Theoretical study of the BINOL–zinc complex-catalyzed asymmetric inverse-electron-demand imino Diels–Alder reaction: mechanism and stereochemistry†
Weiyi Li*a,
Na Yangb and
Yajing Lva
aSchool of Science, Xihua University, Chengdu, 610039, Sichuan, P. R. China. E-mail: weiyili@mail.xhu.edu.cn; Fax: +86-28-87727663; Tel: +86-28-87727663
bInstitute of Nuclear Physics and Chemistry, China Academy of Engineering Physics, Mianyang, 621900, P. R. China
First published on 27th October 2015
Abstract
The mechanism and stereochemistry of an inverse electron-demand imino Diels–Alder (IEDIDA) reaction between a cyclic imine and an electron-poor chromone-derived diene catalyzed by a chiral BINOL–zinc complex have been studied using a combination of DFT calculations, chemical reactivity indices and distortion/interaction analyses. The calculations reveal that the coordination of Lewis acid zinc catalyst to the ester C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group of the electrophilic diene significantly lowers the energy barriers of the cycloaddition reaction by an increase of the electrophilic character of the diene. Herein, both the catalytic mechanism and stereochemistry of the cycloadduct depend on the configuration of the diene. An energy-favored stepwise mechanism is adopted when the ester C
O group of the electrophilic diene significantly lowers the energy barriers of the cycloaddition reaction by an increase of the electrophilic character of the diene. Herein, both the catalytic mechanism and stereochemistry of the cycloadduct depend on the configuration of the diene. An energy-favored stepwise mechanism is adopted when the ester C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group of the diene takes the trans configuration. This configuration allows a stabilizing interaction to form between the zinc center of the catalyst and the phenyl moiety of the dienophile and favors the steric discrimination from the naphthalene ring of the BINOL ligand at the ring-closure transition state that is rate-determining and stereo-controlling for the entire cycloaddition process. The electrophilic/nucleophilic interaction defines the most favored alignment between the dienophile and the diene–catalyst complex, which overwhelms the distortion of these fragments and realizes the exo-selectivity of the cycloadduct.
O group of the diene takes the trans configuration. This configuration allows a stabilizing interaction to form between the zinc center of the catalyst and the phenyl moiety of the dienophile and favors the steric discrimination from the naphthalene ring of the BINOL ligand at the ring-closure transition state that is rate-determining and stereo-controlling for the entire cycloaddition process. The electrophilic/nucleophilic interaction defines the most favored alignment between the dienophile and the diene–catalyst complex, which overwhelms the distortion of these fragments and realizes the exo-selectivity of the cycloadduct.
1. Introduction
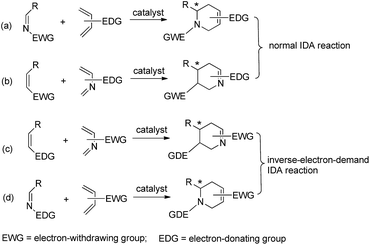
The imino Diels–Alder reaction (IDA),1 initiated by a dienophile (as an imine derivative) or diene (as a 1- or 2-azadiene), is a powerful synthetic route for the construction of various chiral nitrogen-containing six-membered heterocycles, such as piperidines, tetrahydroquinolines, quinolines and so on (Scheme 1). The resultant aza-heterocycles and their derivatives can be widely used as starting materials or intermediates in the total synthesis of many natural products and complex biologically active molecules.2 Consequently, much effort has been devoted to the development of the asymmetric IDA reaction in order to obtain chiral aza-heterocycles with high yields and enantioselectivities.Experimentally, a number of the normal IDA reactions between the electron-poor or electron-neutral imines and the electron-rich dienes have been realized. For example, a series of chiral Lewis acids catalysts combined with the BINOL ligands and a variety of metals (Zr,3a–e Nb,3f–i and Zn3j–k) were found to be effective in the catalytic asymmetric IDA reactions of electron-poor imines with Danishefsky's diene. The metal center of the catalyst serves as a Lewis acid to activate the nitrogen center of the imine and increase the electrophilicity of the dienophile. The 3,3′-positions of the BINOL ring provides the asymmetric environment for the enantioselective cycloaddition of dienes. Shortly after these reports were published, a variety of chiral Lewis catalytic systems like BINAP–Cu,4a,bN,P-oligopeptide–Ag,4c VAPOL–boron,4d and N,N′-α-dioxide–metals4e–i were developed for the IDA reaction between dienophile imines and Danishefsky's diene, which aimed at increasing the enantioselectivities of cycloadducts, decreasing of catalysts loading and expanding the scope of the substrates. In addition, as one kind of useful and powerful catalysts, many environmental-friendly bifunctional organocatalysts were also successfully applied to enhance the enantioselective IDA reactions of imino-dienophiles with dienes. Ohsawa and co-workers5a reported the first example of L-proline-catalyzed IDA reaction of cyclic imines and methyl vinyl ketones, which leads to the production of the precursors of natural indole alkaloids with good yields and excellent enantioselectivities. They also proposed the final cycloadduct was obtained via an enamine active intermediate generated from enone and proline in the Mannich-type fashion.5b Afterward, the modification of the amino acid scaffold on the proline catalyst was performed in order to increase the solubility of the catalyst and expand the scope of the reaction. Differing from the proline-catalyzed IDA reaction, the activation of the dienophile imines by chiral Brønsted phosphoric acids occurs primarily via hydrogen-bonding interactions. Akiyama et al.6 discovered that BINOL-derived phosphoric prove as efficient catalysts for the IDA reaction of imines with both Danishefsky's diene and Brassard's diene. The IDA reaction of an imine with the non-activated diene cyclohexenone was also achieved using a cooperative catalyst combined with the chiral phosphoric acid and acetic acid, in which acetic acid played the essential role in accelerating the reaction rate.7
In addition to the normal IDA reaction, the asymmetric inverse electron-demand imino Diels–Alder (IEDIDA) reaction between the electron-deficient 2-aza-butadiene and an electron-rich dienophile was also investigated in the presence of chiral Lewis acids8 and organocatalysts.9 The catalytic methodologies can be classified into three categories: (i) activation of the diene by Lewis acidic metal complexes or organocatalysts; (ii) activation of dienophiles via the generation of an enamine or enolate intermediate; and (iii) the dual activation strategy of both the dienophiles and of dienes. To the best of our knowledge, aza-dienes were employed as electron-deficient component in the almost reported cases.10 Examples of IEDIDA reaction between electron-rich imines and electron-deficient dienes are rather rare.
Very recently, Kumar and Waldmann reported the first case of an asymmetric IEDIDA reaction that is the cycloaddition of the electron-rich cyclic imine 1a with the chromone-derived diene 2a in the presence of the Lewis acid zinc and (R)-BINOL ligand (Scheme 2).11 The target indoloquinolizine 4a was obtained through the intermediary cycloadduct 3a with moderate yield (51%) and high an enantiomeric excess (ee) value (93%). The authors also suggested a dual activation catalytic model to explain the observed enantioselectivity. In this model, both the nitrogen atom of the imine moiety and the oxygen atom of the vinylogous ester diene are coordinated with the zinc center. However, this model seems not to fit within the three catalytic categories as mentioned previously, because the coordination of the nitrogen atom of the imine moiety with the Lewis acid center might decrease the electron density of the dienophile and disfavor the cycloaddition with an electron-deficient diene. Furthermore, the detailed reaction mechanism, the origin of the regioselectivity, the endo/exo selectivity and the enantioselectivity of the reaction are altogether unclear. Herein, we carried out a comprehensive theoretical investigation on the titled reaction to further understand the mechanism and the stereochemistry of this novel reaction at the molecular level. The catalytic role and the chiral introduction of the BINOL–Zn complex were examined by a combination of DFT calculations, reactivity indices and distortion/interaction analyses, which are expected to provide useful information for the design of more and efficient asymmetric IEDIDA reactions.
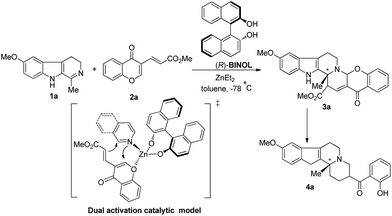 | ||
| Scheme 2 Asymmetric IEDIDA reaction between the imine dienophile 1a and chromone-derived diene 2a in the presence of Lewis acid zinc and (R)-BINOL ligand. | ||
2. Computational methods
Among the correlated density functional methods, the hybrid meta exchange–correlation M05-2X, developed by Zhao and Truhlar, performs well in main group thermochemistry, kinetics, and noncovalent interactions.12 Particularly, the M05-2X method also gives accurate geometry optimization and energetic calculations for zinc compounds, which has been demonstrated in previous theoretical investigations.13 Accordingly, the geometry optimization of all reactants, products, intermediates (IMs) and transition states (TSs) involved in the present system was carried out using the M05-2X functional with the 6-31G(d) basis set.14 The vibrational frequencies were calculated at the same level to characterize each optimized structure is an intermediate (no imaginary frequency) or a transition state (unique imaginary frequency) and obtain the thermal corrections at 298 K. Intrinsic reaction coordinates (IRC)15 scans were conducted when necessary to ensure the transition state correctly connects the two relevant minima. To consider the solvent effect, single-point energy calculation in toluene (experimentally used) was performed on the gas-optimized structure with the SMD16 continuum solvation model at the M05-2X/6-311+G(d,p) level. Unless otherwise specified, the relative free energies (ΔG) including the thermal corrections in the gas phase and the single-point energies in the solvent were reported.In addition, exhaustive theoretical studies on a series of DA reactions by Domingo et al.17 shown that the reactivity indices defined within the conceptual DFT framework were very useful tools to understand the reaction mechanism and explain the chem- and regioselectivity in the polar cycloaddition reactions. Accordingly, the electronic chemical potential μ, chemical hardness η, and the global electrophilicity ω and nucleophilicity N were calculated, respectively, at the ground state (GS) of the molecules.18,19 The local reactivity electrophilicity ωk and nucleophilicity indices Nk for the reactants and molecular complex were also calculated based on the Parr function, recently proposed by Domingo,20 using the following equations,
| ωk = ωPk+ | (1) |
| Nk = NPk− | (2) |
The global electron density transfer (GEDT)21 is a global flux of electron density taking place from the nucleophile to the electrophile, which could be one of the key factors in determining the activation energy. This value at the TS was computed by sharing the nature charge through natural bond orbital (NBO)22 analysis between the nucleophilic and the electrophilic frameworks.
To gain an insight into the origin of the endo/exo selectivity in the reaction, distortion/interaction analysis,23 recommended by Houk and co-workers in cycloaddition reactions, was carried out on the corresponding the TSs. By definition, the activation energy (ΔE≠) is decomposed into the distortion energy (ΔE≠dist) and the interaction energy (ΔE≠int), ΔE≠ = ΔE≠dist + ΔE≠int. The ΔE≠dist is the energy required to distort the reactants and catalysts into the geometry they have in the transition states and ΔE≠dist is a negative value which indicates favorable interaction between the reactants and catalysts.
All DFT calculations were carried out with the Gaussian 09 software package.24 The three-dimensional molecular structures were drawn using the CYLVIEW program.25
3. Results and discussion
3.1. Background reaction without catalyst
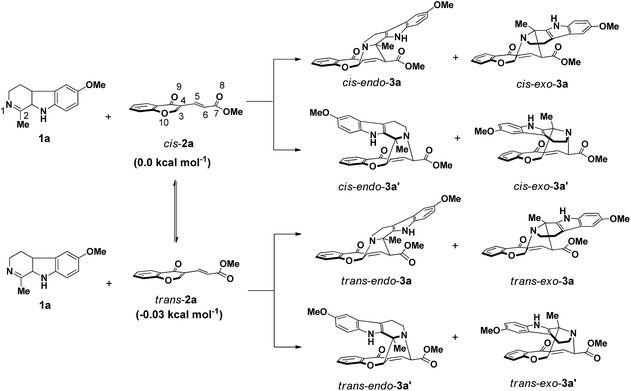
For the chromone-derived diene 2a, the C7![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O8 double bond of the ester group might adopt a cis or trans configuration relative to the C5
O8 double bond of the ester group might adopt a cis or trans configuration relative to the C5![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C6 double bond (Scheme 3). In this work, the cycloaddition of imine dienophile 1a with both cis- and trans-2a was simulated because the free energy difference between these two isomers is less than 0.1 kcal mol−1. Moreover, the reaction mechanism and the stereochemical outcome of this reaction were also found to be very sensitive to the configuration of the diene (vide infra). Hence, four possible reaction channels (cis-endo, cis-exo, trans-endo, and trans-exo) are in principle available in the background reaction, resulting in eight different products: cis or trans isomer for each of the two regio-isomers (3a and 3a′), and an endo and exo approach for each of the two regio-isomers. To make a concise expression, trans reaction channels will be discussed with emphasis in what follows. The details along cis reaction channels are provided in the ESI (Section S2.1†).
C6 double bond (Scheme 3). In this work, the cycloaddition of imine dienophile 1a with both cis- and trans-2a was simulated because the free energy difference between these two isomers is less than 0.1 kcal mol−1. Moreover, the reaction mechanism and the stereochemical outcome of this reaction were also found to be very sensitive to the configuration of the diene (vide infra). Hence, four possible reaction channels (cis-endo, cis-exo, trans-endo, and trans-exo) are in principle available in the background reaction, resulting in eight different products: cis or trans isomer for each of the two regio-isomers (3a and 3a′), and an endo and exo approach for each of the two regio-isomers. To make a concise expression, trans reaction channels will be discussed with emphasis in what follows. The details along cis reaction channels are provided in the ESI (Section S2.1†).
Initially, the reactivity indices of the reactants were analyzed (Table 1). The electronic chemical potential of imine 1a (μ = −3.7 eV) is found to be higher than that of trans-2a (μ = −4.8 eV). Indeed, 1a with high global nucleophilicity index (N = 4.0 eV) and low global electrophilicity index (ω = 1.0 eV) can be classified as a strong nucleophile (N > 3.0).26 For diene trans-2a, the global nucleophilicity index (N = 2.8 eV) is also larger than its electrophilicity index (ω = 1.6 eV). However, trans-2a can only serve as the electrophile in this cycloaddition reaction. Consequently, the GEDT will flux from the nucleophile 1a to the electrophile trans-2a during the cycloaddition process. These results confirm that this [4 + 2] cycloaddition reaction requires inverse-electron-demand. Additionally, the computed Parr Fukui functions based on the atomic spin density suggest a higher nucleophilic site at the N1 position of the imine group, Pk− = 0.27, than the C2 atom with Pk− = 0.13. In trans-2a, the conjugated C3 position is predicted to be more electrophilical than the C6 position, as verified by the higher Parr Fukui functions electrophilic Pk+ (0.57 for the C3 atom a 0.10 for the C6 atom). As a consequence of the activity differences between the reaction sites, the relative free energies of TSs trans-endo-TSa, trans-exo-TS1a and trans-exo-TS2a, corresponding to the construction of N1–C3 and C2–C6 bonds, are less energetic than TSs trans-endo-TSb and trans-exo-TSb, leading to the formation of N1–C6 and C2–C3 bonds (Fig. 1). Therefore, there is a pronounced regioselectivity for this cycloaddition reaction, yielding the cycloadduct trans-endo-3a or trans-exo-3a as the major product. The calculations well reproduced the experimental observations that trans-endo-3a′ and trans-exo-3a′ were not yielded in this cycloaddition reaction.11 Along the more favourable region-isomeric reaction channel, the trans-endo stereoisomer is generated via a one-step two-stage mechanism. At TS trans-endo-TSa, the length of the N1–C3 forming bond (1.812 Å) is shorter than that of the C2–C6 forming bond (2.798 Å). This large difference (Δd = 0.986 Å) between the lengths of the two forming bonds suggests that TS trans-endo-TSa correspond with concerted but highly asynchronous bond-formation process where the N1–C3 forming bond is being formed in a larger extension than the C2–C6 one. The GEDT from the nucleophilic 1a to the electron-deficient trans-2a at TS trans-endo-TSa is 0.24 e, indicating this cycloaddition process belongs to a polar DA reaction (0.15 e < GEDT < 0.40 e).17f The comparison of the GEDT values with the unfavorable TSs trans-endo-TSb and trans-exo-TSb (0.01 e) suggests that the GEDT is the main factor controlling the reaction rate of the DA reactions.17f With respect to separate reactants, the activation energy (ΔE≠), activation enthalpy (ΔH≠) and activation free energy (ΔG≠) barrier for TS trans-endo-TSa is 8.4, 7.6, and 24.4 kcal mol−1, repetitively (Table S2†). These results can well account for the reaction temperature (80 °C) required for the uncatalyzed reaction.11
| Species | μ [eV] | η [eV] | ω [eV] | N [eV] | P k + (Mulliken) | ω k [eV] | P k − (Mulliken) | N k [eV] | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C3 | C6 | C3 | C6 | N1 | C2 | N1 | C2 | |||||
| 1a | −3.7 | 6.6 | 1.0 | 4.0 | — | — | 0.27 | 0.13 | 1.1 | 0.5 | ||
| COM-I | −4.7 | 6.8 | 1.6 | 3.0 | — | — | 0.27 | 0.03 | 1.0 | 0.3 | ||
| COM-II | −4.8 | 6.1 | 1.7 | 3.2 | — | — | 0.21 | 0.36 | 0.7 | 1.2 | ||
| trans-2a | −4.8 | 7.0 | 1.6 | 2.8 | 0.57 | 0.10 | 0.9 | 0.2 | — | — | ||
| trans-COM-III | −5.5 | 7.1 | 2.1 | 2.1 | 0.50 | 0.06 | 1.1 | 0.1 | — | — | ||
| trans-COM-IV | −5.7 | 6.7 | 2.5 | 2.0 | 0.42 | −0.02 | 1.1 | 0.0 | — | — | ||
| trans-COM-V | −5.7 | 6.4 | 2.5 | 2.1 | 0.47 | 0.20 | 1.2 | 0.5 | — | — | ||
| trans-COM-VI | −5.4 | 6.8 | 2.1 | 2.3 | 0.36 | 0.15 | 0.8 | 0.3 | — | — | ||
| trans-COM-VII | −5.5 | 6.6 | 2.3 | 2.2 | 0.43 | 0.07 | 1.0 | 0.2 | — | — | ||
Alternatively, calculations show that the trans-exo cycloadduct is yielded through a stepwise mechanism, with the forming a zwitterionic intermediate trans-exo-IM1a. This result is different with the trans-endo channel as well as the cis-exo channel when diene cis-2a is employed (Fig. S1 in ESI†). The geometric optimizations in toluene using SMD solvation model also gave the same result, indicating that the inclusion of solvent effect does not alter the reaction mechanism. Based on the analysis of molecular orbitals (Fig. S2 and 3 in ESI†), this discrepancy might be attributed to the fact that the phase of the π orbital of the pyrrole and phenyl moieties in the HOMO of 1a cannot match with that of the p orbital of the ester carbonyl oxygen atom in the LUMO of trans-2a. Consequently, the N1–C3 bond formation is initially formed via TS trans-exo-TS1a, in which the length of the forming N1–C3 is 1.772 Å, whereas the bond distance between C2 and C6 atom is still long as 3.599 Å. This long bond distance suggests that there is no interaction between C2 and C6 centers during the N1–C3 bond formation. Compared with TS trans-endo-TSa, TS trans-exo-TS1a is predicted with a larger polarity, as reflected by the larger GEDT value of 0.39 e. Thus, the relative free energy of TS trans-exo-TS1a is calculated to be 3.0 kcal mol−1 lower than that of trans-endo-TSa. After TS trans-exo-TS1a, the N1–C3 bond is formed in IM trans-exo-IM1a, and the GEDT value of IM trans-exo-IM1a is increased to 0.50 e. This high value indicates that IM trans-exo-IM1a has zwitterionic characteristics (GEDT > 4.0 e). Finally, the trans-exo cycloadduct can be generated via a ring-closure TS trans-exo-TS2a, which requires a higher energy barrier of 26.9 kcal mol−1. The GEDT value at TS trans-exo-TS2a is 0.38 e, which is similar that of TS trans-exo-TS1a (0.39 e). Hence, the increased energy barrier at the ring-closure step might be caused by the electrostatic repulsion between the pyrrole and phenyl moieties of 1a and the ester carbonyl group of trans-2a when C2 and C6 centers approach one another at TS trans-exo-TS2a. Additionally, since this cycloaddition process presents a stepwise mechanism, the diradical structures could in principle be involved. The stability of wave functions for the TSs and IMs involved was tested with the unrestricted UM05-2X method. The UM05-2X/6-31G* calculations predict that wave functions are stable and the optimized-structures are the same as the corresponding ones obtained from the restricted M05-2X/6-31G*. The triplet structures exist but lies about 10.0–15.0 kcal mol−1 above the singlet ones in free energy. Therefore, diradical mechanism can be ruled out for the trans-exo channel. Overall, for the background reaction between 1a and trans-2a, the endo selectivity is kinetically preferred to the exo selectivity by 2.5 kcal mol−1 in kinetics. The reaction Gibbs free energies (ΔGrxn) for generation of trans-endo-3a and trans-exo-3a are endothermic by 8.4 and 5.6 kcal mol−1, respectively. Thus, these intermediary cycloadducts will spontaneously convert to indoloquinolizine trans-endo-4a and trans-exo-4a with the release of 14.0 and 13.0 kcal mol−1 in free energy, respectively.
3.2. Catalytic reaction mechanism
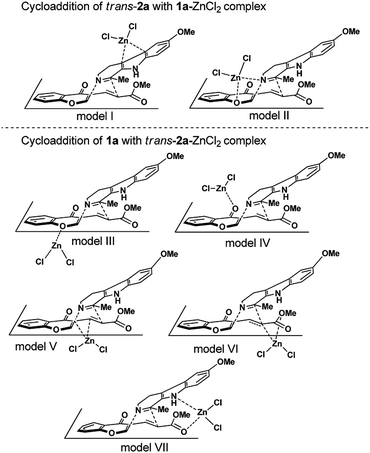
Kumar's experiment showed that either the reaction temperature could be lowered or the reaction time could be shortened when Lewis acid catalyst zinc catalyst was added into the reaction system.11 To shed light on the actual catalytic role of this Lewis acid catalyst, the mechanistic investigation on the IEDIDA reaction of 1a with both cis and trans-2a in the presence of the simple ZnCl2 were carried out in this section. Seven modes of activation, depending on where ZnCl2 coordinates to the two substrates are considered (Scheme 4).![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond in the pyrrole ring of 1a is η2-bonded to the zinc center, complex COM-I is formed, lying 5.7 kcal mol−1 below the two individual species in free energy. As compared with free 1a, the chemical potential μ is deceased to −4.7 eV, while the global electrophilicity index ω for complex COM-I is increased to 1.6 eV. Thus, the electrophilicity difference (Δω) between complex COM-I and trans-2a is decreased to zero, indicating a very lower polar character for the cycloaddition of complex COM-I with trans-2a.17b The calculations show that the cycloaddition of complex COM-I with trans-2a undergoes a one-step two-stage concerted mechanism for both endo and exo approaches. As expected, the GEDT values at TSs trans-endo-TS-I and trans-exo-TS-I are decreased to 0.14 e and 0.18 e, respectively, which are even lower than those in the background reaction. Meanwhile, differences between the lengths of the two forming bonds (Δd) at these two TSs are also decreased to 0.855 and 0.780 Å, respectively. These changes indicate that the polarity for these cycloaddition processes and asynchronicity at the two TSs are both lowered. The relative free energies of trans-endo-TS-I and trans-exo-TS-I are reduced to 18.8 and 22.2 kcal mol−1, respectively, with respect to the corresponding ones in the background reaction. However, the energy barriers for these two TSs measured from complex COM-I are comparable or even higher (24.5 and 28.9 kcal mol−1, respectively). On the other hand, ZnCl2 acting as a Lewis acid, extremely trends to combine with the basic center (N1 atom) of 1a, which leads to the generation of a stable complex COM-II by exothermic of 23.8 kcal mol−1 in free energy. This complexation also makes the decrease of the chemical potential μ (μ = −4.7 eV) and the increase of the global electrophilicity for the complex COM-II, and therefore might disfavor the polarity for the cycloaddition process between complex COM-II and trans-2a. From complex COM-II, the cycloaddition reaction with trans-2a also takes place along a concerted but weakly asynchronous mechanism for both endo and exo channels, as suggested by the smaller difference between the lengths of the two forming bonds (Δd = 0.576 Å for trans-endo-TS-II and 0.309 for trans-exo-TS-II). At TS trans-endo-TS-II, it can be noticed that the vinylogous ester oxygen (O7) atom in diene trans-2a is coordinated to the zinc center of the Lewis acid with a Zn–O bond distance of 2.240 Å, which corresponds to the dual activation model proposed by Kumar.11 This interaction is slightly weaker in TS trans-exo-TS-II, as suggested by the longer Zn–O bond distance of 2.782 Å. With this stabilization, the relative free energy of the endo TS is 5.7 kcal mol−1 more favorable than the exo one, although the GEDT value at TS trans-exo-TS-II is found to be 0.1 e larger than that at TS trans-exo-TS-II. Nevertheless, the free energy barrier for overcoming TS trans-endo-TS-II is increased to 43.5 kcal mol−1, meaning that this dual activation catalytic model cannot be responsible for the catalytic role of ZnCl2 in the reaction.11 The coordination of the Lewis acid zinc catalyst to the nucleophilic 1a increases the electrophilicity of 1a–ZnCl2 molecular complex and decreases the polarity for the cycloaddition reaction, and thereby disfavors for lowing the activation free energy.17
C double bond in the pyrrole ring of 1a is η2-bonded to the zinc center, complex COM-I is formed, lying 5.7 kcal mol−1 below the two individual species in free energy. As compared with free 1a, the chemical potential μ is deceased to −4.7 eV, while the global electrophilicity index ω for complex COM-I is increased to 1.6 eV. Thus, the electrophilicity difference (Δω) between complex COM-I and trans-2a is decreased to zero, indicating a very lower polar character for the cycloaddition of complex COM-I with trans-2a.17b The calculations show that the cycloaddition of complex COM-I with trans-2a undergoes a one-step two-stage concerted mechanism for both endo and exo approaches. As expected, the GEDT values at TSs trans-endo-TS-I and trans-exo-TS-I are decreased to 0.14 e and 0.18 e, respectively, which are even lower than those in the background reaction. Meanwhile, differences between the lengths of the two forming bonds (Δd) at these two TSs are also decreased to 0.855 and 0.780 Å, respectively. These changes indicate that the polarity for these cycloaddition processes and asynchronicity at the two TSs are both lowered. The relative free energies of trans-endo-TS-I and trans-exo-TS-I are reduced to 18.8 and 22.2 kcal mol−1, respectively, with respect to the corresponding ones in the background reaction. However, the energy barriers for these two TSs measured from complex COM-I are comparable or even higher (24.5 and 28.9 kcal mol−1, respectively). On the other hand, ZnCl2 acting as a Lewis acid, extremely trends to combine with the basic center (N1 atom) of 1a, which leads to the generation of a stable complex COM-II by exothermic of 23.8 kcal mol−1 in free energy. This complexation also makes the decrease of the chemical potential μ (μ = −4.7 eV) and the increase of the global electrophilicity for the complex COM-II, and therefore might disfavor the polarity for the cycloaddition process between complex COM-II and trans-2a. From complex COM-II, the cycloaddition reaction with trans-2a also takes place along a concerted but weakly asynchronous mechanism for both endo and exo channels, as suggested by the smaller difference between the lengths of the two forming bonds (Δd = 0.576 Å for trans-endo-TS-II and 0.309 for trans-exo-TS-II). At TS trans-endo-TS-II, it can be noticed that the vinylogous ester oxygen (O7) atom in diene trans-2a is coordinated to the zinc center of the Lewis acid with a Zn–O bond distance of 2.240 Å, which corresponds to the dual activation model proposed by Kumar.11 This interaction is slightly weaker in TS trans-exo-TS-II, as suggested by the longer Zn–O bond distance of 2.782 Å. With this stabilization, the relative free energy of the endo TS is 5.7 kcal mol−1 more favorable than the exo one, although the GEDT value at TS trans-exo-TS-II is found to be 0.1 e larger than that at TS trans-exo-TS-II. Nevertheless, the free energy barrier for overcoming TS trans-endo-TS-II is increased to 43.5 kcal mol−1, meaning that this dual activation catalytic model cannot be responsible for the catalytic role of ZnCl2 in the reaction.11 The coordination of the Lewis acid zinc catalyst to the nucleophilic 1a increases the electrophilicity of 1a–ZnCl2 molecular complex and decreases the polarity for the cycloaddition reaction, and thereby disfavors for lowing the activation free energy.17
 | ||
| Fig. 3 The M05-2X/6-31G(d)-optimized trans-2a–ZnCl2 complexes (the bond distances are labeled in Å and relative Gibbs free energies are given in kcal mol−1). | ||
Among these five complexes, complexes trans-COM-IV and trans-COM-VII, formed by the coordination of the ester carbonyl oxygen atom (O8) and the ketone carbonyl oxygen atom (O9) of trans-2a to the zinc center, are predicted to be more stable in energy than the others, which lie 9.5 and 9.0 kcal mol−1 below separate trans-2a and ZnCl2, respectively. The presence of ZnCl2 coordinated to the functional groups of trans-2a decrease the chemical potential μ of the trans-2a–ZnCl2 molecular complexes, but increases their electrophilicity, as shown in Table 1. These changers enhances both chemical potential difference (Δμ) and electrophilicity difference (Δω) between 1a and ZnCl2–trans-2a molecular complex, and will in consequence favors the charge transfer from nucleophilic 1a to the electrophilic ZnCl2–trans-2a molecular complex and increase the polar character of the cycloaddition processes.17 The calculations show that the cycloaddition between the ZnCl2–trans-2a molecular complexes and 1a nearly takes place along a stepwise process, except for the trans-endo model III from complex trans-COM-III (Fig. S11 in ESI†). The first step corresponds to the nucleophilic attack of the N1 atom of 1a to the C3 atom of the ZnCl2–trans-2a complex to give acyclic zwitterionic intermediate. The second step corresponds to the ring-closure process at the zwitterionic intermediate with the production of the final [4 + 2] cycloadduct. The diradical mechanism was also ruled out by the unrestricted DFT calculations. The exhaustive exploration of the potential energy profiles for these five reaction channels (Fig. S11–S14†) allows us to determine the most energy-favorable reaction channel. As shown in Fig. 4, the most energy-favorable reaction channel starts from complex trans-COM-VII, in which the ester C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O oxygen atom is bonded to the Lewis acid center. As this complex gets close to the imine moiety of 1a, the intermolecular nucleophilic attack of the N1 atom of imine moiety on the C3 position initially occurs, leading to the formation of N1–C3 bond in IM trans-endo-IM1-VII or trans-exo-IM1-VIIvia TS trans-endo-TS1-VII or trans-exo-TS1-VII, respectively. In TS trans-endo-TS1-VII, N1–C3 bond length of 1.841 Å is longer than in TS trans-exo-TS1-VII, while the distance between C2 and C6 (2.902 Å) is shorter than in TS trans-exo-TS1-VII (3.040 Å). At the same time, it can be noticed that the nitrogen atom of the pyrrole ring is strongly coordinated to the zinc center in the endo TS with the Zn–N bond distance of 2.307 Å, while a relatively weaker interaction (2.432 Å for Zn–C bond distance) between the carbon atom of phenyl ring and the zinc center is formed in the exo TS. As a result, the GEDT value at the endo TS trans-endo-TS1-VII (0.37 e) about 0.1 e smaller than in the exo one trans-exo-TS1-VII (0.48 e). The relative free energy of the exo TS is predicted to be 2.9 kcal mol−1 preferred than that of the endo one. At TSs trans-endo-TS1-VII and trans-exo-TS1-VII, the N1–C3 Wiberg bond index (WBI) values are 0.48 and 0.56, respectively, while the C2–C6 WBI values are 0.02 and 0.02, respectively. At IMs trans-endo-TS1-VII and trans-exo-TS1-VII, the N1–C3 WBI values increase to 0.84 and 0.80, indicating these N1–C3 bonds are nearly formed, while the C2–C6 WBI values are remain to 0.04 and 0.02, respectively. The C6–C7 WBI values at these IMs, 1.29 and 1.44, point out a certain π character for the C6–C7 bond as a result of the large delocalization of the negative charge on the C6 atom (−0.40 e in trans-endo-IM1-VII and −0.49 e in trans-exo-IM1-VII) belonging to the ester group. The interactions between the ester carbonyl oxygen atoms (O8) and the zinc center are also stronger, as verified by the shorter bond lengths of 1.938 and 1.940 Å. Thus, the accumulated negative charge on the C6 atoms can be dispersed and stabilized in these two zwitterionic IMs. Finally, the highly active zwitterionic IM1s can undergo an intramolecular nucleophilic addition of the electronegative C6 atom to the positive C2 atom via ring-closure TS2strans-endo-TS2-VII and trans-exo-TS2-VII, respectively, allowing the production of [4 + 2] ring-formation products complexed with Lewis acid ZnCl2. In the two ring-closure TS2s, the C2 and C6 atoms get closer with the bond distances of 2.479 and 2.456 Å, while the N1–C3 bond lengths are further shortened to 1.505 and 1.471 Å. The C2–C6 WBI values are 0.18 and 0.17, respectively, while the N1–C3 WBI values increase to 0.86 and 0.91, respectively. The GEDT values at the two ring-closure TS2s are 0.44 and 0.51 e, respectively, which are larger than those at the nucleophilic attack TS1s (0.37 and 0.48 e) but smaller than those at the zwitterionic IM1s (0.43 and 0.57 e). These trends show an increase of the GEDT along the nucleophilic attack of 1a to the ZnCl2-coordinated trans-2a up to formation of the zwitterionic IM1s, which is similar to the observation in the Lewis acid-catalyzed DA reaction.17a However, the relative free energies of the ring-closure TS2s are predicted to 10.1 and 10.2 kcal mol−1, which are higher than the nucleophilic attack TS1s. Thus, ring-closure step can be regarded as the rate-determining step (RDS) for the entire reaction, which is different with the calculations that RDS is the formation of zwitterionic IM in most of Lewis acid-catalyzed DA reactions.17a,27 In addition, although the GEDT value at exo TS for the ring-closure step is larger than that at the endo one, the relative free energies of these TSs are comparable. These observations cannot be explained by the differences of the GEDT values at the TSs. Consequently, the distortion/interaction analyses were performed on these four TSs in order to explore other factors that may influence the reaction activation barrier. The distortion energy ΔE≠dist and the interaction energy (ΔE≠int) are summarized in Table 2. From the results of distortion/interaction analyses, it is clearly that the distortion energies ΔE≠dist for the three fragments 1a, trans-2a and ZnCl2 decomposed from the two nucleophilic attack TS1s are comparable with one another. The total distortion energies (ΔE≠dist) of three fragments are identical. The preference for the exo TS in energy can be attributed to the favorable interaction energy ΔE≠int (by 3.6 kcal mol−1). This is in good accordance with the result of the higher GEDT value at the exo TS. For the ring-closure step, three fragments 1a, trans-2a, and ZnCl2 all suffer more heavily distortion at both endo and exoTS2s, than the corresponding ones at nucleophilic attack TS1s. Especially, for the diene moiety, the distorted energy is increased to 80.3 kcal mol−1 at the exo TS, implying that high distortion energy is required for the diene moiety to achieve ring-closure TS overtake the favorable interaction. As a result, although the GEDT value at the exo TS is higher and the interaction energy ΔEint is more favorable, the activation energy for the exo TS is similar to that for the endo one. The distortion energy should also be an important factor that controlled the reaction rate and the endo/exo selectivity.
O oxygen atom is bonded to the Lewis acid center. As this complex gets close to the imine moiety of 1a, the intermolecular nucleophilic attack of the N1 atom of imine moiety on the C3 position initially occurs, leading to the formation of N1–C3 bond in IM trans-endo-IM1-VII or trans-exo-IM1-VIIvia TS trans-endo-TS1-VII or trans-exo-TS1-VII, respectively. In TS trans-endo-TS1-VII, N1–C3 bond length of 1.841 Å is longer than in TS trans-exo-TS1-VII, while the distance between C2 and C6 (2.902 Å) is shorter than in TS trans-exo-TS1-VII (3.040 Å). At the same time, it can be noticed that the nitrogen atom of the pyrrole ring is strongly coordinated to the zinc center in the endo TS with the Zn–N bond distance of 2.307 Å, while a relatively weaker interaction (2.432 Å for Zn–C bond distance) between the carbon atom of phenyl ring and the zinc center is formed in the exo TS. As a result, the GEDT value at the endo TS trans-endo-TS1-VII (0.37 e) about 0.1 e smaller than in the exo one trans-exo-TS1-VII (0.48 e). The relative free energy of the exo TS is predicted to be 2.9 kcal mol−1 preferred than that of the endo one. At TSs trans-endo-TS1-VII and trans-exo-TS1-VII, the N1–C3 Wiberg bond index (WBI) values are 0.48 and 0.56, respectively, while the C2–C6 WBI values are 0.02 and 0.02, respectively. At IMs trans-endo-TS1-VII and trans-exo-TS1-VII, the N1–C3 WBI values increase to 0.84 and 0.80, indicating these N1–C3 bonds are nearly formed, while the C2–C6 WBI values are remain to 0.04 and 0.02, respectively. The C6–C7 WBI values at these IMs, 1.29 and 1.44, point out a certain π character for the C6–C7 bond as a result of the large delocalization of the negative charge on the C6 atom (−0.40 e in trans-endo-IM1-VII and −0.49 e in trans-exo-IM1-VII) belonging to the ester group. The interactions between the ester carbonyl oxygen atoms (O8) and the zinc center are also stronger, as verified by the shorter bond lengths of 1.938 and 1.940 Å. Thus, the accumulated negative charge on the C6 atoms can be dispersed and stabilized in these two zwitterionic IMs. Finally, the highly active zwitterionic IM1s can undergo an intramolecular nucleophilic addition of the electronegative C6 atom to the positive C2 atom via ring-closure TS2strans-endo-TS2-VII and trans-exo-TS2-VII, respectively, allowing the production of [4 + 2] ring-formation products complexed with Lewis acid ZnCl2. In the two ring-closure TS2s, the C2 and C6 atoms get closer with the bond distances of 2.479 and 2.456 Å, while the N1–C3 bond lengths are further shortened to 1.505 and 1.471 Å. The C2–C6 WBI values are 0.18 and 0.17, respectively, while the N1–C3 WBI values increase to 0.86 and 0.91, respectively. The GEDT values at the two ring-closure TS2s are 0.44 and 0.51 e, respectively, which are larger than those at the nucleophilic attack TS1s (0.37 and 0.48 e) but smaller than those at the zwitterionic IM1s (0.43 and 0.57 e). These trends show an increase of the GEDT along the nucleophilic attack of 1a to the ZnCl2-coordinated trans-2a up to formation of the zwitterionic IM1s, which is similar to the observation in the Lewis acid-catalyzed DA reaction.17a However, the relative free energies of the ring-closure TS2s are predicted to 10.1 and 10.2 kcal mol−1, which are higher than the nucleophilic attack TS1s. Thus, ring-closure step can be regarded as the rate-determining step (RDS) for the entire reaction, which is different with the calculations that RDS is the formation of zwitterionic IM in most of Lewis acid-catalyzed DA reactions.17a,27 In addition, although the GEDT value at exo TS for the ring-closure step is larger than that at the endo one, the relative free energies of these TSs are comparable. These observations cannot be explained by the differences of the GEDT values at the TSs. Consequently, the distortion/interaction analyses were performed on these four TSs in order to explore other factors that may influence the reaction activation barrier. The distortion energy ΔE≠dist and the interaction energy (ΔE≠int) are summarized in Table 2. From the results of distortion/interaction analyses, it is clearly that the distortion energies ΔE≠dist for the three fragments 1a, trans-2a and ZnCl2 decomposed from the two nucleophilic attack TS1s are comparable with one another. The total distortion energies (ΔE≠dist) of three fragments are identical. The preference for the exo TS in energy can be attributed to the favorable interaction energy ΔE≠int (by 3.6 kcal mol−1). This is in good accordance with the result of the higher GEDT value at the exo TS. For the ring-closure step, three fragments 1a, trans-2a, and ZnCl2 all suffer more heavily distortion at both endo and exoTS2s, than the corresponding ones at nucleophilic attack TS1s. Especially, for the diene moiety, the distorted energy is increased to 80.3 kcal mol−1 at the exo TS, implying that high distortion energy is required for the diene moiety to achieve ring-closure TS overtake the favorable interaction. As a result, although the GEDT value at the exo TS is higher and the interaction energy ΔEint is more favorable, the activation energy for the exo TS is similar to that for the endo one. The distortion energy should also be an important factor that controlled the reaction rate and the endo/exo selectivity.
| Structures | ΔE≠dista | ΔE≠intb | ΔE≠c | |||
|---|---|---|---|---|---|---|
| 1a | trans-2a | ZnCl2 | Total | |||
| a ΔE≠dist is the energy required to distort the reactants and catalysts into the geometry they have in the TSs. b ΔE≠dist is a negative value, indicating favourable interaction between the reactants and catalysts. c ΔE≠ is the reaction activation energy. | ||||||
| trans-endo-TS1-VII | 3.7 | 19.4 | 5.1 | 28.2 | −42.1 | −14.1 |
| trans-exo-TS1-VII | 2.6 | 21.3 | 4.3 | 28.2 | −45.7 | −17.5 |
| trans-endo-TS2-VII | 10.1 | 50.5 | 7.6 | 68.2 | −83.1 | −14.9 |
| trans-exo-TS2-VII | 9.5 | 80.3 | 6.2 | 96.0 | −110.0 | −15.0 |
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group adopts the cis-configuration relative to the C5
O group adopts the cis-configuration relative to the C5![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C6 double bond, the interaction of the zinc center to the dienophile and the catalytic mechanism slightly changes (Fig. 5).
C6 double bond, the interaction of the zinc center to the dienophile and the catalytic mechanism slightly changes (Fig. 5).
Along the endo channel, the zinc center is simultaneously bonded with the ester carbonyl oxygen atom of cis-2a and the phenyl carbon atom of 1a. The cis-endo cycloadduct is formed via a one-step two-stage concerted mechanism, without the generation of a zwitterionic IM. When the cycloaddition reaction takes place along the exo approach, the zinc center is only bonded with ester carbonyl oxygen atom. The cis-exo cycloadduct is generated via a stepwise mechanism, and the ring-closure TS is also predicted to be RDS. The relative free energies of the RDS TSs (10.4 kcal mol−1 for cis-endo-TS-VII and 9.5 kcal mol−1 for cis-exo-TS-VII) along the cis-endo and cis-exo channels are similar to the corresponding ones along the trans-endo and trans-exo channels.
In summary, there is a notable deceasing of the activation energy in the Lewis acid ZnCl2-catalyzed cycloaddition reaction relative to the uncatalyzed one. The reaction mechanism of nucleophilic 1a with trans- or cis-COM-VII complex formed by the coordination of Lewis acid zinc to the ester C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group of the trans- or cis-2a diene can be responsible for the experimental observation that reaction rate was accelerated as the Lewis acid catalyst was added.11 The Lewis acid zinc catalyst plays a central role in increasing the electrophilicity the diene and stabilizing the zwitterionic intermediate by delocalizing the negative charge on the C6 atom of the diene.
O group of the trans- or cis-2a diene can be responsible for the experimental observation that reaction rate was accelerated as the Lewis acid catalyst was added.11 The Lewis acid zinc catalyst plays a central role in increasing the electrophilicity the diene and stabilizing the zwitterionic intermediate by delocalizing the negative charge on the C6 atom of the diene.
3.3. The origin of stereochemistry
After understanding the mechanism of the cycloaddition of 1a with 2a catalyzed by simple Lewis acid ZnCl2, our attention turns to the chiral BINOL-ligated-Zn complex-mediated reaction, because the origin of stereochemistry of the reaction is more attractive in asymmetric catalysis. Although an excellent enantioselectivity of 93% ee was achieved in the presence of the chiral (R)-BINOL ligand and the Lewis acid catalyst ZnEt2,11 the TS model assumed by Kumar and co-worker seems not be rationale to account for the origin of the stereochemical course of the reaction. In terms of the energetic comparison (Section S4.1 in ESI†), the energy-favorable reaction channel is the addition of 1a with BINOL–Zn–trans-2a complex in which the zinc center is coordinated to the ester carbonyl oxygen atom of the diene, which is the same as the outcome of the achiral system.Starting from the complex formed between the BINOL–Zn catalyst and trans-2a, four possible reaction channels are located theoretically when the attack of the BINOL–Zn–trans-2a complex on the Re or Si face of the imine 1avia the endo or exo approach is considered. These four channels are denoted as trans-endo-Re, trans-endo-Si, trans-exo-Re, and trans-exo-Si (Scheme 5). The optimized-structures and relative energies of the key TSs and IMs involved in the entire reaction are shown in Fig. S17–19 in ESI.† Calculations indicate that each of the reaction pathway proceeds through a stepwise mechanism with the formation of the zwitterionic intermediates. Along the PES of the four channels, the relative free energies of the ring-closure TS2s are higher than those of TS1s. This trend is the same as the activation energy calculated in achiral system. Meanwhile, it is obviously that the chiral carbon atom (C6) in cycloadduct is generated in the ring-closure step. Therefore, the ring-closure step should be the stereo-controlling step for the entire reaction, and the enantioselectivity of the cycloadduct should depend on the relative energies of TS2s. The optimized-structures and the relative energies of TS2s are presented in Fig. 6 and Table 3, respectively.
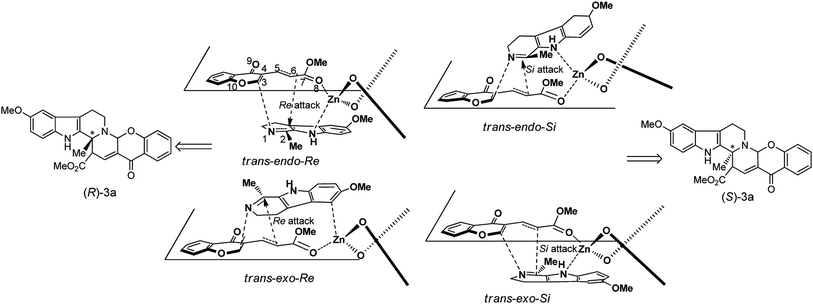 | ||
| Scheme 5 Proposed reaction models for the chiral BINOL–Zn-catalyzed the asymmetric IEDIDA reaction between 1a and trans-2a. | ||
| Species | ΔE≠dist | ΔE≠int | ΔE≠ | ΔG≠ | τ a/% | GEDT | |||
|---|---|---|---|---|---|---|---|---|---|
| 1a | 2a | Catalyst | Total | ||||||
a τ: occupied probability based on Boltzmann distribution. τ = N*i/N = [gi![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−εi/kT)]/[∑gi exp(−εi/kT)]/[∑gi![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−εi/kT)] (T = 298 K), ee = (∑τR − ∑τS) − (∑τR + ∑τS). exp(−εi/kT)] (T = 298 K), ee = (∑τR − ∑τS) − (∑τR + ∑τS). |
|||||||||
| trans-endo-TS2-Re | 19.7 | 58.5 | 2.5 | 73.9 | −98.9 | −18.2 | 12.0 | 1.2 | 0.39 |
| trans-endo-TS2-Si | 18.5 | 56.7 | 2.6 | 77.8 | −97.3 | −19.5 | 13.1 | 0.2 | 0.42 |
| trans-exo-TS2-Re | 12.1 | 88.2 | 3.1 | 103.4 | −123.8 | −20.5 | 9.7 | 98.3 | 0.52 |
| trans-exo-TS2-Si | 15.6 | 93.2 | 2.8 | 111.7 | −129.3 | −17.6 | 12.8 | 0.3 | 0.50 |
| cis-endo-TS-Re | 6.4 | 25.4 | 2.4 | 34.2 | −52.1 | −18.0 | 12.4 | 37.6 | 0.35 |
| cis-endo-TS-Si | 7.7 | 24.2 | 2.4 | 34.3 | −52.2 | −17.9 | 12.1 | 62.4 | 0.35 |
| cis-exo-TS2-Re | 10.9 | 61.5 | 3.6 | 76.0 | −81.5 | −5.5 | 22.0 | 0.0 | 0.40 |
| cis-exo-TS2-Si | 11.3 | 58.9 | 5.2 | 75.4 | −80.8 | −5.4 | 23.4 | 0.0 | 0.40 |
Of these four competing TSs, TSs trans-endo-TS2-Re and trans-exo-TS2-Re, corresponding to the Re face attack on the imine moiety of 1a, are the lowest structures, which are favored over the other two structures by 1.1 and 3.1 kcal mol−1 in free energy, respectively. Hence, the R configuration of the cycloadduct 3a is predicted to be the major product, corresponding to the theoretically expected ee value of 99% using Boltzmann distribution of the four TSs at 298 K (Table 3). The computed result is qualitatively in agreement with the experimental observed outcome that the R configuration indoloquinolizine was obtained with the enantioselectivity of 90% ee value. For the two energy-favorable TSs, the exo structure is 2.3 kcal mol−1 preferred to the endo one, meaning that the exo cycloadduct should be the predominate product, which is different from the endo preference in the most of DA and HDA reactions.28
To further elucidate the origin of the enantioselectivity and the unusual exo selectivity, the four competing TSs were investigated by the combination of the structural and the distortion/interaction analyzes. The result of the distortion/interaction energies for the four TSs indicates that the total distortion energies of the TSs mainly arise from the distortion energies of diene fragment, which suffers heavier distortion compared with the dienophile and catalyst fragments during the ring-closure process. With respect to the endo TSs, the distortion of the diene fragment at the exo TSs is more serious, and therefore the total distortion energies of the exo TSs are disfavored by approximately 20–30 kcal mol−1. However, the ring-formation process proceeding through the exo pathway favors the electrophilic/nucleophilic interaction between the dienophile and diene–catalyst fragments, as demonstrated by greater GEDT values (0.50 and 0.52 e) at the two exo TSs. The interaction energies completely overwhelm the distortion energies in the exo TSs, which might account for the observed exo preference in the reaction. For the two exo TSs, the zinc Lewis acid center adopts a tetra-coordination. When the attack of the diene–catalyst complex occurs on the Re face of the imine moiety in 1a, the pyrrole carbon atom is weakly interacted to the zinc center in TS trans-exo-TS2-Re, in which the Zn–C bond distance is 2.518 Å. This loose bonding mode places the imine moiety in the empty pocket of the catalyst and then can avoid less steric repulsion from the naphthalene ring of the BINOL ligand. In contract, the pyrrole nitrogen atom in trans-exo-TS2-Si is strongly coordinated to the zinc center with a Zn–N bond distance of 2.355 Å, which forces the dienophile moiety to orientate toward the bulky pocket of the BINOL–Zn catalyst, here it suffers larger steric repulsion from one naphthalene ring of the BINOL ligand. This repulsion is also reflected by distortion energy of the dienophile fragment (12.1 kcal mol−1 in TS trans-exo-TS2-Re and 15.6 kcal mol−1 in TS trans-exo-TS2-Si). As a result, TS trans-exo-TS2-Si is destabilized relative to trans-exo-TS2-Re by 3.1 kcal mol−1 in free energy, leading to the preference for the R configuration product.
Furthermore, the cycloaddition of 1a with diene cis-2a under the chiral catalytic system was investigated to explore the influence of the configuration of diene on the stereochemistry of this reaction. Calculations show that the mechanisms are similar to those obtained in the achiral ZnCl2-catalzyed reaction system, in which the cis-endo channel proceeds through a one-step concerted mechanism, whereas the cis-exo channel undergoes a stepwise mechanism (see PES in Fig. S20 and 21†). The difference of these two channels lies in the coordination mode at the zinc center (Fig. 7). When the cycloaddition of 1a with the diene–catalyst complex takes places via the endo approach, the carbon atom of the phenyl moiety in 1a is bonded with the zinc center of the catalyst, leading to formation of a stable tetra-coordination zinc center. However, either the nitrogen atom of the pyrrole moiety or the carbon atom of the phenyl moiety cannot interact with zinc center when the exo approach occurs. Hence, the exo TSs are approximately 10.0 kcal mol−1 destabilized in comparison to the endo ones, and could not contribute to the product distribution. For the energy-favored endo TSs, it is clear that the dienophile moieties are located in the empty pocket of the BINOL ligand, regardless of whether the attack of the diene–catalyst complex originates on the Re or Si face of imine moiety. The dienophile moieties suffer less steric hindrance from the naphthalene ring of the BINOL ligand, as indicated by similar distortion energies that deformed from the two endo TSs (Table 3). Thus, the energy difference between the two competing TSs is only 0.3 kcal mol−1, suggesting that no preference for producing the (R)-3a and (S)-3a in kinetics. That is to say, the BINOL ligand cannot allow efficient chiral induction for 1a when the ester C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group of diene adopts the cis configuration.
O group of diene adopts the cis configuration.
Overall, it can be predicted that the stereochemistry of the IEDIDA reaction is controlled by a combination of electrophilic/nucleophilic interaction between 1a and diene–catalyst complex, the configuration of the diene and the asymmetric induction from the BINOL ligand. The cycloaddition via the exo approach favors GEDT from the nucleophilic 1a to the electrophilic, even though the two fragments suffer large distortion. The ester C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group of diene 2a with the trans configuration permits the formation of a stabilizing interaction between the zinc center of the chiral catalyst and the dienophile moiety, leading to the generation of an energy-favored four-coordinated TS. As a result, the steric hindrance from the naphthalene ring of the BINOL ligand can effectively shields the Si face of the imine moiety in 1a, making the attack of the Re face of the imine moiety more accessible.
O group of diene 2a with the trans configuration permits the formation of a stabilizing interaction between the zinc center of the chiral catalyst and the dienophile moiety, leading to the generation of an energy-favored four-coordinated TS. As a result, the steric hindrance from the naphthalene ring of the BINOL ligand can effectively shields the Si face of the imine moiety in 1a, making the attack of the Re face of the imine moiety more accessible.
4. Conclusions
The reaction mechanism and stereochemistry of the IEDIDA reaction between the cyclic imine 1a and the electron-poor chromone diene 2a catalyzed by chiral BINOL–zinc complex has been theoretically investigated by a combination of DFT calculations, chemical reactivity indices and distortion/interaction analyses. The major conclusions are listed below:(1) The cycloaddition of imine 1a with diene–ZnCl2 complex that formed by the coordination of Lewis acid zinc to the ester C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group of the diene 2a is the most energy-preferable channel for the generation of [4 + 2] cycloadduct. The computed activation energy is much lower than that in the uncatalyzed reaction as well as the catalytic model hypothesized in the original experimental paper.11
O group of the diene 2a is the most energy-preferable channel for the generation of [4 + 2] cycloadduct. The computed activation energy is much lower than that in the uncatalyzed reaction as well as the catalytic model hypothesized in the original experimental paper.11
(2) The Lewis acid zinc catalyst plays an important role in increasing the electrophilicity of diene and changes the mechanism from a concerted to a stepwise one because a large stabilization of the corresponding zwitterionic intermediate via the charge delocalization.
(3) The catalytic reaction mechanism as well as the stereochemistry of the cycloadduct is sensitive to the configuration of the diene. When the ester C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group adopts trans configuration relative to the C
O group adopts trans configuration relative to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond, a stabilizing interaction between zinc center of the catalyst and the phenyl moiety of the dienophile can be formed, which favors the steric discrimination by the BINOL ligand. The imine suffers less repulsion from the naphthalene ring of the BINOL ligand, when the electrophilic attack of diene–catalyst complex takes place from its Re face.
C double bond, a stabilizing interaction between zinc center of the catalyst and the phenyl moiety of the dienophile can be formed, which favors the steric discrimination by the BINOL ligand. The imine suffers less repulsion from the naphthalene ring of the BINOL ligand, when the electrophilic attack of diene–catalyst complex takes place from its Re face.
(4) The exo-selectivity is predicted for the cycloadduct, which is attributed to the favorable electrophilic/nucleophilic interaction between the imine and diene–catalyst complex.
Acknowledgements
The authors are grateful for the financial support from the National Natural Science Foundation of China (No. 21402158), the Scientific Research Fund of Education Department of Sichuan Province (No. 14ZB0131), Key Scientific Research Found of Xihua University (No. Z1313319) and Open Research Subject of Key Laboratory (Research center for advanced computation) of Xihua University (No. szjj2015-051).Notes and references
- (a) H. Waldmann, Synthesis, 1994, 535 CrossRef CAS; (b) G. R. Heintzelman, I. R. Y. Meigh, R. Mahajan and S. M. Weinreb, Org. React., 2005, 65, 141 CAS; (c) G. Masson, C. Lalli, M. Benohoud and G. Dagousset, Chem. Soc. Rev., 2013, 42, 902 RSC; (d) V. V. Kouznetsov, Tetrahedron, 2009, 65, 2721 CrossRef CAS; (e) G. B. Rowland, E. B. Rowland, Q. Zhang and J. C. Antilla, Curr. Org. Chem., 2006, 10, 981 CrossRef CAS; (f) P. R. Girling, T. Kiyoib and A. Whiting, Org. Biomol. Chem., 2011, 9, 3105 RSC; (g) Y. Kumar, P. Singh and G. Bhargava, Synlett, 2015, 26, 363 CrossRef CAS.
- (a) P. M. Weintraub, J. S. Sabol, J. M. Kane and D. R. Borcherding, Tetrahedron, 2003, 59, 2953 CrossRef CAS; (b) M. G. P. Buffat, Tetrahedron, 2004, 60, 1701 CrossRef CAS; (c) F.-X. Felpin and J. Lebreton, Curr. Org. Synth., 2004, 1, 83 CrossRef CAS; (d) M. Bazin and C. Kuhn, J. Comb. Chem., 2005, 7, 302 CrossRef CAS; (e) A. R. Katritzky, S. Rachwal and B. Rachwal, Tetrahedron, 1996, 52, 15031 CrossRef CAS; (f) V. Sridharan, P. A. Suryavanshi and J. C. Menéndez, Chem. Rev., 2011, 111, 7157 CrossRef CAS.
- (a) S. Kobayashi, S. Komiyama and H. Ishitani, Angew. Chem., Int. Ed., 1998, 37, 3121 CrossRef; (b) S. Kobayashi, K.-I. Kusakabe, S. Komiyama and H. Ishitani, J. Org. Chem., 1999, 64, 4220 CrossRef CAS; (c) S. Kobayashi, K.-I. Kusakabe and H. Ishitani, Org. Lett., 2000, 2, 1225 CrossRef CAS PubMed; (d) S. Kobayashi, M. Ueno, S. Saito, Y. Mizuki, H. Ishitani and Y. Yamashita, Proc. Natl. Acad. Sci. U. S. A., 2004, 101, 5476 CrossRef CAS; (e) Y. Yamashita, Y. Mizuki and S. Kobayashi, Tetrahedron Lett., 2005, 46, 1803 CrossRef CAS; (f) V. Jurčík, K. Arai, M. M. Salter, Y. Yamashita and S. Kobayashi, Adv. Synth. Catal., 2008, 350, 647 CrossRef; (g) S. Kobayashi, K. Arai, H. Shimizu, Y. Ihori, H. Ishitan and Y. Yamashita, Angew. Chem., Int. Ed., 2005, 44, 761 CrossRef CAS; (h) K. Arai, M. M. Salter, Y. Yamashita and S. Kobayashi, Angew. Chem., Int. Ed., 2007, 46, 955 CrossRef CAS; (i) K. Arai, S. Lucarini, M. M. Salter, K. Ohta, Y. Yamashita and S. Kobayashi, J. Am. Chem. Soc., 2007, 129, 8103 CrossRef CAS PubMed; (j) S. Guillarme and A. Whiting, Synlett, 2004, 711 CAS; (k) L. Di Bari, S. Guillarme, J. Hanan, A. P. Henderson, J. A. K. Howard, G. Pescitelli, M. R. Probert, P. Salvadori and A. Whiting, Eur. J. Org. Chem., 2007, 5771 CrossRef.
- (a) S. Yao, M. Jøhannsen, R. G. Hazell and K. A. Jørgensen, Angew. Chem., Int. Ed., 1998, 37, 3121 CrossRef CAS; (b) S. Yao, S. Saaby, R. G. Hazell and K. A. Jørgensen, Chem.–Eur. J., 2000, 6, 2435 CrossRef CAS; (c) N. S. Josephsohn, M. L. Snapper and A. H. Hoveyda, J. Am. Chem. Soc., 2003, 125, 4018 CrossRef CAS; (d) C. A. Newman, J. Antilla, C. P. Chen, A. V. Predeus, L. Fielding and W. D. Wulff, J. Am. Chem. Soc., 2007, 129, 7216 CrossRef CAS PubMed; (e) X. Liu, L. Lin and X. Feng, Acc. Chem. Res., 2011, 44, 574 CrossRef CAS PubMed; (f) D. J. Shang, J. Y. Xin, L. Liu, X. Zhou, X. Liu and X. Feng, J. Org. Chem., 2008, 73, 630 CrossRef CAS PubMed; (g) Z. Chen, L. Lin, D. Chen, J. Li, X. Liu and X. Feng, Tetrahedron Lett., 2010, 51, 3088 CrossRef CAS; (h) Y. L. Liu, D. J. Shang, X. Zhou, X. Liu and X. Feng, Chem.–Eur. J., 2009, 15, 2055 CrossRef CAS PubMed; (i) Y. L. Liu, D. J. Shang, X. Zhou, Y. Zhu, L. Lin, X. Liu and X. Feng, Org. Lett., 2010, 12, 180 CrossRef CAS.
- (a) T. Itoh, M. Yokoya, K. Miyauchi, K. Nagata and A. Ohsawa, Org. Lett., 2003, 5, 4301 CrossRef CAS; (b) T. Itoh, M. Yokoya, K. Miyauchi, K. Nagata and A. Ohsawa, Org. Lett., 2006, 8, 1533 CrossRef CAS.
- (a) T. Akiyama, Y. Tamura, J. Itoh and K. Fuchibe, Synlett, 2006, 141 CrossRef CAS; (b) J. Itoh, K. Fuchibe and T. Akiyama, Angew. Chem., Int. Ed., 2006, 45, 4796 CrossRef CAS PubMed.
- M. Rueping and C. Azap, Angew. Chem., Int. Ed., 2006, 45, 7832 CrossRef CAS PubMed.
- (a) J. Esquivias, R. Gómez Arrayás and J. C. Carretero, J. Am. Chem. Soc., 2007, 129, 1480 CrossRef CAS PubMed; (b) J. Esquivias, I. Alonso, R. Gómez Arrayás and J. C. Carretero, Synthesis, 2009, 113 CAS.
- (a) M. He, J. R. Struble and J. W. Bode, J. Am. Chem. Soc., 2006, 128, 8418 CrossRef CAS; (b) T.-Y. Jian, P.-L. Shao and S. Ye, Chem. Commun., 2011, 47, 2381 RSC; (c) S. Dong, X. Liu, X. Chen, F. Mei, Y. Zhang, B. Gao, L. Lin and X. Feng, J. Am. Chem. Soc., 2010, 132, 10650 CrossRef.
- (a) P. Buonora, J. C. Olsen and T. Oh, Tetrahedron, 2001, 57, 6099 CrossRef; (b) X. X. Jiang and R. Wang, Chem. Rev., 2013, 113, 5515 CrossRef PubMed.
- V. Eschenbrenner-Lux, P. Küchler, S. Ziegler, K. Kumar and H. Waldmann, Angew. Chem., Int. Ed., 2014, 53, 2134 CrossRef PubMed.
- (a) Y. Zhao and D. G. Truhlar, Theor. Chem. Acc., 2008, 120, 215 Search PubMed; (b) Y. Zhao and D. G. Truhlar, Acc. Chem. Res., 2008, 41, 157 CrossRef PubMed; (c) M. D. Wodrich, C. Corminboeuf, P. R. Schreiner, A. A. Fokin and P. V. R. Schleyer, Org. Lett., 2007, 9, 1851 CrossRef PubMed.
- (a) E. A. Amin and D. G. Truhlar, J. Chem. Theory Comput., 2008, 4, 75 CrossRef PubMed; (b) A. Sorkin, D. G. Truhlar and E. A. Amin, J. Chem. Theory Comput., 2009, 5, 1254 CrossRef PubMed; (c) L. Schiaffino and G. Ercolani, Chem.–Eur. J., 2010, 16, 3147 CrossRef PubMed; (d) W. Li, Z. Su and C. Hu, Chem.–Eur. J., 2013, 19, 124 CrossRef PubMed.
- (a) R. Ditchfield, W. J. Hehre and J. A. Pople, J. Chem. Phys., 1971, 54, 724 CrossRef; (b) W. J. Hehre, R. Ditchfield and J. A. Pople, J. Chem. Phys., 1972, 56, 2257 CrossRef; (c) P. C. Hariharan and J. A. Pople, Mol. Phys., 1974, 27, 209 CrossRef; (d) M. S. Gordon, Chem. Phys. Lett., 1980, 76, 163 CrossRef; (e) P. C. Hariharan and J. A. Pople, Theor. Chem. Acc., 1973, 28, 213 Search PubMed; (f) J. P. Blaudeau, M. P. McGrath, L. A. Curtiss and L. Radom, J. Chem. Phys., 1997, 107, 5016 CrossRef; (g) M. M. Francl, W. J. Pietro, W. J. Hehre, J. S. Binkley, M. S. Gordon, D. J. DeFrees and J. A. Pople, J. Chem. Phys., 1982, 77, 3654 CrossRef; (h) R. C. Binning Jr and L. A. Curtiss, J. Comput. Chem., 1990, 11, 1206 CrossRef; (i) V. A. Rassolov, J. A. Pople, M. A. Ratner and T. L. Windus, J. Chem. Phys., 1998, 109, 1223 CrossRef; (j) V. A. Rassolov, M. A. Ratner, J. A. Pople, P. C. Redfern and L. A. Curtiss, J. Comput. Chem., 2001, 22, 976 CrossRef.
- (a) C. Gonzalez and H. B. Schlegel, J. Chem. Phys., 1989, 90, 2154 CrossRef; (b) C. Gonzalez and H. B. Schlegel, J. Phys. Chem., 1990, 94, 5523 CrossRef.
- A. V. Marenich, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2009, 113, 6378 CrossRef PubMed.
- (a) L. R. Domingo, Tetrahedron, 2002, 58, 3765 CrossRef; (b) L. R. Domingo, M. J. Aurell, P. Pérez and R. Contreras, Tetrahedron, 2002, 58, 4417 CrossRef CAS; (c) L. R. Domingo, A. Asensio and P. Arroyo, J. Phys. Org. Chem., 2002, 15, 660 CrossRef CAS; (d) L. R. Domingo and J. Andrés, J. Org. Chem., 2003, 68, 8662 CrossRef CAS PubMed; (e) L. R. Domingo, E. Chamorro and P. Pérez, J. Org. Chem., 2008, 73, 4615 CrossRef CAS; (f) L. R. Domingo and J. A. Sáez, Org. Biomol. Chem., 2009, 7, 3576 RSC.
- (a) R. G. Parr, L. von Szentpály and S. Liu, J. Am. Chem. Soc., 1999, 121, 1922 CrossRef CAS; (b) Y. Yamaguchi, Y. Osamura and H. F. Schaefer, J. Am. Chem. Soc., 1983, 105, 7512 CrossRef.
- The global electrophilicity index ω, which measures the stabilization energy when the system acquires an additional electronic charge ΔN from the environment, is given in terms of the electronic chemical potential μ and chemical hardness η by the following simple expression: ω [eV] = (μ2/2η). Both quantities can be calculated in terms of the HOMO and LUMO electron energies, εH and εL, as μ ≈ (εH + εL)/2 and η ≈ (εH − εL), respectively. The nucleophilicity index N,18b based on the HOMO energies obtained within the Kohn–Sham scheme, is defined as N = EHOMO(Nu) − EHOMO(TCE). The nucleophilicity is taken relative to tetracyanoethylene (TCE) as a reference, because it has the lowest HOMO energy in a large series of molecules already investigated in the context of polar cycloadditions.
- L. R. Domingo, M. J. Aurell, P. Pérez and J. A. Sáez, RSC Adv., 2013, 3, 1486 RSC.
- L. R. Domingo, RSC Adv., 2014, 3, 32415 RSC.
- (a) A. E. Reed and F. Weinhold, J. Chem. Phys., 1985, 83, 1736 CrossRef CAS; (b) A. E. Reed, R. B. Weinstock and F. Weinhold, J. Chem. Phys., 1985, 83, 735 CrossRef CAS; (c) A. E. Reed, L. A. Curtiss and F. Weinhold, Chem. Rev., 1988, 88, 899 CrossRef CAS; (d) A. E. Reed and P. R. Schleyer, J. Am. Chem. Soc., 1990, 112, 1434 CrossRef CAS.
- (a) G. Zhong, B. Chan and L. Radom, THEOCHEM, 2007, 811, 13 CrossRef CAS; (b) D. H. Ess and K. N. Houk, J. Am. Chem. Soc., 2007, 129, 10646 CrossRef CAS PubMed; (c) C. Gordon, J. L. Mackey, J. C. Jewett, E. M. Sletter, K. N. Houk and C. R. Bertozzi, J. Am. Chem. Soc., 2012, 134, 9199 CrossRef CAS; (d) D. H. Ess, G. O. Jones and K. N. Houk, Org. Lett., 2008, 10, 1633 CrossRef CAS; (e) F. Liu, R. S. Paton, S. Kim, Y. Liang and K. N. Houk, J. Am. Chem. Soc., 2013, 135, 15642 CrossRef CAS PubMed; (f) H. V. Pham, R. S. Paton, A. G. Ross, S. J. Danishfsky and K. N. Houk, J. Am. Chem. Soc., 2014, 135, 2397 CrossRef.
- M. J. Frisch, et al., Gaussian 09, Reversion A.02, Gaussian, Inc., Wallingford CT, 2009 Search PubMed.
- C. Y. Legault, CYLview, 1.0b, Université de Sherbrooke, Sherbrooke, Québec, Canada, 2009, http://www.cylview.org Search PubMed.
- P. Jaramilio, L. R. Domingo, E. Chamorro and P. Pérez, THEOCHEM, 2008, 865, 68 CrossRef.
- (a) M. Roberson, A. S. Jepsen and K. A. Jørgensen, Tetrahedron, 2001, 57, 90 CrossRef; (b) L. R. Domingo, M. Oliva and J. Andrés, J. Org. Chem., 2001, 66, 6157 Search PubMed; (c) A. Bongini and M. Panunzio, Eur. J. Org. Chem., 2006, 972 CrossRef CAS; (d) Z. Pi and S. Li, J. Phys. Chem. A, 2006, 110, 9225 CrossRef CAS PubMed; (e) N. Çelebi-Ölçüm, D. H. Ess, V. Aviyente and K. N. Houk, J. Org. Chem., 2008, 73, 7472 CrossRef PubMed; (f) Z. Su, S. Qin, C. Hu and X. Feng, Chem.–Eur. J., 2010, 16, 4359 CrossRef CAS PubMed.
- (a) G. Ujaque, P. S. Lee, K. N. Houk, M. F. Hentemann and S. J. Danishefsky, Chem.–Eur. J., 2002, 8, 3423 CrossRef CAS; (b) H. Audrian, J. Thorhauge, R. G. Hazell and K. A. Jørgensen, J. Org. Chem., 2000, 65, 4487 CrossRef; (c) S. Danishefsky, E. Larson, D. Askin and N. Kato, J. Am. Chem. Soc., 1985, 107, 1246 CrossRef CAS; (d) D. H. Ess, G. O. Jones and K. N. Houk, Adv. Synth. Catal., 2006, 348, 2337 CrossRef CAS; (e) S. A. Cinar, S. Ercan, S. E. Gunal, I. Dogan and V. Aviyente, Org. Biomol. Chem., 2014, 12, 8079 RSC; (f) P. Rooshenas, K. Hof, P. R. Schreiner and C. M. Williams, Eur. J. Org. Chem., 2011, 983 CrossRef CAS; (g) N. Nikbin, P. T. Do, S. Caratzoulas, R. F. Lobo, P. Dauenhauer and D. G. Vlachos, J. Catal., 2013, 297, 35 CrossRef CAS; (h) Z. Su and C. K. Kim, Org. Biomol. Chem., 2015, 13, 6313 RSC.
Footnote |
| † Electronic supplementary information (ESI) available: Computational details, optimized geometries, calculated energies and the full citation of Gaussian 09 program. See DOI: 10.1039/c5ra17981e |
| This journal is © The Royal Society of Chemistry 2015 |