Phthalazinone structure-based covalent triazine frameworks and their gas adsorption and separation properties†
Kuanyu Yuanabc,
Cheng Liu*abc,
Jianhua Hanabc,
Guipeng Yud,
Jinyan Wangabc,
Hongmin Duane,
Zhonggang Wangabc and
Xigao Jianabc
aState Key Laboratory of Fine Chemicals, Department of Polymer Science and Materials, School of Chemical Engineering, Dalian University of Technology, Dalian, People's Republic of China 116024. E-mail: liuch1115@dlut.edu.cn
bDepartment of Polymer Materials & Engineering, Dalian University of Technology, Dalian, People's Republic of China 116024
cLiaoning Province Engineering Research Centre of High Performance Resins, Dalian, People's Republic of China 116024
dCollege of Chemistry and Chemical Engineering, Central South University, Changsha, People's Republic of China 410083
eDalian Institute of Chemical Physics, Chinese Academy of Science, Dalian, People's Republic of China 116023
First published on 18th January 2016
Abstract
In this work, new classes of phthalazinone-based covalent triazine frameworks (PHCTFs) were prepared by ionothermal synthesis from two full rigid dicyano building blocks with rigid, thermostable and asymmetric N-heterocycle-containing structures. The surface and internal morphologies of PHCTFs were examined by FE-SEM and TEM. The resultant microporous polymers, PHCTFs, exhibited BET specific surface areas up to 1845 m2 g−1 and a moderately narrow pore size distribution. According to the sorption measurements, the CO2 uptake can be up to 17.1 wt% (273 K/1 bar) and the H2 uptake can be up to 1.92 wt% (77 K/1 bar). Moreover, the initial slopes of the single component gas adsorption isotherms in the low pressure range were used as the gas separation ratios. The obtained polymer networks possess satisfactory CO2/N2 selectivity performance up to 52 and CO2/CH4 selectivity up to 12. Combining the relationship of the structure and performance, it can be concluded that a twisted and non-coplanar topology conformation can be used to improve the porosity of microporous organic polymers. At the same time, the nitrogen- and oxygen-rich characteristics of the phthalazinone core endow the networks with a strong affinity for CO2 and thereby high CO2 adsorption capacity. So the pore structure and chemical composition may play very important roles on the adsorption properties of small gas molecules.
Introduction
The porous materials including macroporous (>50 nm), mesoporous (2–50 nm) and microporous (<2 nm) materials are classified by IUPAC according to their pore size.1 For their special porous architectures, in the micro- and nanoscale range, these materials have attracted much attention in many fields of science and technology, especially in advanced functional material design.2 Apart from the traditional types of porous materials, such as activated carbon, zeolites,3 or mesoporous silicates,4 metal–organic frameworks5 (MOFs) containing larger organic linkers and metal ions have formed one of the fastest growing fields in chemistry during the past decades for their high surface areas and large range levels of porosity. However, the low physicochemical stability of MOFs may limit their wider applications.6In the past few years, purely microporous organic polymers (MOPs) materials, which are constructed through strong covalent bonding via polymerization of building blocks that are composed of non-metallic elements such as C, H, O, N and B,7 have attracted considerable attention. The relatively high surface area, permanent porosity, low mass densities and wide structure turnability8 make MOPs show great potential in gas storage9 and separation,10 catalysis11 and sensors.12 Several different classes of MOPs have been developed, including covalent organic frameworks (COFs),13 conjugated microporous polymers (CMPs),14 porous aromatic frameworks (PAFs),15 porous polymer networks (PPNs),16 polymers of intrinsic microporosity (PIMs),17 hypercrosslinked polymers (HCPs),9a,18 element–organic frameworks (EOFs)11b,19 and organic cage frameworks (OCFs).20 In consequence of the remarkably potential application of MOPs, exploiting new structure or new performance remains a research hot issue in this field.
Due to the plethora of organic reactions and building blocks,17b,21 which provide flexibility for the materials to be designed to achieve desirable pore properties,22 various reactions7b of organic functional groups make contribution to the construction of MOPs, such as reversible borate chemistry, palladium-catalyzed Sonogashira–Hagihara cross-coupling, homocoupling of aromatic bromides, Friedel–Crafts reaction, dioxane-forming polymerization, oxidative coupling, amide or imide formation and Schiff-base chemistry. Recently, an emerging class of microporous organic materials called covalent triazine frameworks23 (CTFs), which own the novel polymer network based on triazine linkage through the trimerization of nitriles, provides a new method to construct the MOPs.
Covalent triazine frameworks (CTFs) were first developed by Antonietti, Thomas and co-workers via ionothermal synthesis23d in zinc chloride (ZnCl2) salt melts medium in 2008. Though, the reaction was performed at high temperature (400–700 °C) for long time and may deteriorate the structure of some monomers as well as carbonize the derived polymers.24 In 2012, Zhu and co-authors25 reported an alternative method which used strong Brønsted acid-trifluoromethanesulfonic acid to catalyze the cyclotrimerization reaction at mild conditions. Compared with the latter, however, the ionothermal method shows such significant advantages26 as being cheaper and experimentally simpler, and yielding materials with high surface areas.
Apart from the specialty of the synthesis of CTFs, they possess very large specific surface area together with27 exceptional chemical inertness and high thermal stability owing to their graphite-like composition and robust carbon–carbon and carbon–nitrogen linkages.
The large specific surface areas of CTFs can be ascribed to the abundant pore structure, primarily micropores and mesopores. And the micropores, especially the narrow ultramicropores (<1 nm), make greater contribution to the gas molecules, such as the small thermodynamic size gas molecule CO2. Also, the incorporation of the nitrogen-rich polar triazine moieties makes CTFs electron-rich, which may enhance the affinity and the isosteric heat between the adsorbent and specific sorbate molecule by the significantly promoted dipole–quadrupole interaction.28 The co-contribution of above factors, microporoes and nitrogen-rich, enables CTFs material to be a promising candidate for gas adsorption and separation. Meanwhile, CTFs with N-doping show potential catalysts supporter by nitrogen–metal interactions in large amounts owing to the large accessible surface areas.27 All of the characterizes and advantages in structure and functionality make CTFs be considered as a promising candidate for energy gas storage and catalytic support materials, and have spurred the scientific interest in searching novel building blocks to construct versatile CTFs.
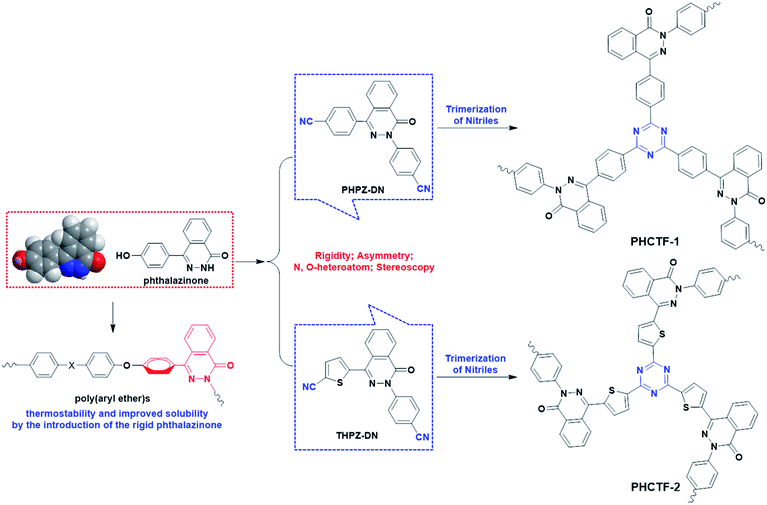
Motivated by the above-mentioned distinctions and advantages of CTFs, we wonder the effect of N-heterocycle-containing, rigid, twisted and asymmetric structure on the properties of CTFs. We have previously demonstrated the synthesis and properties of aromatic polymers containing phthalazinone moiety,29 such as poly(aryl ether)s,30 polyamides,31 polyimides,32 and so on, which possess rigid and twisted backbones and remain excellent thermostability coupled with improved solubility due to the introduction of the rigid phthalazinone with twisted, non-coplanar conformation. Therefore, we consider that this stereoscopic phthalazinone core, which favors the construction of CTFs and limits the stacking of the polymers networks, may help to increase the available space accessible for gas adsorption and storage. To the best of our knowledge, there are no same building blocks, which simultaneously possess rigidity, asymmetry, N,O-heteroatoms and stereoscopy, have been selected to construct CTFs.
In our work, we designed and synthesized two fully rigid dicyano building blocks containing phthalazinone structure, 2-(4′-cyanophenyl)-4-(4′-cyanophenyl)-2,3-phthalazin-1-one (PHPZ-DN) and 2-(4′-cyanophenyl)-4-(4′-cyanothiophene)-2,3-phthalazin-1-one (THPZ-DN). Then, new classes of phthalazinone core-based covalent triazine frameworks (PHCTFs) (Scheme 1) were prepared by ionothermal reaction and their porosity, gas storage capacities and selectivity have also been characterized by different pure gas adsorption/desorption measurements. The conformation of THPZ-DN exhibits more coplanarity, because the dihedral angle calculated by Gaussian 09W between thiophene and naphthyridine of THPZ-DN is smaller than phenyl and naphthyridine of PHPZ-DN. This is another aim to demonstrate how the change of conformation of phthalazinone influences the porosity of the targeted polymer networks.
Experimental
Material
All the starting materials were purchased from commercial suppliers and used as received unless otherwise indicated. Zinc chloride was refluxed and distilled over thionyl chloride to remove water, and then excess thionyl chloride was azeotropically distilled with toluene. After the removal of thionyl chloride and toluene, anhydrous zinc chloride was obtained after being dried at 180 °C under vacuum for 24 h.Instrumentation
Fourier transform infrared spectra (FT-IR) were recorded using a Nicolet 20 DXB FT-IR spectrophotometer in the 400–4000 cm−1 region. Samples were prepared by dispersing the complexes in potassium bromide (KBr) and compressing the mixtures to form disks. 1H-NMR (400 Hz) spectra were obtained with a Bruker spectrometer at an operating temperature of 25 °C using tetramethylsilane as an internal standard. Elemental analyses were determined with an Elementar Vario EL III elemental analyzer. Thermogravimetric analysis (TGA) of the polymers was performed on a Mettler TGA/SDTA851 thermogravimetric analysis instrument in flowing nitrogen atmosphere at a heating rate 20 °C min−1. The temperature range was from room temperature to 800 °C. The powder X-ray diffraction patterns (XRD) of the samples were recorded with a SmartLab (9) diffractometer using CuKα radiation operated at 45 kV and 200 mA, performing from 5° to 80° at a speed of 8° min−1. Field-emission scanning electron microscopy (FE-SEM) experiments were carried on a Nova NanoSEM 450. Transmission electron microscopy (TEM) images were carried out using Tecnai12 transmission electron microscopy (FEI, Eindhoven, The Netherlands) at an operating voltage of 120 kV. Adsorption and desorption measurements for all the gases were conducted on an Autosorb iQ (Quantachrome) analyzer. Before sorption measurements, the samples were degassed at 120 °C under high vacuum for 12 h. The nitrogen adsorption and desorption were measured at 77 and 273 K up to 1 bar. The specific surface areas were calculated according to the Brunauer–Emmett–Teller (BET) model in the relative pressure (P/P0) ranging from 0.01 to 0.1. Pore size distributions were derived from the N2 adsorption isotherms at 77 K using the nonlocal density functional theory (NLDFT) method. High-purity gas (99.999%) was used for adsorption experiment.Synthesis of 2-(4-cyanophenyl)-4-(4-bromophenyl)(2H)phthalazin-1-one (PHPZ-CN)
A 250 mL three-necked round-bottom flask equipped with a mechanical stirrer, a Dean–Stark trap outfitted with a condenser, and nitrogen inlet and outlet, was charged with 1,2-dihydro-4-(4-bromophenyl)(2H)phthalazin-1-one (PHPZ-Br) (9.03 g, 0.03 mol), potassium carbonate (K2CO3, 2.82 g, 0.02 mol), N,N-dimethylacetamide (DMAc, 60 mL) and toluene (50 mL). Under an atmosphere of nitrogen, the reaction mixture was heated and maintained at 140 °C for 4–5 h to remove all water by means of azeotropic distillation with toluene. Then, the temperature was increased to 150 °C to remove toluene for 4 h. After the mixture was cooled to room temperature, 4.95 g (0.04 mol) of 4-chlorobenzonitrile was added and the temperature was maintained at 180 °C for 8 h. The mixture was then poured into an ethanol/water mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v), 200 mL). The precipitate was collected on a filter and crystallized from N,N-dimethylformamide (DMF) to give white solid (10.15 g, yield: 84%), mp: 278–279 °C; 1H-NMR (400 MHz, DMSO-d6/TMS int, ppm) δ: 8.47 (d, 1H), 8.03–7.96 (m, 6H), 7.82–7.74 (m, 3H), 7.67 (d, 2H); HRMS calculated for C21H12BrN3O, 401.0164; found, 401.0159.
1 (v/v), 200 mL). The precipitate was collected on a filter and crystallized from N,N-dimethylformamide (DMF) to give white solid (10.15 g, yield: 84%), mp: 278–279 °C; 1H-NMR (400 MHz, DMSO-d6/TMS int, ppm) δ: 8.47 (d, 1H), 8.03–7.96 (m, 6H), 7.82–7.74 (m, 3H), 7.67 (d, 2H); HRMS calculated for C21H12BrN3O, 401.0164; found, 401.0159.
Synthesis of 2-(4′-cyanophenyl)-4-(4′-cyanophenyl)(2H)phthalazin-1-one (PHPZ-DN)
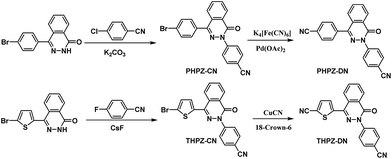
A 250 mL three-necked round-bottom flask equipped with a magnetic stirrer a condenser was charged with PHPZ-CN (7.240 g, 0.018 mol), potassium hexacyanoferrate (K4[Fe(CN)6]·3H2O, 1.673 g, 0.004 mol, 0.22 equiv.), sodium carbonate (Na2CO3, 1.908 g, 0.018 mol, 1.0 equiv.), palladium acetate (Pd(OAc)2, 0.020 g, 0.5%), and 1-methyl-2-pyrrolidinone (NMP, 100 mL). The flask was evacuated and filled with nitrogen (two times) and heated to 140 °C. After maintaining the temperature for 12 h, the reaction mixture was then poured into diluted water. The precipitate was collected on the filter and crystallized from DMAc and dimethyl sulfoxide (DMSO) to give white solid (3.564 g, 57%), mp: 312–313 °C; 1H-NMR (400 MHz, CDCl3/TMS int, ppm) δ: 8.64 (d, 1H), 7.98 (d, 2H), 7.88 (d, 4H), 7.78 (d, 4H), 7.71 (d, 1H); HRMS calculated for C22H12N4O, 348.1011; found, 348.1020.Synthesis of 2-(4′-cyanophenyl)-4-(4′-bromothiophene)-2,3-phthalazin-1-one (THPZ-CN)
A 250 mL three-necked round-bottom flask equipped with a mechanical stirrer, nitrogen inlet and outlet and a condenser was charged with 1,2-dihydro-4-(4-bromothiophene)(2H)phthalazin-1-one (THPZ) (9.22 g, 0.03 mol), cesium fluoride (CsF, 5.47 g, 0.036 mol), 4-fluorobenzonitrile (4.36 g, 0.036 mol) and DMAc (30 mL). Under the N2 atmosphere, the reaction mixture was heated and maintained at 150 °C for 12 h. Then, the mixture was poured into an ethanol/water mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 200 mL). The precipitate was collected on a filter and crystallized from DMF to give yellow solid (10.42 g, 85%); mp: 258–259 °C; 1H-NMR (400 MHz, DMSO-d6/TMS int, ppm) δ: 8.43–8.49 (d, 1H), 8.22–8.27 (d, 1H), 7.92–8.10 (m, 6H), 7.58–7.62 (d, 1H), 7.39–7.43 (d, 1H). HRMS calculated for C19H10BrN3OS: 406.9728; found: 406.9723.
1, 200 mL). The precipitate was collected on a filter and crystallized from DMF to give yellow solid (10.42 g, 85%); mp: 258–259 °C; 1H-NMR (400 MHz, DMSO-d6/TMS int, ppm) δ: 8.43–8.49 (d, 1H), 8.22–8.27 (d, 1H), 7.92–8.10 (m, 6H), 7.58–7.62 (d, 1H), 7.39–7.43 (d, 1H). HRMS calculated for C19H10BrN3OS: 406.9728; found: 406.9723.
Synthesis of 2-(4′-cyanothiophene)-4-(4′-cyanophenyl)(2H)phthalazin-1-one (THPZ-DN)
THPZ-CN (4.08 g, 10 mmol), cuprous cyanide (CuCN, 4.48 g, 50 mmol) and 18-crown-6 (90 mg) were dissolved in dry DMF (100 mL) under N2 atmosphere. After stirred at 150 °C for 24 h, the mixture was poured into a cold dilute ammonia water and stirred for 12 h. The brown solid was collected by filtration, washed with water and ethanol several times, dried under vacuum and crystallized from DMF to give a light brown solid (2.32 g, 66%); mp: 270–271 °C. 1H-NMR (400 MHz, DMSO-d6/TMS int, ppm) δ: 8.44–8.50 (d, 1H), 8.17–8.22 (d, 1H), 7.94–8.10 (m, 6H), 7.88–7.91 (d, 1H). HRMS calculated for C20H10N4OS: 354.0575; found: 354.0569.Synthesis of PHCTFs
PHCTFs were synthesized by heating a mixture of the resultant phthalazinone-based monomers, PHPZ-DN or THPZ-DN, with ZnCl2 (Table 1). The two kinds of PHCTFs were synthesized by a similar method, so only the synthesis procedure for PHCTF-1a is afforded here as an example. A quartz tube (3 × 5 cm) was charged with PHPZ-DN (0.16 g, 0.45 mmol) and ZnCl2 (0.61 g, 4.5 mmol). The tube was evacuated to a high vacuum and then sealed rapidly. After a temperature program (250 °C for 10 h, 300 °C for 10 h, 350 °C for 10 h, and 400 °C for 20 h), the quartz tube was cooled to room temperature, and the resultant mixture was subsequently ground and then washed thoroughly with water for 72 h to remove most of the catalyst. After that, the product was isolated by filtration and again stirred with 100 mL hydrochloric acid (HCl, 2 mol L−1) for 24 h to remove the residual salt. The resulting black powder was filtered and washed successively with water and alcohol, followed by an overnight Soxhlet extraction using acetone and methyl alcohol as eluting solvents sequentially, and finally dried in vacuum at 150 °C. Yield: 80%.| PHCTF | Monomer![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ZnCl2 (molar ratio) ZnCl2 (molar ratio) |
Temperature (°C)/(time) |
|---|---|---|
| 1a | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
250 (10 h), 300 (10 h), 350 (10 h), 400 (20 h) |
| 1b | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
250 (10 h), 300 (10 h), 350 (10 h), 400 (20 h) |
| 1c | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
250 (10 h), 300 (10 h), 350 (10 h), 400 (20 h), 600 (20 h) |
| 2a | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
250 (10 h), 300 (10 h), 350 (10 h), 400 (20 h) |
| 2b | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
250 (10 h), 300 (10 h), 350 (10 h), 400 (20 h) |
Results and discussion
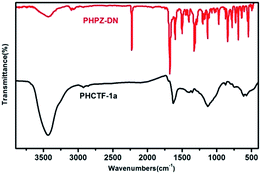
Two analogous phthalazinone-containing dicyano monomers were selected as building blocks, which were synthesized by nucleophilic substitution reaction, followed by cyanation (the PHPZ-DN was prepared from the cyanation of PHPZ-Br according to the procedure of literature33 with some modifications). The synthetic routes are illustrated in Scheme 2 and their proposed structures were confirmed by 1H-NMR (Fig. S1 and S2 of the ESI†). Although using strong Brønsted acid as catalyst, trifluoromethanesulfonic acid etc., may avoid decomposition and condensation reactions such as C–H bond cleavage and carbonization,34 this method generally leads to lower surface areas accompanied with the breakage within C–N and C–O linkage of the nitrile monomers.35 In contrast, CTFs constructed from 1,4-dicyanobenene at the presence of ZnCl2 as Lewis acid catalyst, solvent and porogen, exhibited high surface areas (1710 m2 g−1 to 3000 m2 g−1),23a,23c but the dicyano monomer should suffer continuous heating at 400 °C or even higher temperature (600 °C) during the reaction. The relative excellent thermostability (Fig. S3†) of the building blocks seems to also be necessary to afford complete and good physicochemical stabile polymer networks. The 10% decomposition temperatures under N2 atmosphere recorded at 336 °C for PHPZ-DN and 364 °C for THPZ-DN may be a little lower compared to previous reported dicyano monomers, o-DCB and DCBP23d (the temperatures for 10 wt% mass loss under N2 atmosphere exceed 400 °C), so the trimerization reaction was conducted under a high vacuum system to avoid the rapid decomposition under high temperature and long reaction time.According to Yu's research,35 the reaction temperature less than 350 °C results in low-molecular-weight compounds. So, to avoid the probable complete carbonization by the one-step procedure to high polymerization temperature, the stepwise heating procedure35 was applied to prepare the powdered PHCTFs successfully in good yield (Scheme 1). The obtained materials, PHCTFs, are insoluble in any common organic solvents, such as DMSO, NMP and tetrahydrofuran (THF), implying good chemical stability. The formation of PHCTFs frameworks was confirmed by Fourier transform infrared (FT-IR). Notably, taking PHCTF-1a for example (Fig. 2), the strong intense characteristic C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N stretching band of the building block (PHPZ-DN) around 2238 cm−1 disappeared, as well as the formation the new characteristic C–N stretching bands for triazine at 1352 cm−1 and 1497 cm−1 (Fig. 2).
N stretching band of the building block (PHPZ-DN) around 2238 cm−1 disappeared, as well as the formation the new characteristic C–N stretching bands for triazine at 1352 cm−1 and 1497 cm−1 (Fig. 2).
As indicated by the X-ray diffraction (XRD) measurements (Fig. S4†), they are amorphous and have less structural order as most reported CTFs materials.26,27 The variations of the of contents of N-heteroatom during high temperature polymerization reaction were detected by elemental analysis (Table S1†), giving relative lower nitrogen content and concomitantly an increasing trend in C/N ratio compared with the theoretically calculated values. Referring to related researches,34 an amount of nitrile decomposition involving C–H and Ar-CN elimination should be taken into account. For the potential broad application of the porous frameworks, the thermal stability of the obtained frameworks should be considered. The thermogravimetric analysis (Fig. S5†) under nitrogen atmosphere of PHCTFs indicates that the polymer skeleton decomposition starts at 520 °C, suggesting its good thermal stability. Some mass amount before the skeleton decomposition may be attributed to the hydrated water and solvents. Surface morphologies of PHCTFs were evaluated by field-emission scanning electron microscopy (FE-SEM) (Fig. 3), showing the similar surface morphology in line with their respective similar porosity characteristics from N2 sorption studies except PHCTF-1c. The local structure was observed through transmission electron microscopy (TEM) (Fig. 3). From the TEM images, various irregular-shaped microparticles can be seen, which are agglomerated and interconnected with a continuous polymeric phase. Such disordered and amorphous structures shown in the TEM imagines are consistent with other amorphous microporous organic polymers.36
 | ||
| Fig. 3 FE-SEM images of (a) PHCTF-1a, (b) PHCTF-1b, (c) PHCTF-1c, (d) PHCTF-2a and (e) PHCTF-2b at 500 nm. TEM images of (a′) PHCTF-1a, (b′) PHCTF-1b, (c′) PHCTF-1c, (d′) PHCTF-2a and (e′) PHCTF-2b. | ||
The porosity parameters and surface areas (Fig. 4) of the PHCTFs were investigated by nitrogen sorption at 77 K, and the pore sizes and distributions were calculated by the nonlocal density functional theory (NLDFT) from their N2 adsorption–desorption isotherms. The isotherms demonstrated rapid nitrogen uptake and high gas uptake at relative pressure (P/P0) less than 0.01, indicative of the characteristics of permanent micropores. Fig. 4 shows that PHCTFs more or less display apparent hysteresis loop, demonstrating the presence of mesopores, especially for PHCTF-1c, which may result from the softness of the organic polymer skeleton and swelling effect should be the dominant reason, resulting in combination of type I and IV characteristics and co-existence of micropores and mesopores of PHCTF-1c. On the one hand, the breakage of the polymer network is more likely to happen because of its organic nature. On the other hand, triazine retrotrimerization should be taken into account and opening triazine cross-links leads to local expansions of the network.23a The apparent surface areas calculated from Brunauer–Emmett–Teller (BET) models within the pressure range of P/P0 = 0.05–0.1 are shown in Table 2. The BET surface areas seem to be associated closely with the polymerization reaction temperature, consequently the highest surface area of PHCTF-1c. A comparison of pore size distribution (PSD) obtained by fitting the nitrogen uptake branch of the isotherms with the non-local density function theory (NLDFT) is illustrated in Fig. 4. Although PHCTF polymer networks exhibit amorphous nature, they have relative uniform micropores with a diameter less than 2 nm except for PHCTF-1c, which PHCTF-1c simultaneously possesses micropores centering at 1.3 nm and mesopores at 5.8 nm.
 | ||
| Fig. 4 Nitrogen adsorption (filled)–desorption (empty) isotherms and NLDFT pore size distribution (PSD) curves of PHCTFs. | ||
| PHCTF | SBETa (m2 g−1) | SLangb (m2 g−1) | Vmicroc (cm3 g−1) | Vtotd (cm3 g−1) | Vmicro/Vtot |
|---|---|---|---|---|---|
| a Calculated BET surface area over the pressure range 0.05–0.1 P/P0.b Langmuir surface area over the pressure range 0.05–0.3 P/P0.c Micropore volume was calculated using the t-plot method.d Total pore volume at P/P0 = 0.9. | |||||
| 1a | 1062 | 1499 | 0.44 | 0.56 | 0.79 |
| 1b | 955 | 1223 | 0.40 | 0.46 | 0.89 |
| 1c | 1845 | 3046 | 0.28 | 1.32 | 0.21 |
| 2a | 731 | 989 | 0.20 | 0.42 | 0.48 |
| 2b | 887 | 1267 | 0.28 | 0.52 | 0.54 |
The above results of porosity parameters and surface areas of PHCTFs indicate that the phthalazinone-based building blocks with a rigid, twisted and non-coplanar structure can be used in the fabrication of the MOPs with relatively high surface areas (maximum to 1845 m2 g−1) and narrow pore size distributions. In this study, the two phthalazinone-based building blocks have similar structures, but not identical geometry conformation (Fig. 1). The dihedral angle (θ1) of the phenyl ring and naphthyridine of PHPZ-DN is about 48°, which is much larger than the dihedral angle (θ2) between thiophene and naphthyridine of THPZ-DN. So the steric conformation of PHPZ-DN may theoretically impart much more advantage to increasing inter-chain porosity from the network interpenetrating of polymer network than the approximately plane V-shaped conformation of THPZ-DN. The fact of the higher BET surface areas of PHCTF-1 than that of PHCTF-2, as well as the pore volumes, also reflects this influence trend. This is because the plane V-shaped conformation of THPZ-DN makes the polymer incline to form two-dimensional network resulted from the possible π–π stacking between layers.37 While the more twisted and non-coplanar conformation of PHPZ-DN is helpful to form the network with more and a little larger micropores, which results in the increased BET surface areas of it. Thus, these results offered us a novel strategy to design and fabricate the phthalazinone-based microporous organic polymers with varied porosity.
Considering the special N,O-heteroatom structure, the relatively high surface areas and the microporous nature of PHCTFs, we were prompted to further explore their property of CO2 capture and separation from other gases. Fig. 5 shows the adsorption isotherms of CO2 at 273 K and 298 K up to a pressure of 1 bar for PHCTFs, and the CO2 uptakes displayed a rapid rise in the initial stage which may result from the favorable interaction between CO2 molecule and the polymer skeleton.37 According to the adsorption behavior curves of the porous networks, PHCTFs, the adsorbed CO2 amount continually increased with the pressure, implying that the adsorption had not reached its equilibrium or saturated state in the investigated pressure range.38 Among the PHCTs, PHCTF-1c exhibited the best performance on the adsorption of CO2 reaching 17.1 wt% at 273 K and 1 bar, which may fall behind that of CPOP-1 and PPF-1 (up to 21.2 (ref. 39) and 26.7 wt% (ref. 6) respectively), but is still comparable to other porous polymers, such as CMPs (4.0–17.0 wt%),40 BLPs (7.4–12.8 wt%)41 and POFs (12.2–18.0 wt%).42 Generally, pores less than 1.0 nm may be more effective towards CO2 capture at low pressure since the molecular size of CO2 is 0.36 nm.43 However, PHCTF-1c still illustrated the highest CO2 uptake for its dominant highest BET surface area among the resultant CTFs. Due to the high affinity of polymer skeleton toward CO2 resulted from the naphthyridine containing abundant electron-rich nitrogen and oxygen atoms, PHCTF-1a, PHCTF-1b, PHCTF-2a and PHCTF-2b also possess satisfactory CO2 uptakes at 273 K and 1 bar, up to 10.2, 9.4, 13.5 and 13.6 wt%, respectively. These uptakes may be inferior to materials such as FCTF-1-600 (24.3 wt%)44 and BILP-4 (23.5 wt%),45 but still notably exceed or are competitive with a lot of organic porous organic polymer networks, such as PAF-3 (8 wt%),15a CMP-1 (9 wt%),46 TBI-2 (11.8 wt%).47 The PHCTF-2 were constructed from THPZ-DN through the same polymerization process as PHCTF-1a and -1b, but exhibited the less BET surface areas in comparison to that of PHCTF-1a and -1b because of the less stereoscopic conformation. Nevertheless, it is noted that the higher CO2 uptakes are obtained for PHCTF-2 with the relatively lower surface areas compared to PHCTF-1a and -1b. First of all, the narrow, uniform pore size distribution and smaller pore size less than 1 nm of PHCTF-2 resulted from the possible π–π stacking between layers may be one of the important factors. As we know, the narrow pore size distribution and small pore size play the same important roles compared to the high surface areas, sometimes even more through improved molecular interaction28b and the trapping-effect.48 Another possibility can be attributed to their higher charge density at the sulfur site of networks (Table S1†) that can facilitate local-dipole/quadrupole interactions with carbon dioxide.49 Through the comparison and analysis of the adsorption performance to CO2 of PHCTFs, it is apparent that the N, O and S atoms in PHCTFs create a high electric field on the network surface leading to a high binding force with quadrupolar CO2 molecules.35 Moreover, the further optimized conformation leads to the narrow, uniform pore size distribution, which contributes to improve the molecular interaction between polar heteroatoms (N, O and S) and CO2 molecules.
To understand and gain further insights from the host–guest interaction, Qst (CO2 isosteric enthalpies) of PHCTFs toward CO2 were calculated from the CO2 adsorption isotherms at 273 K and 298 K in term of Clausius–Clapeyron equation.50 Fig. 6 shows the plots of the functions of adsorbed amount of CO2 to isosteric enthalpies, and PHCTFs exhibited a relative high Qst, approximate or even far exceed 30 kJ mol−1, which are parallel to some reported heterocyclic rings-containing MOPs, such as CMPs (27.0–33.0 kJ mol−1)51 and COFs (15–30 kJ mol−1).52 Though PHCTFs have high enthalpies on low adsorption capacities, the Qst values decrease gradually with the increase of CO2 adsorption, indicating the obvious adsorption behaviors of CO2 molecule on the PHCTFs network skeleton rather than aggregate. PHCTF-2 have relative lower surface areas, however their higher CO2 uptakes than that of PHCTF-1a and -1b might be partly attributed to the high values of heat of adsorption at high coverage, because of the introduction of polar S atom apart from N and O atoms in phthalazinone core. The highest CO2 capacity refers to its high BET surface area, though PHCTF-1c shows the lowest isosteric heat. The virial plots of CO2 for PHCTFs show quite good straight lines (Fig. 6). The interaction between CO2 molecule and pore surface of the polymers networks represents by the first virial coefficients, A0, which are the intercepts of the lines. And according to A0, we can calculate the Henry's law constants (KH) through KH = exp(A0). Thus, the limiting enthalpy of adsorption (Q0), which is the Qst at zero surface CO2 coverage, can be obtained from the plot slope of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KH versus 1/T. From the values at 273 and 298 K shown in Table 3, it can be seen that the A0 and KH values of PHCTF-2s exceed those of PHCTF-1s, the same as the Q0.
KH versus 1/T. From the values at 273 and 298 K shown in Table 3, it can be seen that the A0 and KH values of PHCTF-2s exceed those of PHCTF-1s, the same as the Q0.
 | ||
| Fig. 6 Variations of enthalpies of adsorption with the adsorbed amount of CO2 (up) and virial plots (down) for PHCTFs. | ||
| PHCTF | T (K) | KH (mol g−1 Pa−1) | A0 Ln (mol g−1 Pa−1) | Q0 (kJ mol−1) |
|---|---|---|---|---|
| 1a | 273 | 5.43 × 10−5 | −9.820 | 22.2 |
| 298 | 2.39 × 10−5 | −10.640 | ||
| 1b | 273 | 5.91 × 10−2 | −2.828 | 21.6 |
| 298 | 2.66 × 10−2 | −3.628 | ||
| 1c | 273 | 5.48 × 10−2 | −2.904 | 16.0 |
| 298 | 3.03 × 10−2 | −3.495 | ||
| 2a | 273 | 1.42 × 10−4 | −8.860 | 25.0 |
| 298 | 5.64 × 10−5 | −9.783 | ||
| 2b | 273 | 9.78 × 10−5 | −9.233 | 27.6 |
| 298 | 3.52 × 10−5 | −10.254 |
As the presence of abundant N,O-heteroatoms of phthalazinone core, we suppose that the PHCTFs may possess the potential in gas separation. Besides the CO2 adsorption capacity, the single component gas adsorption isotherms of CH4 and N2 at 273 K up to 1 bar were also measured. The ratios of the Henry law constants were calculated from the initial slopes of the single component gas adsorption isotherms in the low pressure range (Table 3). Then, the ratios were used to estimate the selective performance of the CO2/CH4 and CO2/N2. The adsorption isotherms of CO2, N2 and CH4 at 273 K, 0–1 bar are shown in Fig. 7. The uptakes of CO2 in the adsorption isotherms exhibit considerably higher than N2 and CH4 in the whole pressure range. The selectivity factors of the CO2/N2 of these PHCTFs are up to 52 at 273 K, comparable to zeolitic imidazole frameworks (ZIFs, 20–50)53 and porous polymers, such as APOPs (23.8–43.4)54 and BILPs (59–113).45,55 For the adsorption and selectivity performance of PHCTFs, it can be found that PHCTF-2 simultaneously possesses relatively higher CO2 uptakes and CO2/N2 selectively than PHCTF-1a and -1b. The higher CO2 uptake can be derived from the introduction polar S atom apart from N,O-heteroatoms (Table S1†). The narrow pore size distribution also leads to fine selectivity. Although PHCTF-1c exhibited the highest CO2 uptake, however the more mesoporous area and the wider pore size distribution may be disadvantageous for the recognition of the small CO2 (3.30 Å) from the large N2 (3.64 Å) molecule which encumber its high CO2 uptake simultaneously with excellent selectivity. However, PHCTFs exhibited the different adsorption behaviors toward CH4 and N2. Apparently, the higher uptake of CH4 than N2 for PHCTFs results from the higher critical temperature of CH4 (191 K) than N2 (126 K), because the gas solubility coefficient in a polymer is positively correlated with its critical temperature.56 So, the selectivity of CO2/CH4 may be lower than that of CO2/N2 for PHCTFs, which is up to 12 and comparable to some microporous polymers55a and some ZIFs (Table 4).53
| Sample | CO2 uptakea (wt%) | CO2 selectivity (initial slope) | H2 uptakeb (wt%) | ||
|---|---|---|---|---|---|
| 273 K | 298 K | CO2/N2 | CO2/CH4 | 77 K | |
| a Uptakes for CO2 at 1 bar.b Uptakes for H2 at 1 bar. | |||||
| PHCTF-1a | 10.2 | 6.2 | 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1.12 |
| PHCTF-1b | 9.4 | 6.0 | 49![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
0.66 |
| PHCTF-1c | 17.1 | 10.0 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1.92 |
| PHCTF-2a | 13.5 | 8.6 | 38![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
0.93 |
| PHCTF-2b | 13.6 | 7.2 | 52![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
0.96 |
Hydrogen, with a large specific energy, is a kind of green fuel and suitable candidate to replace gasoline and other fossil fuels. In recent years, microporous organic polymers with high specific surface area, narrow pore distribution and electron-rich systems are of interest to be invaluable in the fields of gas storage, especially for the storage of hydrogen. So the hydrogen physisorption isotherms of PHCTFs were measured at 77 K and 1 bar, and are shown in Fig. 8. Overall, for PHCTF-1c with the highest BET specific surface area, its hydrogen uptake is up to 1.92 wt% at 77 K and 1 bar, which is comparable to Trip(Me)-PIMs57 (1.79 wt%, SBET = 1760 m2 g−1) and P(Fe-TTPP)58 (∼1.5 wt%, SBET = 1248 m2 g−1) under the same condition. Also, this result is satisfactory and superior to some other MOPs with higher specific surface area, such as PPN-3 (ref. 16a) (1.58 wt%, SBET = 2840 m2 g−1), PAF-1 (ref. 15b) (1.50 wt%, SBET = 5600 m2 g−1) and COF-102 (ref. 59) (1.2 wt%, SBET = 3620 m2 g−1) at 77 K and 1 bar. These indicate that the rational design and selection of the building blocks are very important to afford excellent capacity of the hydrogen. On the one hand, the high specific surface area is very necessary. On the other hand, the molecular structure and chemical nature of the building blocks still play crucial roles.60
Conclusions
The phthalazinone-based microporous organic polymer networks, PHCTFs, were constructed by ionothermal reaction with relatively high specific surface area and uniform pore size distribution. The chemical structures of PHCTFs have been well confirmed by FTIR and elemental analysis. The surface morphologies were evaluated by field-emission scanning electron microscopy (FE-SEM), and the local structure was observed through transmission electron microscopy (TEM). The measurements of sorption of nitrogen at 77 K showed that the polymers have large BET surface up to 1845 m2 g−1. Besides, their CO2 adsorption capacities are up to 17.1 wt% (273 K/1 bar), and the polymers possess uptake of hydrogen (1.92 wt%, 77 K/1 bar). Also, PHCTFs exhibit good selectivity of CO2/N2 (52, 273 K/1 bar) and CO2/CH4 (12, 273 K/1 bar). These results indicate that the rigid, twisted and asymmetric phthalazinone structure can be used to construct satisfactory PHCTFs with good adsorption and separation properties. At the same time, the pore size distributions of the afforded PHCTFs can be adjusted through the change of the conformation of phthalazinone. The introduction of polar electron-rich S atom can further increase the CO2 uptake of PHCTFs apart from abundant N,O-heteroatoms of phthalazinone structure. These results indicate that the obtained the phthalazinone-based microporous organic polymer networks, PHCTFs, are promising functional materials for gas separation and storage.Acknowledgements
The present research was financially supported by National Natural Science Foundation of China (No. 51473025) and Chinese Universities Scientific Fund (DUT13LK20). The authors acknowledge the High Performance Computing Centre of Dalian University of Technology for providing computational resources which have contributed to the research results.Notes and references
- K. S. Sing, Pure Appl. Chem., 1985, 57, 603–619 CrossRef CAS.
- Y. Xu, S. Jin, H. Xu, A. Nagai and D. Jiang, Chem. Soc. Rev., 2013, 42, 8012–8031 RSC.
- R. M. Barrer, Zeolites, 1981, 1, 130–140 CrossRef CAS.
- Z. ALOthman, Materials, 2012, 5, 2874–2902 CrossRef CAS.
- (a) S. Ma and H.-C. Zhou, Chem. Commun., 2010, 46, 44–53 RSC; (b) J. R. Long and O. M. Yaghi, Chem. Soc. Rev., 2009, 38, 1211214 RSC; (c) H. Li, M. Eddaoudi, M. O'Keeffe and O. M. Yaghi, Nature, 1999, 402, 276–279 CrossRef CAS.
- Y. Zhu, H. Long and W. Zhang, Chem. Mater., 2013, 25, 1630–1635 CrossRef CAS.
- (a) J. Germain, J. M. J. Fréchet and F. Svec, Small, 2009, 5, 1098–1111 CrossRef CAS PubMed; (b) S. Xu, Y. Luo and B. Tan, Macromol. Rapid Commun., 2013, 34, 471–484 CrossRef CAS PubMed.
- X. Zhang, J. Lu and J. Zhang, Chem. Mater., 2014, 26, 4023–4029 CrossRef CAS.
- (a) J. Y. Lee, C. D. Wood, D. Bradshaw, M. J. Rosseinsky and A. I. Cooper, Chem. Commun., 2006, 2670–2672 RSC; (b) C. D. Wood, B. Tan, A. Trewin, F. Su, M. J. Rosseinsky, D. Bradshaw, Y. Sun, L. Zhou and A. I. Cooper, Adv. Mater., 2008, 20, 1916–1921 CrossRef CAS.
- H. B. Park, C. H. Jung, Y. M. Lee, A. J. Hill, S. J. Pas, S. T. Mudie, E. Van Wagner, B. D. Freeman and D. J. Cookson, Science, 2007, 318, 254–258 CrossRef CAS PubMed.
- (a) X. Du, Y. Sun, B. Tan, Q. Teng, X. Yao, C. Su and W. Wang, Chem. Commun., 2010, 46, 970–972 RSC; (b) J. Schmidt, J. Weber, J. D. Epping, M. Antonietti and A. Thomas, Adv. Mater., 2009, 21, 702–705 CrossRef CAS; (c) J. Luo, X. Zhang and J. Zhang, ACS Catal., 2015, 5, 2250–2254 CrossRef CAS.
- (a) Y. Wang, N. B. McKeown, K. J. Msayib, G. A. Turnbull and I. D. W. Samuel, Sensors, 2011, 11, 2478–2487 CrossRef CAS PubMed; (b) J. C. Thomas, J. E. Trend, N. A. Rakow, M. S. Wendland, R. J. Poirier and D. M. Paolucci, Sensors, 2011, 11, 3267–3280 CrossRef PubMed.
- (a) X. Feng, X. Ding and D. Jiang, Chem. Soc. Rev., 2012, 41, 6010–6022 RSC; (b) A. P. Cote, A. I. Benin, N. W. Ockwig, M. O'Keeffe, A. J. Matzger and O. M. Yaghi, Science, 2005, 310, 1166–1170 CrossRef CAS PubMed; (c) H. M. El-Kaderi, J. R. Hunt, J. L. Mendoza-Cortes, A. P. Cote, R. E. Taylor, M. O'Keeffe and O. M. Yaghi, Science, 2007, 316, 268–272 CrossRef CAS PubMed.
- (a) F. Vilela, K. Zhang and M. Antonietti, Energy Environ. Sci., 2012, 5, 7819–7832 RSC; (b) J. X. Jiang, F. Su, A. Trewin, C. D. Wood, N. L. Campbell, H. Niu, C. Dickinson, A. Y. Ganin, M. J. Rosseinsky, Y. Z. Khimyak and A. I. Cooper, Angew. Chem., Int. Ed., 2007, 46, 8574–8578 CrossRef CAS PubMed.
- (a) T. Ben, C. Pei, D. Zhang, J. Xu, F. Deng, X. Jing and S. Qiu, Energy Environ. Sci., 2011, 4, 3991–3999 RSC; (b) T. Ben, H. Ren, S. Ma, D. Cao, J. Lan, X. Jing, W. Wang, J. Xu, F. Deng, J. M. Simmons, S. Qiu and G. Zhu, Angew. Chem., Int. Ed., 2009, 48, 9457–9460 CrossRef CAS PubMed; (c) Y. Yuan, F. Sun, H. Ren, X. Jing, W. Wang, H. Ma, H. Zhao and G. Zhu, J. Mater. Chem., 2011, 21, 13498–13502 RSC.
- (a) W. Lu, D. Yuan, D. Zhao, C. I. Schilling, O. Plietzsch, T. Muller, S. Bräse, J. Guenther, J. Blümel, R. Krishna, Z. Li and H.-C. Zhou, Chem. Mater., 2010, 22, 5964–5972 CrossRef CAS; (b) D. Yuan, W. Lu, D. Zhao and H.-C. Zhou, Adv. Mater., 2011, 23, 3723–3725 CrossRef CAS PubMed.
- (a) P. M. Budd, B. S. Ghanem, S. Makhseed, N. B. McKeown, K. J. Msayib and C. E. Tattershall, Chem. Commun., 2004, 230–231 RSC; (b) N. B. McKeown and P. M. Budd, Chem. Soc. Rev., 2006, 35, 675–683 RSC; (c) N. B. McKeown, B. Gahnem, K. J. Msayib, P. M. Budd, C. E. Tattershall, K. Mahmood, S. Tan, D. Book, H. W. Langmi and A. Walton, Angew. Chem., Int. Ed., 2006, 45, 1804–1807 CrossRef CAS PubMed.
- (a) J. Germain, J. Hradil, J. M. J. Fréchet and F. Svec, Chem. Mater., 2006, 18, 4430–4435 CrossRef CAS; (b) M. P. Tsyurupa and V. A. Davankov, React. Funct. Polym., 2006, 66, 768–779 CrossRef CAS; (c) C. D. Wood, B. Tan, A. Trewin, H. Niu, D. Bradshaw, M. J. Rosseinsky, Y. Z. Khimyak, N. L. Campbell, R. Kirk, E. Stöckel and A. I. Cooper, Chem. Mater., 2007, 19, 2034–2048 CrossRef CAS; (d) O. W. Webster, F. P. Gentry, R. D. Farlee and B. E. Smart, Macromol. Symp., 1992, 54, 477–482 CrossRef.
- (a) M. Rose, W. Bohlmann, M. Sabo and S. Kaskel, Chem. Commun., 2008, 21, 2462–2464 RSC; (b) S. Yuan, S. Kirklin, B. Dorney, D.-J. Liu and L. Yu, Macromolecules, 2009, 42, 1554–1559 CrossRef CAS; (c) O. K. Farha, A. M. Spokoyny, B. G. Hauser, Y.-S. Bae, S. E. Brown, R. Q. Snurr, C. A. Mirkin and J. T. Hupp, Chem. Mater., 2009, 21, 3033–3035 CrossRef CAS; (d) F. J. Uribe-Romo, J. R. Hunt, H. Furukawa, C. Klöck, M. O'Keeffe and O. M. Yaghi, J. Am. Chem. Soc., 2009, 131, 4570–4571 CrossRef CAS PubMed.
- (a) T. Tozawa, J. T. Jones, S. I. Swamy, S. Jiang, D. J. Adams, S. Shakespeare, R. Clowes, D. Bradshaw, T. Hasell, S. Y. Chong, C. Tang, S. Thompson, J. Parker, A. Trewin, J. Bacsa, A. M. Slawin, A. Steiner and A. I. Cooper, Nat. Mater., 2009, 8, 973–978 CrossRef CAS PubMed; (b) Y. Jin, B. A. Voss, R. McCaffrey, C. T. Baggett, R. D. Noble and W. Zhang, Chem. Sci., 2012, 3, 874–877 RSC.
- A. P. Cote, H. M. El-Kaderi, H. Furukawa, J. R. Hunt and O. M. Yaghi, J. Am. Chem. Soc., 2007, 129, 12914–12915 CrossRef CAS PubMed.
- P. Pandey, A. P. Katsoulidis, I. Eryazici, Y. Wu, M. G. Kanatzidis and S. T. Nguyen, Chem. Mater., 2010, 22, 4974–4979 CrossRef CAS.
- (a) P. Kuhn, A. Thomas and M. Antonietti, Macromolecules, 2009, 42, 319–326 CrossRef CAS; (b) P. Kuhn, A. Forget, J. Hartmann, A. Thomas and M. Antonietti, Adv. Mater., 2009, 21, 897–901 CrossRef CAS; (c) P. Kuhn, A. Forget, D. Su, A. Thomas and M. Antonietti, J. Am. Chem. Soc., 2008, 130, 13333–13337 CrossRef CAS PubMed; (d) P. Kuhn, M. Antonietti and A. Thomas, Angew. Chem., Int. Ed., 2008, 47, 3450–3453 CrossRef CAS PubMed.
- Y. P. Tang, H. Wang and T. S. Chung, ChemSusChem, 2015, 8, 138–147 CrossRef CAS PubMed.
- X. Zhu, C. Tian, S. M. Mahurin, S. H. Chai, C. Wang, S. Brown, G. M. Veith, H. Luo, H. Liu and S. Dai, J. Am. Chem. Soc., 2012, 134, 10478–10484 CrossRef CAS PubMed.
- A. Bhunia, I. Boldog, A. Möller and C. Janiak, J. Mater. Chem. A, 2013, 1, 14990–14999 CAS.
- S. Hug, M. E. Tauchert, S. Li, U. E. Pachmayr and B. V. Lotsch, J. Mater. Chem., 2012, 22, 13956–13964 RSC.
- (a) Y. Liu, S. Wu, G. Wang, G. Yu, J. Guan, C. Pan and Z. Wang, J. Mater. Chem. A, 2014, 2, 7795–7801 RSC; (b) Q. Chen, M. Luo, P. Hammershoj, D. Zhou, Y. Han, B. W. Laursen, C. G. Yan and B. H. Han, J. Am. Chem. Soc., 2012, 134, 6084–6087 CrossRef CAS PubMed; (c) H. Lim, M. C. Cha and J. Y. Chang, Macromol. Chem. Phys., 2012, 213, 1385–1390 CrossRef CAS.
- (a) G. Yu, C. Liu, J. Wang, J. Xu and X. Jian, Polym. Int., 2010, 59, 1233–1239 CrossRef CAS; (b) G. Yu, C. Liu, J. Wang, G. Li, Y. Han and X. Jian, Polymer, 2010, 51, 100–109 CrossRef CAS; (c) G. Yu, J. Wang, C. Liu, E. Lin and X. Jian, Polymer, 2009, 50, 1700–1708 CrossRef CAS; (d) G. Yu, C. Liu, H. Zhou, J. Wang, E. Lin and X. Jian, Polymer, 2009, 50, 4520–4528 CrossRef CAS.
- S. Yoshida and A. S. Hay, Macromolecules, 1995, 28, 2579–2581 CrossRef CAS.
- Q. Liang, P. Liu, C. Liu, X. Jian, D. Hong and Y. Li, Polymer, 2005, 46, 6258–6265 CrossRef CAS.
- J. Y. Wang, G. X. Liao, C. Liu and X. G. Jian, J. Polym. Sci., Part A: Polym. Chem., 2004, 42, 6089–6097 CrossRef CAS.
- S. A. Weissman, D. Zewge and C. Chen, J. Org. Chem., 2005, 70, 1508–1510 CrossRef CAS PubMed.
- A. Bhunia, V. Vasylyeva and C. Janiak, Chem. Commun., 2013, 49, 3961–3963 RSC.
- S. Wu, Y. Liu, G. Yu, J. Guan, C. Pan, Y. Du, X. Xiong and Z. Wang, Macromolecules, 2014, 47, 2875–2882 CrossRef CAS.
- Y. C. Zhao, L. M. Zhang, T. Wang and B. H. Han, Polym. Chem., 2014, 5, 614–621 RSC.
- G. Li and Z. Wang, Macromolecules, 2013, 46, 3058–3066 CrossRef CAS.
- Y. Yang, Q. Zhang, S. Zhang and S. Li, Polymer, 2013, 54, 5698–5702 CrossRef CAS.
- Q. Chen, M. Luo, P. Hammershøj, D. Zhou, Y. Han, B. W. Laursen, C.-G. Yan and B.-H. Han, J. Am. Chem. Soc., 2012, 134, 6084–6087 CrossRef CAS PubMed.
- (a) A. Thomas, Angew. Chem., 2010, 49, 8328–8344 CrossRef CAS PubMed; (b) A. I. Cooper, Adv. Mater., 2009, 21, 1291–1295 CrossRef CAS.
- (a) K. T. Jackson, M. G. Rabbani, T. E. Reich and H. M. El-Kaderi, Polym. Chem., 2011, 2, 2775–2777 RSC; (b) T. E. Reich, S. Behera, K. T. Jackson, P. Jena and H. M. El-Kaderi, J. Mater. Chem., 2012, 22, 13524–13528 RSC.
- A. P. Katsoulidis and M. G. Kanatzidis, Chem. Mater., 2011, 23, 1818–1824 CrossRef CAS.
- D. Cazorla-Amorós, J. Alcañiz-Monge and A. Linares-Solano, Langmuir, 1996, 12, 2820–2824 CrossRef.
- Y. Zhao, K. X. Yao, B. Teng, T. Zhang and Y. Han, Energy Environ. Sci., 2013, 6, 3684–3692 CAS.
- M. G. Rabbani and H. M. El-Kaderi, Chem. Mater., 2012, 24, 1511–1517 CrossRef CAS.
- J. X. Jiang, F. Su, A. Trewin, C. D. Wood, H. Niu, J. T. A. Jones, Y. Z. Khimyak and A. I. Cooper, J. Am. Chem. Soc., 2008, 130, 7710–7720 CrossRef CAS PubMed.
- Y. C. Zhao, Q. Y. Cheng, D. Zhou, T. Wang and B.-H. Han, J. Mater. Chem., 2012, 22, 11509–11514 RSC.
- J. Germain, F. Svec and J. M. J. Fréchet, Chem. Mater., 2008, 20, 7069–7076 CrossRef CAS.
- M. Y. Jiang, Q. Wang, Q. Chen, X. M. Hu, X.-L. Ren, Z. H. Li and B. H. Han, Polymer, 2013, 54, 2952–2957 CrossRef CAS.
- V. Krungleviciute, L. Heroux, A. D. Migone, C. T. Kingston and B. Simard, J. Phys. Chem. B, 2005, 109, 9317–9320 CrossRef CAS PubMed.
- R. Dawson, D. J. Adams and A. I. Cooper, Chem. Sci., 2011, 2, 1173–1177 RSC.
- S. Y. Ding and W. Wang, Chem. Soc. Rev., 2013, 42, 548–568 RSC.
- R. Banerjee, H. Furukawa, D. Britt, C. Knobler, M. O'Keeffe and O. M. Yaghi, J. Am. Chem. Soc., 2009, 131, 3875–3877 CrossRef CAS PubMed.
- W. C. Song, X. K. Xu, Q. Chen, Z. Z. Zhuang and X. H. Bu, Polym. Chem., 2013, 4, 4690–4696 RSC.
- (a) M. G. Rabbani and H. M. El-Kaderi, Chem. Mater., 2011, 23, 1650–1653 CrossRef CAS; (b) M. G. Rabba, T. E. Reich and H. M. El-Kaderi, Abstr. Pap. Am. Chem. Soc., 2012, 243, 1156 Search PubMed.
- V. M. Shah, B. J. Hardy and S. A. Stern, J. Polym. Sci., Part B: Polym. Phys., 1993, 31, 1185 CrossRef.
- B. S. Ghanem, M. Hashem, K. D. M. Harris, K. J. Msayib, M. Xu, P. M. Budd, N. Chaukura, D. Book, S. Tedds, A. Walton and N. B. McKeown, Macromolecules, 2010, 43, 5287–5294 CrossRef CAS.
- J. Xia, S. Yuan, Z. Wang, S. Kirklin, B. Dorney, D. J. Liu and L. Yu, Macromolecules, 2010, 43, 3325–3330 CrossRef CAS.
- H. Furukawa and O. M. Yaghi, J. Am. Chem. Soc., 2009, 131, 8875–8883 CrossRef CAS PubMed.
- Q. Chen, J. X. Wang, Q. Wang, N. Bian, Z. H. Li, C. G. Yan and B. H. Han, Macromolecules, 2011, 44, 7987–7993 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: This section contains ten figures, including the TGA curves of aromatic cyanide monomers and PHCTFs, 1H-NMR spectra of aromatic cyanide monomers, XRD patterns, elemental analysis and adsorption selectivity of CO2 over CH4 and N2 at 273 and 298 K of PHCTFs. See DOI: 10.1039/c5ra23148e |
| This journal is © The Royal Society of Chemistry 2016 |