Elucidation of the role of estradiol and progesterone in regulating reproduction in the edible crab, Oziothelphusa senex senex†
CH. Swetha‡
ab,
B. P. Girish‡ab and
P. Sreenivasula Reddy *b
*b
aDepartment of Biotechnology, Sri Venkateswara University, Tirupati-517 502, India
bDepartment of Zoology, Sri Venkateswara University, Tirupati-517 502, India. E-mail: psreddy1955@gmail.com; Fax: +91-877-2249611; Tel: +91-877-2249320
First published on 1st March 2016
Abstract
Vertebrate sex steroids are ubiquitous and important bioactive mediators for many physiological functions. Progesterone and 17β-estradiol have been detected in few crustaceans and it has been hypothesized that they are involved in the regulation of ovarian maturation. However, in the freshwater crab, Oziothelphusa senex senex, the presence of 17β-estradiol and progesterone, as well as their mechanisms of action on ovarian maturation, have not yet been investigated. In this study, we report the presence of 17β-estradiol and progesterone in the haemolymph of O. senex, and their levels were significantly higher during late vitellogenesis. Moreover, we report that administration of these hormones increased ovarian index, oocyte diameter, and ovarian vitellin levels in the crab. Additionally, the mRNA levels of ecdysteroid receptor and retinoid receptor in the hepatopancreas and ovary, and vitellogenin mRNA levels in the hepatopancreas also increased after administration of progesterone and 17β-estradiol. In silico analysis revealed interaction between vertebrate steroid hormones and the ecdysteroid receptor. The ability of female sex steroids to mimic the effects of ecdysteroids suggests that the vertebrate steroids may represent another group of reproductive endocrines in crustaceans besides peptides (gonad inhibiting/stimulating hormones), terpenoids (methyl farnesoate) and ecdysteroids.
1. Introduction
Steroids have been speculatively viewed as very ancient ‘bio-regulators’ that are omnipresent throughout the biosphere and evolved prior to the appearance of eukaryotes.1 Thus, the steroid structure is characteristic not only of steroid hormones, but also of a variety of materials that occur in the natural environment. Their use as hormones is well established in two phylogenetically unrelated lines: the arthropods, where ecdysteroids contribute to the regulation of growth (molting) and reproduction,2–4 and the vertebrates, where androgens and estrogens regulate sexual characteristics and reproduction. Although androgens and estrogens are highly characteristic of the vertebrate endocrine system, they also occur in invertebrates and in plants.Vertebrate steroids viz., pregnenolone, progesterone, 17α-hydroxyprogesterone, 17α-hydroxypregnenolone, 6β-hydroxyprogesterone, 20α-hydroxyprogesterone, 17β-estradiol, estrone, testosterone, etc., have also been discovered in the hepatopancreas, ovary and haemolymph of several crustaceans.5–14 Moreover the enzymes involved in steroid biosynthesis especially 17β-hydroxysteroid dehydrogenase and 3β-hydroxysteroid dehydrogenase have also been discovered in the tissues of several crustaceans.15–21 Exogenous administration of estradiol and progesterone induced ovarian maturation in crabs, crayfishes and prawns22–27 by stimulating vitellogenin (Vg) synthesis28–33 and secretion.27 Indeed, a positive correlation between circulating Vg levels and haemolymph progesterone and 17β-estradiol levels have been reported for crabs,34–36 shrimps32,37 and crayfish.38
Despite several reports on the occurrence of vertebrate steroid hormones and their receptors in crustaceans,39 their modes of action in crustaceans remain to be elucidated. The present study reports the profiles of the two steroid hormones namely 17β-estradiol and progesterone during vitellogenesis in the haemolymph of the fresh water edible crab, Oziothelphusa senex senex. Furthermore, this study also examines the effect of exogenous administration of these hormones on ovarian growth and vitellin levels. In addition, in silico docking studies were conducted to explore the possible mechanism of action of vertebrate steroids in crustaceans. Finally, mRNA levels of ecdysteroid receptor (EcR), retinoid receptor (RXR), ecdysteroid responsive gene (E75) and vitellogenin in the tissues were determined after administration of 17β-estradiol and progesterone. Knowledge on the precise role of vertebrate steroid hormones on ovarian maturation and oocyte development could be used to develop technology to induce the production of gametes and spawning of commercially important crustaceans.
2. Material and methods
2.1. Determination of the levels of 17β-estradiol and progesterone in the haemolymph of previtellogenic and vitellogenic stage III crabs
Sexually mature female crabs, O. senex, (NCBI Taxon ID: 324906) at various stages of ovarian maturation, with average weight of 32–37 g were obtained from irrigation canals around Tirupati (13° 36′ N, 79° 21′ E), India. Ovarian stages were determined according to Swetha et al.40 Further, morphological and histological description of the different stages of ovarian development in the crab O. senex senex was presented as a ESI data (Fig. S1; Table S1†). Crabs were acclimatized for 48 h before the haemolymph was collected for the determination of 17β-estradiol and progesterone. The crabs were anesthetized on ice for 10 min before the collection of haemolymph. Haemolymph was collected through the arthrodial membrane of the coxa of the third pair of walking legs of the crab. To 1 ml of the haemolymph, 3 ml of diethyl ether was added. All the samples were vortexed for 30 s and kept for 2 min. The samples were then plunged in liquid nitrogen for 2 min and the upper ether phase containing the steroids was recovered. The residue was subjected to ether extraction for three more times to extract the steroids completely. The extracts were pooled and evaporated to dryness at 40 °C. The extracts of haemolymph were used for estimation of 17β-estradiol levels by competitive chemiluminescent enzyme immunoassay (CLEIA) using Immunolite 2000 Estradiol kit (Diagnostic Products Corporation, Los Angeles, CA, USA). The sensitivity or detection limit of the assay was 15 pg ml−1. Progesterone was measured using the Progesterone EIA kit (Euro-Diagnostica B.V, Arnhem, The Netherlands) following manufacturer's instructions. Samples were analyzed in duplicate. The inter-assay variation was 5.4% and 6.0% for 17β-estradiol and progesterone respectively. The sample size for determination of hormone level is relatively small, since only limited numbers of previtellogenic stage and vitellogenic stage III crabs (out of 60 sexually mature crabs collected in three instances, 5 in each group) were obtainable in collection.2.2. In vivo effect of 17β-estradiol and progesterone on ovarian maturation
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) width
width![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) height = 60
height = 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30
30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 cm) with sufficient ambient medium (salinity: 0.5 ppt) and water was replaced daily. Animals were maintained under controlled laboratory conditions (temperature 27 ± 1 °C and a light period of 12 h) for at least one week before beginning the experiments. During their sojourn, the crabs were fed with sheep meat daily ad libitum.
30 cm) with sufficient ambient medium (salinity: 0.5 ppt) and water was replaced daily. Animals were maintained under controlled laboratory conditions (temperature 27 ± 1 °C and a light period of 12 h) for at least one week before beginning the experiments. During their sojourn, the crabs were fed with sheep meat daily ad libitum.Group 1: control crabs, did not receive any treatment and were sacrificed on day 1 of experiment.
Group 2: crabs injected with 10 μl of castor oil on 1st, 7th, 14th and 21st day of experiment at the base of the coxa of the third walking leg and served as concurrent control.
Group 3: crabs injected with 17β-estradiol at a dose of 10−7 mol per crab, in 10 μl volume on 1st, 7th, 14th and 21st day of experiment at the base of the coxa of the third walking leg.
Group 4: crabs injected with and progesterone at a dose of 10−7 mol per crab, in 10 μl volume on 1st, 7th, 14th and 21st day of experiment at the base of the coxa of the third walking leg.
The dose employed here falls within the physiological concentrations reported for estradiol and progesterone in the haemolymph of the mud crab Scylla serrata35 and the prawn Marsupenaeus japonicus41 and the crayfish Procambarus clarkii.23 The crabs were sacrificed with ice anesthetization on day 28 of the experiment. The experiments were carried out in accordance with the guidelines of the Committee for the Purpose of Control and Supervision on Experiments on Animals42 and approved by the Institutional Animal Ethical Committee, Sri Venkateswara University, Tirupati (Regd. no. IAEC/No438/01/a/CPCSEA dt: 04.03.2002).
Left ovary was used for determination of vitellogenin levels (n = 10) and right ovary was used for histological studies (n = 5) and mRNA isolation (n = 5). The ovarian vitellogenin levels were determined as described by Reddy et al.22 For histological studies, the ovaries were placed in Bouin's fixative (picric acid![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) formaldehyde
formaldehyde![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) acetic acid, 75
acetic acid, 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25
25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) immediately after isolation. After 24 h, they were washed in water and dehydrated with ascending alcohol series. Dehydrated ovaries were embedded in paraffin wax after clearing in xylene, sectioned transversely at 5 μm and stained with haematoxylin and counter stained with eosin,44 the maximum and minimum diameters of up to 50 randomly selected oocytes of each sectioned ovary were measured using an ocular micrometer under a compound microscope (Olympus, Model-BX41TF HB, Japan) through the nucleus and the mean diameter was calculated.
5) immediately after isolation. After 24 h, they were washed in water and dehydrated with ascending alcohol series. Dehydrated ovaries were embedded in paraffin wax after clearing in xylene, sectioned transversely at 5 μm and stained with haematoxylin and counter stained with eosin,44 the maximum and minimum diameters of up to 50 randomly selected oocytes of each sectioned ovary were measured using an ocular micrometer under a compound microscope (Olympus, Model-BX41TF HB, Japan) through the nucleus and the mean diameter was calculated.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 ratio with coating buffer (carbonate buffer: 1.59 g sodium carbonate, 2.93 g sodium bicarbonate in 1000 ml distilled water, diethyl dithio carbomate (DIECA) 56 mg per 25 ml buffer). Blank values were obtained from wells coated with coating buffer alone. The plate is covered with a lid and placed in a humid chamber at 37 °C for 2 h. After emptying, the plate was washed five times with 0.1 M PBS containing 0.05% Tween 20, pH 7.2 (PBST). 200 μL of 1
10 ratio with coating buffer (carbonate buffer: 1.59 g sodium carbonate, 2.93 g sodium bicarbonate in 1000 ml distilled water, diethyl dithio carbomate (DIECA) 56 mg per 25 ml buffer). Blank values were obtained from wells coated with coating buffer alone. The plate is covered with a lid and placed in a humid chamber at 37 °C for 2 h. After emptying, the plate was washed five times with 0.1 M PBS containing 0.05% Tween 20, pH 7.2 (PBST). 200 μL of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000 dilution of primary antibody (antibodies raised in rabbits against Vg isolated from vitellogenic stage III ovary) in 0.1 M PBST with 2% polyvinyl pyrrolidine, 0.2% ovalbumin (PBST-PO) is added to each well. The plate is covered with a lid and placed in a humid chamber at 37 °C for 2 h. After emptying, the plate was washed 5 times with PBST. 200 μL of horseradish peroxidase (HRP) conjugated anti IgG antibody (1
1000 dilution of primary antibody (antibodies raised in rabbits against Vg isolated from vitellogenic stage III ovary) in 0.1 M PBST with 2% polyvinyl pyrrolidine, 0.2% ovalbumin (PBST-PO) is added to each well. The plate is covered with a lid and placed in a humid chamber at 37 °C for 2 h. After emptying, the plate was washed 5 times with PBST. 200 μL of horseradish peroxidase (HRP) conjugated anti IgG antibody (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000 dilution with PBST-PO) was added to each plate and kept in dark for 1 h at 37 °C. The plate was washed 5 times with PBST, and 200 μL Tetra Methyl Benzidene (TMB in 0.015% hydrogen peroxide) was added to each plate. The plate was kept in dark for 1 h at 37 °C. The reaction was stopped by adding 50 μL of 1 M phosphoric acid per well. Absorbance was measured with ELISA reader at 450 nm. Secondary antibody (anti IgG) conjugated with HRP was purchased from Genei, Bangalore. All standards and samples measurement were performed in duplicate.
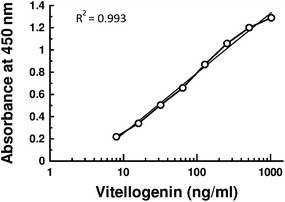
1000 dilution with PBST-PO) was added to each plate and kept in dark for 1 h at 37 °C. The plate was washed 5 times with PBST, and 200 μL Tetra Methyl Benzidene (TMB in 0.015% hydrogen peroxide) was added to each plate. The plate was kept in dark for 1 h at 37 °C. The reaction was stopped by adding 50 μL of 1 M phosphoric acid per well. Absorbance was measured with ELISA reader at 450 nm. Secondary antibody (anti IgG) conjugated with HRP was purchased from Genei, Bangalore. All standards and samples measurement were performed in duplicate.The robustness of the ELISA was assessed with the measurement of inter- and intra-assay variation, where the variation of intra-assay was measured by adding the same concentration of standard to one microtiter plate 12 times, and the inter-assay variation was measured by comparing various dilutions of the standard added to 12 different microtiter plates. The detection limit of the ELISA was calculated as the lowest standard with an absorbance at least three times higher than the standard deviation of the first value that was significantly different from the ELISA control wells. The data were analyzed with Kyplot (version 2.0). This software determines the polynominal equation that best fits the standards. Vg-ELISA assay parameters such as the range of standard curves, dilution of antibodies, matrix effect, incubation time and buffer, were investigated and optimized. After optimization of these parameters, a linear response was observed in the range from 8.0 ng purified Vg to 1.0 μg (R2 = 0.993). Sensitivity of the ELISA was as little as 5.0 ng of Vg; intra-assay variation was 4% to 6% (n = 12) and the inter-assay variation was 9% to 10.6% (n = 12). A typical standard curve using purified O. senex Vg was presented in Fig. 2.
2.3. Protein structure modeling and docking analysis
A search of the RCSB protein data bank confirmed that none of the crustacean EcR crystal structures was publically available. Homology modelling and structural validation of O. senex EcR ligand binding domain (LBD) (AGY56107.1) was carried out using SWISS MODEL workspace and RAMPAGE47,48 using EcR LBD of Bovicola ovis (PDB ID: 4OZT) as the template. Missing side chains from the finalized model was modeled by “Build/Check and Repair” tool of the WHATIF web server (http://swift.cmbi.ru.nl/servers/html/index.html). Finally, the model was verified by using the structural analysis and verification server (SAVES), a meta-server having various programs such as PROCHECK, WHATCHECK and VERIFY3D (http://nihserver.mbi.ucla.edu/SAVES). To reveal the major differences among built homology models and to assess their stability and reliability, the modeled structure was further subjected to energy assessment and structural analysis using Atomic Non-Local Environment Assessment (ANOLEA).49The constructed model was subjected to energy minimization by steepest descent, using GROMOS96 43b1 force field. The structures of ecdysteroid (CID: 5459840), estradiol (CID: 5757) and progesterone (CID: 5994) were downloaded from PubChem in SDF format and then converted into PDB format using openbabel. Docking and interaction analysis of O. senex EcR LBD were performed using PyRx virtual screening software.50 Lamarckian genetic algorithm (numbers of run, 30; population size, 150; maximum number of energy evaluations, 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000
000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000; number of generations, 27
000; number of generations, 27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000; rate of gene mutation, 0.02; rate of cross over, 0.8) was used as a search parameter. Ligand interactions with protein were visualised using Pymol.51
000; rate of gene mutation, 0.02; rate of cross over, 0.8) was used as a search parameter. Ligand interactions with protein were visualised using Pymol.51
2.4. Sequence retrieval and sequence based homology and phylogeny
The representative protein sequences retrieved from NCBI protein database were subjected to multiple sequence analysis using CLUSTALW with default settings. Estrogen related receptor (ERR) from Scylla paramamosain (ADB43256.1) is selected since it is the only ERR sequence available in genbank from crustaceans. EcR sequences from D. melanogaster, S. paramamosain, O. senex senex and H. virescens (NP_724456.1; AFN08569.1, KF319123, CAA70212.1 respectively) were taken for multiple sequence analysis, whereas EcR and ERR from crustacean S. paramamosain (AFN08659.1, ADB43256.1 respectively); insect D. melanogaster (NP_724456.1, NP_729340.1 respectively); estrogen receptor (ER) from mollusc Mytilus edulis (BAF34366.2); reptile Crocodylus niloticus (BAE45626.1); bird Columba livia (AFU48567.1); rodent Rattus norvegicus (CAA43411.1); human Homo sapiens (AEP43755.1) were randomly selected and used for phylogenetic analysis.2.5. Statistical analysis
Statistical analysis were performed by one-way analysis of variance (ANOVA) followed by Tukey's post-test using SPSS (student version 7.5, SPSS Inc., UK). Probability values of 0.05 or below were considered significant.3. Results
3.1. Determination of 17β-estradiol and progesterone levels in the haemolymph of pre-vitellogenic and vitellogenic stage III crabs
Progesterone and estrogen levels in the haemolymph of pre-vitellogenic females were 2.73 ± 0.18 ng ml−1 and 27.89 ± 6.52 pg ml−1 respectively. The circulatory levels of progesterone and estrogen were significantly (p < 0.05) higher in the haemolymph of vitellogenic stage III females when compared to pre-vitellogenic levels (Table 1).| Hormone | Pre-vitellogenic stage | Vitellogenic stage III |
|---|---|---|
| a Values are mean ± SD (n = 5). Values in the parentheses are fold increase from pre-vitellogenic stage. *p < 0.05. | ||
| 17β-Estradiol (pg ml−1) | 27.89 ± 6.52 | 134.23* ± 17.37 (4.81) |
| Progesterone (ng ml−1) | 2.73 ± 0.18 | 11.89* ± 2.34 (4.36) |
3.2. Effect of 17β-estradiol and progesterone on ovarian index and oocyte diameter
The ovarian index and oocyte diameter in control and concurrent control crabs were 0.39 ± 0.01 (w/w%), 20.18 ± 5.71 μm and 0.382 ± 0.02 (w/w%), and 21.13 ± 4.21 μm, respectively (Table 2). The ovarian index or oocyte diameter in both control and concurrent control crabs were not significantly different in a 28-day experimental period. Administration of 17β-estradiol and progesterone resulted in a significant increase (p < 0.001) in the ovarian index and oocyte diameter in crabs when compared with control crabs. The ovarian index were 3.90 and 3.92, and oocyte diameters were 3.71 and 4.17 fold more in group 3 and group 4 crabs respectively when compared with the controls.| Parameters | Control | Concurrent control | Estradiol injected | Progesterone injected |
|---|---|---|---|---|
| a Values are mean ± SD of 10 individuals unless mentioned.b Values represent mean of 50 randomly selected oocytes of each sectioned ovary from 5 crabs.c Values in the parentheses are fold change from control.d p < 0.05. | ||||
| Ovarian index (w/w%) | 0.39 ± 0.01 | 0.38 ± 0.02 (0.98) | 1.52d ± 0.4 (3.90) | 1.53d ± 0.45 (3.92) |
| Oocyte diameterb (μm) | 20.18 ± 5.71 | 21.13 ± 4.21 (1.05) | 74.76d ± 6.76 (3.71) | 84.20d ± 7.95 (4.17) |
| Vg levels (mg per g tissue) | 0.069 ± 0.003 | 0.070 ± 0.004 (1.01) | 0.357d ± 0.004 (5.17) | 0.188d ± 0.01 (2.72) |
3.3. Effect of 17β-estradiol and progesterone on ovarian Vg levels
The ovarian Vg levels were 0.069 ± 0.003 and 0.07 ± 0.004 mg per g tissue in the control and concurrent control crabs. Vg levels in the ovaries of crabs administered with 17β-estradiol and progesterone were significantly (p < 0.05) higher than the concurrent control group (Table 2).3.4. Effect of 17β-estradiol and progesterone on histological changes in the ovaries
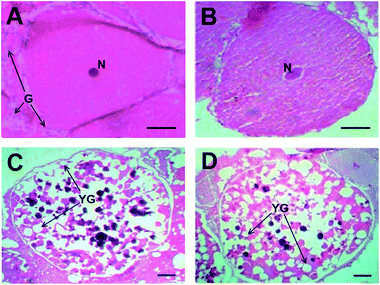
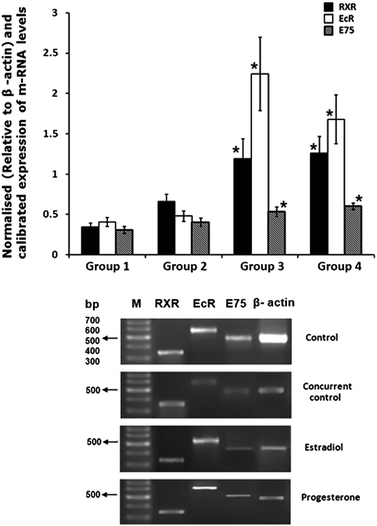
Transverse section of ovary from concurrent control females revealed that the ovaries were in pre-vitellogenic stage with thick ovarian wall and centrally located germanium (Fig. 3A), whereas the ovaries from 17β-estradiol and progesterone injected crabs were in vitellogenic stage-III as evidenced by accumulation of yolk globules in the entire oocyte (Fig. 3B and C).3.5. Effect of 17β-estradiol and progesterone on RXR, EcR, E75 and Vg mRNA levels in hepatopancreas and ovary
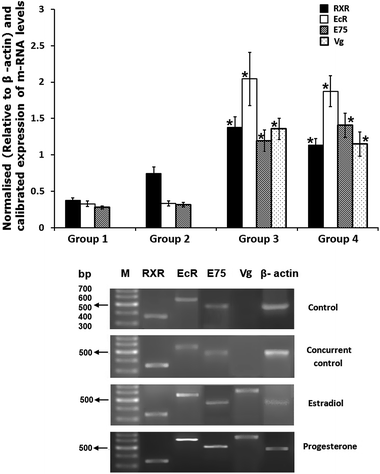
Significant increase in RXR, EcR and E75 mRNAs was observed in hepatopancreas (Fig. 4) and ovary (Fig. 5) of experimental crabs when compared to concurrent controls. Vg mRNA levels were detected in the hepatopancreas of both experimental groups but not in the hepatopancreas of concurrent control crabs (Fig. 4).3.6. Modelling and validation of LBD of EcR
3D model generated by SWISS Workspace is validated by using PROCHECK, WHATCHECK and VERIFY3D. Ramachandran plot drawn through PROCHECK program validated the model with 98% of the total residues in the allowed region and 2.0% in the generously allowed region. None of the residues were located in the disallowed region confirming that the protein backbone dihedral angles phi (Φ) and psi (Ψ) occupied reasonably accurate positions in the selected 3D model.3.7. Ecdysone, 17β-estradiol and progesterone interaction with EcR
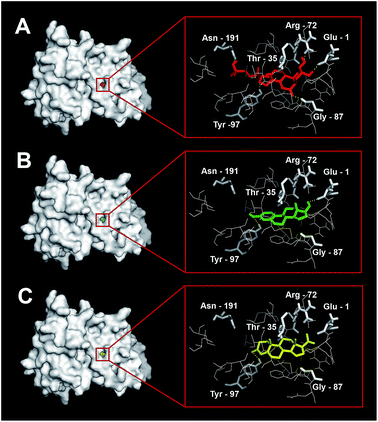
Amino acids interacting with ecdysteroid, 17β-estradiol and progesterone were presented in Table 3 and Fig. 6. The binding affinity of progesterone (−10.5 kcal mol−1) to simulated EcR-LBD was greater than 17β-estradiol (9.9 kcal mol−1) whereas the binding affinity of natural ligand, ecdysteroid (−10.6 kcal mol−1) was comparable with the binding affinity of progesterone. Progesterone and 17β-estradiol interact with Glu1, Thr35, Arg72, Gly87, Tyr97 and Asn191 at the binding site of EcR-LBD, which are also involved in binding of ecdysteroid (Fig. 6).| Hormone | Binding affinity | Interacting amino acids |
|---|---|---|
| Ecdysteroid | −10.6 kcal mol−1 | Glu1, Thr35, Arg72, Gly87, Tyr97, Asn191 |
| 17β-Estradiol | −9.9 kcal mol−1 | Glu1, Tyr97 |
| Progesterone | −10.5 kcal mol−1 | Tyr97 |
3.8. Phylogenetic analysis of EcR and ERR
To detect the regions of variability or conservation among EcR and ERR in the arthropods, a multiple sequence alignment was performed. The phylogenetic analysis also showed a clear demarcation between different groups and early branching of EcR protein (Fig. 7).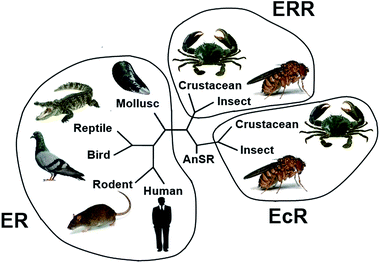 | ||
| Fig. 7 Phylogenetic tree of EcR, ERR and ER from selected organisms. AnSR: ancestral steroid receptor; EcR: ecdysteroid receptor; ERR: estrogen related receptor; ER: estrogen receptor. | ||
4. Discussion
The ovarian index, oocyte diameter and ovarian Vg levels increased significantly in crabs injected with 17β-estradiol and progesterone. Considering the above and the fact that the 17β-estradiol and progesterone were detected in the haemolymph of the crabs and the levels of these hormones increased during vitellogenesis, it seems likely that the ovarian growth and maturation in the crab is under the control of steroid hormones.It is well established that vertebrate steroids have been shown to induce significant oocyte growth in several crustaceans.26,27,33,52 A direct stimulatory effect of 17α-hydroxyprogesterone and progesterone on ovarian growth and ovarian Vg synthesis in the freshwater crab O. senex22 and freshwater crayfish Cherax albidus38 has been demonstrated. Recently we have demonstrated that hepatopancreas, but not ovary is the source of Vg in the crab O. senex.45 Hepatopancreas explants of the shrimp Metapenaeus ensis incubated in vitro with steroid hormones demonstrated that both 17β-estradiol and progesterone stimulated Vg gene expression, although 17β-estradiol was more effective.53,54
In the present study, though the levels of ovarian Vg and Vg mRNA levels in hepatopancreas increased significantly in experimental crabs; and more pronounced in 17β-estradiol injected crabs when compared to progesterone injected crabs. Coccia et al.38 reported that both 17β-estradiol and progesterone caused an increase in Vg expression, although co-administration were not synergic in Cherax albidus and hypothesized that, progesterone induces an increase of Vg synthesis while 17β-estradiol induces an increase of gene transcription and/or stabilizes immature mRNA, regulating its translation.
To determine the mechanism of action of 17β-estradiol and progesterone, we performed docking studies between EcR-LBD protein and 17β-estradiol and progesterone. It is evident from the present study that progesterone possesses comparable binding affinity when compared with natural ligand, ecdysteroid. From the data, it is also evident that 17β-estradiol and progesterone interacts at the binding site of EcR-LBD similar to ecdysteroid. EcR-LBD amino acids interacting with ecdysteroid observed in the present study are similar to those reported in Metopograpsus messor.55
Nagaraju et al.56 demonstrated that silencing of RXR results in decreased expression of vitellogenin and hypothesised that RXR heterodimerizes with EcR to bring Vg expression in Carcinus maenas. Further, direct evidence for heterodimerization of RXR and EcR in Uca pugilator was demonstrated recently.57 Hopkins et al.58 also showed that loading of MF onto RXR facilitates binding of EcR to RXR. In the present study, increased co-expression of RXR, E75, Vg with EcR in hepatopancreas shows inter-relation among these genes in bringing out vitellogenesis. In O. senex, hepatopancreas is the exclusive site for vitellogenesis.45 Increased expression of EcR with RXR and E75 in ovary indicates their role in the regulation of other functions (such as vitellogenin uptake, morphogenesis etc.). In the present study, increased expression of EcR and its partner RXR along with its primary target E75 in the tissues of experimental crabs indicates that vertebrate steroids may act through EcR.
Nuclear receptors are ancient ubiquitous proteins that have important functions in regulating many aspects of metazoan physiology. Genetic and phylogenetic analyses suggest that vertebrate steroid receptors arose from a common ancestral gene in deuterostomes59–62 similar to an estrogen receptor.39,62 This supposition is supported by the fact that no androgen- or progestin-like receptors have been found in invertebrates63 though estrogen related receptor is reported in the mollusc Aplysia californica.64 Estrogen receptors have not been reported in crustaceans, although nuclear receptors sharing high amino acid similarity with estrogen receptors have been discovered in Drosophila.64
It is generally believed that sex steroid receptors have been lost in the lineage leading to arthropods64 since there is no ortholog of these genes in the genome of the insect, Drosophila melanogaster64 and the amphipod Hyalella azteca.39 Maglich et al.65 identified estrogen related receptor (ERR) in Drosophila which shares high amino acid sequence homology with human ER but not ortholog. The ERR in D. melanogaster is considered as a pseudo gene.65 ERRs have not been reported in any crustaceans except in the mud crab Scylla paramamosain (NCBI accession number: ADB43256.1, direct submission). Kohler et al.39 identified an estrogen receptor α (ERα)-like protein in the amphipod, Gammarus fossarum but its regulation by estrogens and involvement in estrogen signal transduction remains unclear. However, the ancient origin of estrogen signaling and the cited reports on physiological action of sex steroids in crustaceans suggest that possibly some ecdysozoans have retained sex steroid receptors during their evolution.
On the other hand, binding proteins for vertebrate steroids have been described in several species, including nematodes, echinoderms, crustaceans, and molluscs66–71 but are not coupled to a biological response. The only steroid receptor present in arthropods, is ecdysteroid receptor, which is also belong to a nuclear receptor clade that seem to have branched-off from the common ancestral deuterostome receptor protein58,72 from which the estrogen receptor evolved. Accordingly we hypothesize that 17β-estradiol and progesterone may render their action through EcR in crustaceans, a possible non-ER mediated signalling. The in silico docking experiments of the present study also supported this hypothesis, revealing that EcR in arthropods is not terminally evolved and retained its plasticity to accommodate the action of vertebrate steroids viz., 17β-estradiol and progesterone.
5. Conclusion
The present study indicates that 17β-estradiol and progesterone not only detected in the haemolymph of crab and their levels elevated in circulation during vitellogenesis. Administration of 17β-estradiol and progesterone induced vitellogenesis and also increased oocyte diameter, ovarian index and ovarian vitellogenin levels. In silico studies revealed interaction between EcR and sex steroids suggesting the mediation of EcR in inducing vitellogenesis. In support to this, the mRNA levels of RXR, EcR, E75 and Vg in hepatopancreas and RXR, EcR and E75 in ovary also increased after 17β-estradiol and progesterone administration. This also indicates the involvement RXR and EcR in the regulating reproduction in crustaceans. Piecing these evidences together, the present data demonstrate that 17β-estradiol and progesterone, well-established steroid hormones with known importance in mammalian reproduction, are also important molecules in signaling the ovarian maturation in crustaceans. Besides, this study provides evidence to support the ancient origin of steroid hormonal regulation and suggests that endocrine regulation of mammalian reproduction may be derived from primitive regulatory pathways. It is obviously a successful system in an evolutionary sense, since arthropods appeared on earth several hundred millions of years before mammals, and the ancestors of both groups diverged at least 100 million years before that.6. Live subject statement
The experiments were conducted in accordance with CPCSEA and approved by Institutional Animal Ethical Committee, Sri Venkateswara University, India. The authors declare that the experiments conducted during these studies comply with the current laws of their country.Acknowledgements
C. H. Swetha and B. P. Girish are highly thankful to University Grants Commission (BSR-RFSMS) and Council of Scientific and Industrial Research (CSIR-JRF) respectively, for providing financial support in the form of research fellowship. This work is also supported by grants from DST, New Delhi to PSR.References
- J. Sandor and A. Z. Mehdi, in Hormones and Evolution, ed. E. J. Barrington, Academic Press, NewYork, 1979, pp. 1–72 Search PubMed.
- V. Gunamalai, R. Kirubagaran and T. Subramoniam, Gen. Comp. Endocrinol., 2004, 138, 128–138 CrossRef CAS PubMed.
- T. Okumura, C. H. Han, Y. Suzuki, K. Aida and I. Hanyu, Zool. Sci., 1992, 9, 37–45 CAS.
- M. N. Wilder, T. Okumura and K. Aida, Zool. Sci., 1991, 8, 919–927 CAS.
- B. G. Burns, G. B. Sangalang, H. C. Freeman and M. McMenemy, Gen. Comp. Endocrinol., 1984, 54, 422–428 CrossRef CAS PubMed.
- E. F. Couch, N. Hagino and J. W. Lee, Comp. Biochem. Physiol., Part A: Mol. Integr. Physiol., 1987, 87, 765–770 CrossRef.
- N. J. Fairs, R. P. Evershed, P. T. Quinlan and L. J. Goad, Gen. Comp. Endocrinol., 1989, 4, 199–208 CrossRef.
- M. Fingerman, R. Nagabhushanam and R. Sarojini, Zool. Sci., 1993, 18, 13–29 Search PubMed.
- M. Oetken, J. Bachmann, U. Schulte-Oehlmann and J. Oehlmann, Int. Rev. Cytol., 2004, 236, 1–44 CrossRef CAS PubMed.
- E. T. Quinitio, K. Yamauchi, A. Hara and A. Fuji, Gen. Comp. Endocrinol., 1991, 81, 343–348 CrossRef CAS PubMed.
- T. Subramoniam, Comp. Biochem. Physiol., 2000, 125, 135–156 CAS.
- B. Tsukimura, Am. Zool., 2001, 41, 465–476 CAS.
- E. Van Beek and A. De Loof, Comp. Biochem. Physiol., Part A: Mol. Integr. Physiol., 1988, 89, 595–599 CrossRef.
- M. N. Wilder, T. Subramoniam and K. Aida, in Reproductive Biology of Invertebrates ed. A. S. Raikhel and T. W. Sappington, Science Publishers Inc., Plymouth, UK, 2002, pp. 131–174 Search PubMed.
- W. S. Baldwin and G. A. LeBlanc, Environ. Toxicol. Chem., 1994, 13, 1013–1021 CrossRef CAS.
- M. F. Blanchet, R. Ozon and J. J. Meusy, Comp. Biochem. Physiol., Part B: Biochem. Mol. Biol., 1972, 41, 251–261 CrossRef CAS.
- D. Ghosh and A. K. Ray, Gen. Comp. Endocrinol., 1993, 89, 248–254 CrossRef CAS PubMed.
- A. Kanazawa and S. I. Teshima, Bull. Jpn. Soc. Sci. Fish., 1971, 37, 891–898 CrossRef CAS.
- E. Oberdorster, D. Rittschof and P. McClellan-Green, Mar. Pollut. Bull., 1998, 36, 144–151 CrossRef CAS.
- L. Swevers, J. G. D. Lambert and A. De Loof, Comp. Biochem. Physiol., Part B: Biochem. Mol. Biol., 1991, 99, 35–41 CrossRef.
- T. Verslycke, K. De Wasch, H. F. De Brabander and C. Jansen, Gen. Comp. Endocrinol., 2002, 126, 190–199 CrossRef CAS PubMed.
- P. R. Reddy, P. Kiranmayi, K. T. Kumari and P. S. Reddy, Aquaculture, 2006, 254, 768–775 CrossRef CAS.
- E. M. Rodriguez, D. A. Medesani, L. L. Greco and M. Fingerman, J. Exp. Zool., 2002, 292, 82–87 CrossRef CAS PubMed.
- R. Sarojini, M. S. Mirajkar and R. Nagabhushanam, Comp. Physiol. Ecol., 1985, 10, 7–11 CAS.
- R. Sarojini, K. Jayalakshmi and S. Sambashivarao, J. Adv. Zool., 1986, 7, 50–53 CAS.
- I. Yano, Aquaculture, 1985, 47, 223–229 CrossRef.
- I. Yano, Aquaculture, 1987, 61, 49–57 CrossRef CAS.
- G. K. Kulkarni, R. Nagabhushanam and P. K. Joshi, Indian J. Exp. Biol., 1979, 17, 986–987 CAS.
- L. S. Quackenbush and L. L. Keeley, Am. Zool., 1987, 26, 810 Search PubMed.
- L. S. Quackenbush and L. L. Keeley, Biol. Bull., 1988, 175, 321–331 CrossRef CAS.
- L. S. Quackenbush, Comp. Biochem. Physiol., Part A: Mol. Integr. Physiol., 1992, 103, 711–714 CrossRef.
- E. T. Quinitio, A. Hara, K. Yamauchi and S. Nakao, Comp. Biochem. Physiol., Part C: Pharmacol., Toxicol. Endocrinol., 1994, 109, 21–26 CrossRef.
- I. Yano and N. Itakura, Proceedings and Abstracts of Fourth International Crustacean Congress, Amsterdam, 1998, vol. 81, p. 64 Search PubMed.
- J. T. Shih, Zool. Stud., 1997, 36, 136–145 CAS.
- S. R. Warrier, R. Tirumalai and T. Subramoniam, Comp. Biochem. Physiol., Part A: Mol. Integr. Physiol., 2001, 130, 283–294 CrossRef CAS.
- V. Zapata, L. S. L. Greco, D. Medesani and E. M. Rodriguez, Aquaculture, 2003, 224, 339–352 CrossRef CAS.
- I. Yano, in Reproductive Biology of Invertebrates, ed. K. G. Adiyodi and R. G. Adiyodi, Wiley, New York, 2000, pp. 161–194 Search PubMed.
- E. Coccia, E. De Lisa, C. Di Cristo, A. Di Cosmo and M. Paolucci, Biol. Bull., 2010, 218, 36–47 CAS.
- H. R. Kohler, W. Kloas, M. Schirling, I. Lutz, A. L. Reye, J. S. Langen, R. Triebskorn, R. Nagel and G. Schonfelder, Ecotoxicology, 2007, 16, 131–143 CrossRef PubMed.
- C. H. Swetha, B. P. Girish and P. Sreenivasula Reddy, J. Aquacult. Res. Dev., 2015, 6, 7 Search PubMed.
- T. Okumura and K. Sakiyama, Fish. Sci., 2004, 70, 372–380 CrossRef CAS.
- CPCSEA, Indian J. Pharmacol., 2003, 35, 257–274 Search PubMed.
- A. V. Van Harreveld, Proc. Soc. Exp. Biol. Med., 1936, 34, 408–432 Search PubMed.
- J. D. Bancroft and A. Stevens, Theory and Practice of Histological Techniques, Churchill Livingstone, New York, 2nd edn, 1982 Search PubMed.
- B. P. Girish, C. H. Swetha and P. S. Reddy, Biochem. Biophys. Res. Commun., 2014, 447, 323–327 CrossRef CAS PubMed.
- B. P. Girish, C. H. Swetha and P. S. Reddy, Science of Nature, 2015, 102, 20 CrossRef CAS PubMed.
- S. C. Lovell, I. W. Davis, W. B. Arendall, P. I. de Bakker, J. M. Word, M. G. Prisant, J. S. Richardson and D. C. Richardson, Proteins, 2002, 50, 437–450 CrossRef PubMed.
- L. Bordoli, F. Kiefer and K. Arnold, Nat. Protoc., 2009, 4, 1–13 CrossRef CAS PubMed.
- F. Melo and E. Feytmans, J. Mol. Biol., 1998, 277, 1141–1152 CrossRef CAS PubMed.
- O. Trott and A. J. Olson, J. Comput. Chem., 2010, 31, 455–461 CAS.
- W. L. Delano, The PyMOL Molecular Graphics System, DeLano Scientific, San Carlos, CA, USA, 2002 Search PubMed.
- R. Nagabhushanam, P. K. Joshi and G. K. Kulkarni, Indian J. Mar. Sci., 1980, 9, 227 Search PubMed.
- S. H. K. Tiu, J. G. He, S. S. Tobe and S. M. Chan, Mol. Reprod. Dev., 2006, 73, 424–436 CrossRef CAS PubMed.
- I. Yano and R. Hoshino, Comp. Biochem. Physiol., Part A: Mol. Integr. Physiol., 2006, 144, 18–23 CrossRef PubMed.
- S. Shyamal, G. Anilkumar, R. Bhaskaran, G. P. Doss and D. S. Durica, Gen. Comp. Endocrinol., 2015, 211, 39–51 CrossRef CAS PubMed.
- G. P. C. Nagaraju, B. Rajitha and D. W. Borst, J. Endocrinol., 2011, 210, 379–390 CrossRef CAS PubMed.
- S. Das and D. S. Durrica, Mol. Cell. Endocrinol., 2013, 365, 249–259 CrossRef CAS PubMed.
- P. M. Hopkins, D. Durica and W. Tracy, Comp. Biochem. Physiol., Part A: Mol. Integr. Physiol., 2008, 151, 602–614 CrossRef PubMed.
- M. E. Baker, Mol. Cell. Endocrinol., 1997, 135, 101–107 CrossRef CAS PubMed.
- M. E. Baker, J. Mol. Endocrinol., 2001, 26, 119–125 CrossRef CAS PubMed.
- H. Escriva, R. Safi, C. Hanni, M. C. Langlois, P. Saumitou-Laprade, D. Stehelin, A. Capron, R. Pierce and V. Laudet, Proc. Natl. Acad. Sci. U. S. A., 1997, 94, 6803–6808 CrossRef CAS.
- H. Escriva, F. Delaunay and V. Laudet, Bioassays, 2000, 22, 717–727 CrossRef CAS.
- M. E. Baker, Bioassays, 2003, 25, 396–400 CrossRef CAS PubMed.
- J. W. Thornton, E. Need and D. Crews, Science, 2003, 301, 1714–1717 CrossRef CAS PubMed.
- J. M. Maglich, A. Sluder, X. Guan, Y. Shi, D. D. McKee, K. Carrick, K. Kamdar, T. M. Willson and J. T. Moore, Genome Biol., 2001, 2, 29.1–29.7 CrossRef.
- M. De Waal, J. Portman and P. A. Voogt, Mar. Biol. Lett., 1982, 3, 317–323 CAS.
- A. Di Cosmo, M. Paolucci, C. Di Cristo, V. Botte and G. Ciarcia, Mol. Reprod. Dev., 1998, 50, 451–460 CrossRef CAS PubMed.
- A. Di Cosmo, C. Di Cristo and M. Paolucci, Mol. Reprod. Dev., 2002, 61, 367–375 CrossRef CAS PubMed.
- B. Keshan and A. K. Ray, Gen. Comp. Endocrinol., 2001, 123, 23–30 CrossRef CAS PubMed.
- C. S. Kiser, E. J. Parish and L. W. Bone, Comp. Biochem. Physiol., Part B: Biochem. Mol. Biol., 1986, 83, 787–790 CrossRef.
- G. Paesen and A. De Loof, Invertebr. Reprod. Dev., 1989, 14, 267–277 CrossRef.
- V. Laudet, J. Mol. Endocrinol., 1997, 19, 207–226 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra23637a |
| ‡ The authors wish it to be known that, in their opinion, the first two authors should be regarded as joint first authors. |
| This journal is © The Royal Society of Chemistry 2016 |