pH-induced recovery and redispersion of shape-controlled gold nanorods for nanocatalysis†
Abstract
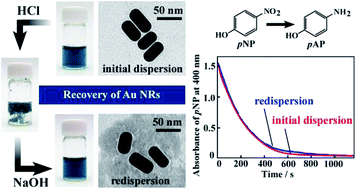
The amphiphilic, pH-responsive amine derivative, 3-[(2-carboxy-ethyl)-hexadecyl-amino]-propionic acid (C16CA) was used for the functionalization of gold nanorods (Au NRs) prepared with cetyltrimethylammonium bromide (CTAB). The Au NRs could also be stabilized with C16CA owing to the selective adsorption of the amino moiety, and the Au NRs were well dispersed following one-step ligand exchange using C16CA. Based on a change in the nature of C16CA, self-assemblies of spherical micelles (pH > 5) or lamellar precipitates (pH 2–5) are formed in dispersion. Au NRs were incorporated into the precipitates at pH 2–5, but could be redispersed by redissolution of C16CA at pH > 5. The pH-induced recovery–redispersion of Au NRs was successfully accomplished without affecting the morphology of the Au NRs, the amount of Au in the dispersion, or the catalytic activity of the Au NRs for the reduction of p-nitrophenol.

 Please wait while we load your content...
Please wait while we load your content...