Fabrication and characterization of in situ graphene oxide reinforced high-performance shape memory polymeric nanocomposites from vegetable oil
Rakesh Das†
,
Sovan Lal Banerjee† and
P. P. Kundu*
Department of Polymer Science and Technology, University of Calcutta, 92, A.P.C. Road, Kolkata-700009, India. E-mail: ppk923@yahoo.com
First published on 11th March 2016
Abstract
Polymeric nanocomposites have been fabricated via in situ cationic polymerization of linseed oil in the presence of surface-modified graphene oxide (SGO). The interfacial interactions between SGO nanoplatelets and the polymer matrix were investigated by field emission scanning electron microscopy (FESEM) and transmission electron microscopy (TEM), which revealed a uniform distribution of SGO throughout the polymer matrix at a lower concentration (<2 wt%), but that higher loading of SGO (2 wt%) results in agglomeration. Nanomechanical characterization revealed the enhancement of the mechanical properties of nanocomposites at low concentrations of nanofillers (<2 wt%). Under dynamic mechanical analysis, the nanocomposites exhibited superior dynamic modulus with respect to the polymer matrix, demonstrating the great reinforcement potential of SGO nanofillers. Also, the thermal stabilities of the nanocomposites effectively improved with an increase in the filler loading. In shape memory tests, the nanocomposites fully recovered their original shape under different stimuli, and the fast shape recovery was observed with an increase in the content of SGO nano fillers.
Introduction
In recent years, bio-based materials have become the center of attention as feedstock chemicals for the preparation of potential polymeric materials.1 This is an alternative pathway to develop environmentally friendly materials, replacing petroleum-derived raw materials. Vegetable oil is the most abundant, cheap and versatile renewable resource among all bio-based resources.2 In polymer nanocomposites, the inclusion of nanoscale particles into the polymer matrix produces a series of significantly enhances mechanical, thermal, electrical, optical properties.3,4 The high aspect ratio of nanoscale filler causes a strong interfacial interaction between fillers and polymer matrix. Thus, incorporation of very low volume fraction filler measurably enhances the composite properties.A lot of functional groups present in vegetable oils can be utilized to develop potential polymeric materials through different chemical reactions.5 Focusing into specific applications, vegetable oil based polymeric bio-composites or bio-nanocomposites have been synthesized using inorganic and organic particles or fibers as reinforcing agent.6 For the preparation of vegetable oil based polymeric nanocomposites or composites, the reinforcing agents like nano clay,7 glass fibre8 or natural fibres9–11 become point of attraction because their incorporation dramatically improves different properties in compare to the matrix polymers.
Recently, graphene based fillers find application in the development of advanced polymer nanocomposites because of their unique properties like high aspect ratio, elastic modulus, fracture strength and thermal conductivity.12–17 Graphene oxide (GO), generated after oxidation of graphite, has numerous superior characteristics than graphite like larger interlayer spacing, wide possibility of chemical surface modifications by utilizing its various surface functional groups such as epoxides, hydroxyls and carboxylic acids.18 The strong interfacial interaction between polymer matrix and GO is required for the effective load transmission from polymer to GO. The surface modification of GO tremendously improves its dispersion, interaction and compatibility into polymer systems.19
Shape memory polymers (SMP) exhibit behavior of fixing a temporary shape and restore its original shape under the action of external stimuli like temperature, humidity, pH, light, electric field and magnetic field.20 The thermally stimulated shape memory behavior of SMP is most extensively used. The SMPs have gained increasing interest in numerous applications, like actuators, smart fabrics, packaging, biomedical devices etc.
The static mechanical properties of polymers, like hardness and elastic modulus are very important for any stress or pressure related application. The nano-indentation is a non-destructive advanced tool for very precise measurement of mechanical properties at nano-level. It has the ability to derive mechanical properties through generating small deformation into the sample surface, especially valuable for the system having limited dimensionality (thin films and coatings), or system of small mass, or heterogeneous materials like polymer nanocomposites.21
In the present research work, polymeric nanocomposites have been developed from linseed oil based polymers reinforced with modified graphene oxide (SGO) through in situ cationic polymerization technique. The SGO nanocomposites of the polymers based on vegetable oils are prepared with the view to improve the properties of the polymers. The evaluation of elastic modulus, hardness enhancement due to incorporation of fillers have been analyzed through nano-indentation test. The dynamic mechanical properties of these nanocomposites have been evaluated in a wide range of temperature and frequency. The various stimuli (thermal, sunlight and micro wave (MW) irradiation) induced shape memory behaviors of nanocomposites have been investigated.
Experimental
Materials
Linseed oil was purchased from the local market of Kolkata. Styrene (ST), divinylbenzene (DVB) (55 mol% DVB and 45 mol% ethylvinylbenzene) and boron trifluoride diethyl etherate complex were purchased from Sigma-Aldrich, USA. Methanol, dichloromethane, concentrated sulphuric acid and hydrogen peroxide were purchased from Merck, India. Potassium hydroxide, potassium permanganate and sodium nitrate was purchased from SRL Chemical Co, India. Cetyltrimethyl ammonium bromide (CTAB) was purchased from Aldrich Chemical Company, USA. Graphite flakes (60 mesh, purity 99%) was purchased from Loba Chemie, India.Preparation of graphene oxide (GO) and its modification
The GO was prepared through the oxidation of graphite powder using a mixture of concentrated sulphuric acid and KMnO4 following modified Hummers method.22 Then, the CTAB modified GO (SGO) was prepared by the following steps: 50 mg GO was dispersed into 250 mL of deionised (DI) water, followed by the addition of 150 mg CTAB; the whole mixture was ultrasonically agitated for 4 hours. The obtained suspension was left to stand for 12 hours and allowed to separate into layers. The obtained SGO suspension was centrifuged at 4000 rpm. The collected precipitate was washed with water for several times and dried in vacuum at 80 °C for 12 hours and then grounded it using mortar and pestle to obtain the fine powder of surface modified GO. The SGO was further characterized through Fourier transform infrared spectroscopy (FTIR) and zeta potential study.Synthesis of GO nanocomposites
A mixture of fixed composition of ST, DVB and linseed oil (40 wt% of linseed oil, 27 wt% of ST, 18 wt% of DVB) was taken in which the SGO was dispersed through ultrasonication. The ultrasonication was performed for 2 hours to get a stable dispersion. Then, the reaction mixture was kept in ice bath and a desired amount of initiator (15 wt%) was added drop wise into it with vigorous stirring. The initiator was prepared through mixing the 5 wt% BFE initiator with 10 wt% methyl ester of linseed oil for its homogeneous distribution into the reaction mixture. Methyl ester of linseed oil was prepared by following the double step transesterification process.23 After proper mixing, the reaction mixture was poured into a rectangular glass mold, which was further sealed by silicon adhesives. At room temperature, the mold was kept for 2 hours and then it was heated at 60 °C to 80 °C for 2 hours, followed by heating at 110 °C for 12 hours, finally post cured at 120 °C for 3 hours. At fixed matrix composition, the SGO concentration was changed from 0.5 to 2 wt% to obtain a variety of polymeric nanocomposites.Characterization
The Fourier transform infrared (FTIR) spectrometer of Bruker Germany (Model: Alpha-E) was utilized to analyze the SGO and nanocomposites in attenuated total reflectance (ATR) mode. The Fourier transform Raman (FT Raman) spectra for GO, SGO and nanocomposites were measured on Jobin Yvon Horiba spectrometer (France, Model T64000) at an excitation wavelength of λmax = 647 nm. The X-ray diffraction experiment for GO, polymer and nanocomposites were conducted on Panalytical X-ray diffractometer (model: X'pert Powder X-ray) using Cu Kα radiation (λ = 0.154 nm) in the range of 4–45° (2θ) at 40 kV and 30 mA. The zeta potential of GO and SGO were measured by a Zetasizer Nano ZS 90 (Malvern Instrument, UK) dynamic light scattering (DLS) instrument at a scattering angle of 90° and at room temperature (25 °C). The microstructure of the nanocomposites was analyzed through Field Emission Scanning Electron Microscope (FESEM) of JEOL (model JSM-7600F) at an acceleration voltage of 5 kV. Before scanning, the samples were sputtered with gold by an ion sputter coater of Hitachi, Japan (Model: E1010). The High Resolution Transmission Electron Microscopy (HRTEM) images of nanocomposites were observed on JEOL, Japan (JEM-2100 HRTEM) with an accelerating voltage of 200 kV. The ultrathin sections (25–50 nm) for HRTEM analysis were microtomed cryogenically by CM-12 microtome of Philips, Holland using a diamond knife.The nanocomposites having different concentration of modified GO along with the pure polymer were tested by TI 950 Tribo-Indenter, Hysitron Inc., USA to evaluate the micromechanical properties. The rectangular specimen having dimension of 5 × 5 × 2 mm was tested in displacement controlled indentation test of fixed maximum penetration depth (hmax) of 200 nm. Berkovich tip was used for nano-indentation and the tip area function was calibrated using a standard quartz sample as per standard practice. Since the loads used for indenting polymer films are very small, the tip area function is calibrated in the low depth ranges for precise determination of the modulus and hardness. Indents at different points on the surface were done for loading and unloading time of 5 s each without any holding time. The load versus penetration depth curve, average elastic modulus (E) and hardness (H) were obtained from this test. A typical loading and unloading flow in nano-indentation process is schematically presented in Fig. 1.24
The dynamic mechanical properties of the polymer and nanocomposites were analyzed by a Perkin Elmer dynamic mechanical analyzer (DMA 8000). In DMA, the temperature sweep (−30 °C to 60 °C, increasing rate 5 °C min−1) in compression mode was measured using the rectangular sections (10 mm × 5 mm × 2 mm) of samples at a fixed frequency of 1 Hz and dynamic strain of 0.001%. The storage modulus (E′), loss modulus (E′′) and damping loss factor (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) were obtained from this measurement.
δ) were obtained from this measurement.
The cross-linking densities (ve) were determined from the following equation;25
| E′ = 3veRT | (1) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) as a function of frequency were recorded in compression mode at dynamic strain of 0.0005% and frequency range of 1 Hz to 50 Hz at room temperature (25 °C).
δ) as a function of frequency were recorded in compression mode at dynamic strain of 0.0005% and frequency range of 1 Hz to 50 Hz at room temperature (25 °C).
The tensile stress–strain characterization of nanocomposites was carried out by universal testing machine of Lloyd Instruments Ltd, England (Model: LR10K PLUS) at room temperature (25 °C) following ASTM D412.
For determining thermal stability of nanocomposites, thermogravimetric analysis (TGA) were carried out by Perkin-Elmer, USA thermogravimeter (Model: Pyris Diamond DSC), under nitrogen (20 ml min−1). The samples were heated from 40 °C to 700 °C at a heating rate of 20 °C min−1.
To determine the shape memory behavior of nanocomposites inducted by thermal, MW and direct sunlight, sample having initial shape of rectangular bar was deformed into a secondary transient form of ‘U’ at the Tα (primary relaxation temperature) plus 50 °C applying external force. At this condition, the deformed samples were quenched in an ice bath for 5 min. When the external force was released, the sample was shrunk and this shape remained fixed. Finally, the recovery process occurred when the specimen was heated in water bath at Tα +50 °C for observing thermally stimulated shape memory behavior. Also, the shape recovery of the samples was occurred under exposure to MW irradiation (400 W) for 30–60 s and direct sunlight (11 am–2 pm, at Calcutta University campus, Kolkata, longitude 88.30° E-22° 33′ N, altitude 9 m 30′) in the month of August on sunny days, (average temperature: 33.5 °C and humidity: 85%) under ambient condition.
The shape fixity ratio which defines the ability of the materials to fix the temporal deformation after unloading is obtained from the following equation,
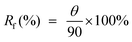 | (2) |
The percentage of shape recovery was calculated using the following equation,
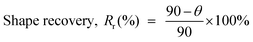 | (3) |
Results & discussions
FTIR spectroscopic analysis of GO, SGO and nanocomposites
The FTIR spectra of GO and SGO and nanocomposite are shown in Fig. 2. In GO, the peak at 3325 cm−1 and 1100 cm−1 are assigned for the O–H stretching vibration and C–O stretching vibration of epoxy group, whereas the peak at 1730 cm−1 is assigned for the for C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration.26 These peaks are significantly diminished in SGO, indicating an interaction between CTAB and different functional groups of GO.27–29 For SGO, the bands at 2924 cm−1 [asymmetric stretching vibration modes of methylene group (–CH2)] and 2853 cm−1 [symmetric stretching vibration modes of methylene group (–CH2)] are designated to the characteristic bands of CTAB. Both in GO and SGO, the peak at 1600 cm−1 is due to skeletal vibrations from unoxidized graphitic domains.30 In the spectrum of nanocomposite, the peak at 1741 cm−1 appears for the stretching vibration of ester linkage (C
O stretching vibration.26 These peaks are significantly diminished in SGO, indicating an interaction between CTAB and different functional groups of GO.27–29 For SGO, the bands at 2924 cm−1 [asymmetric stretching vibration modes of methylene group (–CH2)] and 2853 cm−1 [symmetric stretching vibration modes of methylene group (–CH2)] are designated to the characteristic bands of CTAB. Both in GO and SGO, the peak at 1600 cm−1 is due to skeletal vibrations from unoxidized graphitic domains.30 In the spectrum of nanocomposite, the peak at 1741 cm−1 appears for the stretching vibration of ester linkage (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) of linseed oil and the aromatic out of plane bending vibration of ST and DVB is detected at 699 cm−1. From the FTIR spectra of nanocomposites it is also observed that the intensity of peaks near 2924 cm−1 and 2853 cm−1 is increased due to overlapping of functional groups present in both of pure polymeric matrix and SGO. From this result, we can also predict that there is a hydrophobic–hydrophobic interaction between the long hydrocarbon chain of CTAB and linseed oil.
O) of linseed oil and the aromatic out of plane bending vibration of ST and DVB is detected at 699 cm−1. From the FTIR spectra of nanocomposites it is also observed that the intensity of peaks near 2924 cm−1 and 2853 cm−1 is increased due to overlapping of functional groups present in both of pure polymeric matrix and SGO. From this result, we can also predict that there is a hydrophobic–hydrophobic interaction between the long hydrocarbon chain of CTAB and linseed oil.
FT-Raman spectroscopic analysis of GO & SGO
The FT-Raman spectra of GO and SGO are displayed in Fig. 3. D band comes at 1360 cm−1 whereas G band is observed at 1590 cm−1 which are characteristic peaks for GO.31 The G band is usually come due to the E2g phonon of C sp2 atoms and the D band comes from activation in the first-order scattering process of sp3 carbons in GO sheets.32 Surface modification of GO results many defects in the graphene structure that affects the G band. The formation of defects can be assigned by calculating the intensity ratio of D and G band (ID/IG). As in our case GO surface was ornamented with CTAB molecules ID/IG ratio got increased. In case of GO ID/IG ratio was 0.94 whereas ID/IG ratio in SGO was 1.02.XRD analysis
The XRD patterns of GO, polymer and nanocomposites having 1 wt% SGO and 2 wt% SGO are shown in Fig. 4. In GO, the diffraction peaks at 2θ = 10.7° and 2θ = 42° correspond to its (002) and (001) planes respectively.33 In case of polymer, the broad amorphous peak is detected at 2θ = 20.77°, whereas from the XRD pattern of the nanocomposite it is observed that a broad peak appears near 22° which is due to the presence of polymer matrix in nanocomposite. Peak for GO is not appeared in the XRD pattern of nanocomposite. It may be due to the exfoliation of the GO layer or dilution effect of the filler. To further clarify the morphology of GO sheets inside nanocomposites, the TEM analysis has been performed.Zeta potential measurement of GO and SGO
The zeta potential distribution curve of GO and SGO in water as dispersant medium is shown in Fig. 5. The negative zeta potential value (−25.4 mV) of GO is due to the presence of electronegative functional groups (mainly the epoxy group on the basal plane and hydroxyl and carboxylic groups at the edge side). The functional groups at the edges of the graphene oxide sheets develop negative charges in the solution due to deprotonation, yielding a hydrophilic material. But, in case of SGO, the zeta potential turns into a positive value of 11.6 mV due to incorporation of tertiary ammonium cation on GO surface.FESEM analysis of nanocomposites
FESEM images of pure polymer and nanocomposite containing 1.0 and 2.0 wt% SGO are shown in Fig. 6. As FESEM image can only explain the surface morphology, the dispersion of GO inside the polymer can't be explained. From the figure it is clearly observed that for pure polymer there is no surface roughness whereas the incorporation of GO inside the polymeric phase generates surface roughness. With an increase in filler concentration, wrinkling of the surface increases which clarify the presence of GO.TEM analysis of nanocomposites
The high resolution TEM analysis was performed to study the dispersion state of SGO into polymer matrix. TEM observation provides direct visualization of the morphology and spatial distribution of GO sheet which FESEM could not provide. The HRTEM images of nanocomposites having 1 wt% and 2 wt% SGO are shown in Fig. 7. From the Fig. 7, it is observed that a partial exfoliation of the GO sheets occurs within the polymeric matrix when the polymer is cured after solution mixing of monomers and SGO. A layer like morphology with some extent of wrinkling is also observed in the image. Some regions with single-striation thicknesses are observed for the nanocomposite with a filler loading of 1 wt%, indicating the presence of partial exfoliated structure of GO sheets. From the XRD image of the nanocomposite the same conclusion can be drawn. The d-spacing calculated from XRD for the 1 wt% SGO content nanocomposite is 4.64 Å, for 2 wt% SGO content nanocomposite the d-spacing is 4.20 Å. In case of 2 wt% SGO loading, the presence of stacked layer morphology of SGO indicated a reduction in degree of exfoliation. Thus, the d-spacing for 2 wt% SGO content is decreased in XRD analysis. It is observed that with an increase in the modified GO contents (e.g., from 1 to 2 wt%), the orientation degree is increased, resulting in more compact and distinct stacks in structure of the SGO. The stacking morphology of the SGO inside the polymeric nanocomposites can be explained by the fact that during bulk polymerization process, the neighboring SGO layers hold the polymerizing system within its layers. But during curing, the chains try to recoil. Also, an electrostatic attraction between the positively charged quaternary ammonium ion and negatively charged GO layer as well as the van-der-Waal's force of attraction between the organic fragments of surfactants and polymer are generated. For this reason, the polymer formed inside the GO layer cannot relax, instead, they generate high elastic force. If this elastic force exceeds the internal attractive force and viscous force arise due to medium viscosity, the GO layer effectively dispersed in the system, essentially leading to exfoliation of the layers. Otherwise, they will try to stack with each other and form intercalated structure inside the polymer matrix. This is exactly observed in 2 wt% SGO containing nanocomposites.Nano mechanical analysis of nanocomposites
The load-penetration depth curves of SGO reinforced linseed oil based nanocomposites are shown in Fig. 8. The elastic modulus (E) and hardness (H) and final penetration depth (hf) calculated from the curve are listed in Table 1. The variation of these properties is plotted against SGO content in Fig. 9. It is evident from the figure that both E and H of nanocomposites increases remarkably with an increase in SGO content. The maximum increment of E by 4.5 times, whereas increase in H by 4.6 times with respect to the matrix polymer is observed when 2 wt% of SGO is incorporated in the polymer matrix. | ||
| Fig. 8 Loading and unloading curves of nanocomposites of different filler content under nano-indentation test. | ||
| SGO (wt%) | hf (nm) | E (MPa) | H (MPa) |
|---|---|---|---|
| 0 | 186 | 64.4 | 2.7 |
| 0.5 | 164 | 104 | 3.8 |
| 1 | 147 | 140 | 5.3 |
| 1.5 | 115 | 246 | 10.8 |
| 2 | 98 | 288 | 12.4 |
 | ||
| Fig. 9 The variation of elastic modulus and penetration depth as a function of CTAB–GO content in nanocomposites. | ||
To achieve effective load transfer between SGO and polymer matrix under mechanical stress, non-agglomeration, highly exfoliation, uniformly dispersion of GO in polymer matrix and also great interfacial adhesion between GO and the polymer matrix are essential. In case of weak interfacial bonding, the transmission of load from the polymer matrix to the nano filler will not occur effectively and this will cause failure of the composite even under a low stress.
In comparison to GO, the dispersion of SGO into the linseed oil based polymer matrix increases tremendously. Also, better intermolecular specific attraction and lower interfacial tension between cetylammonium ions and polymer matrix effectively enhances the adhesion between polymer matrix system and SGO. Also, the exfoliated GO nano sheets having free functional groups on the basal plane or edge strongly interact with polymer matrix, leading to an extra reinforcement. Thus, a more effective load transfer across the interface of filler-polymer matrix is obtained, resulting in a significant enhancement of the mechanical properties. Thus, the mechanical properties of nanocomposites improves significantly with an increase in the SGO content in nanocomposites. With an increase in SGO content, the mobility of polymer chain greatly restricts, leading to the improved hardness of nanocomposites. Also, the aligned nano sheet in the nanocomposites may serve as connecting bridges, which prevent the polymer from deformation under loading. As observed from the figure, the final penetration depth (hf) of nanocomposites decreases with an increase in SGO content in nanocomposites. During loading in the nano-indentation test, the Berkovich indenter of triangular geometry is pressed into the specimen, causing both elastic and plastic deformation. Thus a hardness impression into the specimen surface has been formed, conforming to the shape of the indenter as shown in real time image under indentation process in Fig. 10.
After loading of the optional load period, the applied load is reduced and the resulting penetration depth is recorded. During unloading, beside plastic deformation, there is usually some degree of elastic recovery. In specimen, a plastic zone surrounded by elastically strained region is generated during the indentation process. The elastically strained region tries to recover its original shape, whereas the plastic deformation creates a residual impression into the specimen surface. The residual impression is denoted by hf. Generally, the plastic deformation of polymers occurs through bursting of highly localized strain, denoted as shear bands. In the unreinforced polymer matrix, the shear band propagates comfortably without any barrier for their movement. In contrast, the presence of SGO resists its propagation in nanocomposites. Thus, the penetration depth decreases in nanocomposites with respect to the unreinforced polymer as evident from Fig. 10.
Dynamic mechanical analysis of nanocomposites
The plot of storage modulus (E′) and loss factor (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) as a function of temperature for different nanocomposites having varied SGO contents is shown in Fig. 11. The different significant dynamic mechanical parameters are listed in Table 2. The storage moduli fall with increasing temperature whereas a very sharp fall is monitored between 6 °C and 55 °C. The phenomenon of this sharp fall is designated as the primary relaxation process. The tan
δ) as a function of temperature for different nanocomposites having varied SGO contents is shown in Fig. 11. The different significant dynamic mechanical parameters are listed in Table 2. The storage moduli fall with increasing temperature whereas a very sharp fall is monitored between 6 °C and 55 °C. The phenomenon of this sharp fall is designated as the primary relaxation process. The tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ value becomes maximum at primary relaxation process.
δ value becomes maximum at primary relaxation process.
| SGO (wt%) | Tα (°C) | E′ at −30 °C (MPa) | E′ at 25 °C (MPa) | tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δmax δmax |
(tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ)rt δ)rt |
ΔT (°C) | Cross-linking density × 102 (mol m−3) |
|---|---|---|---|---|---|---|---|
| 0 | 33.4 | 41.7 | 3.05 | 1.01 | 0.94 | 78 | 4.2 |
| 0.5 | 41.5 | 53.3 | 31.5 | 0.7 | 0.61 | 77.7 | 7.6 |
| 1 | 45.8 | 60.5 | 40 | 0.66 | 0.53 | 68.8 | 9.1 |
| 1.5 | 54.1 | 74.6 | 53.8 | 0.39 | 0.21 | 30.2 | 15.3 |
| 2 | 58 | 79 | 62.5 | 0.33 | 0.14 | 11.34 | 17.4 |
After the primary relaxation process, slight decrease in E′ is observed. This region is called the rubbery plateau region. E′ is related to the ability of material to store energy when an oscillatory force is applied and it indicates the elastic behavior of materials. As shown in Fig. 11, the storage modulus of SGO reinforced nanocomposites increases enormously with an increase in SGO loading over the whole range of temperature. From the Table 2, it is clear that, at −30 °C, the E′ improves by 1.3 times to 1.9 times and at room temperature (25 °C), it improves by 10.3 times to 20.5 times with respect to matrix polymer when the SGO loading is increased from 0.5 to 2 wt%. This implies that the SGO greatly enhances the rigidity and thermal stability of nanocomposites.
Also, the cross-linking density of the nanocomposites significantly increases with an increase in SGO content due to the formation of physical cross-linking between free functional groups of SGO and polymer chains. Beside interfacial adhesion at the filler matrix interface, this cross-linking leads to a significant enhancement of dynamic modulus over a wide range of temperature. In a previous study,34 using identical polymer matrix and organoclay as filler, the storage modulus increased by 1.2 to 1.53 times at −30 °C and 1.56 to 2 times at room temperature (25 °C) with an increase in clay content from 1 to 4 wt%. In case of SGO nanocomposites, better improvement of dynamic modulus is obtained even at very low weight fraction of filler (up to 2 wt%). Because of high aspect ratio and good miscibility into the polymer matrix, the SGO is established as the most effective reinforcing agent than organoclay.
The primary relaxation temperature (Tα) is widely determined by the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ peak temperature in DMA. The peak of tan
δ peak temperature in DMA. The peak of tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ versus temperature curve i.e. tan
δ versus temperature curve i.e. tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δmax measures the vibration damping behavior of polymers. The Tα is increased from 41.5 °C to 58 °C with an increase in SGO content 0.5 to 2 wt%. The transition is delayed because of the ability of the SGO in resisting the segmental mobility of polymer chain. The materials which exhibit an effective damping (tan
δmax measures the vibration damping behavior of polymers. The Tα is increased from 41.5 °C to 58 °C with an increase in SGO content 0.5 to 2 wt%. The transition is delayed because of the ability of the SGO in resisting the segmental mobility of polymer chain. The materials which exhibit an effective damping (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ ≥ 0.3) in a wide temperature range (ΔT) can be used in practical vibration damping applications.35 As shown in Table 2, ΔT is sufficient up to the SGO loading of 1 wt%, beyond this it decreases significantly. The tan
δ ≥ 0.3) in a wide temperature range (ΔT) can be used in practical vibration damping applications.35 As shown in Table 2, ΔT is sufficient up to the SGO loading of 1 wt%, beyond this it decreases significantly. The tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ values are found to be comparable with the tan
δ values are found to be comparable with the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ values of different polymeric damping materials such as neoprene rubber, polytetrafluoroethylene (PTFE), poly(methyl methacrylate) (PMMA), epoxy etc.36
δ values of different polymeric damping materials such as neoprene rubber, polytetrafluoroethylene (PTFE), poly(methyl methacrylate) (PMMA), epoxy etc.36
The frequency responsive dynamic mechanical behavior was also studied. The variation of storage modulus and loss factor with respect to frequency is plotted in Fig. 12. In frequency response behavior, E′ demonstrates the frequency responsive of elastic behavior. From the figure, the E′ is found to increase steeply with an increase in frequency. Thus, the stiffness of nanocomposites increases with an increase in frequency. The loss factor, tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ which is the ratio of loss modulus and storage modulus slightly decreases with an increase in frequency; this further denotes an improved elastic response within the measured range of frequency.
δ which is the ratio of loss modulus and storage modulus slightly decreases with an increase in frequency; this further denotes an improved elastic response within the measured range of frequency.
Tensile stress–strain characterization of nanocomposites
The tensile stress–strain curve of nanocomposite is shown in Fig. 13A. The curves have two characteristic regions, at lower strain (<10%), a very rapid increment of stress with respect to strain is observed, and after that the stress increases with strain slowly until fracture occurs. Young's modulus in tension (ET), ultimate tensile strength (σT), elongation at break (εT) are evaluated from tensile stress–strain curve. The Young's modulus and ultimate tensile strength with varied SGO content are plotted in Fig. 13B. Both of the ET and σT measurably increases whereas εT decreases with an increase in SGO loading in nanocomposites. With respect to matrix polymer, the ET is improved from 9.6 to 21.4 times and σT is improved from 1.92 to 2.7 times when the SGO content is varied from 0.5 to 2 wt%. The εT of matrix polymer is 117%. It is decreased to 51% at a SGO loading of 2 wt%. Generally, the Young's modulus and ultimate tensile strength improve with an increase in cross-link density.37 The dynamic mechanical analysis of nanocomposites reveals that the cross-link density of nanocomposite increases with an increase in SGO loading. Thus, both of the ET and σT increases with an increase in SGO loading in nanocomposites. | ||
| Fig. 13 (A) Tensile stress versus strain plot for nanocomposites, (B) variation of Young's modulus and ultimate tensile strength as a function of SGO content. | ||
Thermogravimetric analysis (TGA) test
Thermogravimetric analysis was employed to investigate the effect of SGO into the thermal stability of nanocomposites. The TGA plots of matrix polymer and nanocomposites are shown in Fig. 14. The thermal stability of nanocomposites is enhanced with the addition of SGO. This might be due to the presence of SGO in nanocomposite system that had an ability to scavenge the formed radical during the thermal degradation of the polymer chain.38 In addition, the higher thermal conductivity of graphene sheet accelerates the heat dissipation within the nanocomposites, resulting in the improved thermal stability of nanocomposites. Also, the thermal stability improvement is attributed to the so called “tortuous path effect” of graphene nano sheets which causes in the delaying of the escape of volatile degradation product and also char formation.39,40 Tmax is the designated temperature at the peak of the differential of the virgin curve (Fig. 14 B). Tend is the last point on the temperature axis of the virgin curve and the temperatures at 10% and 50% weight loss (T10 and T50) were calculated from the virgin curve. These different degradation temperatures along with the char yield are summarized in Table 3. In all of the nanocomposites including the matrix polymer, the decomposition stage between 400 and 550 °C is most distinctive. In case of matrix polymer and 0.5 wt% SGO content nanocomposite, a minor decomposition stage is observed between 200 and 400 °C. The composites obviously have greater char yield with respect to the matrix polymer, which expectedly increases with the SGO concentration. A very small amount of GO has been significantly improved the thermal stability and char yield which was also reported in different GO reinforced nanocomposites.41,42 This is due to presence of large specific surface area of GO, which improves the interfacial interaction between polymer matrix and GO and also promotes graphitization.43 In our linseed oil based polymeric nanocomposite system, the dispersion of GO tremendously improves after its modification by CTAB. Thus a strong interfacial interaction between CTAB–GO and polymer matrix occurs, that promotes a very improved thermal stability as well as T10. At higher temperature the formation of char layers prevent flames from spreading both in the gaseous phase and condensed phase which results an enhanced thermal stability at higher temperature.44| SGO (wt%) | T10 (°C) | T50 (°C) | Tmax (°C) | Char yield (%) |
|---|---|---|---|---|
| 0 | 294 | 451 | 465 | 1.3 |
| 0.5 | 300 | 459 | 472 | 3.9 |
| 1 | 324 | 470 | 486 | 5.6 |
| 1.5 | 357 | 478 | 495 | 7.3 |
| 2 | 396 | 521 | 523 | 9.9 |
Shape memory behavior
The shape memory behavior of nanocomposites was evaluated under thermal stimulus, sunlight and microwave irradiation. The shape recovery behavior of 1 wt% SGO containing nanocomposite under sunlight is shown in Fig. 15. All the nanocomposites exhibit full shape recovery under mentioned stimuli i.e. the shape recovery ratio (Rr) of nanocomposites is 100%. Thus, the developed nanocomposites possess excellent shape memory behavior. The shape recovery times of nanocomposites having varied SGO content under different stimulus are shown in Table 4. The experimental results demonstrate that the composites have the fastest response to MW irradiation and slowest response to thermal stimulus. The fastest shape recovery under MW irradiation was due to the exceptional MW absorbing capacity of SGO. The shape recovery process becomes faster with an increase in SGO content in nanocomposites. In the shape memory test, the samples were deformed at high temperature and then the shape was fixed as a temporal shape in an ice bath. The strain energy was stored in the form of internal stress in this temporary shape. Through applying the stimulus, the stored strain energy was released in the form of recovery force, which drove the samples to retain their original shape. So, the shape recovery time reduces with an increase in stored strain energy. | ||
| Fig. 15 Shape memory experimental process of nanocomposite under (A) sunlight stimulus (B) thermal stimulus. | ||
| SGO (wt%) | Shape fixity ratio (%) | Shape recovery (%) | Shape recovery time (s) under | ||
|---|---|---|---|---|---|
| Microwave | Sunlight | Thermal | |||
| 0 | 89 | 100 | 86 | 134 | 182 |
| 0.5 | 97 | 100 | 74 | 116 | 144 |
| 1 | 98 | 100 | 62 | 103 | 131 |
| 1.5 | 100 | 100 | 48 | 75 | 102 |
| 2 | 100 | 100 | 37 | 53 | 90 |
The stored elastic stain energy in nanocomposites increases with an increase in SGO content. Thus, during the process of recovery, the nanocomposites having higher SGO content exhibited faster recovery as it had the ability to release higher amount of energy. Also, energy absorbing capacity of nanocomposites from different stimuli was improved with an increase in the SGO content, which helped for faster recovery. In otherwise, the amount of elastic energy stored and released during shape memory process improves with an increase in cross-linking density.45 As found in DMA analysis, the cross-linking density of nanocomposite increases with an increase in SGO content. Thus faster recovery was observed in nanocomposite having higher SGO content. The shape fixity ratio was also improved in nanocomposites in comparison with matrix polymer and it increases with an increase in SGO content. The researchers reported the similar type observation.46 The nature of MW stimulus is completely different from other stimulus like conventional heating and sunlight. The MW stimulus has the specialty to heat uniformly at a faster rate beside its sharp penetration ability into the sample.47 Also it has a control over larger region. As a result, under the action of MW, the nanocomposites most rapidly recover its original shape from temporal shape. The sunlight exhibits more effectiveness than of heating in water bath as it is an electromagnetic radiation and it has a faster activation potential. In addition to this, the light radiation tries to align the randomly oriented polar molecules of nanocomposites which trigger a frequently steering of molecules. The friction among these molecules also generates heat.
Conclusions
A new approach for the development of polymeric nanocomposites from renewable resource having exceptionally enhanced mechanical stability is demonstrated. The nanocomposites have been fabricated through in situ cationic polymerization of linseed oil in the presence of surface functionalized GO (SGO) sheet. The SGO sheets were highly dispersed into the polymer matrix due to the strong intermolecular specific attraction between cetylammonium ions and polymer matrix as confirmed by HRTEM. The static mechanical properties of nanocomposites has been effectively analyzed through nano-mechanical characterization. The maximum increment of elastic modulus by 4.5 times, and hardness by 4.6 times with respect to the matrix polymer is observed when the incorporation of SGO is 2 wt%. The nano-mechanical characterization specially depicts the behavior of nanocomposites with increasing content of nano filler under nanoscale deformation. The dynamic mechanical analysis of nanocomposites reveals that the maximum improvement of storage modulus by 1.9 times at −30 °C and by 20.5 times at room temperature when the incorporation of SGO in the polymer matrix is 2 wt%. The nano-mechanical and dynamic mechanical analysis of SGO filled nanocomposites demonstrate the effective reinforcement capability of SGO. The thermal stability of nanocomposites improves with an increase in the SGO content. The developed nanocomposites exhibit excellent multi stimuli responsive shape memory behavior where 100% shape recovery is observed for all nanocomposites. Thus, a very small incorporation of SGO (up to 2 wt%) into linseed oil based polymer matrix results in a series of polymeric high performance shape memory bio-nanocomposites having exceptional mechanical, dynamic mechanical and thermal stability.Acknowledgements
The authors gratefully acknowledge Council for Scientific and Industrial Research (CSIR), Government of India, SRF Research grant-in-aid [09/028(0925)/2014-EMR-I], for its financial support in this experimental work.Notes and references
- A. Gandini, Macromolecules, 2008, 41, 9491–9504 CrossRef CAS.
- J. C. Ronda, G. Lligadas, M. Galià and V. Cádiz, Eur. J. Lipid Sci. Technol., 2011, 113, 46–58 CrossRef CAS.
- L. Zhou, C. Gao and W. Xu, J. Mater. Chem., 2010, 20, 5675–5681 RSC.
- J. R. Potts, D. R. Dreyer, C. W. Bielawski and R. S. Ruoff, Polymer, 2011, 52, 5–25 CrossRef CAS.
- Y. Lu and R. C. Larock, ChemSusChem, 2009, 2, 136–147 CrossRef CAS PubMed.
- M. A. Mosiewicki, I. Mirta and A. Aranguren, Eur. Polym. J., 2013, 49, 1243–1256 CrossRef CAS.
- S. G. Tan, Z. Ahmad and W. S. Chow, Appl. Clay Sci., 2014, 90, 11–17 CrossRef CAS.
- Y. Lu and R. C. Larock, Macromol. Mater. Eng., 2007, 292, 1085–1094 CrossRef CAS.
- M. M. Reddy, S. Vivekanandhan, M. Misra, S. K. Bhatia and A. K. Mohanty, Prog. Polym. Sci., 2013, 38, 1653–1689 CrossRef CAS.
- D. P. Pfister and R. C. Larock, J. Appl. Polym. Sci., 2012, 123, 1392–1400 CrossRef CAS.
- R. L. Quirino, J. Woodford and R. C. Larock, J. Appl. Polym. Sci., 2012, 124, 1520–1528 CrossRef CAS.
- K. Putz, R. Krishnamoorti and P. F. Green, Polymer, 2007, 48, 3540–3545 CrossRef CAS.
- M. Fang, K. G. Wang, H. B. Lu, Y. L. Yang and S. J. Nutt, Mater. Chem., 2009, 19, 7098–7105 RSC.
- H. T. Hu, X. B. Wang, J. C. Wang, L. Wan, F. M. Liu, H. Zheng, R. Chen and C. Xu, Chem. Phys. Lett., 2010, 484, 247–253 CrossRef CAS.
- H. Kim, S. Kobayashi, M. A. AbdurRahim, M. L. J Zhang, A. M. A. Hillmyer, A. A. Abdala and C. W. Macosco, Polymer, 2011, 52, 1837–1846 CrossRef CAS.
- C. Lee, X. Wei, J. W. Kysar and J. Hone, Science, 2008, 321, 385–388 CrossRef CAS PubMed.
- A. A. Balandin, S. Ghosh, W. Bao, I. Calizo, D. Teweldebrhan, F. Miao and C. N. Lau, Nano Lett., 2008, 8, 902–907 CrossRef CAS PubMed.
- S. Stankovich, D. A. Dikin, R. D Piner, K. A. Kohlhaas, A. Kleinhammes, Y. Jia, Y. Wu, S. T. Nguyen and R. S. Ruoff, Carbon, 2007, 45, 1558–1565 CrossRef CAS.
- H. J. Ryu, S. S. Mahapatra, S. K. Yadav and J. W. Cho, Eur. Polym. J., 2013, 49, 2627–2634 CrossRef CAS.
- T. Xie, Polymer, 2011, 52, 4985–5000 CrossRef CAS.
- A. M. Díez-Pascual, M. A. Gómez-Fatou, F. Ania and A. Flores, Prog. Mater. Sci., 2015, 67, 1–94 CrossRef.
- S. Thakur and N. Karak, Carbon, 2012, 50, 5331–5339 CrossRef.
- B. H. Hameed, L. F. Lai and L. H. Chin, Fuel Process. Technol., 2009, 90, 606–610 CrossRef CAS.
- B. J. Briscoe, L. Fiori and E. Pelillo, J. Phys. D: Appl. Phys., 1998, 31, 2395–2405 CrossRef CAS.
- L. E. Nielson and R. F. Landel, Mechanical Properties of Polymer and Composites, Marcel Dekker, New York, 2nd edn, 1994 Search PubMed.
- S. Thakur and N. Karak, J. Mater. Chem. A, 2014, 2, 14867 CAS.
- Y. J. Yang and W. Li, Biosens. Bioelectron., 2014, 56, 300–306 CrossRef CAS PubMed.
- K. Kakaei and K. Hasanpour, J. Mater. Chem. A, 2014, 2, 15428–15436 CAS.
- W. Meng, E. Gall, F. Ke, Z. Zeng, B. Kopchick, R. Timsina and X. Qiu, J. Phys. Chem. C, 2015, 119, 21135–21140 CAS.
- E.-Y. Choi, T. H. Han, J. Hong, J. E. Kim, S. H. Lee, H. W. Kim and S. O. Kim, J. Mater. Chem., 2010, 20, 1907–1912 RSC.
- K. N. Kudin, B. Ozbas, H. C. Schniepp, R. K. Prud'homme, I. A. Aksay and R. Car, Nano Lett., 2008, 8, 36–41 CrossRef CAS PubMed.
- Y. Zhou, Q. Bao, L. A. L. Tang, Y. Zhong and K. P. Loh, Chem. Mater., 2009, 21, 2950–2956 CrossRef CAS.
- A. K. Geim and K. S. Novoselov, Nat. Mater., 2007, 6, 183–191 CrossRef CAS PubMed.
- R. Das, R. Kumar, S. L. Banerjee and P. P. Kundu, RSC Adv., 2014, 4, 59265 RSC.
- T. Trakulsujaritchok and D. J. Hourston, Eur. Polym. J., 2006, 42, 2968 CrossRef CAS.
- D. D. L. Chung, J. Mater. Sci., 2001, 36, 5733–5737 CrossRef CAS.
- H. F. Schnell, D. Goritz and E. J. Schmid, J. Mater. Sci., 1991, 26, 661 CrossRef CAS.
- H. W. Ha, A. Choudhury, T. Kamal, D.-H. Kim and S.-Y. Park, ACS Appl. Mater. Interfaces, 2012, 4, 4623–4630 CAS.
- Y. Cao, J. Feng and P. Wu, Carbon, 2010, 8, 3834–3839 CrossRef.
- X. Wang, Y. Hu, L. Song, H. Yang, W. Xing and H. Lu, J. Mater. Chem., 2011, 21, 4222 RSC.
- M. Zenga, J. Wang, R. Li, J. Liu, W. Chen, Q. Xu and Y. Gu, Polymer, 2013, 54, 3107–3116 CrossRef.
- J. Yang, M. Wu, F. Chen, Z. Fei and M. Zhong, J. Supercrit. Fluids, 2011, 56, 201–207 CrossRef CAS.
- S. Stankovich, D. A. Dikin, R. D. Piner, K. A. Kohlhaas, A. Kleinhammes and Y. Jia, et al., Carbon, 2007, 45, 1558–1565 CrossRef CAS.
- L. Nie, C. Liu, L. Liu, T. Jiang, J. Hong and J. Huang, RSC Adv., 2015, 5, 92318–92327 RSC.
- F. Li and R. C. Larock, J. Appl. Polym. Sci., 2002, 84, 1533–1543 CrossRef CAS.
- S. A. Abdullah, A. Jumahat, N. R. Abdullah and L. Frormann, Procedia Eng., 2012, 41, 1641–1646 CrossRef CAS.
- F. Zhang, T. Zhou, Y. Liu and J. Leng, Sci. Rep., 2015, 5, 11152, DOI:10.1038/srep11152.
Footnote |
| † Both authors equally contributed in this work. |
| This journal is © The Royal Society of Chemistry 2016 |











