Platinum-catalysed cinnamaldehyde hydrogenation in continuous flow†
Lee J. Durndell,
Karen Wilson and
Adam F. Lee*
European Bioenergy Research Institute, Aston University, Birmingham B4 7ET, UK. E-mail: a.f.lee@aston.ac.uk; Tel: +44 (0)121 2044036
First published on 9th September 2015
Abstract
Platinum is one of the most widely used hydrogenation catalysts. Here we describe the translation of batch reactions to continuous flow, affording tunable C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O versus C
O versus C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation over a Pt/SiO2 catalyst, resulting in high steady state activity and single-pass yields in the selective hydrogenation of cinnamaldehyde to cinnamyl alcohol under mild conditions. Negligible catalyst deactivation occurs under extended flow operation due to removal of reactively-formed poisons from the reaction zone. Process intensification imparts a four-fold enhancement in cinnamyl alcohol productivity.
C hydrogenation over a Pt/SiO2 catalyst, resulting in high steady state activity and single-pass yields in the selective hydrogenation of cinnamaldehyde to cinnamyl alcohol under mild conditions. Negligible catalyst deactivation occurs under extended flow operation due to removal of reactively-formed poisons from the reaction zone. Process intensification imparts a four-fold enhancement in cinnamyl alcohol productivity.
Chemoselectivity underpins 21st century catalysis,1,2 permitting the targeted modification of specific functional groups within complex starting materials,3–7 notably from biomass-derived feedstocks.8 Catalytic hydrogenation of organic compounds possessing multiple unsaturated bonds such as α,β-unsaturated aldehydes is particularly challenging,9–11 necessitating active sites able to discriminate and preferentially activate closely related moieties. Platinum is widely employed in heterogeneously catalysed hydrogenation of diverse functional groups including C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C,12 C
C,12 C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C,13 C
C,13 C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O,13 C
O,13 C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N,14 NO2 (ref. 15) and aromatics.15 The selective hydrogenation of allylic and benzylic aldehydes to unsaturated alcohols is a commercially important industrial process within the flavour, fragrance, agrochemical and pharmaceutical sectors,9,16 in which active and selective heterogeneous catalysts for such transformations are essential to circumvent the greater thermodynamic stability of C
N,14 NO2 (ref. 15) and aromatics.15 The selective hydrogenation of allylic and benzylic aldehydes to unsaturated alcohols is a commercially important industrial process within the flavour, fragrance, agrochemical and pharmaceutical sectors,9,16 in which active and selective heterogeneous catalysts for such transformations are essential to circumvent the greater thermodynamic stability of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O relative to C
O relative to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds.9
C bonds.9
The liquid phase, selective hydrogenation of cinnamaldehyde (CinnALD) to cinnamyl alcohol (CinnOH) illustrated in Scheme 1 is of significant interest due to the widespread use of this allylic alcohol in perfumes and flavourants.16–19 Platinum is a promising catalyst for this challenging reaction, in which hydrogenation of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond is both kinetically and thermodynamically more favourable than the C
C bond is both kinetically and thermodynamically more favourable than the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O function,20 and hence the influence of the physicochemical properties of platinum nanoparticles is a topic of much intensive recent investigation in batch reactors. Particle size effects upon CinnOH selectivity have proved controversial, with oleic acid/oleylamine stabilised mono- and bimetallic colloidal Pt nanoparticles reported to exhibit a strong size dependence of CinnOH selectivity, with low coordination sites favoring C
O function,20 and hence the influence of the physicochemical properties of platinum nanoparticles is a topic of much intensive recent investigation in batch reactors. Particle size effects upon CinnOH selectivity have proved controversial, with oleic acid/oleylamine stabilised mono- and bimetallic colloidal Pt nanoparticles reported to exhibit a strong size dependence of CinnOH selectivity, with low coordination sites favoring C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation,21,22 whereas Zhu and Zaera reported that CinnOH selectivity was insensitive to the size of silica supported Pt nanoparticles albeit over a narrow size range.23 Guo et al. have shown that confinement of Pt nanoclusters within the cavity of metal–organic frameworks also promotes CinnOH selectivity; with steric constraints on CinnALD believed to hinder C
C hydrogenation,21,22 whereas Zhu and Zaera reported that CinnOH selectivity was insensitive to the size of silica supported Pt nanoparticles albeit over a narrow size range.23 Guo et al. have shown that confinement of Pt nanoclusters within the cavity of metal–organic frameworks also promotes CinnOH selectivity; with steric constraints on CinnALD believed to hinder C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C planar adsorption with consequent preferential C
C planar adsorption with consequent preferential C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O activation.24 Kinetics of CinnALD hydrogenation are also a function of support properties25 and hydrogen pressure. We recently reported a detailed mechanistic study of the structural and electronic factors controlling the liquid phase hydrogenation of cinnamaldehyde and related benzylic aldehydes over fumed SiO2 and mesoporous SBA-15 supported Pt nanoparticles.26 Kinetic mapping revealed cinnamaldehyde hydrogenation is structure-insensitive over metallic platinum, proceeding with a common Turnover Frequency (TOF) independent of precursor, particle size or support architecture, while selectivity to CinnOH is highly structure sensitive. Large nanoparticles and high hydrogen pressures favored C
O activation.24 Kinetics of CinnALD hydrogenation are also a function of support properties25 and hydrogen pressure. We recently reported a detailed mechanistic study of the structural and electronic factors controlling the liquid phase hydrogenation of cinnamaldehyde and related benzylic aldehydes over fumed SiO2 and mesoporous SBA-15 supported Pt nanoparticles.26 Kinetic mapping revealed cinnamaldehyde hydrogenation is structure-insensitive over metallic platinum, proceeding with a common Turnover Frequency (TOF) independent of precursor, particle size or support architecture, while selectivity to CinnOH is highly structure sensitive. Large nanoparticles and high hydrogen pressures favored C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O over C
O over C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation due to molecular surface crowding, while in situ ATR-IR highlighted the role of support polarity in enhancing C
C hydrogenation due to molecular surface crowding, while in situ ATR-IR highlighted the role of support polarity in enhancing C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O hydrogenation.
O hydrogenation.
 | ||
| Scheme 1 Principal reaction pathways operating in the Pt-catalysed liquid phase hydrogenation of cinnamaldehyde. | ||
Catalytic hydrogenations are traditionally conducted in stirred batch reactors, presenting challenges in respect of the low hydrogen solubility in most solvents, which results in mass transfer limitations.27,28 These are typically offset through the use of high pressures of up to 100 bar, which increases the associated explosion risks. Small-scale microreactors offer a safer, alternative approach to high-pressure hydrogenations with molecular hydrogen, and in conjunction with continuous flow processing offers significant benefits compared to batch processes, related to the unique gas–liquid–solid triphasic reaction conditions present in such transformations, improved safety, and process intensification.29,30 In recent years the chemical and pharmaceutical sectors have thus targeted a switchover from batch to continuous operation to achieve atom economical, and scalable organic synthesis.31,32 To date, the continuous, liquid phase selective hydrogenation of cinnamaldehyde has only been the subject of two peer-reviewed studies, wherein the focus was evaluating the suitability of either pellet string33 or high-pressure membrane34,35 reactor designs for industrial chemical synthesis. In both cases, the respective Pd/Al2O3 and Pd/MCM-41 catalysts employed were only selective to hydrocinnamaldehyde, and only studied at high pressures >10 bar. In contrast, Pt/SiO2 and Pt/SBA-15 catalysts are moderately selective to desirable CinnOH at lower pressures,26 but are prone to on-stream deactivation in batch, and exhibit strong time selectivity variations. Here we demonstrate that continuous flow operation in a packed bed FlowSyn microreactor with a GAM II coil-in-coil gas pre-saturator36 (Scheme 2); greatly increases the activity and selectivity of Pt/SiO2 towards the liquid phase hydrogenation of CinnALD to CinnOH, accompanied by dramatically improved catalyst stability and ease of separation and flexible productivity.
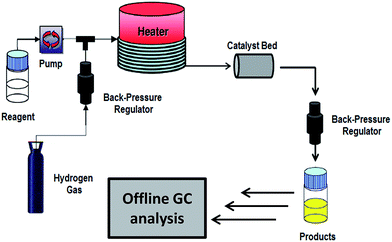 | ||
| Scheme 2 Schematic of FlowSyn continuous flow reactor for heterogeneously catalysed cinnamaldehyde hydrogenation. | ||
Structural properties of the wet-impregnated 2 wt% Pt/SiO2 catalyst are presented in Table S1 and Fig. S1 and S2.† The liquid residence time τ (defined as bed volume/flow rate) was studied with gas flow through determining the system response to the introduction of cinnamaldehyde. The resulting residence time distribution for the normalised response F(t)37 given by eqn (1), showed a step change, and hence near plug-flow behaviour with relatively little broadening from mixing or axial dispersion (Fig. 1). This demonstrates that the catalyst bed was static and filled with well-distributed liquid. At 1 bar, CinnALD hydrogenation was first order in catalyst charge under the conditions employed in this study (Table S2†), confirming the absence of external mass-transport limitations.
| F(t) = {([CinnALD]t + [Products]t)/[CinnALD]inlet}/τ | (1) |
The influence of residence time on CinnALD conversion and product selectivity was first investigated (Fig. 2). At the shortest residence times (τ < 5 min) only trace CinnALD conversion was observed, consistent with the relatively slow batch kinetics (initial rates of 500 mmol g−1 Pt h−1). As the residence time increases, a corresponding monotonic increase in activity was observed, with single pass conversions reaching 45%, however CinnALD hydrogenation attained a plateau at longer residence times above 15 min, likely due to limited hydrogen availability under these conditions; the reaction order in pH2 was 0.5 (Fig. S3†), in excellent agreement with that reported in batch over supported Pt nanoparticles. Product selectivity exhibited a more complex relationship with residence time. CinnOH displayed a volcano dependence, peaking around 60% for τ = 15 min, coincident with the maximum rate of CinnALD hydrogenation, and inversely correlated with the formation of undesired 3-phenylpropionaldehyde and ethylbenzene products via competing C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation and decarbonylation of the resulting saturated aldehyde. Small quantities (3–8%) of 3-phenyl propanol and cinnamyl cinnamate were also formed, mirroring the variation in 3-phenylpropionaldehyde (Fig. S4†). These trends are similar to those seen in batch, wherein CinnALD hydrogenation to 3-phenylpropionaldehyde and 3-phenylpropan-1-ol is favoured at conversions <5 % (the latter a secondary product from rapid hydrogenation of reactively-formed CinnOH over SiO2 supported platinum), with selectivity towards CinnOH increasing to 45% at 40% conversion. The fall in CinnOH selectivity and concomitant rise in 3-phenylpropionaldehyde, and ethylbenzene and 3-phenylpropan-1-ol (latter shown in Fig. S4†) secondary products at longer residence times (high conversion) is attributed to a combination of hydrogen starvation and thermodynamically favoured C
C hydrogenation and decarbonylation of the resulting saturated aldehyde. Small quantities (3–8%) of 3-phenyl propanol and cinnamyl cinnamate were also formed, mirroring the variation in 3-phenylpropionaldehyde (Fig. S4†). These trends are similar to those seen in batch, wherein CinnALD hydrogenation to 3-phenylpropionaldehyde and 3-phenylpropan-1-ol is favoured at conversions <5 % (the latter a secondary product from rapid hydrogenation of reactively-formed CinnOH over SiO2 supported platinum), with selectivity towards CinnOH increasing to 45% at 40% conversion. The fall in CinnOH selectivity and concomitant rise in 3-phenylpropionaldehyde, and ethylbenzene and 3-phenylpropan-1-ol (latter shown in Fig. S4†) secondary products at longer residence times (high conversion) is attributed to a combination of hydrogen starvation and thermodynamically favoured C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation. It is immediately apparent that tuning the residence time affords a simple means to regulate C
C hydrogenation. It is immediately apparent that tuning the residence time affords a simple means to regulate C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O versus C
O versus C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation (Fig. S5†) and hence optimise the CinnOH productivity at constant pH2 and temperature, and that good conversion and selectivity are achievable even for comparatively short reaction times. The sigmoidal increase in cinnamaldehyde conversion (and hence total product yield) with residence time contrasts with the report by Noël et al. for the metal-free perfluoroalkylation of N-methylpyrrole with CF3I, wherein a surprising decrease in perfluoroalkylation rate was observed with increasing residence time, attributed to poorer mixing of the gas and liquid phases,38 highlighting the superior mixing characteristics of the GAM II gas pre-saturator in the present work which ensured efficient hydrogen dissolution even at low flow rates.
C hydrogenation (Fig. S5†) and hence optimise the CinnOH productivity at constant pH2 and temperature, and that good conversion and selectivity are achievable even for comparatively short reaction times. The sigmoidal increase in cinnamaldehyde conversion (and hence total product yield) with residence time contrasts with the report by Noël et al. for the metal-free perfluoroalkylation of N-methylpyrrole with CF3I, wherein a surprising decrease in perfluoroalkylation rate was observed with increasing residence time, attributed to poorer mixing of the gas and liquid phases,38 highlighting the superior mixing characteristics of the GAM II gas pre-saturator in the present work which ensured efficient hydrogen dissolution even at low flow rates.
In order to quantitatively benchmark the performance of our 2 wt% Pt/SiO2 catalyst in flow versus batch CinnALD hydrogenation, we subsequently conducted extended testing over 7 h to evaluate catalyst stability and net CinnOH productivity. Fig. 3a reveals striking differences in catalyst activity, with TOFs at least 50% higher in flow than batch, even during the early stages of reaction. This suggests either that catalyst deactivation is extremely rapid in batch, or that CinnALD hydrogenation in our batch reactor is rate-limited by mass-transport (presumably of hydrogen). The former appears more plausible, since mixing in stirred batch reactors is known to be highly efficient, and furthermore significant deactivation was subsequently observed over the course of 7 h, occurring at an approximately exponential rate which can be extrapolated back to a similar maximum TOF = 1300 h−1 at zero time as observed in flow; exponential activity decay due to catalyst poisoning by strongly bound products has been previously observed in the (semi-)batch hydrogenation of α-methylethylbenzene39 and sitosterol40 over commercial Pd/C catalysts. In contrast, negligible deactivation of the Pt/SiO2 catalyst was observed in flow, consistent with the rapid removal of reactively-formed products and low attendant concentration of potential poisons. The different time-on-stream activity profiles in flow versus batch are mirrored by their corresponding product distributions (Fig. 3b and c). In flow, the desired CinnOH is the major product throughout the course of reaction, with 3-phenylpropionaldehyde and ethylbenzene formed in equal concentrations along with trace and 3-phenylpropan-1-ol, evidencing C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O hydrogenation as the dominant primary reaction of CinnALD. Strong time dependent selectivity variations are observed under batch conditions, with the fresh catalyst strongly favouring C
O hydrogenation as the dominant primary reaction of CinnALD. Strong time dependent selectivity variations are observed under batch conditions, with the fresh catalyst strongly favouring C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation of CinnALD to 3-phenylpropionaldehyde and 3-phenylpropan-1-ol; we have previously shown the latter secondary product only arises via hydrogenation of reactively-formed CinnOH. On-stream catalyst deactivation coincides with a dramatic loss in 3-phenylpropan-1-ol and concomitant rise in selectivity to CinnOH, confirming their interrelationship, and small decrease in 3-phenylpropionaldehyde formation.
C hydrogenation of CinnALD to 3-phenylpropionaldehyde and 3-phenylpropan-1-ol; we have previously shown the latter secondary product only arises via hydrogenation of reactively-formed CinnOH. On-stream catalyst deactivation coincides with a dramatic loss in 3-phenylpropan-1-ol and concomitant rise in selectivity to CinnOH, confirming their interrelationship, and small decrease in 3-phenylpropionaldehyde formation.
The coincidence of deactivation and suppression of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation pathways in batch, strongly implicates a unique Pt active site responsible for C
C hydrogenation pathways in batch, strongly implicates a unique Pt active site responsible for C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation, discrete from that catalysing C
C hydrogenation, discrete from that catalysing C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O hydrogenation. This hypothesis meshes perfectly with our previous studies on size-controlled Pt nanoparticles over silica supports, wherein low coordination sites prevalent on smaller particles were observed to promote C
O hydrogenation. This hypothesis meshes perfectly with our previous studies on size-controlled Pt nanoparticles over silica supports, wherein low coordination sites prevalent on smaller particles were observed to promote C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation of CinnALD, while larger particles and associated Pt terrace promoted C
C hydrogenation of CinnALD, while larger particles and associated Pt terrace promoted C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O hydrogenation. It is well-established in heterogeneous catalysts that edge and vertex sites of transition metal particles are highly reactive due to their undercoordination, and (consequentially) prone to self-poisoning due to the higher adsorption energies of atomic and molecular reactants/products at these sites relative to close-packed facets. Extended Hückel calculations by Delbecq and Sautet have shown that stepped surfaces favour πC
O hydrogenation. It is well-established in heterogeneous catalysts that edge and vertex sites of transition metal particles are highly reactive due to their undercoordination, and (consequentially) prone to self-poisoning due to the higher adsorption energies of atomic and molecular reactants/products at these sites relative to close-packed facets. Extended Hückel calculations by Delbecq and Sautet have shown that stepped surfaces favour πC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C CinnALD adsorption modes and C
C CinnALD adsorption modes and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation relative to Pt(111) terraces which favour di-σCO CinnALD adsorption and C
C hydrogenation relative to Pt(111) terraces which favour di-σCO CinnALD adsorption and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O hydrogenation.41 We therefore propose that during batch CinnALD hydrogenation, one or more reactively-formed products associated with C
O hydrogenation.41 We therefore propose that during batch CinnALD hydrogenation, one or more reactively-formed products associated with C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation at reactive, low coordination sites is strongly adsorbed over the sites responsible for its formation, resulting in self-poisoning of C
C hydrogenation at reactive, low coordination sites is strongly adsorbed over the sites responsible for its formation, resulting in self-poisoning of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogenation pathways. Comparison of the flow versus batch product distributions suggests ethylbenzene as the frontrunner candidate poison, it being formed at low concentrations in flow, but rapidly swept away from the catalyst bed minimising the time for its re-adsorption/equilibration. The absence of ethylbenzene within the bulk reaction media during batch, over the same catalyst to that which forms trace ethylbenzene under identical reaction conditions of temperature, pH2 and [CinnALD] in flow, supports the proposal that catalyst poisoning in batch may reflect adsorption of reactively-formed ethylbenzene over long contact times. This hypothesis was tested by spiking the reaction solution with a constant flow of either 5% or 10% ethylbenzene (Fig. S6†), which resulted in an immediate 14% or 32% decrease in catalyst activity. Cinnamaldehyde conversion rapidly returned to its initial value upon removal of ethylbenzene poison from the reactant stream, evidencing fully reversible deactivation due to site-blocking by strongly-bound ethylbenzene. The absolute CinnOH productivity of our 2 wt% Pt/SiO2 catalyst in flow versus batch is compared in Fig. 4, which highlights an approximately constant four-fold yield enhancement for CinnALD hydrogenation under flow.
C hydrogenation pathways. Comparison of the flow versus batch product distributions suggests ethylbenzene as the frontrunner candidate poison, it being formed at low concentrations in flow, but rapidly swept away from the catalyst bed minimising the time for its re-adsorption/equilibration. The absence of ethylbenzene within the bulk reaction media during batch, over the same catalyst to that which forms trace ethylbenzene under identical reaction conditions of temperature, pH2 and [CinnALD] in flow, supports the proposal that catalyst poisoning in batch may reflect adsorption of reactively-formed ethylbenzene over long contact times. This hypothesis was tested by spiking the reaction solution with a constant flow of either 5% or 10% ethylbenzene (Fig. S6†), which resulted in an immediate 14% or 32% decrease in catalyst activity. Cinnamaldehyde conversion rapidly returned to its initial value upon removal of ethylbenzene poison from the reactant stream, evidencing fully reversible deactivation due to site-blocking by strongly-bound ethylbenzene. The absolute CinnOH productivity of our 2 wt% Pt/SiO2 catalyst in flow versus batch is compared in Fig. 4, which highlights an approximately constant four-fold yield enhancement for CinnALD hydrogenation under flow.
Conclusions
The transition from liquid phase batch to continuous flow cinnamaldehyde hydrogenation over a Pt/SiO2 heterogeneous catalysts affords significant enhancements in activity and selectivity towards C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O activation under optimised (mild) reaction conditions of 5 bar and 90 °C. Superior catalytic performance in flow is attributed to removal of the reactively-formed cinnamyl alcohol and 3-phenylpropionaldehyde primary products from the reaction zone, limiting their over-hydrogenation and/or hydrogenolysis to 3-phenylpropan-1-ol and ethylbenzene which are implicated as strongly adsorbing catalyst poisons under batch operation. The combination of excellent catalyst activity, stability and good selectivity in continuous flow deliver (predictable) cumulative cinnamyl alcohol productivities four times greater than possible under comparable conditions in batch.
O activation under optimised (mild) reaction conditions of 5 bar and 90 °C. Superior catalytic performance in flow is attributed to removal of the reactively-formed cinnamyl alcohol and 3-phenylpropionaldehyde primary products from the reaction zone, limiting their over-hydrogenation and/or hydrogenolysis to 3-phenylpropan-1-ol and ethylbenzene which are implicated as strongly adsorbing catalyst poisons under batch operation. The combination of excellent catalyst activity, stability and good selectivity in continuous flow deliver (predictable) cumulative cinnamyl alcohol productivities four times greater than possible under comparable conditions in batch.
Acknowledgements
We thank the EPSRC for financial support (EP/G007594/4 and EP/K014749/1) and the award of a Leadership Fellowship to AFL. KW thanks the Royal Society for an Industry Fellowship.Notes and references
- G. A. Somorjai and K. McCrea, Appl. Catal., A, 2001, 222, 3–18 CrossRef CAS.
- G. A. Somorjai, H. Frei and J. Y. Park, J. Am. Chem. Soc., 2009, 131, 16589–16605 CrossRef CAS PubMed.
- B. M. Trost and R. J. Kulawiec, J. Am. Chem. Soc., 1993, 115, 2027–2036 CrossRef CAS.
- B. Breit and W. Seiche, Synthesis, 2001, 1–36 CrossRef CAS.
- K. W. Fiori and J. Du Bois, J. Am. Chem. Soc., 2007, 129, 562–568 CrossRef CAS PubMed.
- J. Magano and J. R. Dunetz, Org. Process Res. Dev., 2012, 16, 1156–1184 CrossRef CAS.
- H. U. Blaser, H. Steiner and M. Studer, ChemCatChem, 2009, 1, 210–221 CrossRef CAS PubMed.
- R. A. Sheldon, Green Chem., 2014, 16, 950–963 RSC.
- P. Gallezot and D. Richard, Catal. Rev., 1998, 40, 81–126 CAS.
- H. U. Blaser, C. Malan, B. Pugin, F. Spindler, H. Steiner and M. Studer, Adv. Synth. Catal., 2003, 345, 103–151 CrossRef CAS PubMed.
- P. Maki-Arvela, J. Hajek, T. Salmi and D. Y. Murzin, Appl. Catal., A, 2005, 292, 1–49 CrossRef PubMed.
- I. Lee, F. Delbecq, R. Morales, M. A. Albiter and F. Zaera, Nat. Mater., 2009, 8, 132–138 CrossRef CAS PubMed.
- G. A. Attard, J. A. Bennett, I. Mikheenko, P. Jenkins, S. Guan, L. E. Macaskie, J. Wood and A. J. Wain, Faraday Discuss., 2013, 162, 57–75 RSC.
- C. Poupin, R. Maache, L. Pirault-Roy, R. Brahmi and C. T. Williams, Appl. Catal., A, 2014, 475, 363–370 CrossRef CAS PubMed.
- P. Lara, A. Suárez, V. Collière, K. Philippot and B. Chaudret, ChemCatChem, 2014, 6, 87–90 CrossRef CAS PubMed.
- L. A. Saudan, Acc. Chem. Res., 2007, 40, 1309–1319 CrossRef CAS PubMed.
- J. A. Maga, CRC Crit. Rev. Food Sci. Nutr., 1978, 10, 323–372 CrossRef CAS PubMed.
- C. S. Letizia, J. Cocchiara, J. Lalko, A. Lapczynski and A. M. Api, Food Chem. Toxicol., 2005, 43, 837–866 CrossRef CAS PubMed.
- D. A. Buckley, Br. J. Dermatol., 2007, 157, 295–300 CrossRef CAS PubMed.
- M. A. Vannice and B. Sen, J. Catal., 1989, 115, 65–78 CrossRef CAS.
- W. O. Oduro, N. Cailuo, K. M. Yu, H. Yang and S. C. Tsang, Phys. Chem. Chem. Phys., 2011, 13, 2590–2602 RSC.
- S. C. Tsang, N. Cailuo, W. Oduro, A. T. S. Kong, L. Clifton, K. M. K. Yu, B. Thiebaut, J. Cookson and P. Bishop, ACS Nano, 2008, 2, 2547–2553 CrossRef CAS PubMed.
- Y. Zhu and F. Zaera, Catal. Sci. Technol., 2014, 4, 955–962 CAS.
- Z. Guo, C. Xiao, R. V. Maligal-Ganesh, L. Zhou, T. W. Goh, X. Li, D. Tesfagaber, A. Thiel and W. Huang, ACS Catal., 2014, 1340–1348 CrossRef CAS.
- X. Ji, X. Niu, B. Li, Q. Han, F. Yuan, F. Zaera, Y. Zhu and H. Fu, ChemCatChem, 2014, 6, 3246–3253 CrossRef CAS PubMed.
- L. J. Durndell, C. M. A. Parlett, N. S. Hondow, M. A. Isaacs, K. Wilson and A. F. Lee, Sci. Rep., 2015, 5, 9425 CrossRef CAS PubMed.
- M. Irfan, T. N. Glasnov and C. O. Kappe, ChemSusChem, 2011, 4, 300–316 CrossRef CAS PubMed.
- X. Liu, B. Unal and K. F. Jensen, Catal. Sci. Technol., 2012, 2, 2134–2138 CAS.
- X. Ye, M. D. Johnson, T. Diao, M. H. Yates and S. S. Stahl, Green Chem., 2010, 12, 1180–1186 RSC.
- M. Brzozowski, M. O'Brien, S. V. Ley and A. Polyzos, Acc. Chem. Res., 2015, 48, 349–362 CrossRef CAS PubMed.
- C. Wiles and P. Watts, Green Chem., 2012, 14, 38–54 RSC.
- L. Vaccaro, D. Lanari, A. Marrocchi and G. Strappaveccia, Green Chem., 2014, 16, 3680–3704 RSC.
- A. Müller, M. Ludwig, M. Arlit and R. Lange, Catal. Today, 2015, 241, 214–220 CrossRef PubMed , Part B.
- N. M. Islam, M. Chatterjee, Y. Ikushima, T. Yokoyama and H. Kawanami, Int. J. Mol. Sci., 2010, 11, 164–172 CrossRef CAS PubMed.
- T. Noël and V. Hessel, ChemSusChem, 2013, 6, 405–407 CrossRef PubMed.
- M. O'Brien, N. Taylor, A. Polyzos, I. R. Baxendale and S. V. Ley, Chem. Sci., 2011, 2, 1250–1257 RSC.
- D. S. Mannel, S. S. Stahl and T. W. Root, Org. Process Res. Dev., 2014, 18, 1503–1508 CrossRef CAS PubMed.
- N. Straathof, D. Osch, A. Schouten, X. Wang, J. Schouten, V. Hessel and T. Noël, J. Flow Chem., 2014, 4, 12–17 CrossRef CAS.
- H.-S. Phiong, C. G. Cooper, A. A. Adesina and F. P. Lucien, J. Supercrit. Fluids, 2008, 46, 40–46 CrossRef CAS PubMed.
- T. Salmi, D. Y. Murzin, J. Wärnå, P. Mäki-Arvela and G. Martin, Chem. Eng. Sci., 2013, 104, 156–165 CrossRef CAS PubMed.
- F. Delbecq and P. Sautet, J. Catal., 1995, 152, 217–236 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Details of catalyst synthesis and characterisation, and reaction conditions for batch and continuous flow cinnamaldehyde hydrogenation. See DOI: 10.1039/c5ra14984c |
| This journal is © The Royal Society of Chemistry 2015 |




