A new topology of hexanuclear [MnIII4LnIII2] clusters: syntheses, structures, and magnetic properties†
Lei Sunab,
Hui Chena,
Chengbing Maa and
Changneng Chen*a
aState Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, The Chinese Academy of Sciences, Fuzhou, Fujian 350002, China. E-mail: ccn@fjirsm.ac.cn; Fax: +86 591 83792395
bUniversity of Chinese Academy of Sciences, Beijing 100039, China
First published on 25th January 2016
Abstract
Reactions of manganese nitrate and lanthanide nitrate hexahydrate with 2-(hydroxymethyl)pyridine (hmpH) and sodium propionate as co-ligands in the mixed solutions of acetonitrile and ethanol generated two hexanuclear Mn–Ln compounds [Mn4Ln2O2(OH)(hmp)5(EtCO2)3(MeCN)(NO3)5(H2O)] [Ln = La (1), Nd (2)]. Compounds 1 and 2 are isostructural and possess a core of [MnIII4LnIII2(μ3-OR)3(μ3-O)2(μ2-O)3]5+, which comprises three face-sharing defect cubane units. The core topology represents a new core type of Mn–Ln cluster. The soild-state dc magnetic susceptibility analyses indicate antiferromagnetic interactions within the two compounds. Compound 1 possesses an S = 0 ground state spin, fitting of the dc data for a tetranuclear MnIII4 with the Magpack program gives parameters of J1 = −0.50 cm−1, J2 = −5.03 cm−1, J3 = 3.77 cm−1 and g = 1.95.
Introduction
Growing attention has been given to single molecule magnets (SMMs) in recent decades due to their exotic physical properties and potential applications in information storage.1 SMMs are molecules that exhibit magnetic hysteresis below a certain blocking temperature due to a high energy barrier which is closely related to the combination of a large ground spin state (S) and a large zero field splitting parameter (D).2 Recently, 3d–4f heterometallic clusters as a kind of new SMM have received great interest. Compared with traditional 3d clusters which we have mainly focused on, mixing the transition metal and lanthanide ion in different proportions may modulate magnetic properties as the lanthanide ion often possesses large ground-state spin and significant single-ion anisotropy. Since the first 3d–4f SMM of Cu2Tb2 (ref. 3) was discovered in 2004, various 3d–4f clusters have been investigated, especially Mn/Ln compounds, such as Mn2Ln2,4a Mn3Ln4,4b Mn5Ln4,4c Mn8Ln4,4d Mn11Gd2,4e and Mn21Dy,4f for manganese as a transition metal always displays high spin states as well as large zero field splitting parameters related to the Jahn–Teller effect of MnIII. Nevertheless, the reports on Mn/Ln SMMs incorporating light lanthanide ions are limited by comparison with those containing heavy lanthanide ions.5 To search for new 3d–4f SMMs and further investigate the magnetic interactions within them, more Mn/Ln compounds containing light lanthanide ions need to be explored.It is important to select an appropriate ligand to construct 3d–4f compounds, and the polydentate chelating ligand with nitrogen and/or oxygen atoms could facilitate the coordination affinities of Mn and Ln metal ions. The 2-(hydroxymethyl)pyridine (hmpH), a N/O bidentate chelate and bridging ligand, has been widely employed for the synthesis of high-nuclearity Mn clusters, such as Mn10, Mn12, Mn18, Mn21.6 However, only few examples of Mn/Ln clusters used hmpH as ligand have been reported to date, such as MnIII2LnIII2, MnIII2LnIII4, MnIII8LnIII4 and MnIII4CeIII2.7 The carboxylate ions are flexible ligands and often lead to antiferromagnetic coupling. For Mn–Ln clusters using hmpH and a carboxylate as co-ligands, variation of the carboxylate R groups, from small acetate to bulky phenyl group, generated different kinds of compounds with aesthetical structures and unusual properties.7 In the present work, we used pyridine-based alkoxide and sodium propionate as co-ligands to construct a new family of mixed 3d–4f hexanuclear compounds. The magnetic susceptibility study indicates the presence of antiferromagnetic interactions within the compounds. The syntheses, structures and magnetic properties of these compounds are described in this paper.
Experimental
Syntheses
All reagents and solutions were of commercially available, used without further purification.Preparation of the compounds: Mn(NO3)2 (aq, 50%, 0.36 ml, 1.5 mmol) and Ln(NO3)3·6H2O (1.0 mmol) were added to a colorless stirred solution of hmpH (0.10 ml, 1.0 mmol) and NEt3 (0.14 ml, 1.0 mmol) in MeCN (15 ml) and EtOH (5 ml), which led to a rapid color change to dark red. Then sodium propionate (0.096 g, 1.0 mmol) were added to the vigorous solutions. The mixture was stirred for 5 h, then filtered, and the solution was left undisturbed in a flask. Slow evaporation of the solution at room temperature gave large black crystals in five days, which were collected by filtration, washed thoroughly with MeCN, and dried in vacuum.
Physical measurements
Elemental analyses (C, H and N) were performed on a Vario ELIIII Elemental Analyzer and the content of lanthanide was determined by inductively coupled plasma optical emission spectrometer. IR spectra (400–4000 cm−1) were recorded on a Vertex 70 FT-IR spectrometer in the solid state (KBr pellets). XRPD spectra were measured on a MiniFlexII diffractometer using a Cu-Kα rotating anode source with a wavelength of 1.542 Å (Fig. S1†). Variable temperature dc susceptibility magnetic data for compounds 1 and 2 were obtained by a PPMS-9T superconducting magnetometer with a field of 0.1 T and temperature ranging from 2 to 300 K. AC magnetic susceptibilities were collected on a Quantum Design MPMS-XL magnetometer. This magnetometer operates with an oscillating ac field of 3 G in the ac frequencies range 511–2311 Hz.X-ray crystallography
The single crystal data of the compounds were collected by a Cu kα rotating anode source at 100 K, using a Supernova diffractometer with the ω-scan method. Absorption correction was performed with SCALE3 ABSPACK scaling algorithm, using spherical harmonics. All structures were solved with direct methods and full-matrix least-squares refinement were implemented by the SHELXTL-97 program package.8 The non-H atoms were refined anisotropically, while the H atoms were determined with geometrical calculations riding on the respective C atoms. The solvent molecules of complexes were disordered and could not be refined satisfactorily, which were handled with the SQUEEZE option in PLATON.9 The crystal data and structure refinement details of complexes are displayed in Table 1.| 1 3C2H5OH·3H2O | 2 3C2H5OH·2H2O | |
|---|---|---|
| a R1 = ∑(||Fo| − |Fc||)/∑|Fo|.b wR2 = [∑w(Fo2 − Fc2)2/∑w(Fo2)2]0.5. | ||
| Formula | Mn4La2O36C47N11H75 | Mn4Nd2O35C47N11H73 |
| Fw | 1867.56 | 1860.17 |
| Crystal system | Triclinic | Triclinic |
| Space group | P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
| a, Å | 11.7735(4) | 11.7632(4) |
| b, Å | 16.3609(4) | 16.3266(5) |
| c, Å | 17.8475(5) | 17.7806(3) |
| α, deg | 93.405(2) | 93.1011(18) |
| β, deg | 91.388(3) | 91.1479(19) |
| γ, deg | 108.443(3) | 108.882(3) |
| V, Å3 | 3252.21(17) | 3223.77(15) |
| Z | 2 | 2 |
| T, K | 100 | 100 |
| ρcalcd, g cm−3 | 1.727 | 1.724 |
| μ, mm−1 | 16.82 | 19.049 |
| F(000) | 1656 | 1668 |
| R1a | 0.0621 | 0.0809 |
| wR2b | 0.1514 | 0.1930 |
Results and discussion
Synthesis
Reaction of Mn(NO3)2, Ln(NO3)3·6H2O, EtCO2Na, NEt3 and hmpH in MeCN/EtOH (15/5, v/v) generated two hexanuclear heterobimetallic Mn4Ln2 compounds with general formula [Mn4Ln2O2(OH)(hmp)5(EtCO2)3(MeCN)(NO3)5(H2O)] (Ln = La (1), Nd (2)) were successfully generated. Various reaction conditions have been explored, such as different types of alkali and solvents. The NEt3 is assumed to be the proton acceptor, but also promoted the oxidation reaction of MnII to MnIII with atmospheric O2 by providing alkaline conditions. When KOH, NaOH, and NaN3 were used to replace NEt3, no crystalline products were yielded. While using MeCN, MeCN/CH3OH as the solvent led to the same compounds in low yields. When using MeCN/CH2Cl2 as the solvent, no crystallized products were obtained.To explore the effect of different carboxylate R groups to the structures and properties of compounds, sodium acetate and benzoic acid were replaced sodium propionate in the reactions. When used MeCO2Na and hmpH as co-ligands, no crystallized products were yielded. While used PhCO2H and hmpH as co-ligands, it generated a family of dodenuclear clusters with the formula [MnIII8LnIII4(O)8(hmp)4(O2CPh)12(NO3)4(PhCO2H) (C2H5OH)], which have been reported by our group.7c As compared to the two clusters with different R groups, the structures differ in two parts: (i) compounds with EtCO2− groups are hexanuclear clusters, when PhCO2− as an alternative group to EtCO2− it generated dodenuclear clusters; (ii) the Mn4Ln2 compounds comprise a core of three face-sharing defected cubane units, while the Mn8Ln4 compounds possess a spindle-shaped core.
Description of the crystal structures
Compounds 1 and 2 are isostructural, therefore the structure of compound 1 will be described in detail as a representative. The partially labeled structure and core of compound 1 are shown in Fig. 1. Selected interatomic distances and angles are presented in Table 2. Compound 1 crystallizes in the triclinic space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , and the core [MnIII4LaIII2(μ3-OR)3(μ3-O)2(μ2-O)3]5+ (Fig. 1) comprises three face-sharing defected cubane units of Mn1Mn2La2(μ2-O)2(μ3-OH)(μ3-OR), Mn1Mn2La1(μ3-O)2(μ3-OH)(μ3-OR) and Mn3Mn4La1(μ3-O)2(μ2-OR)2. The four Mn ions form a square plane with the distances of Mn1–Mn2 and Mn3–Mn4 being 3.18 Å and 3.35 Å respectively, which are different from the previous reported butterfly or cubane Mn4 topology. Two La atoms are located above and below the plane. These metals are held together though eight oxygen atoms, of which three are of μ3-O, two are of η1:η2:μ2 hmp− groups, one is of η1:η1:μ2 hmp− group, one is of η1:η3:μ3 hmp− group, and the other is of η1:η2:μ3 hmp− group. The peripheral ligands are completed by five NO3− counterions, three EtCO2− ligands, one MeCN and one H2O terminal groups.
, and the core [MnIII4LaIII2(μ3-OR)3(μ3-O)2(μ2-O)3]5+ (Fig. 1) comprises three face-sharing defected cubane units of Mn1Mn2La2(μ2-O)2(μ3-OH)(μ3-OR), Mn1Mn2La1(μ3-O)2(μ3-OH)(μ3-OR) and Mn3Mn4La1(μ3-O)2(μ2-OR)2. The four Mn ions form a square plane with the distances of Mn1–Mn2 and Mn3–Mn4 being 3.18 Å and 3.35 Å respectively, which are different from the previous reported butterfly or cubane Mn4 topology. Two La atoms are located above and below the plane. These metals are held together though eight oxygen atoms, of which three are of μ3-O, two are of η1:η2:μ2 hmp− groups, one is of η1:η1:μ2 hmp− group, one is of η1:η3:μ3 hmp− group, and the other is of η1:η2:μ3 hmp− group. The peripheral ligands are completed by five NO3− counterions, three EtCO2− ligands, one MeCN and one H2O terminal groups.
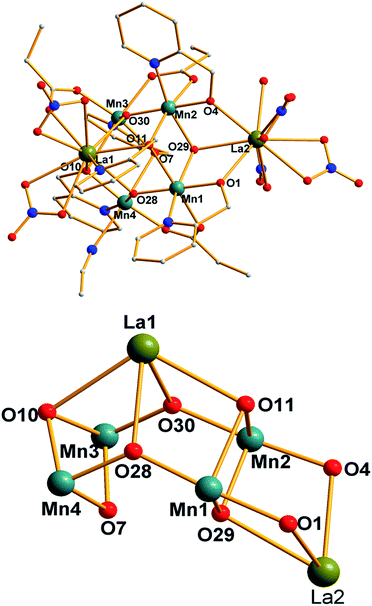 | ||
| Fig. 1 (Top) Partially labelled structure of compound 1. (Bottom) The core of compound 1. Color scheme: La, green; Mn, teal; O, red; N, blue. H atoms have been omitted for clarity. | ||
| Mn1–O1 | 1.894 | Mn3–O6 | 2.134 |
| Mn1–O2 | 2.137 | Mn3–O7 | 1.943 |
| Mn1–O11 | 2.253 | Mn3–O8 | 1.926 |
| Mn1–O28 | 1.838 | Mn3–O10 | 2.462 |
| Mn1–O29 | 1.992 | Mn3–O30 | 1.824 |
| Mn1–N1 | 2.047 | Mn3–N3 | 2.052 |
| Mn2–O4 | 1.899 | Mn4–O3 | 1.927 |
| Mn2–O5 | 2.198 | Mn4–O7 | 2.374 |
| Mn2–O11 | 2.154 | Mn4–O10 | 1.917 |
| Mn2–O29 | 2.044 | Mn4–O28 | 1.823 |
| Mn2–O30 | 1.841 | Mn4–N4 | 2.043 |
| Mn2–N2 | 2.069 | Mn4–N6 | 2.283 |
| La2–O20 | 2.616 | La2–O21 | 2.629 |
| La1–O9 | 2.550 | La1–O10 | 2.637 |
| La1–O11 | 2.536 | La1–O12 | 2.594 |
| La1–O14 | 2.659 | La1–O15 | 2.723 |
| La1–O17 | 2.636 | La1–O28 | 2.496 |
| La2–O1 | 2.484 | La1–O30 | 2.464 |
| La2–O18 | 2.615 | La2–O4 | 2.488 |
| La2–O23 | 2.608 | Mn1–O29–Mn2 | 104.188 |
| Mn1–O1–La2 | 109.882 | Mn1–O28–La1 | 113.912 |
| Mn1–O11–Mn2 | 92.631 | Mn2–O30–La1 | 112.039 |
| Mn1–O28–Mn4 | 126.318 | Mn2–O30–Mn3 | 126.223 |
| Mn1–O29–La2 | 103.266 | Mn2–O4–La2 | 100.589 |
| Mn3–O30–La1 | 113.282 | Mn3–O10–Mn4 | 98.968 |
| Mn3–O7–Mn4 | 101.211 | Mn4–O28–La1 | 109.456 |
The oxidation states of Mn metals and the protonation levels of O atoms were assigned by the charge balance consideration and bond valence sum (BVS)10 calculations (Tables 3 and 4, respectively). All MnIII atoms display a coordination number of six with distorted octahedral geometries. The three MnIII atoms exhibit distinctly Jahn–Teller axial elongations along O(carb)–Mn–O(carb) axes including O2–Mn1–O11 axes, O5–Mn2–O11 axes and O6–Mn3–O10 axes, which avoid the Mn–O2− bonds as expected. The elongated MnIII–O bonds are 0.304–0.418 Å longer than the other MnIII–O bonds. Both La ions are ten coordinate with different types of geometries varying in the coordination environment. La1 is bound by four oxygen atoms from two NO3−, two bridging μ3-O ligands, one nitrogen atom and one oxygen atom from a η1:η1:μ2 hmp− group, one oxygen atom from η1:η1:μ2 hmp− group, one oxygen atom of η1:η1:μ2 EtCO2− group. La2 coordinates with a O10 donor set consisting of six η1:η1:μ NO3−, two η1:η1:μ2 hmp−, one μ3-O and one terminal water. The La–O bond distances range from 2.459 Å to 2.724 Å and the La–N bond distance is 2.734 Å. There exist a hydrogen bond between the terminal water on the La2 and the EtCOO− on the Mn2, the O–H⋯O distance is 2.747 Å and angle is 155.09°. There are no dominant intermolecular interactions within the compound, for the nearest metal distance between different molecules is the separation of Mn–La (7.701 Å), thus it is considered that the magnetic behaviour may be of molecular origin.
| Atom | Mn(II) | Mn(III) | Mn(IV) |
|---|---|---|---|
| a The italic value is the closest to the charge for which it was calculated, and the nearest whole number can be considered as the oxidation state of the atom. | |||
| Mn1 | 3.27 | 3.02 | 3.14 |
| Mn2 | 3.19 | 2.94 | 3.05 |
| Mn3 | 3.21 | 2.98 | 3.08 |
| Mn4 | 3.25 | 3.03 | 3.11 |
| Atom | BVS | Assignment |
|---|---|---|
| a BVS values for O atoms of RO−(μ-O2−) and μ-OH groups are typically 1.8–2.1 and 1.0–1.2 respectively. | ||
| O10 | 1.99 | RO− |
| O11 | 1.94 | RO− |
| O28 | 2.07 | μ3-O2− |
| O29 | 1.26 | μ3-OH |
| O30 | 2.09 | μ3-O2− |
To date, only a small amount family of hexanuclear clusters have been reported, including [MnIV4CeIII2O2(Me-sao)6(NO3)4(OAc)2(H2O)2]11 and [Mn4Ce2O2(ButCO2)5(NO3)5(hmp)4]14 whose cores containing two [Mn2CeO] triangular units, [MnIII4LnIII2(μ3-O)2(Hbeemp)2(OAc)8(μ3-OMe)2(H2O)2] as well as [MnIII4LnIII2(H2L)2(HL)2(CH3COO)4(CH3O)2(CH3OH)4] with butterfly shaped cores,12 a face-fused double-cubane [Mn4Ln2O2(O2CBut)6(edteH2)2(NO3)2],13 [Mn4Ce2O2(O2CMe)6(NO3)4(hmp)4] possessing an octahedral core,15 [MnII2MnIII2LnIII2(Piv)8(thme)2(H2tea)2] containing four face-sharing defected cubane units.16 In addition, the core of compounds 1 and 2 is MnIII4LnIII2, which is distinct from the compound of [MnII2MnIII2LnIII2(Piv)8(thme)2(H2tea)2] with a core of MnII2MnIII2LnIII2. Obviously, the structures of compounds 1 and 2 are completely different from the compounds above, which represent a new topology.
Magnetic properties
The solid-state, variable-temperature dc magnetic susceptibility data of compounds 1 and 2 were measured in the 2–300 K temperature range with an applied field of 0.1 T.Plots of χMT vs. T for compounds 1 and 2 are depicted in Fig. 2. The χMT vs. T plots of compounds 1 and 2 show similar trends. For compound 1, at room temperature, the χMT value is 10.66 cm3 K mol−1, which is slightly lower than the expected value of 12.01 cm3 K mol−1 for the uncoupled four MnIII ions (S = 2, C = 3.00 cm3 K mol−1, g = 2.00) and two diamagnetic LaIII ions. The χMT value decreases slowly upon lowering the temperature until approximately 100 K, and then rapidly decreases down to a value close to zero at 2 K. The susceptibility data in the temperature range of 100–300 K obeys the Curie–Weiss law, and the derived Weiss constant value is −32.04 K. The above mentioned behaviour indicates dominate antiferromagnetic exchanges within compound 1, leading to an S = 0 ground state. As LaIII ion is diamagnetic, the magnetic property of antiferromagnetic for compound 1 is attributed to the interactions of MnIII–MnIII. The curve of M versus H displays an almost linear increase without clear saturation up to 8 T at 2 K (Fig. S2†), which further confirmed this conclusion.17
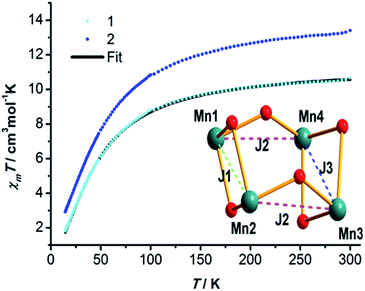 | ||
| Fig. 2 The χMT vs. T plots for compounds 1 and 2 in a 0.1 T dc field. The solid line is fit of the data of compound 1 with the Magpack program using the coupling scheme shown in the inset. | ||
To explore the magnetic exchange interactions within Mn4 core of compound 1, the magnetic susceptibility data in the 15–300 K range were fitted using the Magpack program.18 Data below 15 K were omitted as zero field or Zeeman effects maybe affect the fitting in the low temperature range. The isotropic Heisenberg spin Hamiltonian describing the magnetic interactions within the Mn4 cores could be given as follows:
| Ĥ = −2J1Ŝ1Ŝ2 − 2J2(Ŝ2Ŝ3 + Ŝ1Ŝ4) − 2J3Ŝ3Ŝ4 |
where J1 denotes the Mn1 and Mn2 coupling interaction, the exchange coupling constant of Mn1–Mn4 and Mn2–Mn3 could be considered to be the same as J2 due to similar structural parameters, J3 is the Mn3 and Mn4 exchange constant. The model containing atom labeling and exchange pathways are shown in Fig. 2 (inset). The solid line exhibits a satisfactory fit of the experimental data to the theoretical data (Fig. 2), giving the parameters of J1 = −0.50 cm−1, J2 = −5.03 cm−1, J3 = 3.77 cm−1, and g = 1.95. The exchange interactions within Mn4 core are all weak, and the J3 indicates ferromagnetic interaction between Mn3 and Mn4. The Mn3 and Mn4 are bridged by two μ2-OR− atoms which coming from hmp− groups. The alkoxide O atom of hmpH often supports ferromagnetic coupling between the metal atoms, and the comparison of exchange parameters for the compounds, whose MnIII and MnIII metals are bridged by alkoxide O atom of hmpH, are listed in Table 5.22 In addition, the MnIII–MnIII interaction in Mn4 rhombs with the JT axes containing μ2-OR− groups are often ferromagnetic interactions,19 thus it is reasonable for J3 to be positive. The weak J3 may be related to the longer MnIII–MnIII distance (3.35 Å), comparing with the reported Mn4 cores whose value of J average 8–9 cm−1 and MnIII–MnIII distance span is 3.15 to 3.23 Å.20
For compound 2, the χMT value of 13.40 cm3 K mol−1 at 300 K is much lower than the calculated value of 25.12 cm3 K mol−1 for four independent MnIII ions (S = 2, C = 3.00 cm3 K mol−1, g = 2.00) and two uncoupled NdIII ions (S = 3/2, L = 6, J = 9/2, g = 8/11). The χMT value decreases in a monotonic fashion upon cooling with a value of 0.71 cm3 K mol−1 at 2 K. A negative Weiss constant value of −44.82 K was obtained according to the Curie–Weiss law above 50 K. For compound 2, modelling the magnetic susceptibility with a similar approach as compound 1 is difficult because of the complicated intrinsic magnetic properties of the NdIII ion. In order to investigate more magnetic information of compound 2, we can subtract the plot 1 from 2 plot to explore the interactions between Mn–Nd in complex 2, the interactions of Nd–Nd can be neglected as the distance of two Nd ions being 6.35 Å. The χMT product decrease with decreasing temperature (Fig. S3†) suggesting the interactions between Mn–Nd are antiferromagnetic. The isothermal magnetization curves (M vs. H/T) for compound 2 are not coincide (Fig. 3), displaying the presence of magnetic anisotropy of NdIII or low-lying excited states.21 Furthermore, the magnetization of compound 2 increases slowly with increasing applied field without saturation even at 8 T (Fig. S4†), which is also confirmed the presence of these effects.
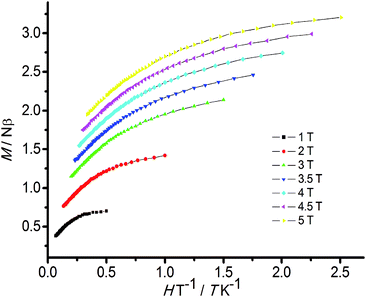 | ||
| Fig. 3 Plot of the magnetization versus H/T for compound 2 in the field of 1–5 T and the temperature of 2–15 K. | ||
The ac susceptibility measurements were performed on compounds 1 and 2 in the temperature range of 2.0 K to 5.0 K with a 3.0 G ac field oscillating in the scope of 511–2311 Hz and a zero-applied dc field. There are no frequency-dependent behaviour in the in-phase signals χ′MT and the out-of-phase signals χ′′M for both compounds (Fig. S5 and S6†). Hence, compounds 1 and 2 do not display SMM behaviour, which may be due to small magnetic anisotropy and low ground state.
Conclusions
In summary, reactions of Mn(NO3)2 and Ln(NO3)3·6H2O with hmpH and sodium propionate as co-ligands have yielded new examples of 3d/4f hexanuclear clusters. Compounds 1 and 2 with aesthetical core of three face-sharing defected cubanes, which are the new additions to the family of Mn4Ln2. Antiferromagnetic interactions within the two compounds were indicated by solid-state dc magnetic susceptibility analyses. Compound 1 has an S = 0 ground state, fitting the χMT versus T data with the Magpack program gives parameters of J1 = −0.50 cm−1, J2 = −5.03 cm−1, J3 = 3.77 cm−1 for the MnIII–MnIII interactions. The ac susceptibility data exhibit no SMM behaviour for both compounds. Further efforts to explore the same cluster with heavy lanthanide are in progress.Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 21173219, 21303201 and 21203195).Notes and references
- (a) R. Bagai and G. Christou, Chem. Soc. Rev., 2009, 38, 1011 RSC; (b) L. Bogani and W. Wernsdorfer, Nat. Mater., 2008, 7, 179 CrossRef CAS PubMed; (c) G. Christou, D. Gatteschi, D. N. Hendrickson and R. Sessoli, MRS Bull., 2000, 25, 66 CrossRef CAS; (d) M. N. Leuenberger and D. Loss, Nature, 2001, 410, 789 CrossRef CAS PubMed; (e) A. K. Powell, Nat. Chem., 2010, 2, 351 CrossRef CAS.
- (a) D. Ruiz, Z. Sun, B. Albela, K. Folting, J. Ribas, G. Christou and D. N. Hendrickson, Angew. Chem., Int. Ed., 1998, 37, 300 CrossRef CAS; (b) A. M. Ako, I. J. Hewitt, V. Mereacre, R. Clerac, W. Wernsdorfer, C. E. Anson and A. K. Powell, Angew. Chem., Int. Ed., 2006, 45, 4926 CrossRef CAS PubMed; (c) M. Murugesu, Nat. Chem., 2012, 4, 347 CrossRef CAS PubMed; (d) M. Nakano and H. Oshio, Chem. Soc. Rev., 2011, 40, 3239 RSC; (e) M. Wang, D. Q. Yuan, C. B. Ma, M. J. Yuan, M. Q. Hu, N. Li, H. Chen, C. N. Chen and Q. T. Liu, Dalton Trans., 2010, 39, 7276 RSC.
- S. Osa, T. Kido, N. Matsumoto, N. Re, A. Pochaba and J. Mrozinski, J. Am. Chem. Soc., 2004, 126, 420 CrossRef CAS PubMed.
- (a) A. Mishra, W. Wernsdorfer, S. Parsons, G. Christou and E. K. Brechin, Chem. Commun., 2005, 2086 RSC; (b) J. Y. Liu, C. B. Ma, H. Chen, M. Q. Hu, H. M. Wen, H. H. Cui, X. W. Song and C. N. Chen, Dalton Trans., 2013, 42(11), 3787 RSC; (c) V. Mereacre, A. M. Ako, R. Clerac, W. Wernsdorfer, I. J. Hewitt, C. E. Anson and A. K. Powell, Chem.–Eur. J., 2008, 14, 3577 CrossRef CAS PubMed; (d) F. Cao, S. N. Wang, D. C. Li, S. Y. Zeng, M. J. Niu, Y. Song and J. M. Dou, Inorg. Chem., 2013, 52, 10747 CrossRef CAS PubMed; (e) V. Mereacre, Y. Lan, R. Clérac, A. M. Ako, W. Wernsdorfer, G. Buth, C. E. Anson and A. K. Powell, Inorg. Chem., 2011, 50, 12001 CrossRef CAS PubMed; (f) C. Papatriantafyllopoulou, W. Wernsdorfer, K. A. Abboud and G. Christou, Inorg. Chem., 2011, 50, 421 CrossRef CAS PubMed.
- R. Sessoli and A. K. Powell, Coord. Chem. Rev., 2009, 253, 2328 CrossRef CAS.
- (a) N. C. Harden, M. A. Bolcar, W. Wernsdorfer, K. A. Abboud, W. E. Streib and G. Christou, Inorg. Chem., 2003, 42, 7067 CrossRef CAS PubMed; (b) C. Boskovic, E. K. Brechin, W. E. Streib, K. Folting, J. C. Bollinger, D. N. Hendrickson and G. Christou, J. Am. Chem. Soc., 2002, 124, 3725 CrossRef CAS PubMed; (c) E. C. Sanudo, E. K. Brechin, C. Boskovic, W. Wernsdorfer, J. Yoo, A. Yamaguchi, T. R. Concolino, K. A. Abboud, A. L. Rheingold, H. Ishimoto, D. N. Hendrickson and G. Christou, Polyhedron, 2003, 22, 2267 CrossRef CAS; (d) E. C. Sanudo, W. Wernsdorfer, K. A. Abboud and G. Christou, Inorg. Chem., 2004, 43, 4137 CrossRef CAS PubMed.
- (a) C. Papatriantafyllopoulou, K. A. Abboud and G. Christou, Inorg. Chem., 2011, 50, 8959 CrossRef CAS PubMed; (b) J. Feuersenger, D. Prodius, V. Mereacre, R. Clérac, C. E. Anson and A. K. Powell, Inorg. Chem. Commun., 2011, 14, 1851 CrossRef CAS; (c) L. Sun, H. Chen, C. B. Ma and C. N. Chen, Dalton Trans., 2015, 44, 20964 RSC; (d) C. Papatriantafyllopoulou, K. A. Abboud and G. Christou, Polyhedron, 2013, 52, 196 CrossRef CAS.
- G. M. Sheldrick, SHELXTL, 5.0, Siemens Analytical X-ray Instruments Inc., Madison, Wisconsin, USA, 1997 Search PubMed.
- P. Van der sluis and A. L. Spek, Acta Crystallogr., Sect. A: Found. Crystallogr., 1990, 46, 194 CrossRef.
- I. D. Brown and D. Altermatt, Acta Crystallogr., Sect. B: Struct. Sci., 1985, 41, 244 CrossRef.
- C. J. Milios, P. A. Wood, S. Parsons, D. Foguet-Albiol, C. Lampropoulos, G. Christou, S. P. Perlepes and E. K. Brechin, Inorg. Chim. Acta, 2007, 360, 3932 CrossRef CAS.
- T. Shiga, N. Hoshino, M. Nakano, H. Nojiri and H. Oshio, Inorg. Chim. Acta, 2008, 361, 4113 CrossRef CAS.
- A. Saha, M. Thompson, K. A. Abboud, W. Wernsdorfer and G. Christou, Inorg. Chem., 2011, 50, 10476 CrossRef CAS PubMed.
- C. Papatriantafyllopoulou, K. A. Abboud and G. Christou, Polyhedron, 2013, 52, 196 CrossRef CAS.
- C. Lampropoulos, A. E. Thuijs, K. J. Mitchell, K. A. Abboud and G. Christou, Inorg. Chem., 2014, 53, 6805 CrossRef CAS PubMed.
- J. Liu, C. Ma, H. Chen, M. Hu, H. Wen, H. Cui, X. Song and C. Chen, Dalton Trans., 2013, 42, 2423 RSC.
- (a) J. Goura, P. Bag, V. Mereacre, A. K. Powell and V. Chandrasekhar, Inorg. Chem., 2014, 53, 8147 CrossRef CAS PubMed; (b) E. M. Pineda, N. F. Chilton, F. Tuna, R. E. P. Winpenny and E. J. L. Mclnnes, Inorg. Chem., 2015, 54, 5930 CrossRef PubMed; (c) H. Ke, L. Zhang, Y. Guo and J. K. Tang, Dalton Trans., 2012, 41, 2314 RSC.
- (a) J. J. Borrás-Almenar, J. M. Clemente-Juan, E. Coronado and B. S. Tsukerblat, Inorg. Chem., 1999, 38, 6081 CrossRef; (b) J. J. Borrás-Almenar, J. M. Clemente-Juan, E. Coronado and B. S. J. Tsukerblat, Comput. Chem., 2001, 22, 985 CrossRef.
- O. Roubeau and R. C. Erac, Eur. J. Inorg. Chem., 2008, 4325 CrossRef CAS and references therein.
- T. C. Stamatatos, K. M. Poole, K. A. Abboud, W. Wernsdorfer, T. A. Brien and G. Christou, Inorg. Chem., 2008, 47, 5006 CrossRef CAS PubMed.
- J. L. Liu, W. Q. Lin, Y. C. Chen, C. C. Silvia, A. Daniel, E. Ruiz, J. D. Leng and M. L. Tong, Chem.–Eur. J., 2013, 19, 17567 CrossRef CAS PubMed.
- (a) L. Lecren, O. Roubeau, C. Coulon, Y.-G. Li, X. F. Le Goff, W. Wernsdorfer, H. Miyasaka and R. Clérac, J. Am. Chem. Soc., 2005, 127, 17353 CrossRef CAS PubMed; (b) L. Lecren, O. Roubeau, Y.-G. Li, X. F. Le Goff, H. Miyasaka, F. Richard, W. Wernsdorfer, C. Coulon and R. Clerac, Dalton Trans., 2008, 755 RSC; (c) H. Miyasaka, K. Nakata, K.-I. Sugiura, M. Yamashita and R. Clérac, Angew. Chem., Int. Ed., 2004, 43, 707 CrossRef CAS PubMed; (d) J. Yoo, A. Yamaguchi, M. Nakano, J. Krzystek, W. E. Streib, L.-C. Brunel, H. Ishimoto, G. Christou and D. N. Hendrickson, Inorg. Chem., 2001, 40, 4604 CrossRef CAS PubMed; (e) H. Miyasaka, K. Nakata, L. Lecren, C. Coulon, Y. Nakazawa, T. Fujisaki, K.-I. Sugiura, M. Yamashita and R. Clérac, J. Am. Chem. Soc., 2006, 128, 3770 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: X-ray crystallographic data for complexes 1 and 2 in CIF format. CCDC 1439267 and 1439268. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c5ra25526k |
| This journal is © The Royal Society of Chemistry 2016 |
