Potential use of alginate beads as a chondrocyte delivery vehicle and stepwise dissolving porogen in a hydrogel scaffold for cartilage tissue engineering†
Abstract
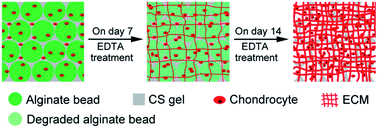
The submicron- or nano-sized pores and uncontrollable degradation of conventional hydrogels have severely constrained cell growth and neo-tissue formation. In this study, alginate beads are explored as both delivery vehicles of chondrocytes and stimuli-responsive porogens within hydrogel scaffolds for cartilage tissue engineering. A typical chondroitin sulfate (CS)–alginate beads composite gel (CS–ABG) is fabricated by photo-encapsulating alginate beads into the CS gel, and subsequently batch-wise dissolution and leaching out of the alginate beads is achieved by twice exposing CS–ABG to chelating agents. The combining and gradual removal of alginate beads effectively modulate the gel's physical properties (e.g. swelling ratio, crosslink density) as well as create macro-scale cavities within CS–ABG. The efficacy of CS–ABG as a scaffold for cartilage tissue engineering is compared with a conventional photocrosslinked CS gel (CS-G). The CS–ABG constructs are developed by co-encapsulating chondrocytes and cell-laden alginate beads within the CS gel body and undergo EDTA treatment on day 7 and 14 of culture, respectively, for stepwise removal of the alginate beads. The chondrocytes cultured in CS–ABG constructs exhibit higher cell viability and proliferation, enhanced cartilage-specific gene expressions as well as ECM production compared with those in CS-G constructs. This study demonstrates the potential of alginate beads as cell delivery vehicles and gradually dissolving porogens within gel scaffolds for cartilage tissue engineering.

 Please wait while we load your content...
Please wait while we load your content...