. It can be observed that in Fig. 2(a), the oxygen evolution potential on the Co-PbO2 electrode was higher than that on the PbO2 electrode in 0.5 mol L−1 H2SO4 aqueous solutions. Thus, the Co-PbO2 electrode exhibited better electrochemical degradation performance for contaminants. As observed in Fig. 2(b), with increasing scan rate, the oxidation peak potential shifted to more positive values, and the peak current increases. The peak current of the Co-PbO2 electrode was proportional to the square root of the scan rate, and the linear equation was expressed as:| I (A) = 0.00089ν (mV s−1) − 0.00068, R2 = 0.98. | (4) |
This suggested that the oxidation process was typically controlled by mass diffusion. This result was similar to the electrocatalytic degradation of methylene blue on PbO2–ZrO2.28
Fig. 2(c) presented the CV of the Co-PbO2 electrode with IBU at different concentrations (0, 80, 320 and 1000 mg L−1), at a scan rate of 50 mV s−1. In the 0.05 mol L−1 Na2SO4 solution, there was an anodic peak at approximately 1.2 V versus SCE before the oxygen evolution potential, whereas the reverse potential scan showed one cathodic peak at approximately 0.9 V. These two peaks corresponded to the reduction–oxidation of Pb(II) and Pb(IV) in the film. However, when IBU (80 mg L−1) was added, the anodic peak exhibited higher intensity owing to the contribution of IBU oxidation, showing that IBU can also be oxidized in the same potential range as Pb(II) before oxygen evolution. Moreover, for higher IBU concentration (320 and 1000 mg L−1), the anodic peak current decreased. Moreover, a new oxidation peak (1.5 V, vs. Hg/Hg2Cl, KClsat) was found, and the current intensity of the peak increased with increasing IBU concentration, which implied that the intermediate species were formed at the potential range. Ciríaco et al. studied the CV of Ti/Pt/PbO2 in different Ibuprofen solutions.15 Similarly, it showed that increasing IBU concentration led to a decrease in the current intensity of the peak. This result was observed because excessive IBU molecules covered the active sites of the Co-PbO2 electrode, and the partial passivation of the electrode surface could occur. This phenomenon ultimately weakened the oxidation of IBU.
3.3 Electrochemical degradation of IBU
| Parameters | k (min−1) | R2 | t1/2 (min) | Ed (W h L−1) | |
|---|---|---|---|---|---|
| Initial concentration (mg L−1) | 40 | 0.073 | 0.964 | 9.6 | 0.12 |
| 80 | 0.073 | 0.998 | 9.5 | 0.11 | |
| 160 | 0.038 | 0.989 | 18.3 | 0.23 | |
| 320 | 0.013 | 0.976 | 53.5 | 0.93 | |
| pH | 4 | 0.082 | 0.940 | 11.0 | 0.09 |
| 6 | 0.073 | 0.998 | 9.5 | 0.11 | |
| 8 | 0.035 | 0.947 | 19.9 | 0.26 | |
| 10 | 0.029 | 0.978 | 23.6 | 0.29 | |
| Current density (mA cm−2) | 3 | 0.073 | 0.998 | 9.5 | 0.11 |
| 10 | 0.060 | 0.910 | 11.6 | 0.54 | |
| 20 | 0.054 | 0.856 | 12.8 | 1.38 | |
| 30 | 0.067 | 0.965 | 10.4 | 2.42 | |
| HA (mg L−1) | 0 | 0.073 | 0.998 | 9.5 | — |
| 10 | 0.049 | 0.950 | 14.3 | — | |
| 20 | 0.039 | 0.983 | 18.0 | — | |
| 40 | 0.023 | 0.891 | 30.2 | — | |
| FA (mg L−1) | 0 | 0.073 | 0.998 | 9.5 | — |
| 10 | 0.117 | 0.973 | 5.9 | — | |
| 20 | 0.047 | 0.923 | 14.8 | — | |
| 40 | 0.029 | 0.917 | 23.9 | — | |
| OA (mmol L−1) | 0 | 0.073 | 0.998 | 9.5 | — |
| 0.1 | 0.025 | 0.936 | 27.5 | — | |
| 0.25 | 0.032 | 0.940 | 21.9 | — | |
| 0.5 | 0.035 | 0.933 | 19.6 | — | |
| CA (mmol L−1) | 0 | 0.073 | 0.998 | 9.5 | — |
| 0.1 | 0.024 | 0.893 | 29.4 | — | |
| 0.25 | 0.030 | 0.889 | 23.2 | — | |
| 0.5 | 0.036 | 0.880 | 19.3 | — |
The effective removal was also indicated by the change in UV absorption spectra (Fig. 4(b)) and the three-dimensional fluorescence experiment (Fig. 4(c and d)). The UV absorption peak at 254 nm declined with electrolysis. Fig. 4(c) showed the three-dimensional fluorescence spectrograms during IBU degradation. A strong peak (named peak 1) at Ex/Em = 220 nm/290 nm before electrocatalysis was observed, which is related to IBU. After 180 min, peak 1 disappeared completely, which indicated that IBU was degraded entirely. Additionally, a new peak (named peak 2) at Ex/Em = 225 nm/340 nm appeared after 180 min, which suggested that new materials had generated and the materials contained C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bonds, benzene rings or C
C double bonds, benzene rings or C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O structures.
O structures.
For the electrochemical degradation, energy demand is an important factor that determines the economic feasibility of the process. The major operating cost is associated with electrical energy consumption during the electrochemical degradation process. Hence, it is necessary to compare the process efficiency. It was observed that within initial IBU concentrations ranging from 40 mg L−1 to 320 mg L−1, Ed decreased slightly from 0.12 W h L−1 at 40 mg L−1 to 0.11 W h L−1 at 80 mg L−1 then increased to 0.93 W h L−1 at 320 mg L−1. The plot of energy efficiency versus initial IBU concentration indicated that the highest energy efficiency was observed at the initial concentration of 80 mg L−1. When the initial concentration of IBU was less than 80 mg L−1, the chance of contact between the pollutants and the hydroxyl radicals increased with increasing initial concentration. Thus, the energy efficiency increased. However, when the initial concentration further increased, many intermediates were accumulated. These intermediates could compete with the degradation of IBU and thus reduce the energy efficiency.
In recent years, the oxidation of IBU on different electrodes has been studied. Ciríaco et al.15 researched the electrochemical degradation of IBU on Ti/Pt/PbO2 and BDD electrodes. The removal efficiency of COD reached approximately 42% at a current density of 30 mA cm−2 for an initial IBU concentration of 0.44 mmol L−1 after 180 min electrolysis (volume of 200 mL and electrode surface area of 10 cm2) on Ti/Pt/PbO2 electrode. This result was similar to that on the Co-PbO2 electrode. When BDD electrode was applied, the removal of COD was about 70% at a current density of 30 mA cm−2 for an initial IBU concentration of 1.75 mmol L−1 after 180 min (volume of 200 mL and electrode surface area of 20 cm2). This demonstrated that BDD electrode exhibited better IBU removal than Co-PbO2 electrode. In addition, Ambuludi et al.30 studied the degradation of IBU on Pt electrode. They found that a complete degradation of IBU (0.2 mmol L−1) on Pt electrode occurred after 180 min reaction at a current density of 20 mA cm−2 (volume of 200 mL and electrode surface area of 25 cm2). This result indicated that Co-PbO2 electrode possessed higher activity for IBU oxidation than Pt electrode. Considering Ti/Pt/PbO2, BDD and Pt electrodes were more expensive, Co-PbO2 electrode was more suitable for decomposition of IBU in terms of degradation efficiency and application cost.
The influence of HA and FA on the IBU degradation reaction was shown in Fig. 6. The additive 10 mg L−1 of FA promoted the removal efficiency of IBU. A similar result was obtained during the initial 20 min while adding in 10 mg L−1 of HA. Analogously, the degradation of 4-chlorophenol was enhanced in the presence of FA using zero-valent iron.31 The improvement of the degradation of IBU might be due to the non-covalent interactions of HA and FA with the aromatic structure of IBU. The reaction of HA and FA with IBU led to the formation of π–π interactions between the aromatic components of the HA and FA and the monoaromatic ring of IBU and then enhanced the density of the electron cloud of IBU, resulting in the improved degradation.32
However, the IBU degradation was inhibited at both 20 mg L−1 and 40 mg L−1 of HA and FA concentration. Moreover, increasing the HA and FA concentration further decreased the degradation of IBU. Similarly, the inhibition of coomassie Brilliant Blue degradation increased by adding more HA in the sonochemical system.33 Moreover, a study showed that FA could inhibit the degradation of IBU by ultrasonic.10 This was because HA and FA may cover active surface sites of the Co-PbO2 electrode or may scavenge OH radicals in competition with IBU.31
Compared with the influence of the addition of FA, HA has a higher inhibition suggesting that HA has a higher OH suppression ability. Generally, HA molecules are often larger and contain more aromatic character, whereas FA molecules typically contain more carbonyl and aliphatic regions. The difference in their structure affects their ability to bind a molecule or sequester it from bulk solution,34 resulting in the slightly different effect of the degradation reaction.
The effect of concentrations of OA and CA on IBU degradation was displayed in Fig. 5. It can be observed that OA and CA can obviously inhibit the degradation of IBU. When the concentration of OA and CA was 0.1 mmol L−1, the degradation efficiency of IBU and COD was minimal. When OA and CA concentration were further increased, the inhibition effect became weak. On the one hand, in the electrochemical oxidation system, OA and CA competed with IBU for the capture of hydroxyl radicals, leading to the decreased IBU removal. On the other hand, the addition of OA and CA could reduce the pH of the electrochemical system. The lower pH inhibited the oxygen evolution reaction, resulting in improved IBU degradation. The effects of OA and CA on IBU degradation depended on the competition between the two factors mentioned above. As the concentration of OA and CA increased, the pH value decreased, resulting in improved IBU degradation. However, this effect was still lower than that of capturing hydroxyl radicals. Therefore, the addition of OA and CA exhibited an inhibition effect on IBU degradation.
In path 1, IBU suffered hydroxylation and then the cleavage of the isobutyl moiety, forming compound P1 (m/z, 164). In path 2, a methyl group of IBU molecule was attacked by ˙OH to form a demethylation product P2 (m/z, 192). Then P2 could be further oxidized into product P3 (m/z, 134). In path 3, IBU underwent hydroxylation and further decarboxylation, forming compound P4 (m/z, 178). In the case of product P4, the oxidation process followed two possible pathways. One pathway was the dehydration, resulting in the production of product P5 (m/z, 160). Then P5 formed compound P6 (m/z, 162) by the addition of oxygen. The other pathway was the deprotonation of product P4, transforming to product P7 (m/z, 176). The demethylation of acetyl chain of compound P7 produced P6 and the decarbonylation and further hydroxylation of P7 led to the production of P8 (m/z, 150). In addition, the hydroxylation on intermediate P8 produced compound P9 (m/z, 166).
Considering real applications in the future, the reusability of Co-doped PbO2 electrode deserved careful attention. Fig. 8(b) showed the removals of IBU and COD reusing eight cycles. It was observed that, although there was a slight decrease on IBU and COD removal, the degradation efficiencies were still up to 94.0% and 47.3% after eight cycles, respectively. Therefore, Co-doped PbO2 electrode had high reusability for degrading IBU.
As Co doped PbO2 electrodes contained toxic metals, including Pb, Sn, Sb and Co, the possible releases of these toxic ions are a potential risk for large scale applications of the electrochemical technology. In order to evaluate the safety of the aqueous solutions after electrolysis, the concentrations of these toxic ions were measured after 3.0 h electrolysis at the current density of 3 mA cm−2 and 30 mA cm−2. It was found that only Co ion was detected at the current density of 30 mA cm−2 while other metal ions were lower than detection line (Pb: 0.004 mg L−1, Sn: 0.002 mg L−1, Sb: 0.004 mg L−1). The dissolved concentration of Co was 0.002 mg L−1, which was below the maximum tolerable limit set by the WHO (World Health Organization).42 As a result, it is safe to use the Co-doped PbO2 electrode to treat waste water.
4 Conclusions
A cheap and highly efficient Co-modified PbO2 electrode was prepared by electrodeposition to oxidize IBU electrocatalytically in aqueous solution. The Co-PbO2 electrode exhibited highly effective electrochemical degradation performance for IBU.Co doping could reduce the crystal size and provide larger specific surface areas. Co-PbO2 electrode had a higher oxygen evolution and lifetime than the PbO2 electrode. The oxidation process of IBU on the electrode was typically controlled by mass diffusion.
The influencing factors, including the initial IBU concentration (40–320 mg L−1), initial pH (4–10), current density (3–30 mA cm−2), HA, FA, OA and CA on IBU degradation and energy demand were systematically investigated. At different conditions, IBU degradation was always in good agreement with apparent first-order kinetics.
The applied current density affected IBU and COD degradation slightly but had great influence on energy demand. Other factors showed an obvious effect on the degradation of IBU, COD and energy demand. When the IBU initial concentration was 80 mg L−1, the highest reaction rate and energy efficiency were observed. The applied current density of 3 mA cm−2 was the best in terms of energy demand and space efficiency. The oxygen evolution reaction was restrained at lower pH, which favored the IBU degradation.
The removal of IBU was enhanced with the addition of low-concentration (10 mg L−1) HA and FA, whereas the elimination of IBU was suppressed by adding high-concentration (20–40 mg L−1) HA and FA. Moreover, the inhibition was enhanced with the increasing concentration of HA and FA. The IBU degradation was also restrained in the presence of OA and CA, and the suppression was weakened with increasing OA and CA.
The possible reaction pathways were proposed. The electrochemical oxidation of IBU mainly involved hydroxylation, decarboxylation and demethylation process.
As a result, the study of influencing factors, reusability and safety would be helpful in providing insight into the application of an electrochemical method for effective IBU wastewater remediation.
Acknowledgements
This work was supported by the Natural Science Foundation of China (No. 51578070 & 21177013) and International S&T Cooperation Program of China (2013DFR90290).References
- C. D. Qi, X. T. Liu, C. Y. Lin, X. H. Zhang, J. Ma, H. B. Tan and W. Ye, Degradation of sulfamethoxazole by microwave-activated persulfate: Kinetics, mechanism and acute toxicity, Chem. Eng. J., 2014, 249, 6–14 CrossRef CAS.
- S. Sarkar, R. Das, H. Choi and C. Bhattacharjee, Involvement of process parameters and various modes of application of TiO2 nanoparticles in heterogeneous photocatalysis of pharmaceutical wastes – a short review, RSC Adv., 2014, 4, 57250–57266 RSC.
- S. Motoc, A. Remes, A. Pop, F. Manea and J. Schoonman, Electrochemical detection and degradation of ibuprofen from water on multi-walled carbon nanotubes-epoxy composite electrode, J. Environ. Sci., 2013, 25, 838–847 CrossRef CAS.
- J. Madhavan, F. Grieser and M. Ashokkumar, Combined advanced oxidation processes for the synergistic degradation of ibuprofen in aqueous environments, J. Hazard. Mater., 2010, 178, 202–208 CrossRef CAS PubMed.
- V. Matozzo, N. Franchi and L. Ballarin, In vitro effects of the nonsteroidal anti-inflammatory drug, ibuprofen, on the immune parameters of the colonial ascidian Botryllus schlosseri, Toxicol. In Vitro, 2014, 28, 778–783 CrossRef CAS PubMed.
- S. Han, K. Choi, J. Kim, K. Ji, S. Kima, B. Ahn, J. Yun, K. Choi, J. S. Khim, X. W. Zhang and J. P. Giesy, Endocrine disruption and consequences of chronic exposure to ibuprofen in Japanese medaka (Oryzias latipes) and freshwater cladocerans Daphnia magna and Moina macrocopa, Aquat. Toxicol., 2010, 98, 256–264 CrossRef CAS PubMed.
- M. Skoumal, R. M. Rodríguez, P. L. Cabot, F. Centellas, J. A. Garrido, C. Arias and E. Brillas, Electro-Fenton, UVA photoelectro-Fenton and solar photoelectro-Fenton degradation of the drug ibuprofen in acid aqueous medium using platinum and boron-doped diamond anodes, Electrochim. Acta, 2009, 54, 2077–2085 CrossRef CAS.
- A. Ziylan and N. H. Ince, Catalytic ozonation of ibuprofen with ultrasound and Fe-based catalysts, Catal. Today, 2015, 240, 2–8 CrossRef CAS.
- C. Sirtori, A. Agüera, W. Gernjak and S. Malato, Effect of water-matrix composition on Trimethoprim solar photodegradation kinetics and pathways, Water Res., 2010, 44, 2735–2744 CrossRef CAS PubMed.
- R. Y. Xiao, Z. Q. He, D. Diaz-Rivera, G. Y. Pee and L. K. Weavers, Sonochemical degradation of ciprofloxacin and ibuprofen in the presence of matrix organic compounds, Ultrason. Sonochem., 2014, 21, 428–435 CrossRef CAS PubMed.
- S. Loaiza-Ambuludi, M. Panizza, N. Oturan and M. A. Oturan, Removal of the anti-inflammatory drug ibuprofen from water using homogeneous photocatalysis, Catal. Today, 2014, 224, 29–33 CrossRef CAS.
- A. Rubio-Clemente, R. A. Torres-Palma and G. A. Peñuela, Removal of polycyclic aromatic hydrocarbons in aqueous environment by chemical treatments: A review, Sci. Total Environ., 2014, 478, 201–225 CrossRef CAS PubMed.
- X. Y. Duan, F. Ma, Z. X. Yuan, L. M. Chang and X. T. Jin, Comparative studies on the electro-catalytic oxidation performance of surfactant–carbon nanotube-modified PbO2 electrodes, J. Electroanal. Chem., 2012, 677, 90–100 CrossRef.
- N. B. Tahar and A. Savall, Electrochemical degradation of phenol in aqueous solution on bismuth doped lead dioxide: a comparison of the activities of various electrode formulations, J. Appl. Electrochem., 1999, 29, 277–283 CrossRef CAS.
- L. Ciríaco, C. Anjo, J. Correia, M. J. Pacheco and A. Lopes, Electrochemical degradation of Ibuprofen on Ti/Pt/PbO2 and Si/BDD electrodes, Electrochim. Acta, 2009, 54, 1464–1472 CrossRef.
- C. I. Brinzila, M. J. Pacheco, L. Ciríaco, R. C. Ciobanu and A. Lopes, Electrodegradation of tetracycline on BDD anode, Chem. Eng. J., 2012, 209, 54–61 CrossRef CAS.
- J. T. Xing, D. H. Chen, W. Zhao, X. L. Peng, Z. L. Bai, W. W. Zhang and X. X. Zhao, Preparation and characterization of a novel porous Ti/SnO2–Sb2O3–CNT/PbO2 electrode for the anodic oxidation of phenol wastewater, RSC Adv., 2015, 5, 53504–53513 RSC.
- J. T. Kong, S. Y. Shi, L. C. Kong, X. P. Zhu and J. R. Ni, Preparation and characterization of PbO2 electrodes doped with different rare earth oxides, Electrochim. Acta, 2007, 53, 2048–2054 CrossRef CAS.
- X. Y. Duan, F. Ma, Z. X. Yuan, L. M. Chang and X. T. Jin, Lauryl benzene sulfonic acid sodium-carbon nanotube-modified PbO2 electrode for the degradation of 4-chlorophenol, Electrochim. Acta, 2012, 76, 333–343 CrossRef CAS.
- J. Yang, S. H. Cui, J. Q. Qiao and H. Z. Lian, The photocatalytic dehalogenation of chlorophenols and bromophenols by cobalt doped nano TiO2, J. Mol. Catal. A: Chem., 2014, 395, 42–51 CrossRef CAS.
- R. Edla, N. Patel, Z. E. Koura, R. Fernandes, N. Bazzanella and A. Miotello, Pulsed laser deposition of Co3O4 nanocatalysts for dye degradation and CO oxidation, Appl. Surf. Sci., 2014, 302, 105–108 CrossRef.
- L. S. Andrade, R. C. Rocha-Filho, N. Bocchi, S. R. Biaggio, J. Iniesta, V. García-Garcia and V. Montiel, Degradation of phenol using Co- and Co, F-doped PbO2 anodes in electrochemical filter-press cells, J. Hazard. Mater., 2008, 153, 252–260 CrossRef CAS PubMed.
- A. B. Velichenko, R. Amadelli, E. A. Baranova, D. V. Girenko and F. I. Danilov, Electrodeposition of Co-doped lead dioxide and its physicochemical properties, J. Electroanal. Chem., 2002, 527, 56–64 CrossRef CAS.
- Y. Wang, Z. Y. Shen and X. C. Chen, Effects of experimental parameters on 2,4-dichlorphenol degradation over Er-chitosan-PbO2 electrode, J. Hazard. Mater., 2010, 178, 867–874 CrossRef CAS PubMed.
- J. F. Niu, Y. P. Bao, Y. Li and Z. Chai, Electrochemical mineralization of pentachlorophenol (PCP) by Ti/SnO2–Sb electrodes, Chemosphere, 2013, 92, 1571–1577 CrossRef CAS PubMed.
- Q. P. Chen, S. Y. Ai, S. S. Li, J. Xu, H. S. Yin and Q. Ma, Facile preparation of PbO2 electrode for the electrochemical inactivation of microorganisms, Electrochem. Commun., 2009, 11, 2233–2236 CrossRef CAS.
- P. Ju, H. Fan, D. D. Guo, X. M. Meng, M. R. Xu and S. Y. Ai, Electrocatalytic degradation of bisphenol A in water on a Ti-based PbO2–ionic liquids (ILs) electrode, Chem. Eng. J., 2012, 179, 99–106 CrossRef CAS.
- Y. W. Yao, C. M. Zhao, M. M. Zhao and X. Wang, Electrocatalytic degradation of methylene blue on PbO2–ZrO2 nanocomposite electrodes prepared by pulse electrodeposition, J. Hazard. Mater., 2013, 263, 726–734 CrossRef CAS PubMed.
- J. F. Niu, D. Maharana, J. L. Xu, Z. Chai and Y. P. Bao, A high activity of Ti/SnO2-Sb electrode in the electrochemical degradation of 2,4-dichlorophenol in aqueous solution, J. Environ. Sci., 2013, 7, 1424–1430 CrossRef.
- S. L. Ambuludi, M. Panizza, N. Oturan, A. Özcan and M. A. Oturan, Kinetic behavior of anti-inflammatory drug ibuprofen in aqueous medium during its degradation by electrochemical advanced oxidation, Environ. Sci. Pollut. Res., 2013, 20, 2381–2389 CrossRef CAS PubMed.
- S. H. Kang and W. Y. Choi, Oxidative Degradation of Organic Compounds Using Zero-Valent Iron in the Presence of Natural Organic Matter Serving as an Electron Shuttle, Environ. Sci. Technol., 2009, 43, 878–883 CrossRef CAS PubMed.
- D. Šmejkalová, R. Spaccini, B. Fontaine and A. Piccolo, Binding of phenol and differently halogenated phenols to dissolved humic matter as measured by NMR spectroscopy, Environ. Sci. Technol., 2009, 43, 5377–5382 CrossRef.
- M. P. Rayaroth, U. K. Aravind and C. T. Aravindakumar, Sonochemical degradation of Coomassie Brilliant Blue: Effect of frequency, power density, pH and various additives, Chemosphere, 2015, 119, 848–855 CrossRef CAS PubMed.
- M. E. Lindsey and M. A. Tarr, Inhibition of hydroxyl radical reaction with aromatics by dissolved natural organic matter, Environ. Sci. Technol., 2000, 34, 444–449 CrossRef CAS.
- J. A. Zimbron and K. F. Reardon, Fenton's oxidation of pentachlorophenol, Water Res., 2009, 43, 1831–1840 CrossRef CAS PubMed.
- H. T. Tian, Y. F. Fan, Y. P. Zhao and L. Liu, Elimination of ibuprofen and its relative photoinduced toxicity by mesoporous BiOBr under simulated solar light irradiation, RSC Adv., 2014, 4, 13061–13070 RSC.
- Y. F. Rao, D. Xue, H. M. Pan, J. T. Feng and Y. J. Li, Degradation of ibuprofen by a synergistic UV/Fe(III)/Oxone process, Chem. Eng. J., 2016, 283, 65–75 CrossRef CAS.
- I. Michael, A. Achilleos, D. Lambropoulou, V. O. Torrens, S. Pérez, M. Petrović, D. Barceló and D. F. Kassinos, Proposed transformation pathway and evolution profile of diclofenac and ibuprofen transformation products during (sono) photocatalysis, Appl. Catal., B, 2014, 147, 1015–1027 CrossRef CAS.
- M. Skoumal, R. M. Rodríguez, P. L. Cabot, F. Centellas, J. A. Garrido, C. Arias and E. Brillas, Electro-Fenton, UVA photoelectron-Fenton and solar photoelectron-Fenton degradation of the drug ibuprofen in acid aqueous medium using platinum and boron-doped diamond anodes, Electrochim. Acta, 2009, 54, 2077–2085 CrossRef CAS.
- J. L. Cao, H. Y. Zhao, F. H. Cao, J. Q. Zhang and C. N. Cao, Electrocatalytic degradation of 4-chlorophenol on F-doped PbO2 anodes, Electrochim. Acta, 2009, 54, 2595–2602 CrossRef CAS.
- L. M. Chang, Y. Zhou, X. Y. Duan, W. Liu and D. D. Xu, Preparation and characterization of carbon nanotube and Bi co-doped PbO2 electrode, J. Taiwan Inst. Chem. Eng., 2014, 45, 1338–1346 CrossRef CAS.
- M. Soylak and L. Elci, Solid phase extraction of trace metal ions in drinking water samples from Kayseri-Turkey, J. Trace Microprobe Tech., 2000, 3, 397–403 Search PubMed.
| This journal is © The Royal Society of Chemistry 2016 |