Influence of removal of organic matter and iron and manganese oxides on cadmium adsorption by red paddy soil aggregates
Zhongwu Li*ab,
Bin Huangab,
Jinquan Huangc,
Guiqiu Chenab,
Chang Zhangab,
Xiaodong Nieab,
Ninglin Luoab,
Hongbo Yaoab,
Wenming Maabd and
Guangming Zengab
aCollege of Environmental Science and Technology, Hunan University, Changsha, 410082, China. E-mail: lizw@hnu.edu.cn; Fax: +86 731 88640078; Tel: +86 731 88640078
bKey Laboratory of Environment Biology and Pollution Control (Hunan University), Ministry of Education, Changsha 410082, China
cDepartment of Soil and Water Conservation, Yangtze River Scientific Research Institute, Wuhan 430010, China
dCollege of Tourism Historical Culture, Southwest University for Nationalities, Chengdu 610041, China
First published on 16th October 2015
Abstract
The ability of soil aggregates to adsorb heavy metals is controlled by their different component contents. However, the influence of specific components on the adsorption behavior is uncertain. In this study, organic matter and iron and manganese oxides in aggregates of different particle sizes from red paddy soil were selectively removed. The influence of these components on cadmium adsorption was also examined. Results showed that Langmuir equation could accurately describe the adsorption characteristics of Cd in bulk soil and aggregates. The <0.002 mm fraction had the largest adsorption capacity for Cd because of its higher content of organic matter and Fe and Mn oxides. Removal of organic matter, amorphous Fe oxides or Mn oxides could cause obvious decrease in Cd adsorption, whereas an opposite result was observed for free Fe oxides removal. Removing unit mass of Fe or Mn oxides showed stronger effect on the adsorption capacity compared with that of organic matter. Desorption characteristics of Cd in various aggregates appeared to be unaffected by organic matter but more determined by free and amorphous Fe oxides.
1. Introduction
Cadmium is a ubiquitous element with high toxicity to organisms. This element is more mobile and bioavailable than other metals. In the past few decades, an increasing number of anthropogenic Cd have entered soil environment. Soil contamination with Cd usually originates from various practices, such as mining, sewage sludge application and chemical fertilization.1,2 Recently, soil Cd pollution has become one of the most serious issues confronting rice production and soil management in some Asian countries.3,4 According to a previous study,5 the area of cultivated land contaminated by Cd reaches nearly 2.8 × 105 hm2 in China. Excessive levels of Cd retained in paddy soil significantly increase the potential risk of uptake by rice, which, in turn, inevitably posts threat to human health.Adsorption/desorption is the most important process that controls the transformation and migration of Cd in soil.6 The major factors influencing adsorption/desorption process include soil physicochemical properties, temperature, and moisture condition.6–8 Aggregates are the basic structure units of soil, and the ability of aggregates to adsorb/desorb heavy metals is different.9 Soil component (e.g., organic matter or iron and manganese oxides) has an important function in deciding the adsorption capacity of soil aggregates for metal ions. Several previous studies10–12 have indicated that the content of organic matter and Fe oxides could strongly influence the accumulation of metals among aggregates of different size fractions in contaminated soils. Huang et al.13 also showed that the maximum adsorption capacities (obtained from the batch experiments) for Cu and Zn of aggregates of different particle sizes were positively correlated with their organic matter and free Fe2O3 contents; and soil organic matter (SOM) was inferred to have a larger sorption capacity toward metals compared with other soil fractions. In summary, all these aforementioned studies were based on analyzing correlations between the mineralogical properties of soil particles and their metal content or capacity to adsorb metals, whereas the effect of specific components on the affinity of metals in soil has been unexplored.
The influence of soil components on adsorption of metals can be examined in several ways, such as studying adsorption capacity of synthetic or semisynthetic pure substances14,15 and selectively removing a specific component from the soil and evaluating its effect by comparing the variation of metal adsorption capacity.16–18 The former method may inaccurately reflect the sorption behavior of different soil components that are naturally formed. The latter method has already been used to evaluate the influence of organic matter and Fe and Mn oxides on Cd adsorption in several previous studies.19–22 Elliott et al.19 found that Fe oxides extraction reduced Cd adsorption on a wide range of soils, whereas some other researchers found that this treatment showed insignificant effect on certain acid soils.20 Removing organic matter usually resulted in the reduction of Cd retention, and the decreasing degree also deepened on the physicochemical characteristics of the used soil.21,22 All these reports indicated that organic matter and oxides are important in Cd adsorption, but their relative importance may be different. Selective removing technology is an effective method in evaluating the influence of various soil fractions on metal adsorption and can be helpful in further understanding the adsorption mechanism of heavy metals in soil.17,18 However, this method has not been applied with the subject of soil aggregates.
In the present study, organic matter and Fe and Mn oxides in aggregates of different particle sizes from red paddy soil were selectively removed by using corresponding chemical reagents. Their influence on Cd adsorption was then evaluated by comparing with the adsorption results of the untreated aggregates through batch experiments. The information provided in this study will be helpful in explaining the different adsorption behavior of various soil aggregates.
2. Materials and methods
2.1. Soil samples and aggregates fractionation
Surface soil samples (0–20 cm) were collected from a paddy field. This area is 15 km away from the center of Changsha City in Hunan Province (28°09′35′′N, 112°52′37.5′′E). The study site has mean monthly air temperatures ranging from 5.26 °C in January to 29.68 °C in August and has mean annual precipitation of 1361 mm. The soil is mainly derived from the Quaternary red clay and recent alluvial deposits. No heavy metal pollution source exists nearby, and the background concentration of Cd in the soil is very low (0.07 mg kg−1).23 Five sampling sites were selected in the paddy field of this area, and approximately 5 kg soil in each site was collected. The sampled soil was transported back to the laboratory, air dried at room temperature, and mixed thoroughly.The soil samples were sieved to pass through a 2 mm nylon sieve and were partitioned into four aggregate-size fractions (2–0.25 mm, 0.25–0.05 mm, 0.05–0.002 mm, and <0.002 mm), as described in a previous study.11 Briefly, 30 g of soil sample was immersed in 150 ml deionized water and dispersed with ultrasonic (100 J ml−1).24 The 2–0.25 mm fraction was obtained by wet sieving; the 0.25–0.05 and 0.05–0.002 mm fractions were obtained by sedimentation and siphoning; the <0.002 mm fraction was finally obtained after centrifugation. The soil particles were collected respectively and freeze-dried. A few basic properties of soil are presented in Table 1.
| Particle size (mm) | SOM (g kg−1) | CEC (cmol(+) kg−1) | Fed (g kg−1) | Feo (g kg−1) | Fe-oxide (g kg−1) | Mn-oxides (g kg−1) | BET surface area (m2 g−1) | Cd (mg kg−1) | % |
|---|---|---|---|---|---|---|---|---|---|
| a SOM: soil organic matter; CEC: cation exchange capacity. %: mass percentage; Fed: free Fe-oxide; Feo: amorphous Fe-oxides. | |||||||||
| Bulk soil | 56.37 | 41.23 | 11.97 | 1.34 | 50.71 | 0.67 | 16.24 | 0.20 | — |
| 2–0.25 | 54.62 | 43.16 | 12.03 | 1.18 | 54.08 | 0.52 | 13.96 | 0.22 | 10.07 |
| 0.25–0.05 | 49.84 | 39.93 | 10.86 | 1.40 | 47.50 | 0.63 | 15.77 | 0.26 | 23.45 |
| 0.05–0.002 | 50.00 | 40.35 | 12.22 | 1.45 | 37.01 | 0.71 | 15.09 | 0.19 | 49.68 |
| <0.002 | 65.30 | 51.05 | 27.95 | 1.01 | 75.29 | 0.94 | 30.10 | 0.27 | 16.80 |
2.2. Removal of organic matter and Fe and Mn-oxides
The prepared soil particles (bulk soil and aggregates) were subjected by using corresponding chemical reagents to remove various soil components. Organic matter was removed by sodium hypochlorite (NaOCl) extraction method.25 NaOCl has several advantages over hydrogen peroxide (H2O2) for SOM removal; NaOCl was found to destroy more organic matter than H2O2 with minimal loss of Fe and Mn and little alteration of sesquioxides.26,27 Free Fe-oxides were removed by using dithionite–citrate–bicarbonate (DCB) method.28 Amorphous Fe-oxides were removed by (NH4)2C2O4–H2C2O4 method.29 Mn-oxides were removed by NH2OH·HCl method.30 A brief description of the above-mentioned methods is presented in Table 2. All the threated residuals were repeatedly washed and then freeze-dried and grounded to pass a 0.25 mm nylon screen.| Soil component | Chemical reagents | Soil/solution | Reaction conditions |
|---|---|---|---|
| Organic matter | 6% NaOCl (pH = 8) | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
Room temperature, 8 h shaking, repeated 4 times |
| Free Fe-oxides | 0.3 M Na3C6HO7·2H2O + 1 M NaHCO3 + Na2S2O4 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
80 °C boiling in H2O bath, 15 min, repeated thrice |
| Amorphous Fe-oxides | 0.2 M (NH4)2C2O4 + 0.2 M H2C2O4 (pH = 3) | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
Room temperature, 4 h shaking in dark, repeated thrice |
| Mn-oxides | 0.1 M NH2·OH·HCl (pH = 2) | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
Room temperature, 30 min shaking, repeated thrice |
2.3. Adsorption and desorption experiments
Original or treated soil particles were placed into a 50 ml polypropylene centrifuge tube, and 25 ml 0.01 M NaNO3 solution containing a series concentration levels (1, 5, 10, 20, 50, 100, 200 mg l−1) of Cd (as Cd(NO3)2) was added to each tube. The suspensions were shaken at 160 rpm for 4.0 h and kept still for 20.0 h at constant temperature of 25 ± 0.2 °C. Then, the suspensions were centrifuged and supernatant were filtrated to measure of Cd concentration. Cd adsorption was calculated as the difference between that added and that remaining in the supernatant. Langmuir and Freundlich equations31 were applied to fit the data from the batch experiment, eqn (1) and (2):
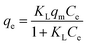 | (1) |
| qe = KFCe1/n | (2) |
Soil residual in centrifuge tubes containing initial Cd(NO3)2 of 200 mg l−1 was used for desorption experiment. After the supernatant was discarded, 25 ml 0.01 M NaNO3 was added to the tubes to replace the adsorbed Cd2+. The followed procedures (shaken, equilibration, centrifugation and separation) were the same as those in the adsorption experiments. The total desorption capacity of the adsorbed Cd was obtained by repeating this sequence of operation for three times.
2.4. Analytical methods
Soil pH was measured at a soil/solution ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 using a HI 3221 pH meter (Hanna Instruments Inc., USA). SOM was determined by K2Cr2O7 oxidation method.32 Cation exchange capacity (CEC) was determined by BaCl2 displaced method.33 Free Fe-oxides (Fed) were determined by Na2S2O4–Na3C6O7 extraction method.28 Amorphous Fe-oxides (Feo) were determined by (NH4)2C2O4–H2C2O4 method.29 Fe, Mn, and Cd (after HNO3–HF–HClO4 digestion) content in soil were determined using atomic absorption spectrophotometer (AA700, PerkinElmer, USA). BET surface area was measured by a NOVA 2200e surface area and pore size analyzer (Quantachrome Inc., USA). All the glassware used in experiments were thoroughly soaked overnight in HNO3 solution (3%, v/v) and were rinsed with deionized water. All the experimental data were the average of triplicate determinations. Statistical analyses were conducted using SPSS 10.0 software. Comparisons of the means were conducted by Duncan's multiple range test at the p ≤ 0.05 level.
2.5 using a HI 3221 pH meter (Hanna Instruments Inc., USA). SOM was determined by K2Cr2O7 oxidation method.32 Cation exchange capacity (CEC) was determined by BaCl2 displaced method.33 Free Fe-oxides (Fed) were determined by Na2S2O4–Na3C6O7 extraction method.28 Amorphous Fe-oxides (Feo) were determined by (NH4)2C2O4–H2C2O4 method.29 Fe, Mn, and Cd (after HNO3–HF–HClO4 digestion) content in soil were determined using atomic absorption spectrophotometer (AA700, PerkinElmer, USA). BET surface area was measured by a NOVA 2200e surface area and pore size analyzer (Quantachrome Inc., USA). All the glassware used in experiments were thoroughly soaked overnight in HNO3 solution (3%, v/v) and were rinsed with deionized water. All the experimental data were the average of triplicate determinations. Statistical analyses were conducted using SPSS 10.0 software. Comparisons of the means were conducted by Duncan's multiple range test at the p ≤ 0.05 level.
3. Results and discussion
3.1. Soil properties
Red paddy soil is a typical agricultural soil that is widely distributed in subtropical area with slight acidity (pH = 5.86). This type of soil is considered to have a strong retention capacity for metal ions because of its relatively high organic matter content (56.37 g kg−1), which is far above the mean level34 (22.90 g kg−1) in northeastern China. The 0.25–0.05 mm (23.45%) and 0.05–0.002 mm (49.68%) aggregates were the dominant fractions of the sampled paddy soil. Among all the fractions, the clay size fraction (<0.002 mm) showed the highest values in content of organic matter, Fe-oxides, free Fe-oxides, Mn-oxides, as well as CEC and BET surface are, but the lowest in amorphous Fe-oxides content. Fine soil particles were also found to have higher organic content in many other types of soil.35,36 Smaller aggregates process stronger ability to protect organic matter and thus retain more carbon matter.37 The Feo/Fed ratio (“activity ratio”), which is used as a relative measure of the degree of aging or crystallinity of free Fe-oxides, indicates that the activity of free Fe oxides in the <0.002 mm fraction was the lowest. Removal of components could reduce the surface area of soil particles to different degrees. For example, the surface areas of bulk soil after the treatment were 8.1 (organic matter removal), 6.65 (free Fe-oxides removal), 10.08 (amorphous Fe-oxides removal), and 11.01 m2 g−1 (Mn-oxides removal). By contrast, free Fe-oxides removal caused the most decrease in surface area.3.2. Adsorption isotherm
The experimental data and adsorption isotherms for Cd adsorption onto original bulk soil and aggregates are shown in Fig. 1. The amount of Cd adsorbed in soil showed a gradual ascending trend in general. Cd was nearly adsorbed completely under the condition of low initial concentration (0–50 mg l−1) addition. Moreover, the adsorption amount in soil showed no apparent increase when the initial concentration exceeded 50 mg l−1, which meant the adsorption had gradually reached the balance value. The variation trend of the adsorption curves was similar to that obtained in many other batch experiments that were used to examine the adsorption characteristic of heavy metals in various soils.38–40 The fitting accuracy of Langmuir (0.9168 ≤ R2 ≤ 0.9978) model is better than that of Freundlich model (0.8417 ≤ R2 ≤ 0.9815) (Table 3). As listed in Table 3, the maximum adsorption capacity (qm) of different particle size fractions decreased in the following order: (<0.002 mm) > bulk soil > 2–0.25 mm > 0.05–0.002 mm > 0.25–0.05 mm. The maximum adsorption capacities (qm) of the <0.002 mm fraction was 1.21–1.44 times than that of other fractions. Compared with the date obtained in our previous study,13 this range was close to the values of Cu (1.14–1.49) but less than that of Zn (1.45–2.22) under the same experimental conditions. In addition, the adsorption capacity of red paddy soil for the three metals followed the order: Cu > Zn ≈ Cd. Cu is generally adsorbed and retained to a higher extent than Zn and Cd in soils.41,42 The different adsorption behavior of the three metals in soil mainly depends on their ionic potential, hydrolysis, and softness.43 | ||
| Fig. 1 Adsorption isotherms for Cd onto bulk soil and aggregates; the experimental data are reported as points and Langmuir model curves. | ||
| Particle size (mm) | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| KL (l mg−1) | qm (mg kg−1) | R2 | KF (l kg−1) | n−1 | R2 | |
| Untreated soil particles | ||||||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Bulk soil Bulk soil |
0.2095 | 2299.66 | 0.9905 | 611.24 | 0.2865 | 0.9364 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2–0.25 mm 2–0.25 mm |
0.2090 | 2149.57 | 0.9662 | 592.10 | 0.2738 | 0.8945 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.25–0.05 mm 0.25–0.05 mm |
0.4141 | 1920.18 | 0.9857 | 688.34 | 0.2233 | 0.8704 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.05–0.002 mm 0.05–0.002 mm |
0.2105 | 2080.14 | 0.9875 | 599.05 | 0.2664 | 0.9115 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) <0.002 mm <0.002 mm |
0.1970 | 2774.35 | 0.9681 | 746.22 | 0.2744 | 0.8417 |
Spearman correlation analysis was performed to evaluate the relationship between qm and the soil component content in bulk soil and aggregates (Table 4). The results showed that the maximum adsorption capacity (qm) was significantly related to the organic matter content (p < 0.01), but had no correlation with the contents of free Fe-oxides, amorphous Fe-oxides, and total Fe- or total Mn-oxides. Organic matter appeared to be a more important factor in determining the adsorption capacity than the other soil components. Similar result was also obtained for Cu and Zn.13 Organic matter has been verified to influence the retention and release ability of metals in soil,43,44 and is also considered to be the major contributor to the ability of soils for retaining heavy metals in an exchangeable form because of its high CEC.45 However, whether the other components had no effect on the adsorption capacity could not be determined based on the result of correlation analysis alone. In fact, clay and hydrous oxides of Fe and Mn can also adsorb foreign metal ions through different binding mechanisms. Ion exchange and surface complexation and precipitation are the main process controlling the adsorption behavior of metals in SOM. As for clay and metal oxyhydroxides, specific adsorption and cation exchange are the main mechanisms.6
3.3. Effect of removal of different components on Cd adsorption
The results of Cd adsorption by the treated bulk soil and aggregates are shown in Fig. 2. Removal of different components (organic matter (Fig. 2(a)), free Fe-oxides (Fig. 2(b)), amorphous Fe-oxides (Fig. 2(c)) and Mn-oxides (Fig. 2(d))) insignificantly influenced the general adsorption characteristics of Cd, and Langmuir model was also more suitable to fit the experimental data (0.9168 ≤ R2 ≤ 0.9978, Table 5). All the treatments caused decrease in Cd adsorption except for the free Fe-oxides removal. Through the calculated qm, the variation in adsorption ability before and after specific component removal treatment can be quantitatively compared. The decrease (p < 0.05) in qm was 456.63–858.66, 200.17–477.75 and 178.69–438.66 mg kg−1 under the condition of organic matter, amorphous Fe-oxides and Mn-oxides removal, respectively. Organic matter removal could reduce the adsorption of Cd to the highest extent. Comparatively, the decrease amounts caused by the removal of amorphous Fe-oxides or Mn-oxides were far less and their values were very close (no significant difference was observed between them). Free Fe-oxides removal caused significant increase (1441.38–2229.39 mg kg−1) in Cd adsorption (p < 0.05). Linear regression analysis reveals that the decrease/increase in qm caused by removal treatment was positive with the corresponding component content in soil particles (b > 0) (Fig. 3). This result reveals that organic matter, and Fe- and Mn-oxides are all influential soil components involved in Cd adsorption in red paddy soil. The decease/increase amount of Cd concentrations in soil phase caused by unit removed component content (judged from the slops in regression equations in Fig. 3) followed the order: amorphous Fe-oxides > Mn-oxides > free Fe-oxides > organic matter.| Particle size (mm) | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| KL (l mg−1) | qm (mg kg−1) | R2 | KF (l kg−1) | n−1 | R2 | |
| Organic matter removal | ||||||
| Bulk soil | 0.2294 | 1608.51 | 0.9718 | 419.84 | 0.2907 | 0.9420 |
| 2–0.25 mm | 0.2390 | 1435.69 | 0.9943 | 398.66 | 0.2722 | 0.9144 |
| 0.25–0.05 mm | 0.2114 | 1463.56 | 0.9835 | 399.75 | 0.2762 | 0.9229 |
| 0.05–0.002 mm | 0.2395 | 1541.82 | 0.9718 | 417.98 | 0.2813 | 0.9213 |
| <0.002 mm | 0.1239 | 1915.69 | 0.9843 | 421.87 | 0.3086 | 0.8672 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Free Fe-oxides removal | ||||||
| Bulk soil | 0.0407 | 3831.81 | 0.9168 | 484.26 | 0.3933 | 0.8495 |
| 2–0.25 mm | 0.1518 | 3684.03 | 0.9704 | 701.85 | 0.3416 | 0.8448 |
| 0.25–0.05 mm | 0.1715 | 3361.56 | 0.9806 | 704.54 | 0.3224 | 0.8628 |
| 0.05–0.002 mm | 0.0435 | 3583.15 | 0.9419 | 416.56 | 0.4099 | 0.8872 |
| <0.002 mm | 0.0815 | 5003.74 | 0.9878 | 694.07 | 0.4093 | 0.9473 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Amorphous Fe-oxides removal | ||||||
| Bulk soil | 0.1335 | 2024.00 | 0.9978 | 424.50 | 0.3163 | 0.9221 |
| 2–0.25 mm | 0.1330 | 1949.40 | 0.9811 | 425.57 | 0.3174 | 0.9257 |
| 0.25–0.05 mm | 0.1188 | 1533.34 | 0.9903 | 337.10 | 0.3168 | 0.9462 |
| 0.05–0.002 mm | 0.1061 | 1602.39 | 0.9915 | 300.25 | 0.3177 | 0.9013 |
| <0.002 mm | 0.0973 | 2520.46 | 0.9639 | 471.24 | 0.3407 | 0.8712 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Mn-oxides removal | ||||||
| Bulk soil | 0.0481 | 2018.29 | 0.9952 | 253.64 | 0.4033 | 0.9658 |
| 2–0.25 mm | 0.0363 | 1970.88 | 0.9871 | 196.88 | 0.4416 | 0.9815 |
| 0.25–0.05 mm | 0.0649 | 1710.36 | 0.9929 | 270.43 | 0.3654 | 0.9399 |
| 0.05–0.002 mm | 0.0476 | 1755.49 | 0.9977 | 219.04 | 0.4033 | 0.9628 |
| <0.002 mm | 0.0851 | 2335.69 | 0.9940 | 421.10 | 0.3477 | 0.9512 |
 | ||
| Fig. 3 Correlation between the decrease/increase amount of qm and different soil component contents. | ||
Both lab and field studies have shown that organic matter is one of the most important soil properties affecting Cd adsorption in soil.21,46,47 Usually, the higher the organic content in soil, the more metal ions can be adsorbed. Zhao et al.22 also found that organic matter removal produced a significant decrease in Cd adsorption of soil, although they used different types of soil and H2O2 as the organic matter removal reagent. He et al.48 indicated that the addition of organic matter increased soil CEC and decreased soil pH, which may result in changes in the solid phase of Cd. The variation trend of Cd adsorption capacity depends on the content of organic matter in soil. Reid and McDuffie20 found that treatment of illite with 30% H2O2 to remove organic matter produced no significant change in adsorption using 100 ppb Cd. Organic matter contents of the soil would be relatively low, and their adsorption capacity for Cd was low. Notably, red paddy soil is in relatively high organic matter content, which would obviously influence the adsorption capacity. For aggregates of different particle-sizes, organic matter removal produced the most variation in the <0.002 mm fraction. This result further demonstrates the importance of organic matter because its content was the highest in this fraction.
Free Fe-oxides in soils mainly present specific adsorption for metal ions.18 However, the obtained result appeared to contradict the widely held view that Fe-oxides have an important function in fixing metal ions in soil. Free Fe-oxides removal by DCB procedure may cause different effects on the adsorption capacity, which also depends on the physicochemical properties of the used soil. Silveira et al.49 observed that Cu adsorption significantly decreased in some tropical soils with high Fe oxide content. Elliot and Liberati50 found similar result for Cd when investigating acidic soils from the Altantic Coastal Plain. Meanwhile, in the considerable research conducted by Elliott et al.,19 Shuman,51 and Agbenin and Latifatu,52 Fe-oxides removal by DCB procedure increased the adsorption capacity of soils for Cd and other metals. As concluded in the above-mentioned works, the increase phenomenon could be attributed to two reasons. First, DCB treatment eliminated Fe oxide/hydroxide, which, in turn, exposed additional sites for heavy metal adsorption; second, the adsorption sites on the free Fe-oxides coating were not as numerous as those released when the coating were removed. On the one hand, Fe oxides in soils are usually positive charged; removal of this component could low the zero points of charges (ZPC) of soil and exposed negative surface charged that was previously combined by Fe-oxides.53 On the other hand, the DCB extraction procedure could release additional exchange sites by removing organically complexed Fe/Al and by stripping interlayer Fe/Al hydroxypolymers from silicate clay minerals.54
Amorphous Fe-oxides are more active fractions in free Fe-oxides, and are characterized as huge surface area and as specific adsorption for cation and anion.55 Similar to organic matter, amorphous Fe-oxides removal decreased the adsorption ability, which was just only contrary to the result obtained for free Fe oxides removal. The possible reason was the affinity of amorphous Fe-oxides for Cd was much higher; extraction of this part may expose some new adsorption sites that were previously combined by amorphous Fe-oxides. However, the amount of the new adsorbed Cd was less than the adsorption capacity of the removed amorphous Fe-oxides. Consequently, a significant decrease was observed after amorphous Fe-oxides were removed. For Mn-oxides, the adsorption ability of different soil particles for Cd also decreased, which may be due to the reduction of available adsorption sites owing to the removal treatment. Mn-oxide minerals exist widely in soils and sediment in various forms of scattered weak crystallized aggregates, nodules, and coatings etc.56 These minerals are commonly believed to have lower ZPC, high negative charge amount, and large surface area; these properties allow these minerals to have high adsorption capacity towards metal ions.57
Although organic matter removal could reduce the adsorption capacity to the highest extent, the removal of Fe- and Mn-oxides exhibited the best ability in influencing the adsorption of Cd in soil. By contrast, the <0.002 mm soil aggregate contained more organic matter, free Fe- and Mn-oxides. Accordingly, this fraction of aggregate can retain more Cd in soil. More attention should be paid to this fraction of aggregate because it can easily migrate in the environment. Furthermore, the combined metals in some specific components can be released as a result of the ambient condition variation. For example, Fu et al.58 found that the adsorption capacities of Mn-oxides for Cd decreased by nearly 80% when pH changed from 8.0 to 5.5.
3.4. Effect on desorption after removal of different components
The calculated maximum adsorption capacity (qm) was approximately equal to the adsorption amount under the initial concentration of 200 mg l−1. Thus, the desorption experiment of different treated soil particles was conducted only with this level of Cd concentration, and the corresponding result was compared with the values in adsorption experiment. Desorption rate can reflect the nonspecific adsorption degree of metals in soil. As shown in Fig. 4, the desorption rates of the 0.25–0.05 mm fraction was the lowest and that of the 0.05–0.002 mm fraction was the highest. Removal of different components could increase or decrease the desorption rates, which depended on the particle size of the aggregates. For example, free Fe-oxides removal increased the desorption rate in the 2–0.25 mm fraction but decreased the rate in the 0.05–0.002 mm fraction. Specific adsorption is usually controlled by Fe- and Mn-oxides. Organic matter appeared to insignificantly affect the desorption rate (the desorption rates of untreated soil and organic matter removal were very close among all particle sizes). Fe-oxides (free and amorphous Fe-oxides) exhibited more influence on the desorption rate in the 0.05–0.002 mm fraction. After the Fe-oxides removal, nonspecific adsorption sites in these fractions decreased, which, in turn, led to a higher desorption rate. Apart from the <0.002 mm fraction that had the extremely high adsorption capacity, the 0.05–0.002 mm fraction had the highest contents of free Fe-oxides and amorphous Fe-oxides content. This phenomenon was the reason why the desorption rate in this fraction was the most influenced.4. Conclusion
Selective removal technology was applied and batch experiments were conducted to study the influence of different components (organic matter and Fe and Mn oxides) on Cd adsorption by soil aggregates. The adsorption characteristics of Cd in bulk soil and aggregates of different particle sizes could be accurately described by Langmuir equation regardless whether a specific component was selectively removed. All the removal treatments caused a decrease in Cd adsorption except for the free Fe-oxides removal. This result revealed that these components played different important roles in controlling adsorption behavior. Organic matter contributed most to the adsorption capacity but appear to insignificantly affect the desorption rate. By contrast, Fe- and Mn-oxides showed stronger influencing ability in determining the adsorption capacity of Cd in red paddy soil. The <0.002 mm soil aggregate contained more organic matter, free Fe- and Mn-oxides. Thus this fraction of aggregates can retain more Cd in soil. More attention should be paid to the fine soil aggregates (the 0.05–0.002 mm and <0.002 mm fractions) that contain higher Fe and Mn-oxides and that are easier to migrate in environment.Acknowledgements
The study was funded by the National Natural Science Foundation of China (41271294), the Program for New Century Excellent Talents in University (NCET-09-330).References
- P. Loganathan, S. Vigneswaran, J. Kandasamy and R. Naidu, Crit. Rev. Environ. Sci. Technol., 2012, 42, 489–533 CrossRef CAS PubMed.
- N. Bolan, A. Kunhikrishnan, R. Thangarajan, J. Kumpiene, J. Park, T. Makino, M. B. Kirkham and K. Scheckel, J. Hazard. Mater., 2014, 266, 141–166 CrossRef CAS PubMed.
- A. L. Seyfferth, S. McCurdy, M. V. Schaefer and S. Fendorf, Environ. Sci. Technol., 2014, 48, 4699–4706 CrossRef CAS PubMed.
- F. J. Zhao, Y. B. Ma, Y. G. Zhu, Z. Tang and S. P. McGrath, Environ. Sci. Technol., 2014, 49, 750–759 CrossRef PubMed.
- P. Li, X. X. Wang, T. L. Zhang, D. M. Zhou and Y. Q. He, J. Environ. Sci., 2008, 20, 449–455 CrossRef CAS.
- H. B. Bradl, J. Colloid Interface Sci., 2004, 277, 1–18 CrossRef CAS PubMed.
- G. M. Zeng, H. P. Wu, J. Liang, S. L. Guo, L. Huang, P. Xu, Y. Y. Liu, Y. J. Yuan, X. X. He and Y. He, RSC Adv., 2015, 5, 34541–34548 RSC.
- S. A. Zheng and M. K. Zhang, J. Environ. Sci., 2011, 23, 434–443 CrossRef CAS.
- M. K. Zhang, Z. L. He, D. V. Calvert, P. J. Stoffella, X. E. Yang and Y. C. Li, Soil Sci. Soc. Am. J., 2003, 67, 1158–1167 CrossRef CAS.
- F. L. Wang, W. Ouyang, F. H. Hao, A. Critto, X. C. Zhao and C. Y. Lin, RSC Adv., 2015, 5, 41238–41247 RSC.
- H. B. Zhang, Y. M. Luo, T. Makino, L. H. Wu and M. Nanzyo, J. Hazard. Mater., 2013, 248, 303–312 CrossRef PubMed.
- C. Gong, L. Ma, H. Cheng, Y. Liu, D. Xu, B. Li, F. Liu, Y. Ren, Z. Liu, C. Zhao, K. Yang, H. Nie and C. Lang, J. Geochem. Explor., 2014, 139, 109–114 CrossRef CAS PubMed.
- B. Huang, Z. W. Li, J. Q. Huang, L. Guo, X. D. Nie, Y. Wang, Y. Zhang and G. M. Zeng, J. Hazard. Mater., 2014, 264, 176–183 CrossRef CAS PubMed.
- E. F. Covelo, F. A. Vega and M. L. Andrade, J. Hazard. Mater., 2007, 140, 308–315 CrossRef CAS PubMed.
- Z. Zhu and W. Li, RSC Adv., 2012, 2, 5178–5184 RSC.
- C. Dumat, M. V. Cheshire, A. R. Fraser, C. A. Shand and S. Staunton, Eur. J. Soil Sci., 1997, 48, 675–683 CrossRef CAS PubMed.
- C. Pérez-Novo, M. Pateiro-Moure, F. Osorio, J. C. Nóvoa-Muñoz, E. López-Periago and M. Arias-Estévez, J. Colloid Interface Sci., 2008, 322, 33–40 CrossRef PubMed.
- D. Sarkar, D. K. de, R. Das and B. Mandal, Geoderma, 2014, 214, 213–216 CrossRef PubMed.
- H. A. Elliott, M. R. Liberati and C. P. Huang, Water, Air, Soil Pollut., 1986, 27, 379–389 CrossRef CAS.
- J. D. Reid and B. McDuffie, Water, Air, Soil Pollut., 1981, 15, 375–386 CrossRef CAS.
- E. Roth, V. Mancier and B. Fabre, Geoderma, 2012, 189, 133–143 CrossRef PubMed.
- X. L. Zhao, T. Jiang and B. Du, Chemosphere, 2014, 99, 41–48 CrossRef CAS PubMed.
- T. G. Dai, X. Li and Q. H. Wu, Environ. Earth Sci., 2008, 36, 321–326 CAS.
- M. Schmidt, C. Rumpel and I. KögelcKnabner, Eur. J. Soil Sci., 1999, 50, 87–94 CrossRef.
- K. Kaiser, K. Eusterhues, C. Rumpel, G. Guggenberger and I. Kögel Knabner, J. Plant Nutr. Soil Sci., 2012, 165, 451–459 CrossRef.
- A. Siregar, M. Kleber, R. Mikutta and R. Jahn, Eur. J. Soil Sci., 2005, 56, 481–490 CrossRef CAS PubMed.
- R. McDowell and L. Condron, Commun. Soil Sci. Plant Anal., 2001, 32, 2531–2547 CrossRef CAS PubMed.
- B. Anderson and E. Jenne, Soil Sci., 1970, 109, 163–169 CrossRef CAS.
- J. A. McKeague and J. H. Day, Can. J. Soil Sci., 1966, 46, 13–22 CrossRef CAS.
- T. T. Chao, Soil Sci. Soc. Am. Proc., 1972, 36, 764–768 CrossRef.
- Y. S. Ho and G. McKay, Water Res., 2000, 34, 735–742 CrossRef CAS.
- D. W. Nelson and L. E. Sommers, in Methods of Soil Analysis, ed. A. L Page, R. H. Miller and D. R. Keeny, ASA, 2nd edn, 1982, pp. 539–579 Search PubMed.
- W. H. Hendershot, H. Lalande and M. Duquette, in Soil Sampling and Methods of Analysis, ed. M. R Carter, Boca Raton, FL, 1993, pp. 141–145 Search PubMed.
- A. Z. Liang, X. M. Yang, X. P. Zhang, N. McLaughlin, Y. Shen and W. F. Li, Soil Tillage Res., 2009, 105, 21–26 CrossRef PubMed.
- S. Mangalassery, S. Sjögersten, D. L. Sparkes, C. J. Sturrock and S. J. Mooney, Soil Tillage Res., 2013, 132, 39–46 CrossRef PubMed.
- G. L. Guo, Y. Zhang, C. Zhang, S. J. Wang, Z. G. Yan and F. S. Li, Geoderma, 2013, 200, 108–113 CrossRef PubMed.
- A. Papadopoulos, N. R. A. Bird, A. P. Whitmore and S. J. Mooney, Eur. J. Soil Sci., 2009, 60, 360–368 CrossRef CAS PubMed.
- Q. Liu, F. Yuan, Y. Liang and Z. Li, RSC Adv., 2015, 5, 16089–16092 RSC.
- Z. F. Meng, Y. P. Zhang and Z. Q. Zhang, J. Hazard. Mater., 2008, 159, 492–498 CrossRef CAS PubMed.
- S. S. Gupta and K. G. Bhattacharyya, RSC Adv., 2014, 4, 28537–28586 RSC.
- M. Abat, M. J. McLaughlin, J. K. Kirby and S. P. Stacey, Geoderma, 2012, 175, 58–63 CrossRef PubMed.
- S. L. Yang, D. Q. Zhou, H. Y. Yu, R. Wei and B. Pan, Environ. Pollut., 2013, 177, 64–70 CrossRef CAS PubMed.
- S. Dragović, N. Mihailović and B. Gajić, Chemosphere, 2008, 72, 491–495 CrossRef PubMed.
- J. H. Chen, F. He, X. H. Zhang, X. Sun, J. F. Zheng and J. W. Zheng, FEMS Microbiol. Ecol., 2014, 87, 164–181 CrossRef CAS PubMed.
- F. R. Zeng, S. Ali, H. T. Zhang, Y. N. Ouyang, B. Y. Qiu, F. B. Wu and G. P. Zhang, Environ. Pollut., 2011, 159, 84–91 CrossRef CAS PubMed.
- P. N. Diagboya, B. I. Olu-Owolabi and K. O. Adebowale, Environ. Sci. Pollut. Res., 2015, 1–9 Search PubMed.
- J. H. Park, D. Lamb, P. Paneerselvam, G. Choppala, N. Bolan and J. W. Chung, J. Hazard. Mater., 2011, 185, 549–574 CrossRef CAS PubMed.
- Q. B. He and B. R. Singh, J. Soil Sci., 1993, 44, 641–650 CrossRef CAS PubMed.
- M. L. A. Silveira, L. R. F. Alleoni, O. A. Camargo and J. C. Casagrande, Commun. Soil Sci. Plant Anal., 2002, 33, 3581–3592 CrossRef CAS PubMed.
- H. A. Elliott and D. L. Sparks, Soil Sci., 1981, 132, 402–409 CrossRef CAS.
- L. M. Shuman, Soil Sci., 1988, 146, 248–254 CrossRef CAS.
- J. O. Agbenin and L. A. Olojo, Geoderma, 2004, 119, 85–95 CrossRef CAS.
- M. A. Donaldson, D. L. Bish and J. D. Raff, Proc. Natl. Acad. Sci. U. S. A., 2014, 111, 18472–18477 CrossRef CAS PubMed.
- C. Shang and L. W. Zelazny, Methods of Soil Analysis, 2008, 5, 33–80 CAS.
- O. Ajouyed, C. Hurel, M. Ammari, L. B. Allal and N. Marmier, J. Hazard. Mater., 2010, 174, 616–622 CrossRef CAS PubMed.
- X. H. Feng, L. M. Zhai, W. F. Tan, F. Liu and J. Z. He, Environ. Pollut., 2007, 147, 366–373 CrossRef CAS PubMed.
- J. E. Post, Proc. Natl. Acad. Sci. U. S. A., 1999, 96, 3447–3454 CrossRef CAS.
- G. M. Fu, H. E. Allen and C. E. Cowan, Soil Sci., 1991, 152, 72–81 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2015 |


