Efficient photo-degradation of dyes using CuWO4 nanoparticles with electron sacrificial agents: a combination of experimental and theoretical exploration
Abstract
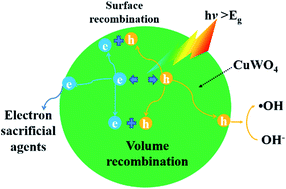
CuWO4 is a promising photocatalytic material, responding in the visible light range, to enhance the utilization of solar energy. Here, CuWO4 nanoparticles have been synthesized via a polyol-mediated synthesis method and subsequently characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), UV-Vis Spectrophotometery combined with theoretical density functional theory (DFT) calculations. For the as-prepared CuWO4 samples, a strong adsorption capacity for the organic pollutant MB rather than photodegradation has been observed. The first-principle calculation with Heyd–Scuseria–Ernzerhof (HSE) screened coulomb hybrid functional results indicate that localization of hybridization of O 2p-orbitals and Cu 3d-orbitals, large electron effective mass and more positive conduction band edge of CuWO4 lead to low carrier mobility and thus the high recombination of excited carriers. Meanwhile, the optical absorption spectrum of experimental observation is consistent with theoretical calculations of pristine CuWO4, demonstrating few defects inhibiting light absorption. To avoid the high rate of recombination of the excited carriers, electron sacrificial agents (H2O2, Na2S2O8) are utilized to suppress the recombination. The photocatalytic activity is thus largely improved.

 Please wait while we load your content...
Please wait while we load your content...