Synthesis, physical properties and ion recognition of a novel larger heteroacene with eleven linearly-fused rings and two different types of heteroatom†
Pei-Yang Gua,
Junkuo Gaob,
Chengyuan Wanga and
Qichun Zhang*ac
aSchool of Materials Science and Engineering, Nanyang Technological University, Singapore 639798, Singapore. E-mail: qczhang@ntu.edu.sg
bThe Key Laboratory of Advanced Textile Materials and Manufacturing Technology of Ministry of Education, College of Materials and Textiles, Zhejiang Sci-Tech University, Hangzhou 310018, China
cDivision of Chemistry and Biological Chemistry, School of Physical and Mathematical Sciences, Nanyang Technological University, Singapore 637371, Singapore
First published on 16th September 2015
Abstract
A novel larger heteroacene, 2,3,15,16-tetradecyloxy-5,8,10,13,18,21,23,26-octaaza-7,11,20,24-tetraoxohendecacene (8N8O) with two different types of heteroatoms (O and N) and eleven linearly fused rings, has been successfully synthesized and can identify iodide over other anions.
In past decades, N-oligoheteroacenes have attracted a lot of researchers' interests because of their potential applications in n-type organic field effect transistors (OFETs), phototransistors, sensors, organic-light emitting diodes (OLEDs), memory devices, and solar cells.1 Compared with their parent systems (plain oligoacenes),2 N-substituted oligoheteroacenes could have more opportunities for scientists to tune their physical properties because the highest occupied molecular orbital (HOMO)–lowest unoccupied molecular orbital (LUMO) gap and HOMO/LUMO positions of oligoheteroacenes strongly depend on the type/position/number of heteroatoms (e.g. N, O, P, B, S). Although significant advancements have been witnessed in synthesis, theoretical study, and applications of short heteroacenes, large heteroacenes (n ≥ 7) is still rare.3 Moreover, introducing two or more types of heteroatoms into the backbone of larger oligoacenes should be more interesting because (1) it should be more challenge to approach them; (2) some unexpected properties could be obtained through carefully selecting the heteroatoms in backbone and tuning their positions; and (3) different heteroatoms could offer more chances to stabilize larger oligoheteroacenes. In this research, we are more interested in large heteroacenes with both oxygen and nitrogen atoms in its backbone due to their relatively-short synthetic steps, reasonable stability, and possible device applications. However, until now, only a few dioxanes derivatives (up to six-fused rings) were reported.4 The curiosity of properties and synthetic possibility strongly encourages us to challenge large heteroacenes (n > 7) with two or more different heteroatoms (N/O).
Since N-substituted heteroacenes are an electron-deficient system, they could act as π acid for anion recognition through anion–π interaction.5 Actually, non-covalent binding between electron deficient aromatic rings and anions, namely anion–π interaction, has been causing a lot of interests in theoretical and experimental investigations because anion–π interactions play a crucial role in many chemical and biological processes and could have potential applications in medicine and environment. Recently, we have demonstrated that azaacenes can selectively recognize F− or H2PO4−.6 Continuing on this direction, we have synthesized a novel heteroacene with eleven linearly-fused rings (2,3,15,16-tetradecyloxy-5,8,10,13,18,21,23,26-octaaza-7,11,20,24-tetraoxohendecacene, abbreviated as 8N8O), which is the longest one now, and found that it can selectively recognize iodide. Among the range of biologically important anions, iodide plays a vital role in many biological activities such as thyroid gland function.7 Although a number of studies, which focus on chromogenic and fluorescent recognition of anions, have been published concerning other anions such as fluoride, there are few reports available for the estimation of iodide.8
In this report, we reported a novel large hetroacene 8N8O, which have two different types of heteroatoms (O and N) and eleven linear fused-rings. Compound 8N8O can optically detect the presence of iodide over a wide range of other anions (F−, Cl−, Br−, PF6−, HSO4−, NO3−, BF4−, CH3COO− (AcO−), CN−, and H2PO4−) based on the “heavy-atom” effect.9
The synthetic procedure for the preparation of compound 8N8O is depicted in Scheme 1. The target compound was synthesized in two-step steps: first, the commercially available 2,5-dihydroxy-1,4-benzoquinone was reacted smoothly with 1,2-bis(decyloxy)-4,5-diaminobenzene (0.9 equiv) in alcohol to afford 7,8-bis(decyloxy)phenazine-2,3-diol with yield 62%. Then the as-prepared product was condensed with 2,3,7,8-tetrachloro-1,4,6,9-tetraazaanthracene (0.33 equiv) using cesium fluoride (10 equiv) as the base to yield compound 8N8O with yield 10%. Compound 8N8O was fully characterized using 1H and 13C NMR spectroscopy, infrared spectroscopy (IR) and mass spectrometry.
 | ||
| Scheme 1 Synthetic route of: (i) CH3CH2OH, N2, reflux, 62%; (ii) 10 equiv CsF, 0.33 equiv 2,3,7,8-tetrachloro-1,4,6,9-tetraazaanthracene, DMF, 80 °C, 10%. | ||
Fig. 1 shows the normalized optical absorption and emission spectra of compound 8N8O. The absorption spectrum of compound 8N8O exhibits three prominent bands at 290 nm, 424 nm, and 451 nm in N-methyl-2-pyrrolidone (NMP) solution, which can be ascribed to a localized aromatic π–π* transition, phenazine core,10 and excimer absorption, respectively. However, there are still three small peaks at shorter wavelength region and the level-off tails at the longer wavelength region, which can be ascribed to the scattering effect of the dye nanoparticles due to the poor solubility of compound 8N8O in NMP solution. When the solvent was changed from NMP to good solvent trifluoroacetic acid (TFA), there are only two peaks at 283 nm and 492 nm and no level-off tails at the longer wavelength region. TFA dramatically shifts the longer wavelength of compound 8N8O due to the protonation of N atoms in the framework of compound 8N8O. Compound 8N8O emits strong green light at 504 nm and 539 nm in NMP solution. The former is monomer emission and the later is excimer emission, which is confirmed through the emission spectra of compound 8N8O in NMP solution with different concentration. Normalized emission spectra of compound 8N8O in NMP solution with different concentration was shown in Fig. S5,† the PL intensity of excimer was decreased with decreasing the concentration. Compound 8N8O exhibits good thermal stability, with an onset decomposition temperature of ∼341 °C (considering the temperature for 5% weight loss, Fig. S6†).
 | ||
| Fig. 1 Normalized absorption/emission of compound 8N8O ((a) and (c) in NMP solution, and (b) in TFA solution, λex = 427 nm). | ||
Fig. S7† shows the cyclic voltammogram (CV) curve of compound 8N8O in a 0.1 M solution of tetrabutylammonium hexafluorophosphate (TBAPF6) in anhydrous NMP solution. Cyclic voltammetry measurements were carried out on a CHI 604E electrochemical analyzer. Glassy carbon (diameter: 1.6 mm; area 0.02 cm2) was used as a working electrode, platinum wires were used as counter electrode and reference electrode, respectively. Fc+/Fc was used as an internal standard. Potentials were recorded versus Fc+/Fc in a solution of anhydrous N-methyl-2-pyrrolidone (NMP) with 0.1 M tetrabutylammonium hexafluorophosphate (TBAPF6) as a supporting electrolyte at a scan rate of 100 mV s−1. The onset oxidation potential for compound 8N8O is 1.0 V, which correspond to the HOMO energy levels of −5.56 eV from the onset oxidation potential with reference to Fc+/Fc (−4.8 eV) using the equation of EHOMO = −[4.8 − EFc + Eonsetox] eV. Thus, the LUMO level is estimated to be −3.13 eV using the equation of ELUMO = (EHOMO + Eoptg) eV (Eoptg = (1240/λonset) nm, λonset = 510 nm). Theoretical calculation of compound 8N8O was performed using the density functional theory (DFT) method of B3LYP with the 6-31G (d) basis set.11 Fig. S8† shows that the electron density distributions of the HOMO mainly locate on aromatic groups while the LUMO orbital is mainly distributed on tetraazaanthracene, which is a strong electron acceptor. The calculated HOMO and LUMO energy levels for compound 8N8O are −5.75 eV and −2.56 eV, respectively, which are in agreement with the experimental values (−5.56 eV and −3.13 eV).
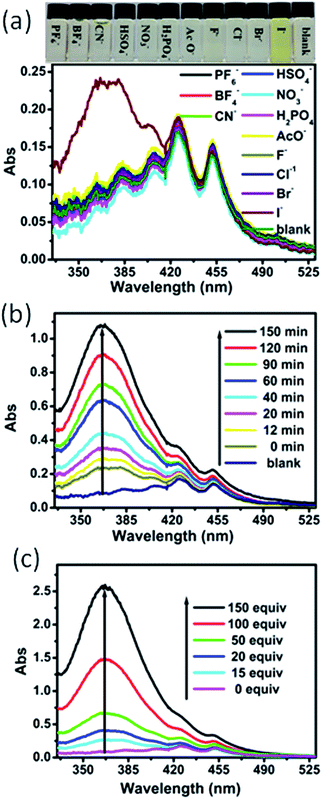
The responses of compound 8N8O to eleven anions (F−, Cl−, Br−, I−, PF6−, HSO4−, NO3−, BF4−, AcO−, CN−, and H2PO4−, 2.0 × 10−4 mol L−1) were investigated by UV-Vis absorption and fluorescence spectroscopy in NMP solution. Fig. 2a shows that there are no color change occurred in the presence of ten anions except iodide. Upon addition of iodide, a new absorption band centre at 368 nm was observed, which induced the color change to yellow. Moreover, the absorption intensity at 368 nm increased with increase in time (Fig. 2b). Fig. 2c displays the absorption titration spectra of compound 8N8O with the addition of tetrabutylammonium iodides. Upon addition of iodide, the absorption intensity at 368 nm was increased. Further competition experiments were performed to demonstrate that the coexistence of these ions does not interfere with the detection of iodide. Fig. S9† shows the absorption response of compound 8N8O towards iodide in the presence of other competitive ions. The absorption spectrum of compound 8N8O in the presence of mixed anions without iodide was similar to the absorption spectrum in the absence, but a new absorption band centre at 368 nm was observed in the presence of mixed anions with iodide. These results suggest that compound 8N8O can function as a selective for iodide. This result was confirmed by the emission spectra of compound 8N8O in the presence of eleven different anions. As shown in Fig. 3a, upon excitation at 427 nm (the compound 8N8O maximum absorption in NMP solution), the free and eleven anions compound 8N8O displays two emission bands centre at 504 nm and 539 nm, respectively, but the PL intensity is quenched in the presence iodide. Fig. 3b and c shows that the PL intensity was decreased with increasing time and concentration of iodide.
The changes in the photophysical properties of compound 8N8O complexation with iodide observed in the UV-Vis absorption and fluorescence spectroscopy results are similar to the previous reports, which exhibited that the changes in photophysical properties are due to the “heavy-atom” effect.9 In addition, there are two types of quenching of the “heavy-atom” effect, one is static quenching through the formation of a complex between a receptor (emitter) and an anion (quencher) and the other is the dynamic quenching due to the random collisions between the emitter and the quencher. In the present investigation, compound 8N8O shows static quenching upon addition of iodide since a new absorption at 368 nm was observed.
In conclusion, we have successfully designed and synthesized a novel heteroacenes, 8N8O, containing two different types of heteroatoms (nitrogen and oxygen atoms) in its backbones. Interestingly, compound 8N8O can act as an efficient anion sensor for iodide over a wide range of other anions (F−, Cl−, Br−, PF6−, HSO4−, NO3−, BF4−, AcO−, CN−, and H2PO4−) due to the “heavy-atom” effect.
Acknowledgements
Q. Z. acknowledges financial support from AcRF Tier 1 (RG133/14) and Tier 2 (ARC 20/12 and ARC 2/13) from MOE, and the CREATE program (Nanomaterials for Energy and Water Management) from NRF, Singapore. Q. Z. also thanks the support from Open Project of State Key Laboratory of Supramolecular Structure and Materials (Grant number: sklssm2015027), Jilin University, China.Notes and references
-
(a) S. Miao, A. L. Appleton, N. Berger, S. Barlow, S. R. Marder, K. I. Hardcastle and U. H. Bunz, Chem.–Eur. J., 2009, 15, 4990 CrossRef CAS PubMed
; (b) U. H. Bunz, Chem.–Eur. J., 2009, 15, 6780 CrossRef CAS PubMed
; (c) U. H. Bunz, J. U. Engelhart, B. D. Lindner and M. Schaffroth, Angew. Chem., Int. Ed., 2013, 52, 3810 CrossRef CAS PubMed
; (d) W. Jiang, Y. Li and Z. Wang, Chem. Soc. Rev., 2013, 42, 6113 RSC
; (e) Z. Liang, Q. Tang, J. Xu and Q. Miao, Adv. Mater., 2011, 23, 1535 CrossRef CAS PubMed
; (f) X. Xu, G. Yu, S. Chen, C. A. Di and Y. Liu, J. Mater. Chem., 2008, 18, 299 RSC
; (g) C. Fu, M. Li, Z. Su, Z. Hong, W. Li and B. Li, Appl. Phys. Lett., 2006, 88, 093507 CrossRef PubMed
; (h) J. Wu, X. Rui, G. Long, W. Chen, Q. Yan and Q. Zhang, Angew. Chem., Int. Ed., 2015, 54, 7354 CrossRef CAS PubMed
; (i) J. Li and Q. Zhang, ACS Appl. Mater. Interfaces, 2015 DOI:10.1021/acsami.5b00113
; (j) G. Li, J. Miao, J. Cao, J. Zhu, B. Liu and Q. Zhang, Chem. Commun., 2014, 50, 7656 RSC
; (k) C. Wang, P. Gu, B. Hu and Q. Zhang, J. Mater. Chem. C, 2015 10.1039/C5TC02080H
.
-
(a) T. K. Mullenbach, K. A. McGarry, W. A. Luhman, C. J. Douglas and R. J. Holmes, Adv. Mater., 2013, 25, 3689 CrossRef CAS PubMed
; (b) J. Li, Y. Zhao, J. Lu, G. Li, J. Zhang, Y. Zhao, X. Sun and Q. Zhang, J. Org. Chem., 2015, 80, 109 CrossRef CAS PubMed
; (c) J. Li and Q. Zhang, Synlett, 2013, 24, 686 CrossRef CAS
; (d) D. Rodriguez-Lojo, D. Perez, D. Pena and E. Guitian, Chem. Commun., 2015, 51, 5418 RSC
; (e) J. Xiao, H. M. Duong, Y. Liu, W. Shi, L. Ji, G. Li, S. Li, X.-W. Liu, J. Ma, F. Wudl and Q. Zhang, Angew. Chem., Int. Ed., 2012, 51, 6094 CrossRef CAS PubMed
; (f) J. Xiao, S. Liu, Y. Liu, L. Ji, X. Liu, H. Zhang, X. Sun and Q. Zhang, Chem.–Asian J., 2012, 7, 561 CrossRef CAS PubMed
; (g) J. Xiao, C. D. Malliakas, Y. Liu, F. Zhou, G. Li, H. Su, M. G. Kanatzidis, F. Wudl and Q. Zhang, Chem.–Asian J., 2012, 7, 672 CrossRef CAS PubMed
.
-
(a) G. J. Richards, J. P. Hill, T. Mori and K. Ariga, Org. Biomol. Chem., 2011, 9, 5005 RSC
; (b) M. M. Payne, S. R. Parkin and J. E. Anthony, J. Am. Chem. Soc., 2005, 127, 8028 CrossRef CAS PubMed
; (c) B. Gao, M. Wang, Y. Cheng, L. Wang, X. Jing and F. Wang, J. Am. Chem. Soc., 2008, 130, 8297 CrossRef CAS PubMed
; (d) S. Chen, F. S. Raad, M. Ahmida, B. R. Kaafarani and S. H. Eichhorn, Org. Lett., 2013, 15, 558 CrossRef CAS PubMed
; (e) H. Qu and C. Chi, Org. Lett., 2010, 12, 3360 CrossRef CAS PubMed
; (f) C. Wang, J. Zhang, G. Long, N. Aratani, H. Yamada, Y. Zhao and Q. Zhang, Angew. Chem., Int. Ed., 2015, 54, 6292 CrossRef CAS PubMed
; (g) P.-Y. Gu, Y. Zhao, J.-H. He, J. Zhang, C. Wang, Q.-F. Xu, J.-M. Lu, X. W. Sun and Q. Zhang, J. Org. Chem., 2015, 80, 3030–3035 CrossRef CAS PubMed
; (h) Q. Zhang, J. Xiao, Z. Y. Yin, H. M. Duong, F. Qiao, F. Boey, X. Hu, H. Zhang and F. Wudl, Chem.–Asian J., 2011, 6, 856 CrossRef CAS PubMed
.
-
(a) G. Li, K. Zheng, C. Wang, K. S. Leck, F. Hu, X. W. Sun and Q. Zhang, ACS Appl. Mater. Interfaces, 2013, 5, 6458 CrossRef CAS PubMed
; (b) P. Y. Gu, F. Zhou, J. Gao, G. Li, C. Wang, Q. F. Xu, Q. Zhang and J. M. Lu, J. Am. Chem. Soc., 2013, 135, 14086 CrossRef CAS PubMed
; (c) C. Wang, J. Wang, P. Li, J. Gao, S. Y. Tan, W. Xiong, B. Hu, P. S. Lee, Y. Zhao and Q. Zhang, Chem.–Asian J., 2014, 9, 779 CrossRef CAS PubMed
; (d) Y. Wu, Z. Yin, J. Xiao, Y. Liu, F. Wei, K. J. Tan, C. Kloc, L. Huang, Q. Yan, F. Hu, H. Zhang and Q. Zhang, ACS Appl. Mater. Interfaces, 2012, 4, 1883 CrossRef CAS PubMed
.
-
(a) M. Mascal, I. Yakovlev, E. B. Nikitin and J. C. Fettinger, Angew. Chem., Int. Ed., 2007, 46, 8782 CrossRef CAS PubMed
; (b) B. P. Hay and V. S. Bryantsev, Chem. Commun., 2008, 2417 RSC
; (c) J. Mareda and S. Matile, Chem.–Eur. J., 2009, 15, 28 CrossRef CAS PubMed
; (d) G. Gil-Ramírez, E. C. Escudero-Adán, J. Benet-Buchholz and P. Ballester, Angew. Chem., Int. Ed., 2008, 120, 4182 CrossRef PubMed
; (e) R. E. Dawson, A. Hennig, D. P. Weimann, D. Emery, V. Ravikumar, J. Montenegro, T. Takeuchi, S. Gabutti, M. Mayor, J. Mareda, C. A. Schalley and S. Matile, Nat. Chem., 2010, 2, 533 CrossRef CAS PubMed
.
- G. Li, Y. Wu, J. Gao, J. Li, Y. Zhao and Q. Zhang, Chem.–Asian J., 2013, 8, 1574 CrossRef CAS PubMed
.
-
(a) B. Ma, F. Zeng, F. Zheng and S. Wu, Chem.–Eur. J., 2011, 17, 14844 CrossRef CAS PubMed
; (b) D. Y. Lee, N. Singh, M. J. Kim and D. O. Jang, Org. Lett., 2011, 13, 3024 CrossRef CAS PubMed
.
-
(a) H. A. Ho and M. Leclerc, J. Am. Chem. Soc., 2003, 125, 4412 CrossRef CAS PubMed
; (b) A. Corma, M. S. Galletero, H. García, E. Palomares and F. Rey, Chem. Commun., 2002, 1100 RSC
; (c) H. J. Jung, N. Singh, D. Y. Lee and D. O. Jang, Tetrahedron Lett., 2010, 51, 3962 CrossRef CAS PubMed
; (d) M. Wang, Z. Wu, J. Yang, G. Wang, H. Wang and W. Cai, Nanoscale, 2012, 4, 4087 RSC
; (e) E. M. Zahran, Y. Hua, S. Lee, A. H. Flood and L. G. Bachas, Anal. Chem., 2011, 83, 3455 CrossRef CAS PubMed
.
-
(a) M. Vetrichelvan, R. Nagarajan and S. Valiyaveettil, Macromolecules, 2006, 39, 8303 CrossRef CAS
; (b) R. M. Duke, E. B. Veale, F. M. Pfeffer, P. E. Kruger and T. Gunnlaugsson, Chem. Soc. Rev., 2010, 39, 3936 RSC
.
-
(a) D.-C. Lee, B. Cao, K. Jang and P. M. Forster, J. Mater. Chem., 2010, 20, 867 RSC
; (b) K. K. McGrath, K. Jang, K. A. Robins and D. C. Lee, Chem.–Eur. J., 2009, 15, 4070 CrossRef CAS PubMed
.
-
(a) S. Kim, Q. Zheng, G. S. He, D. J. Bharali, H. E. Pudavar, A. Baev and P. N. Prasad, Adv. Funct. Mater., 2006, 16, 2317 CrossRef CAS PubMed
; (b) P.-Y. Gu, J. Gao, C.-J. Lu, W. Chen, C. Wang, G. Li, F. Zhou, Q.-F. Xu, J.-M. Lu and Q. Zhang, Mater. Horiz., 2014, 1, 446 RSC
.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental section. See DOI: 10.1039/c5ra17899a |
| This journal is © The Royal Society of Chemistry 2015 |