Switchable solvent based liquid phase microextraction of mercury from environmental samples: a green aspect
Mansoor Khanab and
Mustafa Soylak*a
aErciyes University, Faculty of Sciences, Department of Chemistry, 38039 Kayseri, Turkey. E-mail: soylak@erciyes.edu.tr; Fax: +90 352 4374933
bInstitute of Chemical Sciences, University of Peshawar, Peshawar, Pakistan
First published on 23rd February 2016
Abstract
A novel and environmentally friendly approach using switchable solvent based liquid phase microextraction (SPS-LPME), followed by UV-vis-spectrophotometric determination has been developed for the preconcentration and determination of mercury. Dithizone was used as a complexing reagent that forms a hydrophobic complex with Hg(II) which was determined spectrophotometrically using a UV-vis spectrophotometer at 574.7 nm. The N,N-Dimethyl-cyclohexylamine/protonated N,N-dimethyl-cyclohexylamine carbonate solvent pair was used as a switchable solvent in the presented work. The average relative standard deviation was 0.8% for five repeated determinations at 1.06 μg L−1 of Hg(II) concentration. Analytical parameter like the limit of detection (LOD), limit of quantification (LOQ) and enhancement factor (EF) were calculated as 0.19 μg L−1, 0.62 μg L−1 and 40, respectively. The accuracy of method was evaluated by analyzing NCS ZC81002B human hair certified reference material. The method was successfully applied to the determination of mercury in water and hair samples with satisfactory results.
1. Introduction
Various natural processes such as mineral breakdown, volcanic eruption, soil withering, and anthropogenic sources and a range of industrial catalysts, amalgams, batteries, thermometers, fungicides and pigments are the main cause polluting natural water with mercury, which is a toxic heavy metal.1–3 Mercury has many chemical and physical forms with a wide range of properties. Due to its high affinity towards the SH group in proteins and lipid tissues, accumulation of mercury occurs in consumer organisms and can cause cancer and birth defects.4–8 In natural water, mercury exists in three different forms; inorganic mercury (Hg2+ and its complex form), organic mercury (monomethyl mercury and dimethyl mercury) and elemental mercury (Hg0).5 The permissible limit for mercury in water samples is 10 μg L−1 as prescribed by World Health Organization (WHO).9,10 Also, due to amalgam fillings, the concentration of inorganic mercury in blood is 1.3–4.3 μg L−1, and 1.4–4.8 μg L−1 in urine which cause diverse biological defects.11 Therefore the determination of inorganic mercury is of immense importance in bodies of natural water.12 Due to the low concentration of mercury in natural bodies of water and their high salinity, highly efficient separation and preconcentration methods are required prior to its quantitative determination.13,14Several sophisticated analytical methods have been used for the determination of mercury at trace level which include cold vapor atomic absorption spectrometry (CVAAS),15 inductively coupled plasma mass spectrometry (ICP-MS),16 liquid chromatography coupled with inductively coupled plasma mass spectrometry (LC-ICP-MS)17 and atomic fluorescence spectrometry (AFS).18 But these techniques used for the measurement of mercury are expensive and cannot be operated in ordinary laboratory conditions. UV-visible spectrophotometric determination is a better choice for the determination of mercury in various environmental samples as it is reliable sensitive, cheap and easy to operate in ordinary laboratory conditions.19
In the recent past different methods have been employed for the preconcentration of mercury which include solid phase extraction,20 liquid–liquid phase extraction21 and cloud point extraction.22,23 There are a number of disadvantages to these procedures including long times, expense, and consuming and discharging an excess of sample waste and harmful organic solvents. As a result, secondary hazardous materials are produced that contaminate the environment.24,25 Therefore, special attention has to be made for the development of green analytical methods. To achieve the green chemistry approach, novel solvent and equipment systems and reduced liquid phase sample pretreatment techniques “liquid phase microextraction techniques” have been used to extend the concepts and use of green chemistry.26 In the literature, different models of liquid–liquid microextraction have been reported such as dispersive liquid–liquid microextraction (DLLME),20 single-drop microextraction (SDME),27 supramolecular solvent based microextraction (Ss-ME),28 hollow fiber liquid phase microextraction (HF-LPME)29 and ionic liquid based microextraction (IL-ME).30
Microextraction techniques consist of more than one step; they require a different polarity of the organic solvents for each step with a different solubility in the samples to selectively extract the analyte from the matrix components of the sample. The use of different polarity organic solvents for each step of a microextraction technique makes these techniques expensive, time consuming and harmful to researchers. Jessop et al., has synthesised a new kind of solvent which is called generation “switchable” or “smart” solvents (SS). In switchable solvents, the nonionic liquid becomes an ionic liquid upon exposure to carbon dioxide gas and then returns to its nonionic form when exposed to a nitrogen atmosphere or sodium hydroxide. Due to the reversibility of the reaction, the switched solvent can readily be returned to its original state.31,32
In order to avoid the use of many organic solvents for a single microextraction study, the use of a single switchable-polarity solvent (SP) is of immense importance. Due to their unique properties, switchable-polarity solvents (SPs) are considered green solvents for chemical processes such as extraction and purification of compounds, chemical synthesis, and catalysis due to the abrupt change in their reversible physical properties.33,34 Switchable solvents are liquids that can be reversibly converted from a hydrophilic form in the presence of CO2 at 1 atm pressure to a hydrophobic form with the absence of CO2 after the addition of base. By using the polarity switching feature of these solvents, the number of extraction solutions can be reduced and thus the excessive discharge of organic solvents into waste can be greatly overcome.35,36
The aim of our work was to develop the switchable solvent based liquid phase microextraction of mercury from water and hair samples. A switchable solvent was used for the preconcentration of mercury for the first time in this work. In this work, the switching feature of the protonated N,N-dimethyl cyclohexylamine carbonate–N,N-dimethyl cyclohexylamine solvent pair was used for the microextraction of mercury. Dithizone was used as a chelating agent which forms a colored complex with mercury which was then selectively extracted using the switchable solvent. The concentration of the complex in the extraction phase was measured using UV-visible spectrophotometry at 574.7 nm.
2. Experimental
2.1. Reagents and standards
Water purified with reverse osmosis (18.2 MΩ cm, Millipore) was used to prepare the solution. Analytical reagent grade standard of mercury with a concentration of 1000 mg L−1 was provided by Charleston, SC, USA and was used for the preparation of various standard solutions. Dithizone was purchased from Riedel-deHaen, Germany, and a standard solution of 100 mg L−1 was prepared by dissolving 0.01 g in 100 mL of methanol (Sigma Aldrich). Different combination of salts and solutions were used to achieve the buffer solution of desired pH. Phosphate buffer solutions (pH 2.0–4.0, sodium dihydrogen phosphate/phosphoric acid), phosphate buffer solutions (pH 5.0–7.0, sodium dihydrogen phosphate/disodium hydrogen phosphate) and ammonia buffer solutions (ammonia–ammonium chloride, pH 8.0–9.0) were used. Triethylamine, N,N dimethyl cyclohexylamine, hydrochloric acid (HCl), nitric acid (HNO3), methanol (CH3OH) and acetone (C3H6O) were also provided by E. Merck (Darmstadt, Germany).2.2. Instruments
Absorbance measurements were determined using a Hitachi 150–20 spectrophotometer with a quartz micro-cell with a path length of 10 mm and a volume of 500 μL. pH adjustment of all the sample solutions were carried out using a pH-meter with a Nel pH 900 (Ankara-Turkey) model glass-electrode. A centrifuge with centrifugal vials (ALC PK 120 model, Buckinghamshire, England) was used to achieve quick and complete phase separation. A microwave oven equipped with PTFE vessels, Berghof Speedwave MWS four digestion system (Germany), was used for digestion of the hair samples. The digestion program was given in Table 1.| Step | Program 1 | Program 2 |
|---|---|---|
| T (°C) | 145 | 190 |
| P (bar) | 50 | 50 |
| Power (%) | 70 | 90 |
| Ta (min) | 10 | 5 |
| Time (min) | 5 | 10 |
2.3. The synthesis of the hydrophilic form of the switchable solvent
The protonated N,N-dimethyl-cyclohexylamine carbonate switchable extraction solvent was tested as the extraction solvent in this microextraction study. Protonated N,N-dimethyl-cyclohexylamine carbonate was synthesized by taking 200 mL N,N-dimethyl-cyclohexylamine and 200 mL ultrapure water in 1 L glass beaker. Afterwards, dry ice (∼20 g) as CO2 supplier was added gradually to the beaker, when the dissolution of CO2 was completed, the amine phase became cloudy under the influence of magnetic stirring. The addition of dry ice was repeated until a single phase containing protonated N,N-dimethyl-cyclohexylamine carbonate solution was obtained. Afterward, the mixture was stirred for 2 h at room temperature to ensure the complete protonation of N,N-dimethyl-cyclohexylamine.2.4. General procedure
The proposed switchable solvent based liquid phase microextraction (SPS-LPME) was carried out for the separation and preconcentration of mercury in a 50 mL of centrifuge tube containing 10 μg L−1 of Hg(II). To the tube, 2 mL of pH 7.0 buffer solution and 12 μg of dithizone were added respectively and the solution was allowed to stand for 3 min to ensure that the hydrophobic Hg(II)–dithizone complex formation was complete. A homogenous solution was obtained after the addition of 1.0 mL of protonated N,N-dimethyl-cyclohexylamine carbonate. Then, in order to extract the hydrophobic Hg(II)–dithizone complex, the hydrophilic protonated N,N-dimethyl cyclohexylamine was converted into hydrophobic N,N-dimethyl-cyclohexylamine (microsized extraction phase) after the addition of 1.5 mL of 10 M NaOH solution. The solution was allowed to stand for 2 min to ensure the formation of N,N-dimethyl-cyclohexylamine phase and extraction of Hg(II)–dithizone complex from water phase into the fine droplets of the N,N-dimethyl-cyclohexylamine phase. Complete phase separation was achieved by putting the tube inside the centrifuge for 7 min. The aqueous phase was separated and discarded and the extraction phase (N,N-dimethyl-cyclohexylamine phase) was diluted to 1 mL using methanol. The concentration of the Hg(II) in the last volume was measured quickly to avoid evaporation of methanol at 574.7 nm using a UV-visible spectrophotometer. The schematic representation of the developed SPS-LPME steps is presented in the Fig. 1.2.5. Application to real samples
The developed method was applied to sea water, dam water and dental waste water samples obtained from a dental amalgam restorations department in a hospital (Erciyes University, Turkey). Prior to the preconcentration procedure, all the water samples were filtered through a 0.45 m pore size membrane filter to remove suspended particulate matter.The hair samples and NCS ZC81002B human hair certified reference material were digested in a microwave inside a closed Teflon vessel. 100 mg of hair samples or certified reference material were accurately weighed into a PTFE digestion vessel. The decomposition of the samples was carried out in a microwave digestion system. 10 mL of concentrated HNO3 was added to the vessel and after 20 min, the vessel is closed. A two-step programme was used for the digestion of the samples. The extract was transferred into a centrifuge tube and the developed method was applied the extract. Blank experiments were also carried out in the same way.
3. Result and discussion
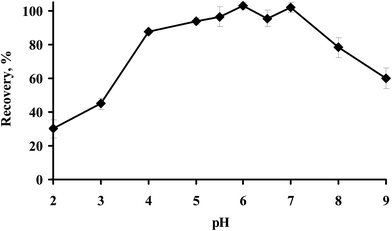
3.1. Effect of pH
Extraction of metal ions from the polar phase to the extraction phase by LPME is greatly influenced by the pH of solution.37,38 It is because the pH of the solution greatly affects the formation of the hydrophobic complex Hg–Dithizone. Therefore, the proposed SPS-LPME of mercury was carried out at different pHs ranging from 2.0 to 9.0, as shown in Fig. 2. It can be concluded from Fig. 2 that the highest percent recoveries of Hg(II) were obtained at the pH range of 5.0–7.0 and reached a maximum at pH 7.0. Therefore, the rest of the other experiments in the presented microextraction method were performed at a solution pH of 7.0.3.2. Selection of switchable extraction solvent
For quantitative recoveries, the selection of suitable switchable solvents is of immense importance in SPS-LPME methods. For the selection of the appropriate extraction solvent, some properties of the switchable solvent should be kept in mind such as (a) the extractive tendency towards the target compounds, (b) it must have two forms (hydrophilic and hydrophobic) which are interconvertible by the addition or removal of CO2 from the solvent system, (c) the hydrophobic form of the switchable solvent should have low density, (d) high solubility is required for the hydrophilic form in water and low solubility is required for the hydrophobic form of the switchable solvent in water so as to establish a stable two-phase system. Based on these criteria, two switchable solvents, triethylamine/protonated triethylamine carbonate and N,N-dimethyl-cyclohexylamine/protonated N,N-dimethyl-cyclohexylamine carbonate switchable extraction solvent pairs were assessed for the extraction of the hydrophobic Hg(II)–dithizone complex from sample solution. The recovery values obtained for triethylamine/protonated triethylamine carbonate solvent pair is (94 ± 3%) and the recovery obtained for N,N-dimethyl-cyclohexylamine/protonated N,N-dimethyl-cyclohexylamine carbonate solvent pair is (99 ± 4%). Both values for the two solvent are similar and quantitative. N,N-Dimethyl-cyclohexylamine/protonated N,N-dimethyl-cyclohexylamine carbonate solvent was used for further works.3.3. Selection of phase transition trigger
Protonated N,N-dimethyl-cyclohexylamine (hydrophobic form) and protonated N,N-dimethyl-cyclohexylamine carbonate (hydrophilic form) are interconvertible by the addition or removal of CO2 from the system. In order to remove CO2, several physical and chemical methods such as the addition of sodium hydroxide or hydrochloric acid solution into the solution, passing of inert gas (Ar) in the solution and heating of the solution were checked. No formation of cloudy solution or phase separation was obtained using hydrochloric acid solution, heating or passing of inert gas (Ar) ways. Due to high tendency of sodium hydroxide towards protonated hexylamine and the tear off hydrogen bond of the amine group, sodium hydroxide gives the best result among all the other procedure used. Hence, 10 M sodium hydroxide solution was used as the phase transition trigger for this microextraction work.3.4. Effect of extraction solvent volume
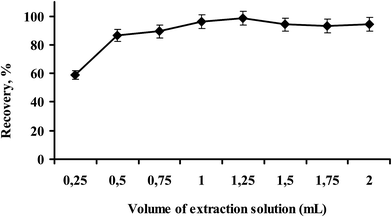
After choosing N,N-dimethyl-cyclohexylamine-protonated N,N-dimethyl-cyclohexylamine carbonate solvent pair as the most suitable switchable solvent system for this microextraction study, it was necessary to optimize the volume of protonated N,N-dimethyl-cyclohexylamine carbonate, which provides a homogenous extraction system for the analyte. The effect of the volume of the protonated N,N-dimethyl-cyclohexylamine carbonate as the extraction solvent on the recovery was investigated under fixed experimental conditions. The microextraction studies were performed at different volumes of extraction solution ranging from 0.25 mL to 2.0 mL, as shown in Fig. 3. It can be concluded from Fig. 3 that there is a gradual increase in the percentage recoveries of Hg(II) from 0.25 mL (59 ± 0.8) to 1 mL (96 ± 0.1). As there is no significant change in the percent recoveries after 1.0 mL of protonated N,N-dimethyl-cyclohexylamine carbonate, that volume was used in further studies.3.5. Effect of NaOH
As 10 M NaOH was found to be the best phase transition trigger as it removes CO2 from the hydrophilic protonated N,N-dimethyl-cyclohexylamine carbonate to form hydrophobic N,N-dimethyl-cyclohexylamine. In order to convert the hydrophilic form of the switchable solvent to its hydrophobic form and also to quantitatively extract the Hg(II)-dithizone complex from sample solution, the proposed switchable solvent based liquid phase microextraction was carried out at different volumes of 10 M of NaOH ranging from 0.25 mL to 2.0 mL, as shown in Fig. 4. Results showed that the quantitative recovery of Hg(II) occurred between 1.5 and 2.0 mL of 10 M NaOH. In this study, 1.5 mL of 10 M NaOH was used for further experiments of the proposed method.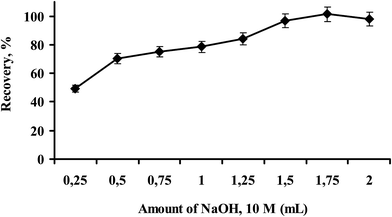 | ||
| Fig. 4 Effect of NaOH volume on the recovery of Hg(II) (experimental conditions; pH: 7.0, volume of extraction solution: 1.0 mL, amount of dithizone: 12 μg, sample volume: 10 mL, N = 3). | ||
3.6. Effect of amount of complexing agent
The selection of the suitable amount of complexing agent (dithizone) is also important as it affect the signals obtained during absorbance measurement in UV-visible spectrometry. Dithizone (diphenylthiocarbazone) is known to be an effective chelating reagent of metal ions for spectrophotometric determination.39,40 Dithizone has been also widely used as a chelating agent for the separation and preconcentration of metal ions at trace levels.41,42 The effect of the amount of dithizone as complexing agent was studied in the range of 2–20 μg as shown from Fig. 5. The obtained results indicate that quantitative recovery of Hg(II) occurred in the range of 12 μg to 20 μg of the amount of complexing agent. Therefore 12 μg of dithizone was used in further switchable solvent based liquid–liquid microextraction experiments for Hg(III).3.7. Effect of sample volume
Determination of the preconcentration factor43–46 is important as it determines the efficiency of the developed method. The highest preconcentration factor of the method can be calculated as the highest sample volume to which the proposed microextraction is successfully applicable. Therefore the proposed switchable solvent based (Ss-LLM) studies were carried out at different volumes of samples in the range of 5–50 mL. Quantitative recoveries of Hg(II) were obtained at a sample volume of below 40 mL, and a preconcentration factor of 40 was obtained, using a 40 mL sample volume and 1 mL final volume.3.8. Matrix effect
Due to the complex matrix nature of samples like soil, ore and water, preconcentration methods are greatly influenced46–49 by various coexisting ions present in the matrices of these samples with analyte metals. Therefore prior to the application of the proposed method to the real samples, assessing the effect of coexisting ions on the % recovery of mercury is of immense importance. In order to investigate the effects of matrix ions on SPS-LPME and UV-vis determination, the proposed microextraction method was carried in the presence of some interfering ions which coexist with analyte metal in the sample matrices. No interference was observed from most of the ions tested except for Cu2+, Mn2+ and Ni2+ ions. To eliminate the interference effects of the ions, the developed SPS-LPME method was carried out in 0.025 M EDTA solution, where the medium acted as the masking agent. The results given in Table 2 show that there is no significant interference by these interfering ions even at high concentrations. This shows that the presented method is highly selective and free of interference.| Matrix ion | Amount added (μg) | Added as | % recovery |
|---|---|---|---|
| a Mean ± standard deviation. | |||
| Na+ | 5000 | NaNO3 | 95 ± 2 |
| K+ | 5000 | KCl | 98 ± 1 |
| Ca2+ | 500 | Ca(NO3)2·4H2O | 94 ± 2 |
| Mg2+ | 500 | Mg(NO3)2·6H2O | 97 ± 1 |
| Zn2+ | 10 | Zn(NO3)2·7H20 | 103 ± 3 |
| Cd2+ | 5 | Cd(NO3)2·6H2O | 103 ± 5 |
| Pb2+ | 5 | Pb(NO3)·6H2O | 99 ± 3 |
| Cu2+ | 2.5 | Cu(NO3)·6H2O | 97 ± 5 |
| Ni2+ | 2.5 | Ni(NO3)2·6H2O | 97 ± 4 |
| Co2+ | 5 | Co(NO3)2·6H2O | 100 ± 3 |
| Mn2+ | 2.5 | Mn(NO3)2·4H2O | 102 ± 3 |
| SO42− | 1000 | Na2SO4 | 101 ± 3 |
| CO32− | 500 | Na2CO3 | 95 ± 1 |
| F− | 1000 | NaF | 96 ± 3 |
3.9. Analytical figures
The analytical performance of the method can be investigated by determining the analytical parameters under optimized experimental conditions. For one sample, the total time is approximately 15 minutes. At the same time, the method could be applied to 20 samples. Calibration standards were subjected to the microextraction procedure. The calibration curve equation was found to be A = 0.0053 + 0.040C (A: the absorbance of the solution, C: determined mercury concentration of the solution). The correlation coefficient (r2) was 0.989. The linear range for calibration curve is 10–300 μg L−1. The limit of detection was found to be 0.19 μg L−1, which was calculated as 3 times the ratio of the signal of 10 blank absorbance’s/slope of calibration curve. The limit of quantification was found to be 0.62 μg L−1, which was calculated as 10 times to the ratio of signal of 10 blank absorbance’s/slope of calibration curve. The relative standard deviations (RSD, %) was calculated as 0.8%, using the analysis of five replicates containing 1.06 μg L−1 of Hg(II). Both the values of preconcentration factor and enhancement factor were found to be 40, which were calculated by determining the ratio of the highest sample volume to the eluent volume and the ratio of Hg(II) concentration in the extraction phase to the initial concentration of Hg(II) in the sample solution respectively.3.10. Applications
The validity of the presented SPS-LPME method was checked by analyzing NCS ZC81002B human hair certified reference material. For the analysis of the mercury content in the certified reference material, 5 mL of 0.05 M EDTA was used as the masking agent in the microextraction procedure. While the certified value of NCS ZC81002B human hair certified reference material was 1.06 ± 0.28 μg g−1, the mercury level was found to be 1.02 ± 0.24 μg g−1 (N = 3). The recovery values were 96%. It was found that there was no significant difference between the result obtained and the certified result. The proposed method can successfully be applied for the quantitative determination of Hg(II) at trace levels in real samples.For the addition–recovery applications, the proposed SPS-LPME method was applied to three water samples (sea water, dam water, dental waste water) and two hair samples (Hair I and Hair II) and the results are given in Table 3. The quantitative % recovery results obtained shows that the developed SPS-LPME is sensitive, precise and accurate for the determination of Hg(II) in real samples.
| Sample | Added, μg | Found, μgb | Recovery, % |
|---|---|---|---|
| a Mean ± standard deviation.b BDL: below detection limit. | |||
| Sea water | 0 | 0.95 ± 0.02a | — |
| 5 | 5.73 ± 0.03 | 96 | |
| 6 | 6.67 ± 0.06 | 95 | |
| Dam water | 0 | BDL | — |
| 3 | 3.00 ± 0.05 | 100 | |
| 6 | 6.29 ± 0.06 | 105 | |
| Waste water | 0 | 2.33 ± 0.05 | — |
| 5 | 7.43 ± 0.05 | 102 | |
| 8 | 10.4 ± 0.03 | 101 | |
| Hair I | 0 | BDL | — |
| 1 | 1.04 ± 0.10 | 104 | |
| 2 | 2.02 ± 0.05 | 102 | |
| Hair II | 0 | BDL | — |
| 2 | 1.90 ± 0.30 | 95 | |
| 3 | 2.87 ± 0.08 | 96 | |
The developed SPS-LPME method was compared with other microextraction methods to assess the extraction efficiency. The LOD, preconcentration factor and additional information are given in Table 4. It can be concluded from Table 4 that the presented method is either comparable or more efficient than other microextraction methods in the literature.54–61
| Method | Analysis | PF/EFa | Detection limit μg L−1 | Sample | Ref. |
|---|---|---|---|---|---|
| a Preconcentration factor/enhancement factor. | |||||
| Ionic liquid phase microextraction | UV-vis | 50 | 18.8 | Water | 1 |
| Solid phase extraction | ICP-MS | — | 0.06 | Biological | 50 |
| Liquid–liquid extraction | Fluorometric | — | 0.2 | Water | 51 |
| Solid phase extraction | UV-vis | — | 0.05 | Water | 52 |
| Cloud point extraction | ICP-MS | 100 | 0.04 | Water | 53 |
| Solid phase extraction | Spectrophotometry | 33 | 2.0 | Water | 54 |
| Ionic liquids microextraction | Stripping voltammetry | 17 | 0.05 | Water | 55 |
| Micelle-mediated extraction | Spectrophotometry | 33.3 | 0.86 | Water | 56 |
| Dispersive liquid–liquid microextraction | Spectrophotometry | 64 | 3.3 | Water | 57 |
| Surfactant-sensitized | Spectrophotometry | — | 6 | Water | 58 |
| Cloud point extraction | Spectrophotometry | 33.3 | 1.65 | Water | 59 |
| Switchable solvent based liquid phase microextraction | Spectrophotometry | 40 | 0.19 | Water, hair | This study |
4. Conclusion
A green approach towards the preconcentration and determination of Hg(II) in water and hair samples was achieved using switchable solvent based liquid phase microextraction (SPS-LPME) associated with UV-vis spectrophotometric determination. The method is highly environmentally friendly as low amounts of organic waste are produced due to the use of switchable solvent. The low LOD value of 0.19 μg L−1 and LOQ of 0.62 μg L−1 reflect the high sensitivity of the method. The preconcentration factor (PF) and enhancement factor (EF) each with a value of 40 shows that the method is highly efficient. The method is also cost effective because there is no need of special laboratory equipment. The proposed method is comparable or better than other preconcentration method with regards to the LOD. The method was successfully applied to water and hair samples.Acknowledgements
Mansoor Khan is thankful to the Scientific and Technological Research Council of Turkey (TUBITAK) under “2216 Research Fellowship Programme for Foreign Citizens” for financial support.References
- M. Gharehbaghi, F. Shemirani and M. Baghdadi, Int. J. Environ. Anal. Chem., 2009, 89, 21–33 CrossRef CAS.
- A. Afkhami, T. Madrakian and H. Siampour, Int. J. Environ. Anal. Chem., 2006, 86, 1165–1173 CrossRef CAS.
- M. Zaki and M. Esmaile, Anal. Lett., 1997, 30, 1579–1590 CrossRef CAS.
- F. Pena-Pereira, I. Lavilla, C. Bendicho, L. Vidal and A. Canals, Talanta, 2009, 78, 537–541 CrossRef CAS PubMed.
- K. Leopold, M. Foulkes and P. Worsfold, Anal. Chim. Acta, 2010, 663, 127–138 CrossRef CAS PubMed.
- E. M. Martinis, P. Bertón, R. A. Olsina, J. C. Altamirano and R. G. Wuilloud, J. Hazard. Mater., 2009, 167, 475–481 CrossRef CAS PubMed.
- A. Abdollahi, M. Amirkavehei, M. M. Gheisari and F. Tadayon, E3S Web of Conferences, EDP Sci. 2013, p. 41006 Search PubMed.
- M. Tuzen, A. Sari, D. Mendil and M. Soylak, J. Hazard. Mater., 2009, 169, 263–270 CrossRef CAS PubMed.
- World Health Organisation, Guidelines for Drinking Water Quality, World Health Organization, Geneva, Switzerland, 2008 Search PubMed.
- S. J. Cobbina, A. B. Duwiejuah, R. Quansah, S. Obiri and N. Bakobie, Int. J. Environ. Res. Public Health, 2015, 12, 10620–10634 CrossRef PubMed.
- J. S. Vamnes, R. Eide, R. Isrenn, P. J. Höl and N. R. Gjerdet, Sci. Total Environ., 2003, 308, 63–71 CrossRef CAS PubMed.
- M. Tuzen and M. Soylak, Bull. Environ. Contam. Toxicol., 2005, 74, 968–972 CrossRef CAS PubMed.
- M. Kawaguchi, K. Inoue, M. Yoshimura, R. Ito, N. Sakui, N. Okanouchi and H. Nakazawa, J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2004, 805, 41–48 CrossRef CAS PubMed.
- H. Ashkenani, S. Dadfarnia, A. M. H. Shabani, A. A. Jaffari and A. Behjat, J. Hazard. Mater., 2009, 161, 276–280 CrossRef CAS PubMed.
- M. Tuzen, I. Karaman, D. Citak and M. Soylak, Food Chem. Toxicol., 2009, 47, 1648–1652 CrossRef CAS PubMed.
- E. V. Alonso, M. S. Cordero, A. G. de Torres, P. C. Rudner and J. C. Pavón, Talanta, 2008, 77, 53–59 CrossRef PubMed.
- L. H. Reyes, G. M. Rahman and H. S. Kingston, Anal. Chim. Acta, 2009, 631, 121–128 CrossRef CAS PubMed.
- V. N. Tirtom, Ş. Goulding and E. Henden, Talanta, 2008, 76, 1212–1217 CrossRef CAS PubMed.
- F. Théraulaz and O. P. Thomas, Microchim. Acta, 2008, 113, 53–59 CrossRef.
- J. P. Snell, W. Frech and Y. Thomassen, Analyst, 1996, 121, 1055–1060 RSC.
- X. Jia, Y. Han, X. Liu, T. Duan and H. Chen, Spectrochim. Acta, Part B, 2011, 66, 88–92 CrossRef.
- C.-G. Yuan, K. Lin and A. Chang, Microchim. Acta, 2010, 171, 313–319 CrossRef CAS.
- Y. Li and B. Hu, Spectrochim. Acta, Part B, 2007, 62, 1153–1160 CrossRef.
- M. Soylak and E. Yilmaz, At. Spectrosc., 2013, 34, 15–19 CAS.
- Z. A. Alothman, E. Yilmaz, M. Habila, A. Shabaka and M. Soylak, Microchim. Acta, 2013, 180, 669–674 CrossRef CAS.
- L. Xia, X. Li, Y. Wu, B. Hu and R. Chen, Spectrochim. Acta, Part B, 2008, 63, 1290–1296 CrossRef.
- H. Bagheri and M. Naderi, J. Hazard. Mater., 2009, 165, 353–358 CrossRef CAS PubMed.
- Y. He, Chem. Pap., 2014, 68, 995–1007 CAS.
- A. Spietelun, Ł. Marcinkowski, M. de la Guardia and J. Namieśnik, Talanta, 2014, 119, 34–45 CrossRef CAS PubMed.
- S. Li, S. Cai, W. Hu, H. Chen and H. Liu, Spectrochim. Acta, Part B, 2009, 64, 666–671 CrossRef.
- E. Yilmaz and M. Soylak, Anal. Chim. Acta, 2015, 886, 75–82 CrossRef CAS PubMed.
- E. Yilmaz and M. Soylak, J. Anal. At. Spectrom., 2015, 30, 1629–1635 RSC.
- P. G. Jessop, D. J. Heldebrant, X. Li, C. A. Eckert and C. L. Liotta, Nature, 2005, 436, 1102 CrossRef CAS PubMed.
- J. R. Vanderveen, J. Durelle and P. G. Jessop, Green Chem., 2014, 16, 1187–1197 RSC.
- Y. Liu, P. G. Jessop, M. Cunningham, C. A. Eckert and C. L. Liotta, Science, 2005, 313, 958–960 CrossRef PubMed.
- I. Anugwom, P. Mäki-Arvela, P. Virtanen, S. Willför, R. Sjöholm and J.-P. Mikkola, Carbohydr. Polym., 2012, 87, 2005–2011 CrossRef CAS.
- K. Pytlakowska, Microchim. Acta, 2016, 183, 91–99 CrossRef CAS.
- M. Tuzen and M. Soylak, J. Hazard. Mater., 2009, 162, 724–729 CrossRef CAS PubMed.
- CRC Handbook of Chemistry and Physics, ed. C. R. Weast, CRC Press, Ohio, 1976 Search PubMed.
- J. Chwastowska and E. Kosiarska, Talanta, 1988, 35, 439 CrossRef CAS PubMed.
- M. Hiraide, M. H. Sorouradin and H. Kawaguchi, Anal. Sci., 1994, 10, 125 CrossRef CAS.
- A. Uzawa, W. Yoshimura and T. Okutani, Bunseki Kagaku, 1995, 44, 391 CrossRef CAS.
- M. Bagtash, Y. Yamini, E. Tahmaseb, J. Zolgharnein and Z. Dalirnasab, Microchim. Acta, 2016, 183, 449–456 CrossRef CAS.
- M. Ghaedi, F. Ahmadi, Z. Tavakoli, M. Montazerozohori, A. Khanmohammadi and M. Soylak, J. Hazard. Mater., 2008, 152, 1248–1255 CrossRef CAS PubMed.
- O. S. Taskin, N. Ersoy, A. Aksu, B. Kiskan, N. Balkis and Y. Yagci, Polym. Int., 2016 DOI:10.1002/pi.5074.
- M. Soylak and O. Ercan, J. Hazard. Mater., 2009, 168, 1527–1531 CrossRef CAS PubMed.
- M. Soylak, L. Elci and M. Dogan, Anal. Lett., 1993, 26, 1997–2007 CrossRef CAS.
- H. Shirkhanloo, M. Falahnejad and H. Z. Mousavi, J. Appl. Spectrosc., 2016, 82, 1072–1077 CrossRef CAS.
- D. Trak, E. Kendüzler and Y. Arslan, Anal. Lett., 2016 DOI:10.1080/00032719.2015.1113424.
- C. Huang and B. Hu, Spectrochim. Acta, Part B, 2008, 63, 437–444 CrossRef.
- C. Zheng, Y. Li, Y. He, Q. Ma and X. Hou, J. Anal. At. Spectrom., 2005, 20, 746–750 RSC.
- M. E. Mahmoud, Anal. Chim. Acta, 1999, 398, 297–304 CrossRef CAS.
- J. C. Wuilloud, R. G. Wuilloud, M. a. F. Silva, R. A. Olsina and L. D. Martinez, Spectrochim. Acta, Part B, 2002, 57, 365–374 CrossRef.
- N. Rajesh and M. S. Hari, Spectrochim. Acta, Part A, 2008, 70, 1104–1108 CrossRef CAS PubMed.
- Z. Li, S. Xia, J. Wang, C. Bian and J. TongState, J. Hazard. Mater., 2016, 301, 206–213 CrossRef CAS PubMed.
- H. I. Ulusoy, J. AOAC Int., 2014, 97, 238–244 CrossRef CAS PubMed.
- V. A. Lemos, L. O. dos Santos and E. D. Silva, J. AOAC Int., 2012, 95, 227–231 CrossRef CAS PubMed.
- R. Gurkan, T. Cepken and H. I. Ulusoy, Turk. J. Chem., 2012, 36, 159–177 CAS.
- H. I. Ulusoy, R. Gurkan and S. Ulusoy, Talanta, 2012, 88, 516–523 CrossRef CAS PubMed.
- E. Beceiro-Gonzalez, A. Guimaraes and M. Alpendurada, J. Chromatogr. A, 2009, 1216, 5563–5569 CrossRef CAS PubMed.
- Z. Gao and X. Ma, Anal. Chim. Acta, 2011, 702, 50–55 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2016 |