Polydiacetylene stabilized gold nanoparticles – extraordinary high stability and integration into a nanoelectrode device†
R. Liffmanna,
M. Homberger*a,
M. Mennickenb,
S. Karthäuserb and
U. Simona
aInstitute of Inorganic Chemistry, JARA – Fundamentals of Future Information Technologies, RWTH Aachen University, D-52074 Aachen, Germany. E-mail: melanie.homberger@ac.rwth-aachen.de
bPeter Grünberg Institut (PGI-7), JARA – Fundamentals of Future Information Technologies, Forschungszentrum Jülich GmbH, D-52425 Jülich, Germany
First published on 26th November 2015
Abstract
A new diacetylene containing photopolymerizable ligand molecule was developed, and tailored for applications in nanoelectronic devices based on gold nanoparticles. This ligand molecule consists of a thiol group, a diacetylene unit and a terminal carboxylic group. The thiol group guarantees preferred binding to the gold nanoparticles surface whereas at the same time the carboxylic group enables electrostatic stabilization. Applying this ligand molecule, gold nanoparticles in the size range of 12–13 nm were prepared. The diacetylene unit was polymerized upon UV irradiation leading to a polymeric ligand shell. Investigations including colloidal stability towards NaCl, DTT displacement reactions, and temperature were performed and indicate an extraordinary high degree of steric and electrostatic stabilization. Individual or at least a few of these particles were immobilized in between nanoelectrodes, thus forming nanoelectronic devices, which were characterized by transport measurements.
Introduction
Ligand stabilized metal nanoparticles are intensively studied objects of the last few decades.1 This extraordinary high research interest arises from the unique size dependent properties in combination with the possibility of implementing tailored features, like selective binding ability, aggregation behavior, or bioresponse, by applying appropriate molecules forming the ligand shell.2–6Ligand stabilized gold nanoparticles (AuNP) exhibit high chemical stability and versatile possibilities to implement distinct, additional functionalities into the ligand shell.7 As a consequence tailored properties of AuNP with respect to various applications ranging from nanoelectronics, to optical sensors, or biomedical applications, can be realized.8–13 The naked gold core itself determines the electrical and optical properties, i.e. Coulomb blockade behavior in the case of AuNP core sizes or plasmon resonance absorption. In this context, we recently studied the pH-dependent reversible aggregation properties as well as the differential adsorption of carboxyl- or amine-terminated AuNP on different metals.14 The possibility to control the adsorption characteristics represents a key aspect for the integration of molecular electronic devices based on functionalized AuNP into traditional CMOS devices. Recently, we succeeded in immobilizing individual ligand stabilized AuNP in between e-beam lithographically fabricated nanoelectrodes by choosing appropriate ligand molecules,15 even in a directed manner.16 Moreover, the resulting I/U-characteristics from these devices directly displayed the molecular transport properties of the ligand molecules. Thus, the electrical properties in such a device depend on the size of the particle, which determines the electrical charging energy, the chemical composition and the length of ligand molecules, as well as the coupling of the molecules to the nanoparticle and the electrodes. So far our results demonstrate that, by choosing tailored ligand molecule-AuNP combinations, the overall transport characteristics in a nanoelectronic device can be adjusted. This fact represents a key step towards the construction of functional nanoelectronic devices like e.g. diodes or switches.
In principle, ligand stabilized AuNP applied in such devices have to fulfill following requirements: (i) the ligand molecules should exhibit not too high electrical resistances in order to allow electrical transport through the device. (ii) Terminal functional groups in the outer sphere of the ligand shell are required that allow proper binding to the electrodes. At the same time, this binding affinity should not exceed the binding affinity of the ligand molecule to the AuNP surface in order to avoid undesired precipitation.17,18 (iii) Moreover, regarding the integration and process ability, the applied ligand stabilized AuNP should exhibit a high degree of stabilization towards ligand displacement reactions due to external stimuli, like temperature or solvent treatment, in order to guarantee reliable and reproducible device fabrication.
Generally, nanoparticle stabilization is achieved either by electrostatic or steric stabilization or combinations of it. Electrostatic stabilization bases on electrostatic repulsion of the diffuse electrical double layer formed around nanoparticles exhibiting surface charges. Steric stabilization bases on geometric constraints around the nanoparticles surface, formed when large and bulky ligand molecules or molecules binding by a polydentate nature to the nanoparticles' surface are applied. According to these concepts highest particle stabilization is achieved when polymeric ligands forming polydentate binding to the AuNP surface and charged groups in the outer ligand sphere, e.g. deprotonated carboxylic groups, are applied.
Up to now several strategies have been developed to enhance the stability of the ligand binding to AuNP and reduce potential ligand desorption or AuNP aggregation. Among these, the predominant strategies are (i) creating crosslinks between terminal functional groups in the outer sphere of the ligand shell, e.g., esterification of terminal carboxylic acid group or (ii) utilizing ligand molecules that form multidentate bonds to the AuNP surface, e.g., molecules bearing multiple thiol groups.19,20 However, the first case is accompanied by losing the accessibility of the terminal carboxylic group and the advantage of additional electrostatic stabilization. In the second case a molecular conformation is required where thiol groups with a short spatial distance point into the same direction, otherwise the multidentate nature of the ligand molecule could lead to interparticle cross linking accompanied by undesired particle aggregation.
In this context Kanaras et al. presented an alternative approach leading to extraordinary high stabilized AuNP utilizing diacteylene containing ligand molecules. Diacetylene containing molecules are known to undergo polymerization via 1,4-addition if properly assembled. Thereby in the first step Kanaras et al. prepared AuNP which were stabilized by monomeric diacetylene containing ligand molecules. The formed ligand shell was then internally cross linked upon UV light irradiation, inducing 1,4-addition between neighboring diacetylene units.21 Thus, a polydentate ligand shell was created. The ligand molecule they used was [46-mercapto-22,43-dioxo-3,6,9,12,15,18-hexaoxa-21,44-diazahexatetraconta-31,33-diyn-1-oic acid], a polyethylene glycol derivative. However, though this molecule fulfills the requirement of high stabilization, the molecular length of about 4.7 nm leads to an expected resistance in the petaohm region. Consequently this ligand molecule is not suitable for applications in nanoelectronic devices.
Another approach published utilized a significantly shorter diacetylene derivative, which seems to be more suitable for nanoelectronic applications, namely 3,5-octadiyne-1-ol-8-thiol. Applying this molecule the authors prepared 5 nm Janus-type AuNP, whereby the photopolymerizable diacetylene ligand covered one hemisphere of the AuNP surface.22 However, according to investigations performed by Evans and co-workers,23 the polymerization degree in self-assembled monolayers (SAMs) consisting of diacetylene derivatives immobilized on gold surfaces, decreases significantly with shorter distances between the diacetylene moiety and the anchoring group (spacer length). They observed nearly no polymerization in SAMs consisting of diacetylenes exhibiting spacer lengths of less than four CH2-units.
Based on these findings we developed a new water soluble photopolymerizable diacetylene (DA) containing ligand molecule tailored for implementation in nanoelectronic devices, exhibiting a spacer length of five CH2 units (DA-COOH, Scheme 1a). Thus polymerization is facilitated according to the investigations of Evans and particles are stabilized by a polymeric ligand shell, which ensures steric stabilization due to geometric constraints of the polymer and additional electrostatic stabilization, if the terminal carboxylic group is deprotonated. Furthermore, the thiol anchor group guarantees a preferred binding to the AuNP's surface, due to the well-known significant high binding affinity of thiol to gold surfaces (Au(0)-S: 167 kJ mol−1).24 The terminal carboxylic acid group ensures the linkage to electrodes in the respective AuNP device.25
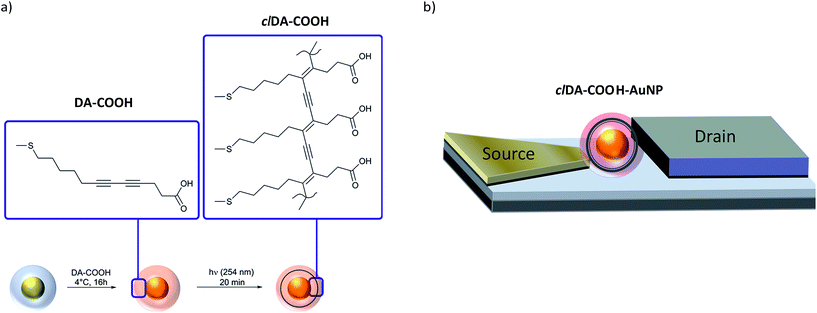 | ||
| Scheme 1 (a) Preparation of clDA-COOH–AuNP. (b) Configuration for the electrical characterization of individual clDA-COOH–AuNP (not drawn to scale). | ||
Here, we report on the preparation and characterization of the tailored ligand, DA-COOH and the respective DA-COOH–AuNP, before (DA-COOH–AuNP) and after photoinduced polymerization (clDA-COOH–AuNP) (Scheme 1a). We applied AuNP in the size range of 12–13 nm.
The particles were characterized by UV-vis spectroscopy, infrared reflection absorption spectroscopy (IRRAS), surface enhanced Raman scattering, ζ-potential as well as scanning electron microscopy in transmission mode (SEM-T) measurements. Stability investigations were performed including stability towards NaCl, ligand displacement against dithiothreithol (DTT) and heating. As the SPR is sensitive to changes in the ligand shell, particle size and interparticle distance,26 these investigations involve mainly UV-vis measurements. Particle aggregation upon decreasing nanoparticle stabilization is reflected by a red-shift and significant broadening, or intensity lost, of the SPR. Transport characteristics of individual clDA-COOH–AuNP were investigated applying the same nanoelectrode setup used in our investigations reported previously (Scheme 1b).15
Experimental
All reagents were purchased either from Acros, Fisher Scientific or Sigma Aldrich and if not mentioned otherwise used without further purification. Ultrapure water with a conductivity < 55 nS cm−1 was used for all procedures.UV-vis absorption spectra were recorded with a JASCO V-630 spectrophotometer using quartz glass cuvettes.
Dynamic light scattering (DLS) measurements and ζ-potential measurements were performed with a Malvern Zetasizer Nano S, He–Ne laser, λ = 633 nm, P = 4 mW, θ = 173°.
IRRAS measurements were performed on a FT-IR spectroscope Vertex 70, Bruker Optics equipped with a MCT detector at a spectral resolution of 4 cm−1.
Scanning electron microscopy (SEM) in transmission mode was conducted with a high-resolution field emission scanning electron microscope (FE-SEM, LEO/ZEISS Supra 35 VP, Oberkochen, Germany). For sample preparation 5 μL of the particle solution were dropped on a carbon-coated copper grid and incubated for 30 min. The remaining solution was removed with a tissue.
Raman and SERS spectra were obtained using a Horiba–Jobin–Yvon Raman spectrometer equipped with Olympus objectives and used at 50 fold magnification and a laser wavelength of 633 nm at 180° backscattering arrangement. The spectral resolution was 7 cm−1 and the power on the sample was 12 mW. Particle solution was dropped and dried on a sputtered Au layer on silicon wafer with a thickness of 100 nm.
TGA measurements were conducted on a Mettler STARe SW 9.20 instrument. 4–5 mg of dried AuNP and pure DA-COOH ligand were weighed in standard alumina pans, and the measurements were carried out between 25 and 600 °C at a heating rate of 10 °C min−1 under a nitrogen flow of 60 mL min−1.
NMR measurements were performed with a Bruker Avance II-400 and Bruker II-HD and referenced to solvent residual signal.
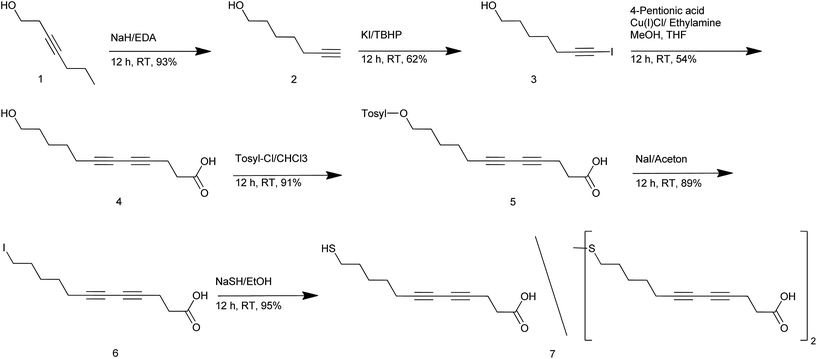
Preparation of diacetylene containing ligand molecules (7)
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CH), 68.3 (–C
CH), 68.3 (–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CH), 62.8 (–CH2–OH), 32.2 (–CH2–CH2–OH), 28.2 (–CH2–(CH2)3–OH), 24.9 (–CH2–(CH2)2–OH), 18.4 (–CH2–C
CH), 62.8 (–CH2–OH), 32.2 (–CH2–CH2–OH), 28.2 (–CH2–(CH2)3–OH), 24.9 (–CH2–(CH2)2–OH), 18.4 (–CH2–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CH).
CH).![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif)
![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) –), 62.71 (HO–
–), 62.71 (HO–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 60.22 (I–
H2–), 60.22 (I–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif)
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–), 32.13 (HO–CH2–
C–), 32.13 (HO–CH2–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2), 28.22 (HO–(CH2)3–
H2), 28.22 (HO–(CH2)3–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2), 24.92 (HO–(CH2)2–
H2), 24.92 (HO–(CH2)2–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2), 20.76 (–
H2), 20.76 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–C
H2–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–).
C–).![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) OOH), 78.05 (–C
OOH), 78.05 (–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif)
![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) –(CH2)2–COOH), 74.79 (HO–(CH2)5–
–(CH2)2–COOH), 74.79 (HO–(CH2)5–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif)
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–), 66.13 (–
C–), 66.13 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif)
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–(CH2)2–COOH), 65.19 (HO–(CH2)5–C
C–(CH2)2–COOH), 65.19 (HO–(CH2)5–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif)
![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) –), 62.73 (–
–), 62.73 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–OH), 32.73 (–
H2–OH), 32.73 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–COOH), 32.01 (–
H2–COOH), 32.01 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–CH2–OH), 27.97 (HO–(CH2)3–
H2–CH2–OH), 27.97 (HO–(CH2)3–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 24.94 (HO–(CH2)2–
H2–), 24.94 (HO–(CH2)2–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 19.13 (HO–(CH2)4–
H2–), 19.13 (HO–(CH2)4–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 14.91 (–
H2–), 14.91 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–CH2–COOH).
H2–CH2–COOH).![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) OOH), 77.79 (–C
OOH), 77.79 (–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif)
![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) –(CH2)2–COOH), 74.74 (I–(CH2)5–
–(CH2)2–COOH), 74.74 (I–(CH2)5–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif)
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–), 66.16 (–
C–), 66.16 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif)
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–(CH2)2–COOH), 65.35 (I–(CH2)5–C
C–(CH2)2–COOH), 65.35 (I–(CH2)5–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif)
![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) –), 32.97 (–
–), 32.97 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–COOH), 32.76 (–
H2–COOH), 32.76 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–CH2–I), 29.68 (I–(CH2)2–
H2–CH2–I), 29.68 (I–(CH2)2–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 27.15 (I–(CH2)3–
H2–), 27.15 (I–(CH2)3–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 19.01 (I–(CH2)4–
H2–), 19.01 (I–(CH2)4–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 14.86 (–
H2–), 14.86 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–CH2–COOH), 6.34 (–
H2–CH2–COOH), 6.34 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–I).
H2–I).![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) OOH), 78.00 (–C
OOH), 78.00 (–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif)
![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) –(CH2)2–COOH), 74.67 (–(CH2)5–
–(CH2)2–COOH), 74.67 (–(CH2)5–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif)
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–), 66.19 (–
C–), 66.19 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif)
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–(CH2)2–COOH), 65.23 (–(CH2)5–C
C–(CH2)2–COOH), 65.23 (–(CH2)5–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif)
![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) –), 38.92 (–S–S–
–), 38.92 (–S–S–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 33.46 (–
H2–), 33.46 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–COOH), 32.81 (–
H2–COOH), 32.81 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–COOH), 32.75 (HS–CH2–
H2–COOH), 32.75 (HS–CH2–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 28.80 (–S–S–CH2–
H2–), 28.80 (–S–S–CH2–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 27.80 (–S–S–(CH2)3–
H2–), 27.80 (–S–S–(CH2)3–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 27.71 (–S–S–(CH2)2–
H2–), 27.71 (–S–S–(CH2)2–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 27.67 (HS–(CH2)3–
H2–), 27.67 (HS–(CH2)3–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 27.52 (HS–(CH2)2–
H2–), 27.52 (HS–(CH2)2–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 24.41 (HS–
H2–), 24.41 (HS–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–), 19.08 (–(CH2)4–
H2–), 19.08 (–(CH2)4–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–
H2–![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–), 14.86 (–
C–), 14.86 (–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H2–CH2–COOH). Neg.-ESI-MS, m/z: calculated: 446.16 (disulfide), 224.1 (thiol), found: 445.2 (disulfide-1), 223.1 (thiol-1).
H2–CH2–COOH). Neg.-ESI-MS, m/z: calculated: 446.16 (disulfide), 224.1 (thiol), found: 445.2 (disulfide-1), 223.1 (thiol-1).Preparation of clDA-COOH–AuNP
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 rpm, 15 min) and redispersed in 2 mL of respective buffer which was either phosphate buffer (pH 8, 0.1 mol L−1 or 0.01 mol L−1) or Hepes/Tris buffer (pH 8, 0.01 mol L−1).
500 rpm, 15 min) and redispersed in 2 mL of respective buffer which was either phosphate buffer (pH 8, 0.1 mol L−1 or 0.01 mol L−1) or Hepes/Tris buffer (pH 8, 0.01 mol L−1).Stability investigations
For heating cycles freshly prepared irradiated DA-COOH–AuNP were sealed in a plastic cuvette and placed in the DLS device. The heating range was 20–70 °C. The heating rate was set to 5 °C/10 min and the final temperature (20 °C, 70 °C) was maintained for 1 h. Three heating/cooling cycles were performed.
Electrical characterization of individual polydiacetylene stabilized AuNP in a heterometallic nanogap
In order to electrically characterize the prepared clDA-COOH–AuNP, individual or at least a few AuNP were immobilized in between heterometallic nanoelectrodes. Nanoelectrode pairs were processed by a multiple step E-beam-lithography process combined with a self-alignment procedure on a SiO2 surface as described previously.15,28 The received heterometallic nanoelectrode pairs consist of one Pt- and one AuPd-electrode and exhibit gap sizes in the range of 10 to 14 nm. In order to deposit AuNPs in between the nanoelectrodes a 15 μL droplet of clDA-COOH–AuNP dissolved in phosphate buffer solution at pH 8 was dropped on a 6 × 6 mm2 sample surface with 48 nanogaps. Individual or at least a few clDA-COOH–AuNP were immobilized using capillary forces during the drying process of the AuNP solution.29 Subsequently, the sample was washed with DI-water to remove residual buffer solution and blown dry with nitrogen. Electrical characterization was performed with a Keithley 6430 sub-femtoampere remote source meter at room temperature under ambient conditions. Cyclic current/voltage (I/U) measurements were recorded with the Pt-electrode connected to ground and a cyclic voltage applied to the AuPd-electrode.Results and discussion
Preparation of DA-COOH
The diacetylene derivative DA-COOH (Scheme 1a) was synthesized involving six reaction steps, as depicted in Scheme 2. In the first step hept-3-yn-1-ol (1) was converted into the respective ω-alkyne-ol (2) adopting the so-called “Alkyne-Zipper”30 reaction. After substituting the acidic alkyne proton of 2 by iodine, we applied the “Cadiot-Chodkiewicz”31 coupling of compound 3 with pent-4-ynoic acid resulting in the diacetylene precursor 4 (DA-OH). This reaction step as well as the following steps was performed under light exclusion in order to avoid uncontrolled photoinduced polymerization. In the following the alcohol functionality was converted into the corresponding thiol involving tosylation, nucleophilic substitution with iodine (NaI) and finally, the reaction with sodium hydrogen sulfide. DA-COOH was obtained as a pale yellow solid. The last step, the conversion into the thiol, was performed in Ar atmosphere applying dry ethanol, due to the well-known tendency of thiols to form disulfides upon oxidation under ambient atmosphere.32 Especially primary alkanethiols are known to be more sensitive towards oxidation than tertiary alkanethiols. However, although we performed the reaction extremely carefully in Ar atmosphere under light exclusion, 1H-NMR measurements of the obtained product always revealed a significant amount of the respective disulfide (about 40%, see Experimental section). Several attempts to get rid of the disulfide or at least to lower the content were not successful. However, since it is well-known that disulfides adsorb on gold surfaces33 in the same way as thiols, the obtained thiol/disulfide mixture was used in the following reactions.Preparation of clDA-COOH–AuNP
In the first step of the preparation of clDA-COOH–AuNP, citrate-AuNP (11.3 nm ± 1.1 nm) were subjected to a homogeneous ligand exchange reaction. The ligand exchange reaction was performed overnight at 4 °C applying an excess of DA-COOH, thereby carefully excluding light in order to avoid uncontrolled photoinduced polymerization at this point.Ligand excess as well as released citrate was removed by repeatedly centrifugation and redispersion in phosphate buffer solution (0.1 M, pH 8) at ambient temperature under light exclusion. The alkaline medium was chosen in order to avoid agglomeration due to poor electrostatic stabilization of the carboxylic acid terminated AuNP at lower pH values. The obtained DA-COOH–AuNP were characterized by UV-vis spectroscopy and infrared reflection absorption spectroscopy (IRRAS). UV-vis spectra (see ESI†) revealed a red-shift of the surface plasmon resonance absorption (SPR) maximum of 4 nm with respect to the starting material, reflecting the modified ligand binding situation (carboxyl towards thiol). For IRRAS measurements the aqueous dispersion was incubated for 2 h on a solid support. In the IRRAS spectrum the characteristic absorption bands for the DA-COOH were detected at 2929 cm−1, 2857 cm−1, 1783 cm−1 and 1573 cm−1, whereby the absorptions at 1783 cm−1 and 1573 cm−1 can be assigned to COOH stretching vibrations (ν(CO) and ν(CO–H)). The absorption at 2929 cm−1 and 2857 cm−1 arise from CH2 stretching vibrations (νas(CH2)) and (νs(CH2)).34 Additionally, the characteristic vibration for the citrate-AuNP at 1394 cm−1 cannot be observed which indicates complete ligand exchange (see ESI†).
In order to induce cross-linking within the DA-COOH ligand shell, the DA-COOH–AuNP were exposed to UV light (254 nm, 6 W) for 15 min. No change of the initially red coloured particle dispersion was observed during irradiation. From scanning electron microscopy investigations in transmission modus (SEM-T) an average particle size of 12.3 nm ± 1.4 nm (231 AuNP counted, Fig. 1) was deduced, confirming that the average particle size was maintained during ligand exchange and illumination.
 | ||
| Fig. 1 Representative SEM-T image of clDA-COOH–AuNP with histogram (inset). The particle size was determined to 12.3 nm ± 1.4 nm (based on 231 AuNP counted). | ||
With respect to further particle characterization, ζ-potential measurements at different pH values were performed.
At pH ≥ 5 the clDA-COOH–AuNP exhibit a negative value for the ζ-potential, reflecting the negative charge of the deprotonated carboxyl group. Whereas lowering the pH < 5 results in values for the ζ-potential approaching zero, resembling protonation of the carboxylate group (see ESI†).
For carboxyl-terminated AuNP a pH-dependent, reversible aggregation behavior upon hydrogen-bond-mediated aggregation processes are characteristic.14,35,36 These aggregation processes were monitored by UV-vis spectroscopy investigations with the clDA-COOH–AuNP in order to prove the existence of terminal COOH-groups after ligand polymerization (Fig. 2). We varied the pH value of the clDA-COOH–AuNP aqueous dispersion between pH 9 and pH 3. At pH 9 one sharp absorption band is observed, corresponding to the SPR of individual AuNP. Lowering the pH to 3 causes a red-shift and significant broadening of the SPR, reflecting the formation of aggregates. This aggregate formation is also observable by a color change of the dispersion from red to blue. Subsequent addition of NaOH, thus raising the pH of the AuNP dispersion again to 9, resulted in a color change back from blue to red and the UV-vis spectrum revealed again the significant absorption band for the SPR of individual AuNP, though with lower intensity. The slight decrease of absorption intensity arises from dilution effects upon adding NaOH or HCl solution, respectively (UV-vis spectra demonstrating the dilution effect on the UV-vis absorption intensity are included in the ESI†). We repeated this process several times and observed always this characteristic switching between aggregated and individual particle state, thus proving the existence of free accessible terminal COOH.
 | ||
| Fig. 2 UV-vis spectra of pH dependent agglomeration of irradiated clDa-COOH–AuNP, while alternately adding acids (blue) and bases (red) starting with pH 9 (pink). | ||
In order to characterize the polymerization of the diacetylene moiety within the ligand shell upon UV irradiation, we performed surface enhanced Raman scattering (SERS) measurements. For this purpose samples of the dispersion containing DA-COOH–AuNP before and after irradiation, respectively, were drop-casted onto Au substrates.
The SERS spectrum of the particles before irradiation shows a signal at 2257 cm−1 (Fig. 3, red graph), which we assigned to the characteristic vibration of the diacetylene moiety. It should be noted that compared with the Raman spectrum of the unbound DA-COOH monomer in the solid state (see ESI†) this signal is shifted by 194 cm−1. We ascribe this effect to the different measurement setups of Raman and SERS. This signal is no longer observed in the SERS spectrum of the irradiated sample (Fig. 3, blue graph). At the same time signals in the region between 1000 and 1600 cm−1 raise in intensity. Between 2800 cm−1 and 3000 cm−1 OH stretching vibrations (ν(OH)) of the COOH group and of methylene groups (νas(CH2) and νs(CH2)) are located which remain unaffected by irradiation. Based on literature data34 these signals (1000–1600 cm−1) are attributed to carbon–carbon double bonds. The absence of the stretching vibrations of the carbon–carbon triple bond after irradiation, accompanied by the significant intensity increase of the vibrations of the carbon–carbon double bond, prove successful 1,4-polymerization. However, the signals assigned to the carbon–carbon double bond vibrations are very broad, which can be attributed to different polymer lengths. This is a consequence of the biradical homopolymerization mechanism.37,38 The fact that the non-irradiated sample (Fig. 3, red curve) showed low intensity signals in the region of the carbon–carbon double bonds may result from measurement initiated polymerization (laser irradiation).
We have also performed TGA measurements in order to quantify the number of DA-COOH ligands on the AuNP surface. From these measurements an average of 2825 ligands per particle is deduced (3.6 ligands per nm2). This value is slightly higher than the coverage of diacetylene-PEG ligands on AuNP (2073/Au15; 3 ligands per nm2).21 With respect to the inaccuracy of the TGA method we conclude that the surface coverage is high enough to ensure a close packing density.
Stability investigations
The stability of the here prepared clDA-COOH–AuNP refers to the concept of steric and electrostatic stabilization. First, due to the presence of terminal COOH-groups, the particles exhibit a pH-dependent surface charge in aqueous solution and are thus electrostatically stabilized, when the COOH group is deprotonated. This electrostatic stabilization is reflected in the previously described pH-dependent agglomeration properties as well as in the ζ-potential measurements. At pH 8 the particles exhibit a ζ-potential of −31 mV. Second, the polydentate nature of the clDA-COOH ligand guarantees steric stabilization due to geometric constraints formed by the polymeric ligand shell.Electrostatically stabilized AuNP are strongly influenced by the ion density present in the solution. Upon addition of halide salts to the aqueous solutions of electrostatically stabilized AuNP, the electrical double layer surrounding the particles is reduced and the electrostatic repulsion is exceeded by van der Waals attraction, thus aggregation is induced.
Accordingly we investigated the influence of NaCl on clDA-COOH–AuNP compared to non-irradiated DA-COOH–AuNP, in order to elucidate the benefit from the polymerization. Typically pure electrostatically stabilized AuNP tend to aggregate at NaCl concentrations of about 100 mM.26
Moreover, recently published literature describes that the addition of a significant excess of halide salts could also lead to an exchange of the thiol ligand against the halide, due to halide anions diffusing through the ligand sphere and adsorbing on the Au-surface. As a consequence the particles are no longer stabilized by electrostatic or steric repulsive forces, finally leading to irreversible aggregation. This effect is also observable in the case of pure sterically stabilized particles, thus the stability towards NaCl also reflects the power of the steric stabilization.39,40
Additional information concerning the steric stabilization power can be gathered from DTT displacement reactions. On the one hand the small molecule DTT is known to have a high affinity to Au surfaces due to its two thiol groups and due to its size DTT should be able to penetrate the ligand shell more easily. On the other hand addition of excess DTT to ligand stabilized AuNP displaces thiol ligands from the particles surface nearly quantitatively, thereby forming aggregates resulting in insoluble precipitate formation. Thus we performed exchange experiments on non-irradiated DA-COOH–AuNP and on clDA-COOH–AuNP with DTT in order to classify the effect of steric stabilization of monomeric and polymerized DA-COOH.
As a third parameter the prepared AuNP were investigated concerning heat stability. Integration of AuNP in a nanoelectronic setup is always accompanied with local heat evolution, thus heat stability is a further benefit with respect of building up nanoelectronic devices.
In the case of the non-irradiated DA-COOH–AuNP in 0.15 M NaCl a slight decrease of the SPR absorption intensity with at the same time slightly increasing absorption intensity in the longer wavelength regime is observed, indicating moderate stability of the particles in this medium. Moreover, in the higher ionic strength solution of 0.3 M NaCl the SPR is red-shifted and significantly broadened immediately after salt addition, reflecting the destabilization resulting in the formation of AuNP aggregates. This trend is consistent with electrostatically stabilized AuNP in a high ionic strength medium.
In contrast to that observation, in the case of clDA-COOH–AuNP, the SPR absorption of the particles in 0.15 M is unaffected over 1 h and even at high ionic strength of 0.3 M NaCl only a low increase in absorption intensity in the long-wavelength regime, pointing to aggregate formation, is observed. The maximum absorption retains at 523 nm in both cases and only a slow, continuous intensity decrease in the case of 0.3 M NaCl of the maximum absorption with time is observed.
This observation hints to additional steric stabilization of the particles which impedes aggregation, as observed in the non-irradiated samples. After 12 h incubation in 0.3 M NaCl the solution of irradiated clDA-COOH–AuNP appeared colorless and the cuvette wall violet, obviously particles were attached to the cuvette surface. We were able to partially detach the particles simply by adding phosphate buffer solution (0.1 M, pH 8) in combination with extensive ultra-sonication (UV-vis spectrum of detached particles see ESI†). Detachment of the particles was visible by reddening of the solution. We suppose that in contrast to the non-irradiated DA-COOH–AuNP, addition of NaCl in this case mainly affects the electrical double layer. The steric stabilization is effective enough, to prevent aggregation, due to the geometric constraints of the nature of the polydentate ligand. As a consequence the particles' interactions of the terminal carboxylic group with the hydrophobic cuvette surface may increase, leading to an attachment of the particles to the cuvette wall. The fact that the particles could be detached in buffer solution additionally proofs high stability.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50, see Experimental section) for 12 h at 60 °C and recorded an UV-vis spectrum in each case. The UV-vis spectra are depicted in Fig. 5a (DA-COOH–AuNP: blue solid line, clDA-COOH–AuNP: red solid line). For reference purposes the UV-vis spectra of the respective particle types without DTT addition (Fig. 5a, DA-COOH–AuNP: blue dashed line, clDA-COOH–AuNP: red dashed line) as well as the UV-vis spectrum of pure DTT after 12 h at 60 °C (Fig. 5a, black solid line) are plotted, too. After incubation of non-irradiated DA-COOH–AuNP with excess DTT, particle aggregation was already observable by the naked eye. The initially red colored particle solution has turned colorless and a black precipitate is formed (Fig. 5a, inset, left sample). Corresponding to that observation the UV-vis spectrum (Fig. 5a, blue solid line) only shows the characteristic absorption signals for DTT itself and the characteristic SPR absorption band of the DA-COOH–AuNP at 522 nm (Fig. 5a, blue dashed line) has completely vanished. This result indicates significant ligand displacement of the DA-COOH-ligand. Thus, steric constraints are obviously not sufficient to hinder the small DTT molecule to penetrate the ligand shell and reach the AuNP surface.
50, see Experimental section) for 12 h at 60 °C and recorded an UV-vis spectrum in each case. The UV-vis spectra are depicted in Fig. 5a (DA-COOH–AuNP: blue solid line, clDA-COOH–AuNP: red solid line). For reference purposes the UV-vis spectra of the respective particle types without DTT addition (Fig. 5a, DA-COOH–AuNP: blue dashed line, clDA-COOH–AuNP: red dashed line) as well as the UV-vis spectrum of pure DTT after 12 h at 60 °C (Fig. 5a, black solid line) are plotted, too. After incubation of non-irradiated DA-COOH–AuNP with excess DTT, particle aggregation was already observable by the naked eye. The initially red colored particle solution has turned colorless and a black precipitate is formed (Fig. 5a, inset, left sample). Corresponding to that observation the UV-vis spectrum (Fig. 5a, blue solid line) only shows the characteristic absorption signals for DTT itself and the characteristic SPR absorption band of the DA-COOH–AuNP at 522 nm (Fig. 5a, blue dashed line) has completely vanished. This result indicates significant ligand displacement of the DA-COOH-ligand. Thus, steric constraints are obviously not sufficient to hinder the small DTT molecule to penetrate the ligand shell and reach the AuNP surface.
In contrast to this observation, the reddening of the solution in the case of clDA-COOH–AuNP was maintained after DTT incubation (Fig. 5a, inset, right sample) and no precipitate was formed. In the UV-vis spectrum the SPR absorption band is only slightly reduced in intensity and shifted from 522 nm to 528 nm (Fig. 5a, red solid line). Furthermore beside the SPR absorption band the characteristic absorption signals for DTT were detected. This observation indicates that for clDA-COOH–AuNP ligand displacement reactions occurred only in a very small amount. The observed intensity loss and red-shift may be a result of marginal ligand displacement. Additional SEM-T investigations of these particles after DTT incubation revealed that the particles are still present as individual particles and the metal core size was unaffected (Fig. 5b).
These results affirm our above described results concerning the stability towards NaCl. Steric shielding of the AuNP metal core in the case of clDA-COOH–AuNP is effective, too. Even the small molecule DTT is significantly hindered to penetrate the ligand shell for ligand replacement.
Thermal stability
Potential effects of heating on irradiated clDA-COOH–AuNP were investigated by time dependent UV-vis spectroscopy of a particle dispersion in buffer solution (pH 8) heated up to 50 °C (see ESI†) and up to 80 °C for 5 h. Fig. 6a depicts the UV-vis spectra taken every hour at 80 °C. Remarkably, during 5 h, which represent harsh conditions for ligand stabilized AuNP, no significant change of the SPR absorption band is observed.Additionally we performed heating cycles and monitored the hydrodynamic radii by DLS measurements during repeatedly warming and cooling between 20 °C and 70 °C. The time dependent evolution of the hydrodynamic radii is displayed in Fig. 6b.
During the 3 times heating and cooling no obvious change of the hydrodynamic diameter is detected, thus aggregation upon temperature induced destabilization of the particles can be excluded. These observations confirm the extraordinary high stability of the prepared clDA-COOH–AuNP.
Electrical characterization of individual clDA-COOH–AuNP
In order to investigate the electrical transport characteristics of the prepared clDA-COOH–AuNP we applied the same setup as already described in our previous investigations on mercaptoundecanoic acid (MOA) stabilized AuNP.16Individual or at least a few clDA-COOH–AuNP were immobilized by drop-casting a solution of clDA-COOH–AuNP on the specially designed electrode structures (see Experimental section).
While drying capillary forces lead to immobilization of a single or at least a few clDA-COOH–AuNP in 10 cases. Fig. 7 depicts an exemplary SEM image of a resulting device used for the electrical characterization.
 | ||
| Fig. 7 Exemplarily SEM image of the setup used to electrically characterize an individual clDA-COOH–AuNP immobilized in between a Pt- and a AuPd-electrode comprising a gap of about 13 nm. | ||
These functional clDA-COOH–AuNP devices exhibited more than 10 reproducible cyclic I/U-measurements between −1 V and +1 V. The obtained I/U-curves revealed a nonlinear current/voltage dependence corresponding to an electron tunneling mechanism which can be described by the Simmons model as introduced previously.15,41,42
In Fig. 8a a characteristic tunneling curve and the corresponding Simmons fit are shown. The current signal gets remarkable noisy for voltages above 0.5 V and the noise rises with higher voltages. Moreover, for voltages above 1 V instabilities are observed pointing to a beginning destruction of the device. This unusual characteristic, compared with investigations performed previously on comparable AuNP-devices based on AuNP capped with several short organic molecules15,16,43 is found for all recorded data on clDA-COOH–AuNP devices.
This finding indicates that the noise is correlated to the specific kind of clDA-COOH–AuNP under investigation here. The experimental conductivity values for all clDA-COOH–AuNP-devices obtained were plotted in a histogram (Fig. 8b). Therefrom, a rather large variation of the conductance values of about 5 orders of magnitude, from 1 pS to 70 nS with a median of 55 pS, can be deduced.
In addition, most of the experimental conductivities are higher than the theoretical value of 11.8 pS for electron tunneling through the clDA-COOH molecular shell that results from applying the single channel Landauer formula like introduced in literature before.16,25,43–45 These higher conductance values indicate a shorter electrical path through the molecular shell of clDA-COOH–AuNP than expected based on the molecular length of DA-COOH.
Particularly, the higher experimental conductance can be interpreted as a mean reduction of the thickness of the molecular shell from 1.53 nm to 1.33 nm which corresponds to the length of 1 to 2 methylene groups.
These results together with the increasing noise of the I/U-curves for voltages above 0.5 V can be interpreted as variations in the cross-linkage within the ligand shell (polymers of different lengths, see Section 2) which leads to slightly different thicknesses of the DA-COOH-ligand shell around the AuNP. Due to the exponential dependence of the conductance through the DA-COOH-device from the tunnelling distance, i.e. the effective length of the clDA-COOH-molecule, an enhancement of the variations results. Here, further optimization is necessary to obtain more uniform polymerization products.
Conclusions
A specially designed ligand molecule consisting of a thiol group, a diacetylene unit and a terminal carboxylic group was prepared, suitable for applications in nanoelectronic devices based on AuNP. This ligand molecule, DA-COOH, was applied in a homogeneous ligand exchange reaction, starting from 11 nm citrate stabilized AuNP, in order to prepare respectively stabilized DA-COOH–AuNP. After ligand exchange the particles were exposed to UV light in order to induce polymerisation between neighbouring ligands containing diacetylene units assembled on the AuNP surface, thus forming a polymeric ligand shell (clDA-COOH–AuNP). Stability investigations towards NaCl, DTT displacement reactions, and temperature were performed and reveal an extraordinary high stability. Additionally, individual particles could be successfully immobilized in between nanoelectrodes comprising gaps of 10 to 14 nm. Electrical transport measurements revealed unexpected variations in conductance values. These could be attributed to small length differences of the tunnelling path through the polymerized ligand shell of clDA-COOH–AuNP.Summarizing a novel extraordinary highly stable ligand-AuNP composite was developed, exhibiting tailored functionalities, which reveal a high potential for nanoelectronic devices based on AuNP. Beyond this specific application scenario, we are sure that the extraordinary stable AuNP introduced here will be of interest for other fields of applications, which require high chemical and thermal particle stability in aqueous solvents.
Acknowledgements
This work was financially supported by the Excellence Initiative of the German federal and state government (Dean's Seed Fund Project “PolyTemp”), by the Jülich Aachen Research Alliance (JARA), and by the German Research Foundation (DFG; contracts Si 609/16-1 and Ka 1819/7-1). We further thank Svenja Bourone (M. Sc.) for performing IRRAS measurements, as well as Sophia Knecht (B. Sc.) and Christina Zitlau for their support in synthetic work as well as Dr Corinna Kaulen and Dr Michael Noyong for helpful discussions on the manuscript.Notes and references
- G. Schmid, Nanoparticles – From Theory to Application, Wiley-VCH, Weinheim, Germany, 2011 Search PubMed.
- X. Liu, M. Yu, H. Kim, M. Mameli and F. Stellacci, Nat. Commun., 2012, 3, 1182, DOI:10.1038/ncomms2155.
- B. Fresch and F. Remacle, J. Phys. Chem. C, 2014, 118(18), 9790–9800 CAS.
- X. Gao, Y. Lu, M. Liu, S. He and W. Chen, J. Mater. Chem. C, 2015, 3, 4050–4056 RSC.
- W. Wei, Y. Lu, W. Chen and S. Chen, J. Am. Chem. Soc., 2011, 133, 2060–2063 CrossRef CAS PubMed.
- J. Broda, G. Schmid and U. Simon, Struct. Bonding, 2014, 161, 189–241 CrossRef.
- M. Homberger and U. Simon, Philos. Trans. R. Soc., A, 2010, 368, 1405–1453 CrossRef CAS PubMed.
- M.-C. Daniel and D. Astruc, Chem. Rev., 2004, 104(1), 293–346 CrossRef CAS PubMed.
- P. M. Tiwari, K. Vig, V. A. Dennis and S. R. Singh, Nanomaterials, 2011, 1, 31–63 CrossRef CAS.
- K. Saha, S. S. Agasti, C. Kim, X. Li and V. M. Rotello, Chem. Rev., 2012, 112(5), 2739–2779 CrossRef CAS PubMed.
- E. C. Dreaden, M. A. Mackey, X. Huang, B. Kang and M. A. El-Sayed, Chem. Soc. Rev., 2011, 40, 3391–3404 RSC.
- E. C. Dreaden, A. M. Alkilany, X. Huang, C. J. Murphy and M. A. El-Sayed, Chem. Soc. Rev., 2012, 41, 2740–2779 RSC.
- A. Buchkremer, M. F. Beckmann, M. Linn, J. Ruff, R. R. Rosencrantz, G. von Plessen, G. Schmitz and U. Simon, Mater. Res. Express, 2014, 1(4), 045015, DOI:10.1088/2053-1591/1/4/045015.
- C. Kaulen, M. Homberger, S. Bourone, N. Babajani, S. Karthäuser, A. Besmehn and U. Simon, Langmuir, 2014, 30(2), 574–583 CrossRef CAS PubMed.
- N. Babajani, P. Kowalzik, R. Waser, M. Homberger, C. Kaulen, U. Simon and S. Karthäuser, J. Phys. Chem. C, 2013, 117(42), 22002–22009 CAS.
- N. Babajani, C. Kaulen, M. Homberger, M. Mennicken, R. Waser, U. Simon and S. Karthäuser, J. Phys. Chem. C, 2014, 118(46), 27142–27149 CAS.
- M. Brust, D. Bethell, D. J. Schiffrin and C. J. Kiely, Adv. Mater., 1995, 7(9), 795–797 CrossRef CAS.
- C. Guarise, L. Pasquato and P. Scrimin, Langmuir, 2005, 21(12), 5537–5561 CrossRef CAS PubMed.
- T. Peterle, A. Leifert, J. Timper, A. Sologubenko, U. Simon and M. Mayor, Chem. Commun., 2008, 3438–3440 RSC.
- L. Srisombat, A. C. Jamison and T. R. Lee, Colloids Surf., A, 2011, 390, 1–19 CrossRef CAS.
- D. Bartczak and A. G. Kanaras, Langmuir, 2010, 26(10), 7072–7077 CrossRef CAS PubMed.
- Y. Song, L. M. Klivansky, Y. Liu and S. Chen, Langmuir, 2011, 27, 14581–14588 CrossRef CAS PubMed.
- H. Menzel, M. D. Mowery, M. Cai and C. E. Evans, Macromol. Symp., 1999, 142, 23–31 CrossRef CAS.
- J. C. Love, L. A. Estroff, J. K. Kriebel, R. G. Nuzzo and G. M. Whitesides, Chem. Rev., 2005, 105(4), 1103–1169 CrossRef CAS PubMed.
- S. Karthäuser, J. Phys.: Condens. Matter, 2011, 23, 013001, DOI:10.1088/0953-8984/23/1/013001.
- S. K. Ghosh and T. Pal, Chem. Rev., 2007, 107(11), 4797–4862 CrossRef CAS PubMed.
- J. Turkevich, P. C. Stevenson and J. Hillier, Discuss. Faraday Soc., 1951, 11, 55–75 RSC.
- M. Manheller, S. Trellenkamp, R. Waser and S. Karthäuser, Nanotechnology, 2012, 23, 125302–125308 CrossRef PubMed.
- Y. Cui, M. T. Björk, A. Liddle, C. Sönnichsen, B. Boussert and A. P. Alivisatos, Nano Lett., 2004, 4(6), 1093–1098 CrossRef CAS.
- Y. Xing and G. A. O'Doherty, Org. Lett., 2009, 11(5), 1107–1110 CrossRef CAS PubMed.
- A. Sevin, W. Chodkiewicz and P. Cadiot, Bull. Soc. Chim. Fr., 1974, 913–917 CAS.
- P. C. B. Page, R. D. Wilkes and D. Reynolds in Comprehensive Organic Functional Group Transformations: Synthesis: carbon with one heteroatom attached by a single bond, ed. A. R. Katritzky, O. Meth-Cohn and C. W. Rees, Elsevier, 1995, pp. 213–214 Search PubMed.
- C. D. Bain, H. A. Biebuyck and G. M. Whitesides, Langmuir, 1989, 5, 723–727 CrossRef CAS.
- G. Sokrates, Infrared and Raman Characterisitc Group Frequencies, John Wiley & Sons Ltd., New York, 2001 Search PubMed.
- G. Li, T. Wang, S. Bhosale, Y. Zhang and J.-H. Fuhrhop, Colloid Polym. Sci., 2003, 281, 1009–1103 Search PubMed.
- J.-Y. Shim and V. K. Gupta, J. Colloid Interface Sci., 2007, 316(2), 977–983 CrossRef CAS PubMed.
- A. Prock, M. L. Shand and R. R. Chance, Macromolecules, 1982, 15, 238–241 CrossRef CAS.
- G. N. Patel, R. R. Chance, E. A. Turi and Y. P. Khanna, J. Am. Chem. Soc., 1978, 100(21), 6644–6649 CrossRef CAS.
- D. Zopes, B. Stein, S. Mathur and C. Graf, Langmuir, 2013, 29, 11217–11226 CrossRef CAS PubMed.
- B. Stein, D. Zopes, M. Schmudde, R. Schneider, A. Mohsen, C. Goroncy, S. Mathur and C. Graf, Faraday Discuss., 2015, 181, 85–102 RSC.
- J. G. Simmons, J. Appl. Phys., 1963, 34(6), 1793–1803 CrossRef.
- J. G. Simmons, J. Appl. Phys., 1963, 34(9), 2581–2590 CrossRef.
- M. Manheller, S. Karthäuser, R. Waser, K. Blech and U. Simon, J. Phys. Chem. C, 2012, 116, 20657–20665 CAS.
- Y. Imry and R. Landauer, Rev. Mod. Phys., 1999, 71, 306–312 CrossRef.
- A. Salomon, D. Cahen, S. Lindsay, J. Tomfohr, V. B. Engelkes and C. D. Frisbie, Adv. Mater., 2003, 15, 1881–1890 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: UV-vis spectra of citrate-AuNP and DA-COOH–AuNP, IRRAS spectra, ζ-potential measurements, Raman spectrum of monomer, UV-vis spectrum of detached particles. See DOI: 10.1039/c5ra17545c |
| This journal is © The Royal Society of Chemistry 2015 |