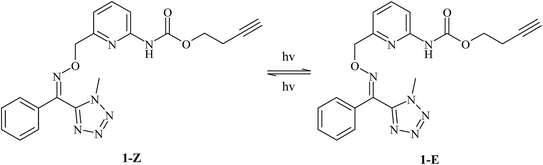
Phototransformation of tetrazoline oxime ethers: photoisomerization vs. photodegradation†
Maxime Fréneauab,
Pascal de Sainte Claireac,
Norbert Hoffmannb,
Jean-Pierre Vorsd,
Julie Geistd,
Michel Euvrardd and
Claire Richard*ae
aEquipe Photochimie CNRS, UMR 6296, ICCF, F-63178 Aubière, France. E-mail: claire.richard@univ-bpclermont.fr; Tel: +33 4 73 40 71 42
bCNRS, Université de Reims Champagne-Ardenne, ICMR, Equipe de Photochimie, UFR Sciences, B.P. 1039, 51687 Reims, France
cUniversité Clermont Auvergne, ENSCCF, Institut de Chimie de Clermont-Ferrand, BP 10448, 63000 Clermont-Ferrand, France
dBayer S.A.S, 14 Impasse Pierre Baizet, 69263 Lyon, France
eUniversité Clermont Auvergne, Université Blaise Pascal, Institut de Chimie de Clermont-Ferrand, BP 10448, F-63000 Clermont-Ferrand, France
First published on 6th January 2016
Abstract
Fungicides showing potent biological activity in green house may have poor activity in the field due to fast photodegradation. This is the case of tetrazoline oxime ethers 1 and 2, active in the Z form, on which very little is known. A comprehensive study was therefore conducted to understand the photochemical behaviour of these compounds. It could be firmly demonstrated that both photoisomerization Z ⇄ E and photodegradation occur, and that interestingly photodegradation only takes place from E forms. Quantum yields of photoisomerization lay within the range 0.38–0.48 and those of E photodegradation in the range 0.06–0.11. During the reaction, the non-fungicidal E forms become the major isomers due to their lower solar light absorptivity than Z forms. The analytical study showed that photodegradation involves the cleavage of the N–O bond and allowed the identification of photoproducts arising from the rearrangement of the iminyl and alkoxyl radicals. Moreover, the proposed mechanism of alkoxyl radical oxidation into corresponding aldehyde and acid was elucidated using a computational study. This fundamental investigation fully explains the fast loss of the fungicidal activity of tetrazoline oxime ethers 1 and 2 in the field.
1 Introduction
Agricultural areas represent about one third of dry lands of the world. However, the growth of global population drives the production of crops grown for human consumption towards an increase of farming yields. Moreover, 50–80% of the crops are discarded in the farming process due to damage inflicted by pests.1 Plant protection treatments can significantly decrease the crop losses, but their use needs to be reduced in order to limit environmental contamination. In the field, pesticides are often submitted to harsh conditions. They are dissipated from the crop leave surfaces by different physical transport processes, such as wash-off, volatilization or plant uptake and by chemical transformation. An important dissipation pathway that weakens treatment efficiency is the photoinduced degradation on the leaves.2,3 Many pesticides absorb solar light,4 and thus can potentially undergo photodegradation. Dose increase or repetitive treatment protocols are generally implemented in the farming procedures to overcome early pesticide degradation. Moreover, the toxicity and persistence of the photoproducts in the environment are a cause of concern.3,5–7 The problem becomes even more urgent because compounds showing highly interesting biological activity but also complex molecular structures and photoreactivity are now available on a large scale. Therefore, to improve pesticide used and reduce their environmental impacts, it is necessary to evaluate and understand the photochemical reactions associated with these processes.About fifty pesticides currently used contain an oxime moiety.4 They belong to the family of cyclohexanediones, strobilurines or methylcarbamate oximes among others. Oxime ethers are stable in the dark but reactive under light exposure.3 Oximes photoisomerization was discovered in the beginning of the twentieth century8,9 and is still a subject of great interest.3,10–16 Chromophores containing the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond can undergo photoinduced isomerization and the resulting photoisomer concentration ratio depends on photoisomerization quantum yields and on the molar absorption coefficient of each photoisomer.17 The photoisomerization reaction can be caused by direct excitation or sensitization.18 While photoisomerization is the major process,19 it often competes with other phototransformation reactions,20–23 such as photofragmentation or photorearrangement. The homolytic photocleavage of the N–O bond of oximes is a common photochemical process.3,12–15,20–25 Irradiated oximes can undergo the Photo-Beckmann rearrangement26 or photocyclization.10,16 They are also used as photochemical or sensitized initiators in photopolymerization reactions.24,27
N bond can undergo photoinduced isomerization and the resulting photoisomer concentration ratio depends on photoisomerization quantum yields and on the molar absorption coefficient of each photoisomer.17 The photoisomerization reaction can be caused by direct excitation or sensitization.18 While photoisomerization is the major process,19 it often competes with other phototransformation reactions,20–23 such as photofragmentation or photorearrangement. The homolytic photocleavage of the N–O bond of oximes is a common photochemical process.3,12–15,20–25 Irradiated oximes can undergo the Photo-Beckmann rearrangement26 or photocyclization.10,16 They are also used as photochemical or sensitized initiators in photopolymerization reactions.24,27
This article describes the detailed photodegradation of oxime ethers 1-Z and 2-Z (see Scheme 1), two fungicides investigated by Bayer Crop Science (WO2003/016303 (DaiNippon); WO2010/000841(Bayer Crop Science)) because they are very active in green house while much less active in the field. Importantly, the E isomers exhibit no fungicide property so that both photoisomerization and photodegradation reactions are expected to inhibit the fungicide activity of 1-Z and 2-Z. Our objective was to understand the photochemical behavior of these compounds from the kinetic and analytical point of views. Most of the work was undertaken on 1-Z but the photochemical behavior of 2-Z was also studied to investigate the potential influence of a distant side chain.
 | ||
| Scheme 1 Structures of the oxime ethers 1-Z and 2-Z, and of potential degradation compounds or model molecules investigated in this study. | ||
2 Experimental part
2.1 Chemicals
Oxime ethers (Z)-but-3-yn-1-yl (6-(((((1-methyl-1H-tetrazol-5-yl)(phenyl)methylene)amino)oxy)methyl)pyridin-2-yl)carbamate 1-Z (purity 99%), (Z)-hexyl (6-(((((1-methyl-1H-tetrazol-5-yl)(phenyl)methylene)amino)oxy)methyl)pyridin-2-yl)carbamate 2-Z (purity 92%), oxime (Z)-(1-methyl-1H-tetrazol-5-yl)(phenyl)methanone oxime 3-Z, 2-benzoyl-N-methyltetrazole (1-methyl-1H-tetrazol-5-yl)(phenyl)methanone 4, substituted pyridine but-3-yn-1-yl (6-(hydroxymethyl)pyridin-2-yl)carbamate 5 and model compound M8 (6-acetamidopicolinic acid) were provided by Bayer Crop Science (La Dargoire Research Center, Lyon, France) and used as received. Acetonitrile (ACN, 99% HPLC grade) was provided by Aldrich (Saint-Quentin Fallavier, France). Milli-Q water was obtained from a Millipore Milli-Q system (Millipore αQ, resistivity = 18 MΩ cm, DOC < 0.1 mg L−1). All other chemicals and solvents were of the highest grade available.2.2 Irradiation devices
For quantum yield and kinetic measurements, solutions were irradiated in a quasi-collimated beam at 278 nm in a cylindrical quartz cell (1 cm optical length) using a high pressure Xe lamp (150 W) equipped with an Oriel monochromator. For sensitizing experiments using xanthone as a sensitizer, solutions were irradiated at 334 nm using a high pressure Hg lamp equipped with an Oriel monochromator (200 W). Ferrioxalate was used as a chemical actinometer. For analytical purposes, polychromatic irradiations were performed in a device equipped with 6 Duke Sunlamp fluorescent tubes (20 W) surrounding the cylindrical Pyrex reactor (internal diameter = 1.4 cm) and emitting between 275 and 350 nm, with a maximum of emission at 313 nm. The Pyrex reactor cut off a great part of radiations having a wavelength below 290 nm (Fig. SI-1†). Irradiations in solid phase were conducted in a solar simulator (Suntest CPS photosimulator from Atlas) set at an irradiance close to the average sunlight intensity received in June in France. A 10 °C cooled water flowed through the bottom of the sample holder to maintain the internal temperature at approximately 35 ± 2 °C. All the dishes were covered by a transparent quartz plate to avoid any displacement due to the vigorous ventilation cooling system. The photon fluence rates in the polychromatic device and in the Suntest were measured using a radiometer QE65000 from Ocean optics.2.3 Irradiation experiments and product studies
Most of the irradiations were conducted in the cylindrical reactor and using the polychromatic tubes. In this case, the same solutions were used for the whole experiments and aliquots were sampled after selected irradiation times. Irradiations were made in duplicate. When solutions were irradiated in parallel beam at 278 nm in the quartz cell, a new solution was used for each irradiation time. The points on the curves were therefore obtained from independent experiments. Irradiations in solid phase were conducted as previously described.2 Solutions of 1-Z (8.2 × 10−5 M) in acetonitrile were deposited as 10 drops of 10 μL on dishes containing paraffinic wax films in the bottom. The initial surfacic concentrations of 1-Z were of 0.41 μg cm−2. Irradiations started after the complete solvent evaporation. Irradiated mixtures were recovered with 1 mL of acetonitrile and directly analyzed by HPLC. Neither volatilization nor dark transformation of 1-Z was observed during manipulations.2.4 Analyses
Absorption spectra were recorded at room temperature using a Varian Cary 3 spectrometer. Scans were performed from 800 to 200 nm with a 1 nm resolution and a 600 nm min−1 rate. For each photolysis experiments, aliquots of solutions were sampled at selected irradiation times and directly analyzed by UPLC. Sample analyses were carried out using a Waters Acquity UPLC equipped with a photodiode array detector and a reversed-phase column (EC 150/2 Nucleodur Sphinx RP, 1.8 μm, Macherey-Nagel) operating at room temperature. Ten μL of samples were injected at a flow rate of 0.2 mL min−1. The following gradient was used: 40% ACN/60% acidified water (pH = 3 using orthophosphoric acid) at the beginning, increase to 45% ACN from 0 to 3 min, and increase to 75% ACN from 3 to 5 min. This method allowed the chromatographic separation of 1-E, 1-Z and their photoproducts. Analyses were made in duplicate for each irradiation time. The same setup was used for the separation of 2-Z and 2-E, but the gradient was modified: 40% ACN/60% acidified water (pH = 3 using orthophosphoric acid) at the beginning, increase to 45% ACN from 0 to 3 min, and increase to 80% ACN from 3 to 5 min. HPLC-mass analyses were carried out at the Research Center of La Dargoire (Bayer Crop Science, Lyon, France). A LCT Premier mass-spectrometer (ESI-TOF) from Waters Company working in positive and negative modes (100 to 1000 u) was used.2.5 Calculations
Time-Dependent Density Functional Theory (TD-DFT) calculations were performed at the B3LYP/6-31G(d,p) level to obtain electronic absorption spectra.283 Results and discussion
3.1 Kinetics of 1-Z and 2-Z phototransformations in acetonitrile
Fig. 1a and b show the spectral changes resulting from the polychromatic irradiation of 1-Z. At very short irradiation times, two isosbestic points are observed at 253 and 306 nm (Fig. 1a) while at longer irradiation times the evolution of the absorption spectra becomes more complex (Fig. 1b). Moreover, HPLC analyses show that after 60 s of irradiation, a unique photoproduct is formed (Fig. SI-2†) against numerous at higher conversion extent (see below). Based on the known photoreactivity of oximes, the photoisomerization of 1-Z into 1-E is highly probable (Scheme 2).8–18 To check this, HPLC-UV-MS analyses were performed. They showed that 1-Z and its photoproduct have the same MS pattern in full agreement with photoisomerization (Fig. SI-3†) and different UV spectra (Fig. SI-4†). The absorption spectrum of 1-E (see Fig. 2) was calculated using the absorption spectrum of the mixture obtained after a conversion extent of 1-Z of 30% and assuming that concentrations of degradation products are negligible behind that of 1-E at this stage of the reaction. After having subtracted the known contribution of 1-Z and after normalization to molarity, the absorption coefficients of 1-E in acetonitrile were obtained and the ratio ε1-Z/ε1-E = 1.5 was calculated at 278 nm. The respective absorption spectra intersected at 253 and 306 nm, wavelengths where isosbestic points were experimentally observed. The shape of the calculated absorption spectrum of 1-E was in full agreement with that obtained by HPLC analyses. Last, TD-DFT calculated spectra confirm that the absorption coefficient of 1-E at 280 nm is significantly lower than that of 1-Z (Fig. SI-5†). | ||
Fig. 2 Absorption spectra of 1-Z and 1-E isomers. ε278 = 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1 for 1-Z and ε278 = 11 000 M−1 cm−1 for 1-Z and ε278 = 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 M−1 cm−1 for 1-E. 400 M−1 cm−1 for 1-E. | ||
The evolution of the concentrations of the isomers during irradiation at 278 nm is shown in Fig. 3. The photoreaction proceeds in two steps. At early stages of the reaction, 1-Z is consumed to give 1-E and a pseudo-equilibrium in which the ratio [1-E]/[1-Z] is around 2 is reached. On a much longer time scale, both 1-Z and 1-E disappear and photoproducts rise through irreversible phototransformation. The existence of a pseudo-equilibrium between 1-E and 1-Z demonstrates that photoisomerization Z ⇄ E is reversible and much faster than the phototransformation. These experimental data were also used to obtain photoisomerization and photodegradation rate constants for both isomers. The set of differential equations for Scheme 3 was solved and the experimental data were fitted simultaneously (see Fig. 3 and Table 1).29,30 In particular, the kinetic modelisation confirms that 1-Z does not photodegrade (kZ→Photoproducts ≈ 0 min−1). The degradation photoproducts only arise from photolysis of 1-E.
 | ||
| Scheme 3 Kinetic model used accounting for Z ⇄ E photoisomerization and Z and E photodegradation. The rate constants were obtained by fitting the solution of the respective set of stiff differential equations by a variable order solver based on numerical differentiation formulas.28,29 | ||
| Compound | kZ→E/min−1 | kE→Z/min−1 | kZ→Photoproducts/min−1 | kE→Photoproducts/min−1 |
|---|---|---|---|---|
| 1-Z | 0.13 ± 0.01 | 0.070 ± 0.01 | 0.00 ± 0.01 | 0.011 ± 0.002 |
| 2-Z | 0.14 ± 0.01 | 0.077 ± 0.01 | 0.00 ± 0.01 | 0.018 ± 0.002 |
| Compound | ΦZ→E | ΦE→Z | ΦZ→Photoproducts | ΦE→Photoproducts |
|---|---|---|---|---|
| 1-Z | 0.45 ± 0.04 | 0.38 ± 0.05 | 0 | 0.06 ± 0.09 |
| 2-Z | 0.48 ± 0.04 | 0.46 ± 0.06 | 0 | 0.11 ± 0.017 |
The quantum yield of 1-Z photoisomerization (ΦZ→E) at 278 nm was measured by using the initial decay rate of 1-Z. Then, the quantum yield of E photoisomerization (ΦE→Z) was obtained by postulating that at the pseudo photostationary equilibrium, the ratio [Z]/[E] is constant and that the rate constants of photodegradation are negligible compared to those of photoisomerization. For solutions showing low absorbances at the excitation wavelength, one can write:
The quantum yield of E photodegradation (ΦE→Photoproducts) was obtained using the relationship:
The respective quantum yields are reported in Table 1. It appears that photoisomerization of 1-Z into 1-E is almost as efficient as that of 1-E into 1-Z with 0.45 against 0.38 and that photodegradation of 1-E is 6.5-fold less efficient than photoisomerization of E into Z.
Irradiation of 1-Z in polychromatic light led to a quantum yield of 1-Z photoisomerization 0.52 ± 0.05 close to the value measured at 278 nm. A difference was however observed in the [1-E]/[1-Z] ratio value. It is of 2.75 in polychromatic light against 2 at 278 nm (Fig. SI-6†).
The same methodology was used to study the 2-Z phototransformation kinetic and the spectral properties of 2-E and similar results were obtained (Fig. SI-7†). Again, we found that 2-Z undergoes clean photoisomerization into 2-E and that phototransformation is only observed from 2-E. The ratio ε2-Z/ε2-E is equal to 1.7 at 278 nm. The quantum yields of 2-Z and 2-E photoisomerisation are very close and around 0.5 and the quantum yield of 2-E photodegradation is 4-fold smaller (see Table 1).
Attempts were made to sensitize the photoisomerization. Xanthone was used as a sensitizer and selectively excited at 334 nm at a concentration of 1.8 × 10−4 M in the presence of 1-Z (1.9 × 10−5 M) in argon saturated acetonitrile. The formation of 1-E is successfully observed and the conversion of 1-Z into 1-E is clean. The ratio [1-E]/[1-Z] is around 2 after 30 min and 100 min of irradiation showing that 1-E also undergoes photoisomerization. It can be concluded that the photoisomerizations are possible from the triplet excited states of 1-Z and 1-E and that their energy levels are below that of xanthone, i.e. 74.1 kcal mol−1.31
3.2 Phototransformation of 1-Z in solid form
To approach the photoreactivity on foliage surface in the field, 1-Z was dropped of on paraffinic wax films and irradiated in a solar simulator. Photoisomerization and photodegradation are observed as in solution and the ratio [1-E]/[1-Z] is 3.05, close to that measured in solution.3.3 Photoproducts identification
 | ||
| Fig. 4 HPLC chromatogram of a deoxygenated acetonitrile solution of 1-Z (8.3 × 10−4 M) irradiated for one hour. UV detection at 278 nm. | ||
| Compound | λmax/nm | ελmax/M−1 cm−1 | MS data molecular peak (mode) |
|---|---|---|---|
| a Value for M8 at 298 nm. | |||
| 1-Z | 278 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
406.15 (ES+) |
| 1-E | 278 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
406.15 (ES+) |
| 2-Z | 278 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
|
| 2-E | 278 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
|
| 5 | 290 | 6400 | 221.09 (ES+) |
| 7 | 298 | 5000a | 219.08 (ES+) |
| 9 | 263 | 2600 | 173 (ES−) and 175 (ES+) |
| 3-E | ≈241 (shoulder) | 8350 | 204.09 (ES+) |
| 3-Z | 255 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 740 740 |
204.09 (ES+) |
| 8 | 305 | 5000a | 215 and 187 (ES−) |
| 6 | 280 | 3000 | |
| 4 | 266 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 900 |
189.08 (ES+) |
In addition, the oxime 3-Z and its photoisomer 3-E were detected in small amounts. The oxime 3-Z (retention time = 1.32 min) could be firmly identified as it was supplied by Bayer. The compound eluted just before 3-Z (retention time = 1.22 min) shows the same m/z = 204 in ES+ and is also present as traces in the synthesized 3-Z. We could confirm that this compound is the isomer 3-E by irradiating acetonitrile solutions of 3-Z with polychromatic light. 3-Z undergoes clean photoisomerization into 3-E and 3-E into 3-Z (Fig. SI-11†) and none of them is photodegraded. For initial and irradiated solutions, we measured HPLC area of each isomer and absorption spectra of solutions. Using this set of data it is possible to determine the extinction coefficient of each isomer (Fig. SI-12†). Absorption spectra of 3-Z and 3-E are significantly different. Data are given in Table 2. We deduce that the ratio [3-E]/[3-Z] is equal to 6.2, the higher concentration of 3-E being due to its lower absorptivity.
Based on the spectral data of Table 2, we can determine the yield of each photoproduct. When the sum 1-Z + 1-E is converted at 65%, 4, 3-Z and 3-E correspond to 52, 1 and 5% respectively of the photoproducts formed from the iminyl radical pathway while the alkoxyl radical pathway leads to 44% of 5. As 1-E is cleaved in two parts, around 50% of photoproducts are identified.
 | ||
| Fig. 5 HPLC chromatogram of air-saturated acetonitrile solution of 1-Z (8.3 × 10−4 M) irradiated for one hour. UV detection at 278 nm. Inset: zoom of the zone 1.5–2.0 min and detection at 305 nm. | ||
In a second step, we irradiated available photoproducts separately in polychromatic light and air-saturated acetonitrile to estimate their own photodegradability. The irradiation of 4 (1.8 × 10−4 M) whose conversion is almost completed after 10 min of irradiation leads to the formation of a major compound eluted at 1.07 min (Fig. SI-16†). This photoproduct, 9, was identified by HPLC-MS as to be the demethylated derivative (Fig. SI-17†). This photoproduct shows a good photostability in the chosen irradiation conditions and is also observed in the HPLC chromatogram of irradiated 1-Z. Its extinction coefficient was obtained by postulating that it is the main photoproduct of 4 (Table 2). Model molecule M8 (1.8 × 10−4 M) is slowly photolyzed, the conversion extent reaching 57% after one hour of irradiation (Fig. SI-18†). No photoproducts could be detected by HPLC analysis. Photoproduct 5 is photostable and 3-Z and 3-E only undergo photoisomerization in air-saturated medium as they do in the absence of oxygen.
Using the extinction coefficients of the different photoproducts (Table 2), we could determine their chemical yields. When the sum 1-Z + 1-E is photodegraded at 91%, the iminyl radical pathway yields 33, 1, 4 and 3% of 4, 3-E, 3-Z and 9 respectively and the alkoxyl radical pathway leads to 12, 30 and 5% of 5, 7 and 8, respectively. Taking into account that the 1-E is cleaved in two parts, around 45% of photoproducts are therefore assigned.
The same photoproducts were obtained by irradiating acetonitrile solutions of 1-Z at 278 nm and with polychromatic light and 1-Z in solid form in the solar simulator.
3.4 Mechanism
This study shows that fungicides 1-Z and 2-Z are readily transformed upon irradiation. They undergo photoisomerization while their photoisomers 1-E and 2-E undergo both photoisomerization and photodegradation. Concerning photoisomerization, one could demonstrate using xanthone as a sensitizer and excitation at 334 nm that this reaction can take place from the triplet excited state. However, this does not rule out the involvement of the singlet excited state when the photoreaction is conducted in the absence of sensitizer. The absence of oxygen effect on the reactions cannot be considered as a criteria to rule out the triplet involvement because this latter may have a too short lifetime to be trapped (<10 ns). Quantum yields of photoisomerization of Z and E are high and close. In these conditions, the ratio [E]/[Z] in the photostationary equilibrium depends on the respective absorption coefficients of the isomers. As E form absorbs less between 250 and 310 nm than its Z counterpart, it is present in the mixture in higher concentration. As a result of these absorption differences, the ratio [E]/[Z] depends on the irradiation conditions: at 278 nm (2), with polychromatic light 280–350 nm (2.7) and in the solar simulator (3). Thus, it is expected to be maximum in real solar irradiation conditions.Concerning photodegradation, we could establish kinetically that photoisomer 1-E is the only one that undergoes phototransformation. The photodegradation mechanism of both photoisomers 1-Z and 1-E was thoroughly studied by DFT quantum calculations. In this work, 8 conformers were characterized. Investigation of the ground state and excited state potential energy surfaces of these species showed that, upon irradiation, the Z-conformers either followed the isomerization path through a conical intersection or returned to the ground state in the Z conformation through intersystem crossing. Contrarily, the major part of the E-conformers can undergo photodegradation by N–O bond dissociation in the singlet or triplet excited states (an activation energy barrier smaller than 5 kcal mol−1 was found for this pathway). This study will be presented in a separate manuscript.
Compounds 1-Z and 2-Z possess side chains with different conformational mobility at the carbamate group. This may have an impact on the photoisomerization of the photodegradation. However, no significant difference concerning the corresponding parameters was observed (Table 1).
The structure of identified photoproducts (Scheme 4) fully confirms the initial cleavage of the N–O bond with formation of the iminyl and alkoxyl radicals in full agreement with the literature data, even though attempts to detect these transient species by laser flash photolysis were unsuccessful. The iminyl radical essentially leads to the imine that is interestingly not observable by reversed-phase HPLC analysis due to its fast acidic hydrolysis into ketone 4. The formation of the imine requires the abstraction of a hydrogen atom by the iminyl radical. This hydrogen may be taken from the starting compound, the alkoxyl radical or the solvent. The abstraction of an H atom from Z and E would generate degradation photoproducts from both. Contrarily only E form undergoes photodegradation. Moreover, the abstraction of an H atom from the alkoxyl radical would give rise to the aldehyde 7. Yet, photoproduct 7 is observed in air-saturated solution and not in nitrogen-saturated solution showing that its production is linked to the presence of oxygen and not to the imine formation. An alternative possibility is a H abstraction from acetonitrile. This case is rarely considered but it was already reported.35
Photolysis of diluted ketone 4 leads to demethylation. This reaction only takes place in oxygenated solution. It can be therefore postulated that the excited carbonyl abstract an H-atom from the methyl to finally yield the alcohol or the acid after oxidation of the CH2 radical. The detection of the demethylated photoproduct suggests the alcohol or the carboxylic acid formed are unstable and loose H2CO or CO2, respectively. The photocatalytic oxidation of phenylurea herbicides was also reported to give rise to such rearrangements after oxidation of the methyl groups.36 The mechanism of this reaction needs to be studied in more details. To rationalize the detection of the demethylated photoproduct in conditions where it is not sure that the ketone 4 is formed, it is also necessary to postulate that it may be also generated by photolysis of the imine 6.
The formations of the alcohol 5, aldehyde 7 and carboxylic acid 8 from the alkoxyl radical appear very logical. The alkoxyl radical needs to abstract a hydrogen atom to yield 5. Again, acetonitrile might be the H-donor. The absence of 7 and 8 in inert atmosphere shows that they are produced subsequently to the reaction of the alkoxyl radical with oxygen. Ground state calculations were therefore performed at the B3LYP/6-31G(d,p) level to understand the mechanisms that led to the formation of the aldehyde (30%) and carboxylic acid (5%) from the alkoxyl radical in acetonitrile and oxygenated continuous irradiation conditions.
The amounts of alcohol and acid species were less significant (respectively 12% and 5%). As mentioned above, these photoproducts were obtained upon photodissociation of the oxime parent molecule in acetonitrile, oxygenated media and continuous irradiation conditions. First, our calculations have shown that the parent oxyl radical O1 was less stable than the secondary alkyl radical O2 (ΔEel = 27.4 kcal mol−1; see Scheme 5 and Fig. 6). However, the electronic activation energy of the elementary intramolecular reaction was high (25.2 kcal mol−1). This rearrangement (O1 → O2) is thus disfavored on kinetic grounds. Alternatively, it may be assisted by the solvent or proceed through the excited state potential energy surface of the oxyl radical.
 | ||
| Scheme 5 The reactivity of oxyl radical O1 in oxygenated medium, continuous irradiation conditions and acetonitrile. R = COOC4H5. | ||
Preliminary TD-DFT calculations that were performed in our laboratory showed that optimization of the excited S1 state of O1 did not lead to any significant change of geometry. Thus, it appeared that further intramolecular rearrangement might not proceed through different adiabatic surfaces. It was then decided to investigate the solvent assisted O1 → O2 reaction (see Scheme 6 and Fig. 6).
 | ||
| Scheme 6 The solvent (acetonitrile) assisted reaction of oxyl radical O1. A unique structure connects both transition states. | ||
Examination of the reaction energies shows that the suite of reactions in Scheme 6 were thermodynamically favored and kinetically competitive with the aldehyde pathway (see discussion below). O1 and O2 may react with oxygen and yield respectively the trioxy radical O1′ or the peroxy radical O2′ (see Scheme 5). The intermediate O1′ was already proposed in an earlier study to explain the negative temperature dependence of the reaction of 2-butoxy radicals with oxygen.37 This mechanism may compete38 with direct hydrogen abstraction from oxygen molecules (O1′ + O2 → aldehyde + HO2). In our study, O1′ was indeed characterized and is an intermediate species. Nevertheless, the respective electronic activation barrier is small (1.3 kcal mol−1; see Fig. 6 and Scheme 7). Thus, the formation of the aldehyde photoproduct follows fast kinetics.
 | ||
| Scheme 7 The formation of the aldehyde photoproduct. O1′ is slightly stabilized by intramolecular hydrogen bonding. | ||
The outcome of O2′ is now investigated. First, a transition state for intramolecular hydrogen abstraction O2′ → acid + HO (see Scheme 8a) was not found at this level of calculation. Peroxyl radical O2′ may react through another intramolecular hydrogen abstraction reaction (see Scheme 8b) and yield the aldehyde photoproduct and radical HO2, provided the latter diffuses out of the solvent cage. The respective activation energy is 10.7 kcal mol−1 (see Fig. 6). However, the in-cage complex may return to the thermodynamically more stable species O2′. Intermolecular hydrogen abstraction reaction from the solvent (Scheme 8c) is not competitive with Scheme 8b because it requires 26.6 kcal mol−1 to overcome the activation barrier.
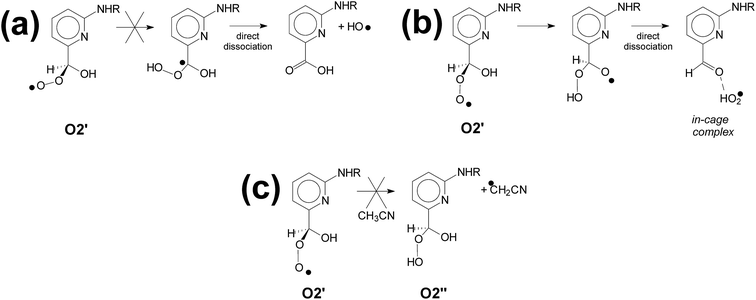 | ||
| Scheme 8 The reactivity of peroxyl radical O2′: intramolecular (a and b) and intermolecular (c) hydrogen abstraction. (a) and (c) are disfavored on kinetic grounds. | ||
Finally, O2 may also react with HO2 radicals released as side-products in aldehyde formation. The second-order rate constant for HO2 recombination with O2 is 6.3 larger than the second-order rate constant for O2 recombination with O2.37 In addition, considering that (1) the initial concentration of 1-Z is 8.3 × 10−4 M and (2) 30% of aldehyde is obtained, the maximum concentration of HO2 is 3 × 10−4 M. This is an order of magnitude less than [O2] (the concentration of oxygen in air-equilibrated acetonitrile is 2.42 × 10−3 M). Thus, 44% (=6.3 × 3 × 10−4/(2.42 × 10−3 + 6.3 × 3 × 10−4)) of species O2 yields O3 (see Fig. 6) and the acid photoproduct. The latter is obtained in hydrogen abstraction reactions with O2 or HO2 radicals. Note that the direct intramolecular reaction RCH(OOH)OH → acid + H2O is not competitive here because the respective activation barrier is 53.9 kcal mol−1 at the B3LYP/6-31G(d,p) level. Alternatively, oxyl radical O1 may also yield the alcohol species RCH2OH by intermolecular H-abstraction reaction from the solvent.
These theoretical results may be resumed as follows: in solution, the alkoxyl radical reacts rapidly with oxygen and yields the aldehyde 7. The respective decay rate of the alkoxyl radical may be obtained from the concentration of oxygen in air-equilibrated acetonitrile37 and k1→aldehyde = 4.8 × 106 L mol−1 s−1,37,39,40 i.e. k1→aldehyde[O2] = 1.2 × 104 s−1. The more stable hydroxyalkyl radical is rather obtained in a solvent assisted reaction. Considering the activation barrier of 12.8 kcal mol−1 for the initial step, the respective solvent assisted decay rate constant of the alkoxyl radical is kRCH2O→RCHOH = 2.6 × 103 s−1. Thus 18% of the alkoxyl radical follows this pathway. RCHOH may react with O2 and yield aldehyde (56%) or react with HO2 radicals released as side-product in the formation of the aldehyde (44%). Small amounts of carboxylic acid (8% overall, i.e. 0.44 × 0.18) are obtained in the reaction with HO2 in good agreement with experimental results: 14% was reported experimentally, i.e. 5/(30 + 5).
To conclude, this study provides a full understanding of the photochemical fate of tetrazoline oxime ethers 1 and 2. Photoisomerization Z ⇄ E takes place with close and high quantum yields for both isomers. The lower absorptivity of E forms compared to Z forms causes the ratio [E]/[Z] to reach values higher than 2 and therefore favours the accumulation of the non-fungicidal isomer. Photodegradation also occurs, and we could firmly demonstrate that this process exclusively takes place from E forms, which is of particular interest. Thus, degradation may be limited if the photoisomerization to the E isomer is inhibited. Photodegradation starts by the cleavage of the N–O bond into the iminyl and alkoxyl radicals from which all the characterized photoproducts are issued. Another important topic of this work was to elucidate the complex mechanism of oxidation of the alkoxyl radical into the corresponding aldehyde and acid with theoretical calculations. In particular, the involvement of the solvent was characterized. On the whole, this detailed study rationalizes the loss of biological activity of these fungicides in the field and may help for the design of less photodegradable phytosanitary products.
References
- E.-C. Oerke, J. Agric. Sci., 2006, 144, 31–43 CrossRef.
- S. Monadjemi, M. El Roz, C. Richard and A. Ter Halle, Environ. Sci. Technol., 2011, 45, 9582–9589 CrossRef PubMed.
- B. Sevilla-Morán, P. Sandín-España, M. J. Vicente-Arana, J. L. Alonso-Prados and J. M. García-Baudín, J. Photochem. Photobiol., A, 2008, 198, 162–168 CrossRef.
- The Pesticide Properties DataBase (PPDB) developed by the Agriculture & Environment Research Unit (AERU), University of Hertfordshire, 2006–2013,http://sitem.herts.ac.uk/aeru/ppdb/en/ Search PubMed.
- E. M. Thurman, M. Meyer, M. Pomes, C. A. Perry and A. P. Schwab, Anal. Chem., 1990, 62, 2043–2048 CrossRef CAS PubMed.
- A. D. Dimou, V. A. Sakkas and T. A. Albanis, J. Agric. Food Chem., 2005, 53, 694–701 CrossRef PubMed.
- G. Durand, D. Barceló, J. Albaigés and M. Mansour, Chromatographia, 1990, 29, 120–124 Search PubMed.
- G. Ciamician and P. Silber, Ber. Dtsch. Chem. Ges., 1903, 36, 4266–4272 CrossRef CAS.
- O. L. Brady and F. P. Dunn, J. Chem. Soc., Faraday Trans., 1913, 103, 1619–1626 RSC.
- R. J. Olsen, J. Photochem. Photobiol., A, 1997, 103, 91–94 CrossRef CAS.
- H. Ikeda, T. Suzue, M. Yukawa, T. Niiya and Y. Goto, Chem. Pharm. Bull., 2002, 50, 969–971 CrossRef CAS PubMed.
- M. A. Brown, D. W. Gammon and J. E. Casida, J. Agric. Food Chem., 1983, 31, 1091–1096 CrossRef CAS.
- D. S. Bohle, Z. Chua, I. Perepichka and K. Rosadiuk, Chem.–Eur. J., 2013, 19, 4223–4229 CrossRef CAS PubMed.
- L. Lapinski, T. Stepanenko, M. F. Rode and M. J. Nowak, J. Mol. Struct., 2010, 976, 181–189 CrossRef CAS.
- T. Stepanenko, L. Lapinski, M. J. Nowak and L. Adamowicz, Vib. Spectrosc., 2001, 26, 65–82 CrossRef CAS.
- M. A. Rodríguez, in Molecular Photochemistry-Various Aspects, ed. S. Saha, InTech, 2012, vol. 12, pp. 265–282 Search PubMed.
- P. Klán and J. Wirz in Photochemistry of Organic Compounds: From Concepts to Practice, Wiley, Chichester, West Sussex, U.K, 2009 Search PubMed.
- Y. Kawamura, R. Takayama, M. Nishiuchi and M. Tsukayama, Tetrahedron Lett., 2000, 41, 8101–8106 CrossRef CAS.
- A. Padwa and F. Albrecht, J. Am. Chem. Soc., 1974, 96, 4849–4857 CrossRef CAS.
- A. Padwa, Chem. Rev., 1977, 77, 37–68 CrossRef CAS.
- A. C. Pratt, Chem. Soc. Rev., 1977, 6, 63–81 RSC.
- H. Roth, in The Chemistry of Hydroxylamines, Oximes and Hydroxamic Acids, ed. Z. Rappoport and J. F. Liebman, John Wiley & Sons, Chichester, 2011, vol. 2, pp. 1–63 Search PubMed.
- H. Suginome, in CRC Handbook of Organic Photochemistry and Photobiology, ed. W. Horspool and F. Lenci, CRC Press, Boca Raton, 2nd edn, 2004, pp. 94/1–94/55 Search PubMed.
- F. Amat-Guerri, R. Mallavia and R. Sastre, J. Photopolym. Sci. Technol., 1995, 8, 205–232 CrossRef CAS.
- C. Kolano, G. Bucher, D. Grote, O. Schade and W. Sander, Photochem. Photobiol., 2006, 82, 332–338 CrossRef CAS PubMed.
- H. Izawa, P. de Mayo and T. Tabata, Can. J. Chem., 1969, 47, 51–62 CrossRef.
- M. Griesser, A. Rosspeintner, C. Dworak, M. Höfer, G. Grabner, R. Liska and G. Gescheidt, Macromolecules, 2012, 45, 8648–8657 CrossRef CAS PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision C.01, Gaussian, Inc., Wallingford CT, 2010 Search PubMed.
- MATLAB and Statistics Toolbox Release 2012b, The MathWorks, Inc., Natick, Massachusetts, United States Search PubMed.
- The solver ode15s in Matlab® was used to achieve these analyses.
- Handbook of Photochemistry, ed. S. T. Murov, I. Carmichael and G. L. Hug, Marcel Dekker, NY, 2nd edn, 1993 Search PubMed.
- M. B. Smith, March's Advanced Organic Chemistry, John Wiley & Sons, Hoboken, 2013, pp. 1077–1079 Search PubMed.
- S. Hammerum and T. I. Sølling, J. Am. Chem. Soc., 1999, 121, 6002–6009 CrossRef CAS.
- A. J. McCarroll and J. C. Walton, J. Chem. Soc., Perkin Trans. 2, 2000, 1868–1875 RSC.
- S. Bouchama, P. de Sainte-Claire, E. Arzoumanian, E. Oliveros, A. Boulkamh and C. Richard, Environ. Sci.: Processes Impacts, 2014, 16, 839–847 CAS.
- C. Richard and S. Bengana, Chemosphere, 1996, 33, 635–641 CrossRef CAS.
- H.-H. Grotheer, G. Riekert, U. Meier and T. Just, Ber. Bunsen-Ges., 1985, 89, 187–191 CrossRef CAS.
- C. Franco and J. Olmsted III, Talanta, 1990, 37, 905–909 CrossRef CAS PubMed.
- G. Falgayrac, F. Caralp, N. Sokolowski-Gomez, P. Devolder and C. Fittschen, Phys. Chem. Chem. Phys., 2004, 6, 4127–4132 RSC.
- P. Warneck, International Geophysics Series, in Chemistry of the Natural Atmosphere, Academic Press, 1999, vol. 71, ch. 6, pp. 264–345 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra24936h |
| This journal is © The Royal Society of Chemistry 2016 |