Cross-coupling reaction of aryl diazonium salts with azodicarboxylate using FeCl2†
M. Khalaj* and
M. Ghazanfarpour-Darjani
Islamic Azad University, Buinzahra Branch, Qazvin, Iran. E-mail: khalaj_mehdi@yahoo.com
First published on 9th September 2015
Abstract
Arene diazonium salts have been employed as the aryl source in reaction with dialkyl azodicarboxylates to form N-aryl hydrazide derivatives. The optimum conditions are developed using FeCl2 in DMSO at 25 °C for 2 h. Various functional groups were tolerated under the optimum conditions.
Introduction
Diazonium compounds are important intermediates due to their high potential for applications in the fields of organic synthesis.1–3 Besides the classical reactions, aryl diazonium salts also serve as an aryl source in transition metals-catalyzed cross-coupling reactions to form carbon–carbon and carbon–heteroatom bonds.4–8 Because of the pioneering study of Kikukawa and Matsuda,9 the coupling reactions of arene diazonium salts have been well developed over the past decades. Regardless of the long history, these substrates still attract attention and new reports have been emerging constantly. Though arene diazonium salts have been established as the aryl source in catalytic reactions, the main drawback is that they are not commercially available and in most cases have to be prepared before use. In this context, one-pot diazotization10–12/cross-coupling is obviously more effective.Dialkyl azodicarboxylates (DAAD) have been employed as the coupling partners in reactions with aryl metals reagents,13a–c arylboronic reagents,14 and arenes15 for the synthesis of N-aryl hydrazides. However, these reactions are typically limited to electron-rich aryl sources. Recently, Yavari and co-workers have also reported novel procedures to form N-aryl hydrazides using various active aryl sources, dialkyl azodicarboxylates and triphenyl phosphine or sodium hydride.16,17 Based on these findings, we seek to examine the efficiency of arene diazonium salts with dialkyl azodicarboxylates to form N-aryl hydrazides using iron(II) salts.
Results and discussion
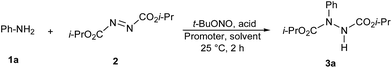
We initially examined the coupling of aniline (1a) with diisopropyl azodicarboxylate (2) using Fe(OAc)2, acetic acid, and tert-butyl nitrite as a model reaction to assess the reaction efficiency. Stirring in DMSO at 25 °C for 2 h resulted diisopropyl 1-(phenyl)-hydrazine-1,2-dicarboxylate in 59% yield together with biphenyl in 19% yield.To develop the reaction conditions a variety of promoters, solvents, and additives were examined (Table 1). No reaction occurred in the absence of an iron(II) salt (Table 1, entry 21). A catalyst screen also showed that FeCl2 gave the best result (Table 1, entry 2). Reaction conducted with anhydrous FeCl2 in extra dry DMSO formed only traces of the desired product (Table 1, entry 3). It could be deduced that H2O involves on reaction pathway. Other iron salts also promoted the reaction; however, the yields were comparatively lowers (Table 1, entries 4 to 8). CuI was not efficient in this transformation, (Table 1, entry 9) whereas reactions conducted with Zn and SmI2 occurred to moderate conversion in more than 4 h (Table 1, entries 10 and 11). It is worth mentioning that eosin Y also promotes the reaction with good yield (Table 1, entry 12). Among the solvents examined, DMSO was superior to other solvents (Table 1, entry 2). Reaction in an apolar solvent like toluene resulted in low conversions (Table 1, entry 13). The desired product was obtained only in 23% yield when H2O was used as the solvent (Table 1, entry 14). This study indicates that the higher amount of H2O inhibits the reaction. Other protic solvents, such as EtOH, also gave the product in low yield (Table 1, entry 15). Note that when the reaction was conducted in DMSO/H2O (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) mixture occurred comparatively in lower yield than those of DMSO. A variety of acids were also considered to evaluate the influence of the acidity and the nature of the counter-ion on reaction progress (Table 1, entries 18 to 20). The results showed that although the tetrafluoroborate anion has been widely used in the literature, it was not the most effective counter-ion for this reaction. Finally, p-TsOH was selected as the acid of choice based on the cost and efficiency. Note that reaction conducted with 1.0 mmol of FeCl2, formed only traces of the desired product which supports the suggested mechanism pathway.
1) mixture occurred comparatively in lower yield than those of DMSO. A variety of acids were also considered to evaluate the influence of the acidity and the nature of the counter-ion on reaction progress (Table 1, entries 18 to 20). The results showed that although the tetrafluoroborate anion has been widely used in the literature, it was not the most effective counter-ion for this reaction. Finally, p-TsOH was selected as the acid of choice based on the cost and efficiency. Note that reaction conducted with 1.0 mmol of FeCl2, formed only traces of the desired product which supports the suggested mechanism pathway.
| Entry | Catalyst | Solvent | Acid | Yield (%) |
|---|---|---|---|---|
a Reaction conditions: 1a (1.0 mmol), 2 (1.0 mmol), promoter (2.0 mmol), acid (1.0 mmol), t-BuONO (1.1 mmol), and solvent (3 mL) were stirred at 25 °C for 2 h, under a N2 atmosphere.b Anhydrous FeCl2 was used.c DMSO/H2O was used in a ratio of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 as the solvent.d 1.0 mmol of FeCl2 was used. 1 as the solvent.d 1.0 mmol of FeCl2 was used. |
||||
| 1 | Fe(OAc)2 | DMSO | TsOH | 59 |
| 2 | FeCl2 | DMSO | TsOH | 94 |
| 3 | FeCl2 | DMSO | TsOH | 6b |
| 4 | FeF2 | DMSO | TsOH | 54 |
| 5 | FeI2 | DMSO | TsOH | 87 |
| 6 | FeC2O4 | DMSO | TsOH | 32 |
| 7 | Fe(BF4)2 | DMSO | TsOH | 88 |
| 8 | FeSO4 | DMSO | TsOH | Trace |
| 9 | CuI | DMSO | TsOH | Trace |
| 10 | Zn | DMSO | TsOH | 64 |
| 11 | SmI2 | DMSO | TsOH | 41 |
| 12 | Eosin Y | DMSO | TsOH | 91 |
| 13 | FeCl2 | Toluene | TsOH | Trace |
| 14 | FeCl2 | H2O | TsOH | 23 |
| 15 | FeCl2 | EtOH | TsOH | 15 |
| 16 | FeCl2 | DMF | TsOH | 71 |
| 17 | FeCl2 | DMSO | TsOH | 46c |
| 18 | FeCl2 | DMSO | HOAc | 69 |
| 19 | FeCl2 | DMSO | MsOH | 80 |
| 20 | FeCl2 | DMSO | HBF4 | 46 |
| 21 | — | DMSO | TsOH | Trace |
| 22 | FeCl2 | DMSO | TsOH | Traced |
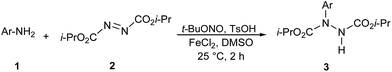
After having defined the optimum reaction conditions, we sought to explore the scope of the reaction (Table 2). Cross-coupling of electron-neutral aryl diazonium salts proceeded with good yields (entries 1 and 2). The reactions conducted with 3- and 4-methyl substituted aniline proceeded in high conversion (entries 1 and 2). This reaction is not sensitive to steric effects as ortho-substituted diazonium salt also afforded the desired product in high yield (entry 5). Electron-poor substrates resulted in slightly lower yields (entries 7 to 11) than those of electron-neutral and electron-rich substrates, which are probably due to the lower nucleophilicity of the aromatic ring. In the presence of 4-bromo and 2-bromo benzenamine (1l, 1m), amination occurred exclusively at the amine position (entry 12). Note that the tolerance for bromide on the aromatic ring offers an opportunity for subsequent cross-coupling, facilitating expedient synthesis of highly complex aryl hydrazides. Heteroaromatic substrate 1n also achieved the desired product in acceptable yield (entry 14). The presence of OH moiety on aromatic ring was not compatible with this amination reaction.
| Entry | 1 | Ar | Yield (%) |
|---|---|---|---|
| a Reaction conditions: 1 (1.0 mmol), 2 (1.0 mmol), FeCl2 (2.0 mmol), TsOH (1.0 mmol), t-BuONO (1.1 mmol), and DMSO (3 mL) were stirred at 25 °C for 2 h, under a N2 atmosphere. | |||
| 1 | a | Ph | 3a, 94 |
| 2 | b | 1-Naphthyl | 3b, 91 |
| 3 | c | 4-Tol | 3c, 94 |
| 4 | d | 3-Tol | 3d, 90 |
| 5 | e | 2-Tol | 3e, 91 |
| 6 | f | 4-MeO–C6H4 | 3f, 97 |
| 7 | g | 2-F3C–C6H4 | 3g, 86 |
| 8 | h | 3-F3C–C6H4 | 3h, 87 |
| 9 | i | 4-NC–C6H4 | 3i, 83 |
| 10 | j | 4-NO2–C6H4 | 3j, 81 |
| 11 | k | 2-NO2–C6H4 | 3k, 86 |
| 12 | l | 4-Br–C6H4 | 3l, 93 |
| 13 | m | 2-Br–C6H4 | 3m, 87 |
| 14 | n | 5-Methyl thienyl | 3n, 75 |
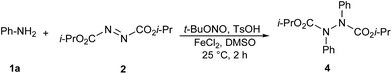
It is worth mentioning that when 2 mmol of phenyl diazonium salt was treated with 2 (1 mmol) at the optimum conditions, diisopropyl 1,2-(diphenyl)-1,2-hydrazinedicarboxylate (4) was achieved in 91% yield (Scheme 1). This result suggests that the mechanism involves reductive coupling of the aryldiazonium salt and azodicarboxylate with the oxidation of the Fe2+ ion.
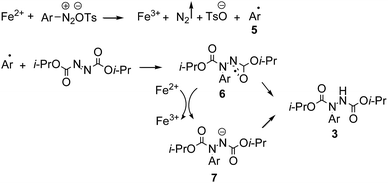
As shown in Scheme 2, the proposed mechanism starts with the formation of an aryl radical 5 by electron transfer from Fe(II) to the aryldiazonium salt. Addition of the aryl radical to diisopropyl azodicarboxylate gives radical intermediate 6. This intermediate is further transformed to the final coupling product 3 by two possible pathways: (a) reduction of the radical intermediate 6 by Fe(II) to give 7, followed by protonation with H2O or (b) direct hydrogen radical transfer from H2O to 6. The radical mechanism is further supported by the fact that 2,2,6,6-tetramethylpiperidinoxyl (TEMPO) effectively inhibits the reaction.
Experimental
Amines, azodicarboxylate, promoters, t-BuONO, acids, and solvents were obtained from Merck and were used without further purification. Mp: electrothermal-9100 apparatus. IR spectra: Shimadzu IR-460 spectrometer. 1H and 13C NMR spectra: Bruker DRX-500 AVANCE instrument; in CDCl3 at 500.1 and 125.7 MHz, respectively; δ in ppm, J in Hz. EI-MS (70 eV): Finnigan-MAT-8430 mass spectrometer, in m/z. Elemental analyses (C, H, N) were performed with a Heraeus CHN-O-Rapid analyzer. The results agreed favourably with the calculated values. All known compounds gave satisfactory spectroscopic data and were consistent with that reported in the literature13,14 (see ESI† for characterization data for all products).General procedure for synthesis of compound 3 or 4
A sample tube containing aryl amine (1.0 or 2.0 mmol), t-BuONO (1.1 mmol), p-TsOH (1.0 mmol), and DMSO (3 mL) was stirred for 10 min at 25 °C. Afterwards, DIAD (1.0 mmol) and FeCl2 (2.0 mmol) were added to the resulting mixture with the ambient conditions. The mixture was then evacuated, and backfilled with N2 (three times) and stirred for 2 h at 25 °C. The crude reaction mixture was diluted with EtOAc (5 mL) and a saturated NH4Cl solution (3 mL). The mixture was stirred for an additional 30 min and the two layers were separated. The aqueous layer was extracted with EtOAc (2 mL × 3). The combined organic layers were dried over MgSO4, filtered, and concentrated in vacuo to yield the crude product 3 (purity 84%). The crude product was further purified by recrystallization from diethyl ether.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 156.1 (C
O), 156.1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O). MS: m/z (%) = 294 (M+, 4), 235 (37), 207 (26), 192 (65), 133 (100), 105 (72), 102 (87), 60 (31). Anal. calcd for C15H22N2O4 (294.35): C, 61.21; H, 7.53; N, 9.52%. Found: C, 61.37; H, 7.70; N, 9.57%.
O). MS: m/z (%) = 294 (M+, 4), 235 (37), 207 (26), 192 (65), 133 (100), 105 (72), 102 (87), 60 (31). Anal. calcd for C15H22N2O4 (294.35): C, 61.21; H, 7.53; N, 9.52%. Found: C, 61.37; H, 7.70; N, 9.57%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 156.1 (C
O), 156.1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O). MS: m/z (%) = 359 (M+, 7), 361 ((M + 2)+, 7), 279 (36), 270 (52), 183 (35), 155 (100), 115 (78), 103 (63), 59 (81). Anal. calcd for C14H19BrN2O4 (359.22): C, 46.81; H, 5.33; N, 7.90; Br, 22.24%. Found: C, 46.89; H, 5.46; N, 7.99; Br, 22.35%.
O). MS: m/z (%) = 359 (M+, 7), 361 ((M + 2)+, 7), 279 (36), 270 (52), 183 (35), 155 (100), 115 (78), 103 (63), 59 (81). Anal. calcd for C14H19BrN2O4 (359.22): C, 46.81; H, 5.33; N, 7.90; Br, 22.24%. Found: C, 46.89; H, 5.46; N, 7.99; Br, 22.35%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 156.0 (C
O), 156.0 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O). MS: m/z (%) = 359 (M+, 2), 361 ((M + 2)+, 5), 279 (21), 270 (38), 183 (30), 155 (100), 115 (78), 103 (63), 59 (81). Anal. calcd for C14H19BrN2O4 (359.22): C, 46.81; H, 5.33; N, 7.90; Br, 22.24%. Found: C, 46.85; H, 5.38; N, 7.94; Br, 22.30%.
O). MS: m/z (%) = 359 (M+, 2), 361 ((M + 2)+, 5), 279 (21), 270 (38), 183 (30), 155 (100), 115 (78), 103 (63), 59 (81). Anal. calcd for C14H19BrN2O4 (359.22): C, 46.81; H, 5.33; N, 7.90; Br, 22.24%. Found: C, 46.85; H, 5.38; N, 7.94; Br, 22.30%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 156.6 (C
O), 156.6 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O). MS: m/z (%) = 300 (M+, 2), 241 (13), 203 (34), 183 (16), 97 (100), 59 (34). Anal. calcd for C13H20N2O4S (300.37): C, 51.98; H, 6.71; N, 9.33; S, 10.68%. Found: C, 52.13; H, 6.89; N, 9.28; S, 10.84%.
O). MS: m/z (%) = 300 (M+, 2), 241 (13), 203 (34), 183 (16), 97 (100), 59 (34). Anal. calcd for C13H20N2O4S (300.37): C, 51.98; H, 6.71; N, 9.33; S, 10.68%. Found: C, 52.13; H, 6.89; N, 9.28; S, 10.84%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O). MS: m/z (%) = 356 (M+, 1), 269 (18), 182 (25), 87 (12), 77 (100). Anal. calcd for C20H24N2O4 (356.42): C, 67.40; H, 6.79; N, 7.86%. Found: C, 67.69; H, 6.84; N, 8.10%.
O). MS: m/z (%) = 356 (M+, 1), 269 (18), 182 (25), 87 (12), 77 (100). Anal. calcd for C20H24N2O4 (356.42): C, 67.40; H, 6.79; N, 7.86%. Found: C, 67.69; H, 6.84; N, 8.10%.Conclusion
We have developed a new cross-coupling reaction for the synthesis of N-aryl hydrazides using readily available and easily prepared starting materials with ambient conditions and a short reaction time. Using this procedure, a simple recrystallization from ether would suffice to isolate the pure titled product. The electronic and steric variations of substrates did not change the yield in an appreciable manner. The reaction is completely selective for a cross-coupling reaction and no compounds arising from benzene homo-coupling are detected by crude GC-MS analysis using the optimal conditions.Acknowledgements
This study was supported by the Islamic Azad University of IRAN, buinzahra branch (research grant).Notes and references
- S. Mahouche-Chergui, S. Gam-Derouich, C. Mangeneya and M. M. Chehim, Chem. Soc. Rev., 2011, 40, 4143 RSC.
- M. Hartmann, Y. Li and A. Studer, J. Am. Chem. Soc., 2012, 134, 16516 CrossRef CAS PubMed.
- A. Wetzel, G. Pratsch, R. Kolb and M. R. Heinrich, Chem.–Eur. J., 2010, 16, 2547 CrossRef CAS PubMed.
- H. Bonin, D. Delbrayelle, P. Demonchaux and E. Gras, Chem. Commun., 2010, 46, 2677 RSC.
- M. B. Andrus and C. Song, Org. Lett., 2001, 3, 3761 CrossRef CAS PubMed.
- F. Mo, D. Qiu, Y. Jiang, Y. Zhang and J. Wang, Tetrahedron Lett., 2010, 52, 518 CrossRef PubMed.
- H. Bonin, E. Fouquet and F. X. Felpin, Adv. Synth. Catal., 2011, 353, 3063 CrossRef CAS PubMed.
- F. X. Felpin, K. Miqueu, J. M. Sotiropoulos, E. Fouquet, O. Ibarguren and J. Laudien, Chem.–Eur. J., 2010, 16, 5191 CrossRef CAS PubMed.
- G. K. Kikukawa and T. Matsuda, Chem. Lett., 1977, 159 CrossRef.
- S. Tang, D. Zhou and Y. C. Wang, Eur. J. Org. Chem., 2014, 3656 CrossRef CAS PubMed.
- L. Malet-Sanz, J. Madrzak, S. V. Ley and I. R. Baxendale, Org. Biomol. Chem., 2010, 8, 5324 CAS.
- L. He, G. Qiu, Y. Gao and J. Wu, Org. Biomol. Chem., 2014, 12, 6965 CAS.
- (a) J. P. Demers and D. H. Klaubert, Tetrahedron Lett., 1987, 28, 4933 CrossRef CAS; (b) A. R. Katritzky, J. Wu and S. V. Verin, Synthesis, 1995, 651 CrossRef CAS; (c) I. Zaltsgendler, Y. Leblanc and M. A. Bernstein, Tetrahedron Lett., 1993, 34, 2441 CrossRef CAS.
- T. Uemura and N. Chatani, J. Org. Chem., 2005, 70, 8631 CrossRef CAS PubMed.
- L. Gu, S. Neo and Y. Zhang, Org. Lett., 2011, 13, 1872 CrossRef CAS PubMed.
- I. Yavari, M. Ghazanfarpour-Darjani, S. Ahmadian and Y. Solgi, Synlett, 2011, 1745 CrossRef CAS.
- I. Yavari, M. Ghazanfarpour-Darjani, M. Bayat and A. Malekafzali, Synlett, 2015, 942 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra15875c |
| This journal is © The Royal Society of Chemistry 2015 |