Scalable simultaneous activation and separation of metal–organic frameworks†
Marta Rubio-Martinez*a,
Thomas Leongbc,
Pablo Julianob,
Trevor D. Hadleya,
Michael P. Battena,
Anastasios Polyzosa,
Kok-Seng Lima and
Matthew R. Hill*a
aCSIRO Mineral Resources, Manufacturing and Energy, Private Bag 33, Clayton South MDC, VIC 3169, Australia. E-mail: marta.rubio@csiro.au; matthew.hill@csiro.au
bCSIRO Food and Nutrition, 671 Sneydes Rd, Werribee, VIC 3030, Australia
cDepartment of Mechanical and Product Design Engineering, Faculty of Science, Engineering and Technology, Swinburne University of Technology, Hawthorn, VIC 3122, Australia
First published on 6th January 2016
Abstract
Despite the many promising applications of Metal–Organic Frameworks (MOFs) the key advances to boost production to industrial scale still remain elusive. Recently, it has been shown that continuous flow chemistry is capable of handling the production of a large variety of MOFs, while also being scalable. However commonly used laboratory methods for postproduction processing, i.e. separation and activation are ineffective and costly at scale. Here we present the use of megasonics, a novel and scalable methodology based on sonication with high frequency sound waves, which performs separation and activation simultaneously, yielding high surface areas that previously were only obtainable with laboratory-scale methods like solvent exchange, supercritrics and calcination.
Metal–Organic Frameworks (MOFs) are one of the most promising porous materials, based on their tuneable functionality, large pore sizes and highest known surface areas.1,2 These characteristics are of high interest for a myriad of applications such as gas storage,3,4 automotive applications,5 carbon capture,6 gas separation,7 drug delivery8 and catalysis.9 Furthermore, they have adsorption properties suitable for dramatically increasing the performance of heat exchange systems, for example, in heat sinks for cooling processes in computing applications.10–12 Consequently, in recent years MOFs as smart materials have experienced an exponential growth of reported publications, as many researchers have focused their efforts on the challenge of developing novel types with new or improved functionalities for specific applications.
However, there are several major obstacles that need to be overcome in order to make these materials viable in commercial scale applications. Large amounts of MOF materials have to be synthesized (kg scale or higher) whilst maintaining the desired product quality i.e. porosity, thermal and chemical stability. This has proven to be difficult on a number of levels. For example, the up-scaling of production capacity using traditional laboratory routes such as the classical hydro(solvo) thermal synthesis, electrochemical or mechanochemical synthesis, is challenging due to long reaction times and low quality materials, caused by different nucleation mechanisms arising from the increment of reaction vessel surfaces.13,14 Furthermore, the large variety of MOFs gives rise to a myriad of potential applications, but also creates an additional obstacle for large scale production. A versatile production method, which can accommodate most of the different synthetic approaches used at the laboratory scale, is required to limit capital costs. Novel approaches like microwave synthesis and flow chemistry could provide solutions to these problems, promising high quality materials with impressive improvement in the Space Time Yields (STY) compared to commercial batch approaches.15,16 To date, continuous flow chemistry is the most promising for large scale production, as it is a paradigm-shifting approach to chemical synthesis, offering continuous production and scalability following green chemistry principles such as recycling of solvents and water-based synthesis.17 Additionally, its large surface to volume ratio and the improved heat and mass transfer available lead to speedy syntheses and new synthetic pathways. Early reports have indicated that it is possible for MOFs to be produced by continuous flow processes, using counter-current mixing,18 microfluidic reactors,19,20 and mesoscale flow production.21 These promising steps towards MOF production at scale have been confirmed by our recent findings that flow chemistry is not only able to produce pure MOFs at a larger scale of kg per day without loss in product quality at drastically reduced reaction times (days to minutes), but also provides the crucial versatility needed given the wide array of known MOFs, and the likelihood of a large range of applications each requiring different MOFs in the future.22
Despite these promising advances, there are still several problems associated with the production process that need to be addressed before the large scale production of MOF materials based on flow chemistry can be achieved. On one hand there is the amount or type of solvent and reactants required by such operations which can be cost prohibitive or cause significant environmental impact. The solvothermal method of producing 11.4 g of aluminium fumarate MOF involves 550 mL of DMF, 300 mL of acetone and 200 mL of methanol.23 On the other hand, the post-synthesis steps to obtain the final product are not trivially solved and can substantially influence product quality and increase the overall cost of the process. In particular, the separation of the small crystals from the mother liquid remains one of the challenges in the production process of MOFs at a larger scale. There are many well established type of equipment for solid–liquid separation such as centrifuges, cyclones, settling chambers, classifiers or filters, in addition to the direct evaporation of the mother liquor. However, the small size of the MOF particles, their low concentration in the solvent, as well as their density approaching that of the solvent (due to the high porosity), makes separation via most conventional methods unfeasible, inefficient or expensive at an industrial scale.24
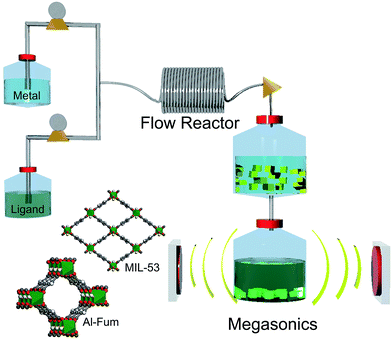
In this communication we address these issues by presenting for the first time the use of megasonics as an alternative strategy for the simultaneous separation and activation of MOF crystals.25–27 Its operating principle is based on the application of high frequency ultrasound to the MOF solution, leading to the separation of the solid MOF particles from the solvent, with an additional improvement of the surface area of the final product with simultaneous removal in occluded reagents, hereby avoiding the time consuming and costly calcination traditionally used in the MIL series. The combination of megasonics and flow chemistry, two cost effective and scalable techniques, with a water-based synthesis is an ideal candidate to elevate MOF production to an industrial scale (Fig. 1).28,29
To demonstrate the feasibility of this approach, we chose two aluminium MOFs which exhibit high thermal stability up to 450 °C and present a reversible uptake/release of water provided by an octahedral aluminium configuration and a strong Al–O bond: aluminium fumarate (Al-fum) and aluminium terephthalate (MIL-53).30,31 Both of these MOFs present very similar structures where the carboxylate groups of the corresponding linkers lead to the formation of a 3D structure with rhombohedral channels interconnected by infinite Al–OH–Al chains. For these materials several attractive applications have been proposed; gas separation, H2 or CH4 storage.32,33 To implement an environmentally friendly and industrially viable synthetic process, we use water as a solvent instead of toxic solvents such as N,N′-dimethylformamide (DMF) commonly used in most MOF synthesis. The strategy for this water-based synthesis consists in the addition of a base (NaOH) to solubilise the ligands, fumaric and terephthalic acid respectively, in the aqueous media, thus promoting a fast coordination between the metal ions with the carboxylic acids of the organic ligands.
The synthetic process commenced by pumping two separate precursor solutions, the organic ligand and the metallic salt, simultaneously through a T-type static mixer into a heated tubular flow reactor using 1.2 min of reaction time (see details in ESI 1 and ESI 2†). To isolate the MOF crystals from the reaction stream megasonics were employed, based on the application of high frequency ultrasound (2 MHz) to create a standing wave, i.e. regions of minimal pressure (nodes) and maximal pressure (antinodes) within a separation chamber (see Fig. 2c). Suspended particles migrated specifically towards these two regions due to local acoustic radiation forces, based on their density, size and compressibility.34,35
 | ||
| Fig. 2 (a) The ultrasonic reactor set-up with a high frequency system using one 2 MHz plate transducer shown in (b). (c) Schematic of the standing wave pattern formed by the superimposition of a reflected sound wave. The separation distance between adjacent nodes or antinodes, is half a wavelength. Depending on the specific density and compressibility of the particles, they will collect either in the nodal (top, black dotted planes) as for the bright yellow particles or antinodal (bottom, red dotted planes) pressure planes as for the darker yellow particles. Reprinted with permission from the work of Leong et al. Copyright 2013 ACS.34 (d) In the left compartment the megasonic separation and purification process of the Al-MOF is shown at specific times during the process. The right compartment shows the same MOF solution without sonication. The settling of the MOF is clearly visible on the left. | ||
This gathering of MOF material enhanced the tendency to form larger aggregates, which then settle at a greatly accelerated rate to the bottom of the separation chamber, where they were collected (see Fig. 2d). In the production rig, a 1.1 L stainless steel container was used for the separation and sonication at a frequency of 2 MHz (305 W) for 10 min which lead to the settling of the MOF crystals at the bottom of the vessel (see Fig. S9 and 10†). The precise mechanism may be related to changes in the local density that the aggregates experience due to varying size distribution of the nanosized MOF crystals.36 Generally, the influence of ultrasound on suspended particles depends on particle size, density and ultrasonic field. However, the resultant separation can be further influenced by possible interactions between MOF particles when they collide (i.e. surface properties). The solvent properties will also influence the specific density of the particles in the field, so that may also affect the separation efficiency as well. For the MOFs structures and solvent studied in this article no appreciable differences were observed. Recently, Lester and co-workers reported the use of ultrasound to activate ZIF-8 obtaining a similar SA reported previously in literature using solvent exchange method.37 On the other hand, megasonic separation has recently been reported to separate immiscible liquid droplets successfully for food applications such as milk fat or palm oil separation with no significant change to the physical nor chemical integrity of the product.38 To the best of our knowledge, no previous study has used megasonics, for the simultaneous separation and activation of MOF materials.
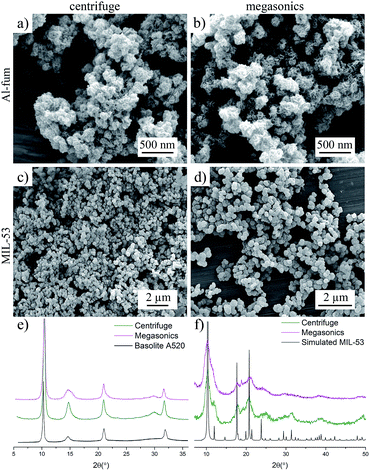
To determine the quality of the crystals, the MOFs separated with megasonics and the standard lab-scale centrifuge were compared by XRPD and SEM measurements. X-Ray powder diffraction (XRPD) confirmed the crystallinity of Al-fum and MIL-53, showing identical patterns to those of crystals synthesized by solvothermal methods. Note that the megasonics separation had no impact on the crystallinity of the materials as demonstrated by identical pattern diffraction (see Fig. 3). From the SEM images it was observed that the high-frequency treatment did not affect the size and shape distribution of the MOFs (see Fig. 3). The thermo gravimetric analysis (TGA) curves showed a continuous weight loss over the temperatures ranges 50 to 100 °C due to water loss and thermal stability up to 450 °C (see Fig. S5†).
ζ-Potentials recorded before and after each processing step (see ESI Table 1†), showed no significant changes to the surface charge, indicating separation based on reversible aggregation. However, the recoverable MOF yield obtained with megasonic separation compared to the conventional centrifuge method was 3% less for each step (see ESI Table 2† and Fig. 3). This difference could be attributed to the fact that centrifuge separation generated a higher G-force compared to settling by gravity in megasonics, leading to a more effective separation of the MOF material. The measurements of the BET surface areas revealed that the MOFs separated and activated with megasonics showed a drastic increase of 21% for the Al-fum and 47% for MIL-53 over standard centrifuge washed MOF, which had BET values similar to literature (see ESI Table 2, Fig. S12† and 4). We attribute this improvement to the enhanced mass transfer that arose from acoustic streaming during megasonic application promoting the removal of excess organic ligands molecules from the pores of the MOF crystals.
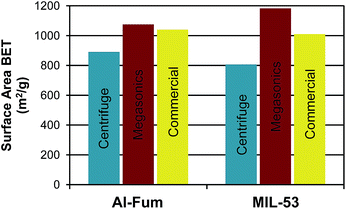 | ||
| Fig. 4 Comparison of the BET surface areas obtained with the different separation methods and that of the commercially available MOF from ref. 39 and 40. Note that all samples were activated with the same procedure. | ||
We have presented the combination of continuous flow chemistry and megasonics as a fast and cost-effective strategy to produce high-quality Al based MOF materials. The methods employed for synthesis, separation and activation are readily scalable and thus capable of producing commercially usable product at large scale, allowing the commercialisation of MOFs for many real world applications.
Notes and references
- S. Kitagawa, R. Kitaura and S. Noro, Angew. Chem., Int. Ed., 2004, 43, 2334–2375 CrossRef CAS PubMed.
- G. Férey, Chem. Soc. Rev., 2007, 37, 191–214 RSC.
- J.-R. Li, Y. Ma, M. C. McCarthy, J. Sculley, J. Yu, H.-K. Jeong, P. B. Balbuena and H.-C. Zhou, Coord. Chem. Rev., 2011, 255, 1791–1823 CrossRef CAS.
- Y. Peng, V. Krungleviciute, I. Eryazici, J. T. Hupp, O. K. Farha and T. Yildirim, J. Am. Chem. Soc., 2013, 135, 11887–11894 CrossRef CAS PubMed.
- D. Feng, K. Wang, Z. Wei, Y.-P. Chen, C. M. Simon, R. K. Arvapally, R. L. Martin, M. Bosch, T.-F. Liu, S. Fordham, D. Yuan, M. A. Omary, M. Haranczyk, B. Smit and H.-C. Zhou, Nat. Commun., 2014, 5, 5723–5731 CrossRef CAS PubMed.
- K. Sumida, D. L. Rogow, J. A. Mason, T. M. McDonald, E. D. Bloch, Z. R. Herm, T.-H. Bae and J. R. Long, Chem. Rev., 2012, 112, 724–781 CrossRef CAS PubMed.
- A. W. Thornton, D. Dubbeldam, M. S. Liu, B. P. Ladewig, A. J. Hill and M. R. Hill, Energy Environ. Sci., 2012, 5, 7637–7646 CAS.
- C. He, K. Lu and W. Lin, J. Am. Chem. Soc., 2014, 136, 12253–12256 CrossRef CAS PubMed.
- D. Farrusseng, S. Aguado and C. Pinel, Angew. Chem., Int. Ed., 2009, 48, 7502–7513 CrossRef CAS PubMed.
- H. V. R. Annapureddy, R. K. Motkuri, P. T. M. Nguyen, T. B. Truong, P. K. Thallapally, B. P. McGrail and L. X. Dang, Mol. Simul., 2014, 40, 571–584 CrossRef CAS.
- S. K. Henninger, F. Jeremias, H. Kummer and C. Janiak, Eur. J. Inorg. Chem., 2012, 2625–2634 CrossRef CAS.
- S. K. Henninger, H. A. Habib and C. Janiak, J. Am. Chem. Soc., 2009, 131, 2776–2777 CrossRef CAS PubMed.
- R. Ameloot, E. Gobechiya, H. Uji-i, J. A. Martens, J. Hofkens, L. Alaerts, B. F. Sels and D. E. de Vos, Adv. Mater., 2010, 22, 2685–2688 CrossRef CAS PubMed.
- N. Stock and S. Biswas, Chem. Rev., 2012, 112, 933–969 CrossRef CAS PubMed.
- S.-H. Jhung, J.-H. Lee and J.-S. Chang, Bull. Korean Chem. Soc., 2005, 26, 880–881 CrossRef CAS.
- P. A. Bayliss, I. A. Ibarra, E. Pérez, S. Yang, C. C. Tang, M. Poliakoff and M. Schröder, Green Chem., 2014, 16, 3796–3802 RSC.
- M. P. Batten, M. Rubio-Martinez, T. Hadley, K.-C. Carey, K.-S. L. A. Polyzos and M. R. Hill, Curr. Opin. Chem. Eng., 2015, 8, 55–59 CrossRef.
- M. Gimeno-Fabra, A. S. Munn, L. A. Stevens, T. C. Drage, D. M. Grant, R. J. Kashtiban, J. Sloan, E. Lester and R. I. Walton, Chem. Commun., 2012, 48, 10642–10644 RSC.
- M. Faustini, J. Kim, G.-Y. Jeong, J. Y. Kim, H. R. Moon, W.-S. Ahn and D.-P. Kim, J. Am. Chem. Soc., 2013, 135, 14619–14626 CrossRef CAS PubMed.
- L. Paseta, B. Seoane, D. Julve, V. Sebastian, C. Tellez and J. Coronas, ACS Appl. Mater. Interfaces, 2013, 19, 9405–9410 Search PubMed.
- K.-J. Kim, Y. J. Li, P. B. Kreider, C.-H. Chang, N. Wannenmacher, P. K. Thallapally and H.-G. Ahn, Chem. Commun., 2013, 49, 11518–11520 RSC.
- M. Rubio-Martinez, M. P. Batten, A. Polyzos, K.-C. Carey, J. I. Mardel, K.-S. Lim and M. R. Hill, Sci. Rep., 2014, 4, 5443–5448 CAS.
- Organometallic Aluminium Fumarate Backbone material Patent WO2007118841, 2008.
- G. D. Ulrich, Chemical engineering process design and economics: a practical guide, Process Pub, Durham, N.H, 2nd edn, 2004 Search PubMed.
- M. Groschl, Acustica, 1998, 84, 432 Search PubMed.
- J. J. Hawkes and W. T. Coakley, Sens. Actuators, B, 2001, 75, 213–222 CrossRef CAS.
- K. Yasuda, S. i. Umemura and K. Takeda, J. Appl. Phys., 1995, 34, 2715–2720 CrossRef CAS.
- S. G. Stephen and F. Klavs Jensen, Green Chem., 2013, 15, 1456 RSC.
- P. Juliano, P. Swiergon, K. H. Lee, P. T. Gee, P. T. Clarke and M. A. Augustin, J. Am. Oil Chem. Soc., 2013, 90, 1253–1260 CrossRef CAS.
- F. Jeremias, D. Fröhlich, C. Janiak and S. K. Henninger, RSC Adv., 2014, 4, 24073–24082 RSC.
- C. Serre, F. Millange, C. Thouvenot, M. Noguès, G. Marsolier, D. Louër and G. Férey, J. Am. Chem. Soc., 2002, 124, 13519–13526 CrossRef CAS PubMed.
- M. Gaab, N. Trukhan, S. Maurer, R. Gummaraju and U. Müller, Microporous Mesoporous Mater., 2012, 157, 131–136 CrossRef.
- P. Rallapalli, K. P. Prasanth, D. Patil, R. S. Somani, R. V. Jasra and H. C. Bajaj, J. Porous Mater., 2011, 18, 205–210 CrossRef CAS.
- T. Leong, L. Johansson, P. Juliano, S. L. McArthur and R. Manasseh, Ind. Eng. Chem. Res., 2013, 52, 16555–16576 CrossRef CAS.
- T. Leong, L. Johansson, P. Juliano, R. Mawson, S. McArthur and R. Manasseh, Ultrason. Sonochem., 2014, 21, 1289–1298 CrossRef CAS PubMed.
- D. de las Heras, N. Doshi, T. Cosgrove, J. Phipps, D. I. Gittins, J. S. van Duijneveldt and M. Schmidt, Sci. Rep., 2012, 2, 789–794 Search PubMed.
- A. S. Munn, P. W. Dunne, S. V. Y. Tang and E. H. Lester, Chem. Commun., 2015, 51, 12811–12814 RSC.
- N. Riley, Annu. Rev. Fluid Mech., 2001, 33, 43–65 CrossRef.
- http://mofapps.com/Order.html.
- http://www.sigmaaldrich.com/catalog/product/aldrich/688738?lang=en%26region=AU.
Footnote |
| † Electronic supplementary information (ESI) available: Synthetic details, characterization, images and schemes. See DOI: 10.1039/c5ra24994e |
| This journal is © The Royal Society of Chemistry 2016 |