Silver-mediated thio-acetoxylation and TFA triggered cyclization of amino disulfides with unactivated alkenes: synthesis of 3-aryl/alkyl-1,4-benzothiazines†
Ch. Durga Prasad,
Ajay Verma ,
Moh. Sattar and
Sangit Kumar*
,
Moh. Sattar and
Sangit Kumar*
Department of Chemistry, Indian Institute of Science Education and Research (IISER) Bhopal, Bhopal By-pass Road, Bhauri, Bhopal, Madhya Pradesh, India-462066. E-mail: sangitkumar@iiserb.ac.in
First published on 25th August 2015
Abstract
A convenient strategy for the synthesis of 3-aryl/alkyl-1,4-benzothiazines has been developed. This reaction proceeds via 1,2-thioacetoxylation of an alkene with silver acetate and acetic acid as an additive, followed by cyclization using TFA. The addition of alkene took place in a regioselective manner, forming exclusively one regioisomer, which was confirmed by a single crystal X-ray study. Various differently substituted alkenes, such as aromatic and aliphatic olefins, were coupled with amino disulfides to form a library of benzothiazine derivatives.
Introduction
Organic molecules containing C–S bonds widely exist in many biologically active compounds.1 They are also frequently used as the building blocks of many drugs, functional materials, and metal complexes.2 Although the methods for C–N3 and C–O4 bond formation receive extensive attention in the field of traditional metal-catalyzed cross-coupling reactions, the efficient construction of a C–S bond through transition-metal catalysis still needs more studies. Recently, progress has been made in C–S cross-coupling reactions catalyzed by Pd,5 Cu,6 and Ni,7 but many of these methods depend on harsh reaction conditions, such as a high reaction temperature, the necessity of a strong base, and the use of expensive ligands, and they suffer from limited substrate scope. Very recently, silver-mediated C–S coupling reactions were realized with ketene aminals,8a,b enamides,8c and in a decarboxylative manner with aliphatic carboxylic acids.8d Consequently, it remains important to explore new approaches for the efficient construction of C–S bonds.Phenothiazines are prevalent motifs in various anti-psychotic and anti-histaminic drugs.9 They also act as norA pump inhibitors. In particular, 3-phenyl-1,4-benzothiazine derivatives, synthesized by the structural minimization of phenothiazines, show modest or no intrinsic antistaphylococcal activity and restore the antibacterial activity of CPX against norA-overexpressing Staphylococcus aureus strains.10 Furthermore, these compounds were proved to be useful in the treatment and/or prevention of various disorders such as arteriosclerosis, cardiac arrhythmias, and inflammatory as well as immunological diseases.11 Despite the diverse biological activities of benzothiazine derivatives (Fig. 1), the synthesis of these compounds has rarely been documented in the literature.12
Benzothiazines can be prepared either by the reaction of the corresponding 2-aminothiophenol/its sodium salt with a substituted α-bromo acetophenone (eqn (1), Scheme 1)11 or by the reaction of benzene sulfenyl chlorides with substituted acetophenones to obtain 2-(2-phenylthio)-1-phenylethanones, which then can be reduced with LiAlH4 to afford, after spontaneous cyclization, benzothiazines (eqn (2), Scheme 1).12b
Moreover, TM-catalyzed/mediated C–S and C–N coupling is difficult and has not been addressed, particularly the addition of S and N atoms to a C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond. Based on our interest in synthesizing phenothiazine derivatives, herein we report a silver-mediated synthesis of 3-phenyl-1,4-benzothiazines from unactivated alkenes, which include enough complexity, substrate scope and avoid the use of pre-functionalized substrates such as sulfenyl chlorides and halogenated substrates.
C bond. Based on our interest in synthesizing phenothiazine derivatives, herein we report a silver-mediated synthesis of 3-phenyl-1,4-benzothiazines from unactivated alkenes, which include enough complexity, substrate scope and avoid the use of pre-functionalized substrates such as sulfenyl chlorides and halogenated substrates.
Results and discussion
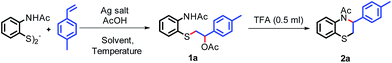
We envisioned that the reaction of 2-acetylaminophenyl disulfide with 4-methyl styrene, using Pd(OAc)2 and AgOAc as an additive, under acidic conditions would result in the benzothiazine derivative 2a via C–N and C–S coupling cascades. Unfortunately, we obtained only the thio-acetoxylated product 1a in 90% yield. However, when a control reaction was performed with AgOAc alone under acidic conditions, it resulted in product 1a in 72% yield (entry 1, Table 1). Interestingly, the addition of 0.5 mL TFA to the thio-acetoxylated compound 1a resulted in intramolecular dehydroacetoxylation at both carbon and nitrogen ends, resulting in 3-tolyl-1,4-benzothiazine (2a) in 63% yield.| Entry | Ag salt | T (°C) | Solvent | Yield of 1ab (%) | Yield of 2ab (%) |
|---|---|---|---|---|---|
| a Reaction was carried out at 0.4 mmol scale using 1 equivalent of 2-acetylaminophenyl disulfide, 3 equivalents of 4-methyl styrene, 1.5 equivalents of silver salt, and 6 equivalents of AcOH in 2 mL of solvent at the mentioned temperature for 6 h, unless otherwise noted; the second step: 0.5 mL of TFA was added at room temperature and stirred for 1 h.b The yields were determined by 1H-NMR analysis of crude products using anisole as the internal standard.c AcOH was not used.d 0.5 equivalents of AgOAc was used.e 1 equivalent of AgOAc was used.f 8 equivalents of AcOH was used.g 20 mol% of AgOAc was used.h Cu(OAc)2 was used. ND = not detected. | |||||
| 1 | AgOAc | 80 | DCE | 72 | 63 |
| 2 | AgNO3 | 80 | DCE | Trace | Trace |
| 3 | AgTFA | 80 | DCE | 39 | 34 |
| 4 | AgOTf | 80 | DCE | Trace | Trace |
| 5 | Ag2CO3 | 80 | DCE | 21 | 18 |
| 6 | AgOAc | 80 | Toluene | 38 | 33 |
| 7 | AgOAc | 80 | MeCN | 48 | 41 |
| 8 | AgOAc | 80 | DMSO | ND | ND |
| 9 | AgOAc | 80 | CHCl3 | Trace | Trace |
| 10 | AgOAc | 80 | THF | Trace | Trace |
| 11 | AgOAc | 100 | DCE | 85 | 73 |
| 12 | AgOAc | 120 | DCE | 80 | 69 |
| 13c | AgOAc | 80 | DCE | 35 | 30 |
| 14 | — | 80 | DCE | ND | ND |
| 15d | AgOAc | 80 | DCE | 16 | 14 |
| 16e | AgOAc | 80 | DCE | 76 | 65 |
| 17f | AgOAc | 80 | DCE | 87 | 75 |
| 18g | AgOAc | 80 | DCE | 5 | Trace |
| 19h | — | 80 | DCE | Trace | Trace |
Encouraged by this result, we varied different parameters for the reaction (Table 1). Various silver salts, such as AgNO3, AgTFA, AgOTf, Ag2CO3 and solvents like toluene, MeCN, DMSO, CHCl3, and THF, were examined to check their viability in the reaction. However, an elevated yield was not obtained (entries 2–5 and 6–10). Performing the reaction at elevated temperatures did not significantly influence the outcome of the reaction (entries 11 and 12). When the reaction was carried out without AcOH as the additive, 35% yield of product 1a was obtained (entry 13). Silver salt is necessary for this transformation, as evident from entry 14. Decreasing the loading of silver salt resulted in lower yields of product 1a (entries 15 and 16). Moreover, on increasing the amount of AcOH, 87% yield of 1a was obtained (entry 17). Furthermore, Cu(OAc)2 could not effectively mediate the thio-acetoxylation reaction (entry 19).
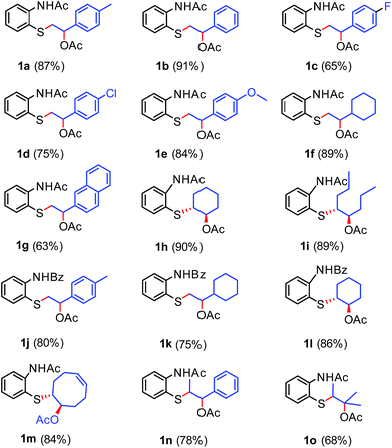
With the optimized reaction conditions in our hand, we then explored the scope of this method with various alkenes. It was found that various aromatic as well as aliphatic alkenes could successfully undergo thio-acetoxylation (Scheme 2), followed by a cyclization reaction to give the desired products (Scheme 3) in modest to good yields.
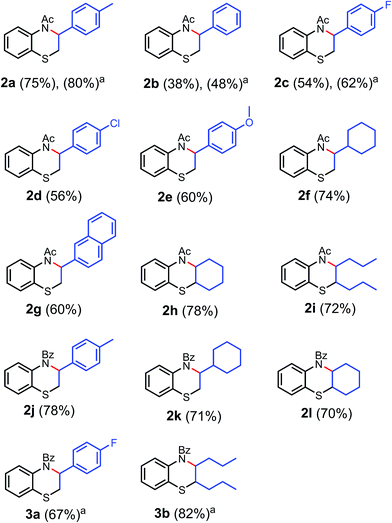 | ||
| Scheme 3 3-Phenyl-1,4-benzothiazine derivatives, yield based on isolated 1. aYield based on disulfide. | ||
The annulation reaction of N-protected aminophenyl disulfide with alkenes proceeded via a thio-acetoxylation reaction succeeded by a cyclization triggered by adding 0.5 mL of TFA. Although most of the thio-acetoxylated products 1a–1o were isolated (Scheme 2), the second step, annulation, can be carried forward without prior purification of the product obtained from first step through column chromatography. The structure of 1h was further confirmed by a single crystal X-ray diffraction study, which reveals an anti-stereochemistry between arylthio and acetoxy groups in 1h (Fig. 2).
First, aromatic alkenes were tested under our reaction conditions (Scheme 3). For example, the reaction of 2-acetylamino phenyl disulfide with methyl styrene gave N-acetyl-2,3-dihydro-3-(p-tolyl)-1,4-benzothiazine in 75% yield (2a), whereas the reaction with styrene gave N-acetyl-2,3-dihydro-3-phenyl-1,4-benzothiazine in 38% yield (2b). The reaction of substituted styrenes, especially with electron-rich substituents, such as methyl, methoxy, and naphthyl groups (2a, 2e and 2g), yielded the desired products in modest to good yields (60–75%), whereas electron-deficient substituents, such as fluoro and chloro groups (2c and 2d), also yielded the products, albeit in moderate amounts (54 and 56%).
Next, the reaction of 2-acetylamino phenyl disulfide with aliphatic alkenes was tested. Interestingly, the reactivity of aliphatic alkenes towards this reaction was higher compared to that of styrenes, which was evident from the yields of the respective products. The reaction of 4-vinyl cyclohexane gave a good yield (74%) of N-acetyl-2,3-dihydro-3-cyclohexyl-1,4-benzothiazine (2f), whereas the reaction of trans-4-octene produced 72% of the corresponding product (2i). To check the versatility of the reaction, cyclohexene, an alicyclic alkene, was treated under the optimized reaction conditions, which indeed resulted in a good yield (78%) of the respective product (2h). 1,5-Cyclooctadiene was also tested as a reactive partner with the disulfide under the same reaction conditions. It gave the thio-acetoxylated product (1m) in 84% yield (Scheme 2); however, the cyclization step resulted in an inseparable mixture (please see ESI, page 31†). The structures of 2a and 2g are confirmed by a single crystal X-ray diffraction study (Fig. 3).
We then explored 2-benzoylamino phenyl disulfide as the coupling partner with various alkenes (Scheme 3). To our delight, this disulfide also reacted smoothly with aromatic and aliphatic as well as alicyclic alkenes, irrespective of their electronic nature under the optimized reaction conditions, to obtain the corresponding benzothiazine derivatives (2j–2l and 3a, 3b) in good yields, as shown in Scheme 3. The reaction of 4-methyl styrene gave a 78% yield of N-benzoyl-2,3-dihydro-3-tolyl-1,4-benzothiazine (2j). 4-Fluoro styrene, an electron poor alkene, reacted smoothly to yield 67% of the respective product 3a, whereas cyclohexene gave 70% of N-benzoyl-2,3-dihydro-3-cyclohexyl-1,4-benzothiazine (2l). Aliphatic alkenes, such as 4-vinyl cyclohexane and trans-4-octene, reacted under the optimized conditions to obtain 71% and 82% yields of the cyclized products 2k and 3b, respectively.
An attempted reaction of 2-aminophenyl disulfide, where the amino group is unprotected, was noticed to be sluggish and the desired product could not be obtained, even in trace amounts. Therefore, it appears that N-protected disulfides are necessary for this transformation.
Mechanistic consideration
A plausible mechanism is depicted in Scheme 4 based on the obtained results. First, 2-acetylamino phenyl disulfide on reaction with styrene and silver acetate forms a sulfenyl intermediate I, which undergoes elimination of silver to form the thio-acetoxylated product 1a. The aryl sulfenyl anion could revert to 2-acetylamino phenyl disulfide. The thio-acetoxylated product on treatment with TFA forms a carbocation II, which undergoes deprotonation with a trifluoroacetate ion to evolve our desired product 2a.Conclusions
We have developed an efficient strategy to obtain the biologically active 3-phenyl benzothiazines using N-protected aminophenyl disulfides and alkenes, which involves thio-acetoxylation followed by a cyclization reaction under acidic conditions. This protocol is a convenient entry towards the synthesis of benzothiazine derivatives, having a wide substrate scope of alkenes. We have used aromatic, aliphatic and alicyclic olefins in this reaction to form 3-phenyl 1,4-benzothiazines. Currently, reactions that involve direct access to phenothiazine are under investigation in our laboratory.Experimental section
General details
All NMR experiments were carried out on 400 or 500 MHz spectrometers in CDCl3 and NMR chemical shifts are reported in ppm, referenced to the solvent peaks of CDCl3 (7.26 ppm for 1H and 77.16 (±0.06) ppm for 13C). Mass analysis is performed on a quadruple-time of flight (Q-TOF) mass spectrometer, equipped with an ESI source (+ve). 2-Aminothiophenol and all alkenes were used as received from Aldrich and Spectrochem India Pvt. Ltd. 2-Aminophenyl disulfide was prepared by oxidation of the respective thiol using tert-butyl hydroperoxide in the presence of a catalytic amount of N,N-dimethylbenzylamine ditelluride. Silica gel (100–200 mesh size) was used for column chromatography. TLC analysis of reaction mixtures was performed using silica gel plates. Synthesis of 2-acetylaminophenyl and 2-benzoylaminophenyl disulfides was carried out using known procedures in the literature.13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 hexane/ethyl acetate to afford 2-((2-acetamidophenyl)thio)-1-(p-tolyl)ethyl acetate, 1a (119 mg, 87% yield).
20 hexane/ethyl acetate to afford 2-((2-acetamidophenyl)thio)-1-(p-tolyl)ethyl acetate, 1a (119 mg, 87% yield).The resulting brown oil was placed in a 10 mL round bottomed flask, charged with 0.5 mL of TFA and stirred for 1 h. After completion of the reaction as analyzed by TLC, the reaction mixture was mixed with ethyl acetate (20 mL). The ethyl acetate layer was washed with a saturated solution of sodium bicarbonate (5 mL × 2). The ethyl acetate layer was dried over anhydrous Na2SO4 and the solvent was evaporated under reduced pressure. The crude product was purified over a column of silica gel and eluted with 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 hexane/ethyl acetate to afford 1-(3-(p-tolyl)-2,3-dihydro-4H-benzo[b][1,4]thiazin-4-yl)ethan-1-one, 2a (49 mg, 75% yield).
10 hexane/ethyl acetate to afford 1-(3-(p-tolyl)-2,3-dihydro-4H-benzo[b][1,4]thiazin-4-yl)ethan-1-one, 2a (49 mg, 75% yield).
Characterization data of the products
Acknowledgements
We are thankful to the Department of Science and Technology (DST), New Delhi, India, Defence Research and Development Organization (DRDO) New Delhi and IISER Bhopal for the financial support. CDP & AV are grateful to UGC New Delhi for fellowship.Notes and references
- (a) G. Liu, J. T. Link, Z. Pei, E. B. Reilly, S. Leitza, B. Nguyen, K. C. Marsh, G. F. Okasinski, T. W. von Geldern, M. Ormes, K. Fowler and M. Gallatin, J. Med. Chem., 2000, 43, 4025 CrossRef CAS PubMed; (b) G. Liu, J. R. Huth, E. T. Olejniczak, R. Mendoza, P. DeVries, S. Leitza, E. B. Reilly, G. F. Okasinski, S. W. Fesik and T. W. von Geldern, J. Med. Chem., 2001, 44, 1202 CrossRef CAS PubMed; (c) M. D. McReynolds, J. M. Dougherty and P. R. Hanson, Chem. Rev., 2004, 104, 2239 CrossRef CAS PubMed; (d) N. D. D'Angelo, T.-S. Kim, K. Andrews, S. K. Booker, S. Caenepeel, K. Chen, D. D'Amico, D. Freeman, J. Jiang, L. Liu, J. D. McCarter, T. S. Miguel, E. L. Mullady, M. Schrag, R. Subramanian, J. Tang, R. C. Wahl, L. Wang, D. A. Whittington, T. Wu, N. Xi, Y. Xu, P. Yakowec, K. Yang, L. P. Zalameda, N. Zhang, P. Hughes and M. H. Norman, J. Med. Chem., 2011, 54, 1789 CrossRef PubMed; (e) X.-B. Xu, J. Liu, J.-J. Zhang, Y.-W. Wang and Y. Peng, Org. Lett., 2013, 15, 550 CrossRef CAS PubMed.
- (a) J. Malmström, M. Jonsson, I. A. Cotgreave, L. Hammars tröm, M. Sjödin and L. Engman, J. Am. Chem. Soc., 2001, 123, 3434 CrossRef PubMed; (b) C. Ericsson and L. Engman, Org. Lett., 2001, 3, 3459 CrossRef CAS PubMed; (c) M. Besev and L. Engman, Org. Lett., 2002, 4, 3023 CrossRef CAS PubMed; (d) H. Yamaguchi and Y. J. Minoura, Appl. Polym. Sci., 2003, 15, 1869 CrossRef PubMed; (e) R. Amorati, G. F. Pedulli, L. Valgimigli, H. Johansson and L. Engman, Org. Lett., 2010, 12, 2326 CrossRef CAS PubMed; (f) S. Johnasson, D. Shanks, L. Engman, R. Amorati, G. F. Pedulli and L. Valgimigli, J. Org. Chem., 2010, 75, 7535 CrossRef PubMed; (g) I. P. Beletskaya and V. P. Ananikov, Chem. Rev., 2011, 111, 1596 CrossRef CAS PubMed.
- (a) S. Guo, B. Qian, Y. Xie, C. Xia and H. Huang, Org. Lett., 2011, 13, 522 CrossRef CAS PubMed; (b) S. E. Creutz, K. J. Lotito, G. C. Fu and J. C. Peters, Science, 2012, 338, 647 CrossRef CAS PubMed; (c) A. C. Bissember, R. J. Lundgren, S. E. Creutz, J. C. Peters and G. C. Fu, Angew. Chem., Int. Ed., 2013, 52, 5129 CrossRef CAS PubMed; (d) K. Shin, J. Ryu and S. Chang, Org. Lett., 2014, 16, 2022 CrossRef CAS PubMed.
- (a) Z. Liang, W. Hou, Y. Du, Y. Zhang, Y. Pan, D. Mao and K. Zhao, Org. Lett., 2009, 11, 4978 CrossRef CAS PubMed; (b) X. Wang, Y. Lu, H.-X. Dai and J. Q. Yu, J. Am. Chem. Soc., 2010, 132, 12203 CrossRef CAS PubMed; (c) B. Xiao, T.-J. Gong, Z.-J. Liu, J.-H. Liu, D.-F. Luo, J. Xu and L. Liu, J. Am. Chem. Soc., 2011, 133, 9250 CrossRef CAS PubMed; (d) A. Modak, D. Dutta, R. Kancherla, R. Maity, M. Bhadra, S. M. Mobin and D. Maiti, Org. Lett., 2014, 16, 2602 CrossRef CAS PubMed.
- (a) T. Itoh and T. Mase, Org. Lett., 2004, 6, 4587 CrossRef CAS PubMed; (b) J.-Y. Lee and P. H. Lee, J. Org. Chem., 2008, 73, 7413 CrossRef CAS PubMed; (c) E. Alvaro and J. F. Hartwi, J. Am. Chem. Soc., 2009, 131, 7858 CrossRef CAS PubMed; (d) N. Taniguchi, Synlett, 2013, 24, 2571 CrossRef CAS.
- (a) N. Taniguchi and T. Onami, J. Org. Chem., 2004, 69, 915 CrossRef CAS PubMed; (b) W. Zhu and D. Ma, J. Org. Chem., 2005, 70, 2696 CrossRef CAS PubMed; (c) N. Taniguchi, Synlett, 2005, 1185 Search PubMed; (d) N. Taniguchi, J. Org. Chem., 2006, 71, 7874 CrossRef CAS PubMed; (e) S. Kumar and L. Engman, J. Org. Chem., 2006, 71, 5400 CrossRef CAS PubMed; (f) N. Taniguchi, J. Org. Chem., 2007, 72, 1241 CrossRef CAS PubMed; (g) J. Gan and D. Ma, Org. Lett., 2009, 11, 2788 CrossRef CAS PubMed; (h) N. Taniguchi, Tetrahedron, 2009, 65, 2782 CrossRef CAS PubMed; (i) Y. Jiang, Y. Qin, S. Xie, V. Zhang, J. Dong and D. Ma, Org. Lett., 2009, 11, 5250 CrossRef CAS PubMed; (j) D. Ma, S. Xie, P. Xue, X. Zhang, J. Dong and Y. Jiang, Angew. Chem., Int. Ed., 2009, 48, 4222 CrossRef CAS PubMed; (k) N. Taniguchi, Eur. J. Org. Chem., 2010, 75, 2670 CrossRef PubMed; (l) N. Taniguchi, Synlett, 2011, 1308 CrossRef CAS; (m) L. Shi, X. Liu, H. Zhang, Y. Jiang and D. Ma, J. Org. Chem., 2011, 76, 4200 CrossRef CAS PubMed; (n) D. Ma, X. Lu, L. Shi, H. Zhang, Y. Jiang and X. Liu, Angew. Chem., Int. Ed., 2011, 50, 1118 CrossRef CAS PubMed; (o) N. Taniguchi, Synlett, 2012, 23, 1245 CrossRef CAS; (p) N. Taniguchi, Tetrahedron, 2014, 70, 1984 CrossRef CAS PubMed.
- (a) H. J. Cristau, B. Chabaud, R. Labaudiniere and H. Christol, J. Org. Chem., 1986, 51, 875 CrossRef CAS; (b) N. Taniguchi, J. Org. Chem., 2004, 69, 6904 CrossRef CAS PubMed; (c) Y. Zhang, K. C. Ngeow and J. Y. Ying, Org. Lett., 2007, 9, 3495 CrossRef CAS PubMed.
- (a) X.-Y. Jiang, Z.-C. Liu, L. Fang, S.-J. Yan and J. Lin, RSC Adv., 2014, 4, 26389 RSC; (b) T.-T. Liu, X.-R. Lin, R. Huang, L.-F. Yang, S.-J. Yan and J. Lin, Tetrahedron, 2014, 70, 8858 CrossRef CAS PubMed; (c) L. Yang, Q. Wen, F. Xiao and G.-J. Deng, Org. Biomol. Chem., 2014, 12, 9519 RSC; (d) P.-F. Wang, X.-Q. Wang, J.-J. Dai, Y.-S. Feng and H.-J. Xu, Org. Lett., 2014, 16, 4586 CrossRef CAS PubMed.
- G. Taurand, Phenothiazine and Derivatives, in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005 Search PubMed.
- S. Sabatini, G. W. Kaatz, G. M. Rossolini, D. Brandini and A. Fravolini, J. Med. Chem., 2008, 51, 4321 CrossRef CAS PubMed.
- H. J. Chao, H. Tuerdi, E. K. Kick and W. Yang, US 2007/0093470 A1, 2007.
- (a) M. Wilhelm and P. Schmidt, J. Heterocycl. Chem., 1969, 6, 635 CrossRef CAS PubMed; (b) N. E. MacKenzie, R. H. Thomson and C. W. Greenhalgh, J. Chem. Soc., Perkin Trans. 1, 1980, 12, 2923 RSC.
- K. M. El-Shaieb, J. Sulfur Chem., 2007, 28, 223 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: 1H, 13C NMR spectra, HRMS data file and X-ray crystallographic data. CCDC 1409837 (1h), 1409829 (2a) and 1409828 (2g). For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c5ra12995h |
| This journal is © The Royal Society of Chemistry 2015 |