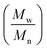Fabrication and properties of hybrid Mo-CPF/P1B from cardanol
Guomei Xuab,
Tiejun Shi*a,
Yu Xianga,
Wei Yuana and
Quan Wanga
aSchool of Chemistry and Chemical Engineering of Hefei University of Technology, Hefei, 230009, People’s Republic of China. E-mail: stjhfut@163.com
bSchool of Materials and Chemical Engineering of West Anhui University, Anhui, Lu’an, 237012, People’s Republic of China
First published on 26th August 2015
Abstract
A Mo-CPF/P1B hybrid has been prepared from molybdenum modified cardanol phenolic resin (Mo-CPF) and cardanol benzoxazine with phosphorus (P1B). Cardanol benzoxazine with phosphorus (P1B) has been synthesized from cardanol-allylamine-based benzoxazine (BZc-a) and DOPO (9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide). Cardanol phenolic resin modified with molybdenum (Mo-CPF) has been synthesized by reacting cardanol-based phenolic resin (CPF) with ammonium molybdate tetrahydrate at 150 °C, where the CPF was synthesized from cardanol and paraformaldehyde in the presence of sodium hydroxide at 65 °C. The synthesized CPF and Mo-CPF were characterized by 1H-NMR and size exclusion chromatography (SEC). Mo-CPF was mixed with P1B, and the mixtures were cured at 110 °C for 4 h and got the Mo-CPF/P1B hybrid. The properties of the Mo-CPF/P1B hybrid were investigated; the results demonstrated that the mechanical and thermal properties, together with flame retardance, were greatly improved. Dynamic Mechanical Analysis (DMA) measurement results indicated that CPF, Mo-CPF and Mo-CPF/P1B could all sustain a large amount of stress and the elongations at break were different. DMA measurements suggested that the Tg of CPF, Mo-CPF and Mo-CPF/P1B was 98 °C, 170 °C and 131 °C respectively. Mo-CPF/P1B exhibited better flame retardance after conjugating with molybdenum and phosphorus. TGA results suggested that Mo-CPF/P1B exhibited better thermal properties. Field emission electron microscopy (FE-SEM) suggested that molybdenum was randomly distributed in Mo-CPF and Mo-CPF/P1B, and EDX indicated that molybdenum and phosphorus were randomly distributed in Mo-CPF/P1B.
1. Introduction
Phenolic resin (PF) was prepared firstly by A. von Baeyer as early as 1872; however, it attracts technical and commercial interests until 1907, which was attributed to Leo H. Baekeland.1 Since then it has played an important role during the process of social development and own century-long history. Nowadays, PF is still very popular and attracts much attention both in experimental and theoretical scientific fields due to its significant advantages,2 such as thermal stability, adhesive properties, and so on. In addition, PF’s properties can be improved by combining it with reinforcing and filling ingredients. For example, Chun-Te Lin et al. reported the preparation of molybdenum/phenolic resin by blending m-AIN with molybdenum/phenolic resin,3 and the mechanical properties of the composite were drastically improved which meant it could be applied as a binder for diamond cutting wheels.Benzoxazine could be normally synthesized from phenol, formaldehyde and primary amines.4 During synthesis, ring-opening polymerization would form a novel phenolic structure in the presence of catalysts or under heating.5 Lin Jin et al.6 have reported that a bis(benzoxazine-maleimide) was synthesized with hydroxyphenylmaleimide, paraformaldehyde and various diamines. 4-Cyanophenol7 was used to synthesize the benzoxazine monomer via Mannich reaction. These materials are all petroleum-based. Long-term use of petroleum-based materials not only constitutes a threat to human health but also aggravates the crises of petroleum. So, considering sustainable development and ecological issues, it is vital to look for and use eco-friendly alternatives, and various alternatives have also been explored in the production of benzoxazine. A typical example is cardanol which is bio-based and requires no consumption of petroleum.8 Cardanol can be obtained from cost-free and sustainable cashew nut shell liquid,9 which is a mixture of non-isoprenoic phenols10 and also contains a meta-substituted long unsaturated alkyl chain.11 Emanuela Calò et al.12 prepared a novel benzoxazine pre-polymer derived from cardanol. Various products have also been prepared from cardanol such as phenolic resin,13 crosslinked polymers,14 and so on.15
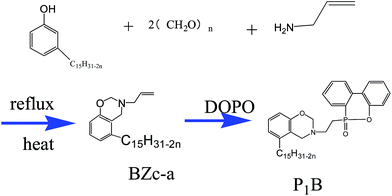
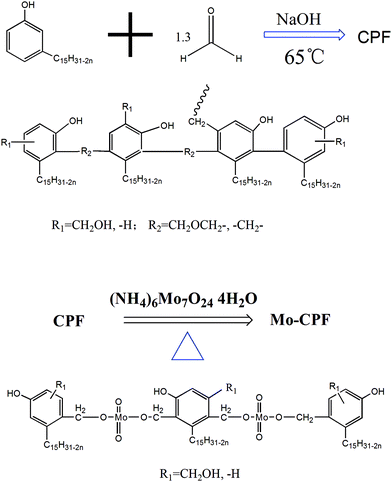
Herein, we propose to prepare a hybrid cardanol-type16 material through reacting PF with benzoxazine, aiming to improve the fragility of benzoxazine with PF (Scheme 1). In addition, ammonium molybdate tetrahydrate was used to modify PF to improve the crosslinking of PF (Scheme 2). Liquid benzoxazine containing DOPO (9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide) is used to endow the hybrid with good flame retardancy.17 The mechanics, flame retardancy and thermal properties of the hybrid material will be investigated.
2. Experimental
2.1 Materials
Cardanol was obtained from Shangdong Haobo Biological Material Co., Ltd (M = 304.52), DOPO and paraformaldehyde were purchased from Shanghai Chemical Reagents Company and used as received, and hexaammonium molybdate tetrahydrate was obtained from Anhui Kehua Fine Chemical Industry Research Institute.A liquid benzoxazine containing DOPO (P1B) was self-prepared; 23 g BZc-a was reacted with 2.8 g DOPO in 200 mL alcohol solution. The reaction continued for 8 h at 65 °C under refluxing in a nitrogen atmosphere, and then stopped reacting and the alcohol was removed under vacuum; a maroon sticky liquid product was isolated. The chemical structure of P1B is shown in Scheme 1 and the other properties are presented elsewhere.18 All other chemicals were used as received.
2.2 Preparation of cardanol-based phenolic resin (CPF)
Typically, 258 g cardanol was placed in a 500 mL three-necked round flask equipped with a mechanical stirrer, thermometer and refluxing condenser. Upon heating up to 65 °C gradually, 32 g of paraformaldehyde was added slowly into the reaction mixture, in the presence of 2 g NaOH. The reaction was conducted under refluxing for six hrs when the solution was very viscous. The synthetic route is shown in Scheme 2. Finally, a maroon sticky product was obtained.142.3 Modification of cardanol-based phenolic resin with molybdenum (Mo-CPF)
The preparation process of Mo-CPF is similar to the synthesis of CPF. Typically, into a three-necked round flask reactor, 100 g CPF and 10 g hexaammonium molybdate tetrahydrate were added and stirred at 150 °C for 4 h. The reaction was stopped and the mixture was washed with water to remove the impurities and side products. Finally, a brown liquid was obtained as the product (Scheme 2).2.4 Preparation of the Mo-CPF/P1B hybrid material
Mo-CPF/P1B was prepared by solution blending. Typically, 20 g Mo-CPF was mixed with 20 g P1B in alcohol at an elevated temperature. Then the mixture was heated up to 80 °C and reacted for 2 h before the final product could be obtained, and ultrasonication was applied to promote the blending rate.Then the blend was heated in a stainless rectangular mold in an air-circulating oven for curing, and then continued to cure at 110 °C for 4 h and the Mo-CPF/P1B hybrid was obtained.
2.5 Measurements
1H-NMR spectra were recorded in DMSO-d6 on a VNMRS600 instrument of 600 MHz (Agilent, America). Size exclusion chromatography (SEC) was conducted on a Waters 515 pump and a Waters 2414 differential refractive index (RI) detector (set at 40 °C) using three linear Styragel HR1, HR2 and HR4 columns. Tetrahydrofuran (THF) was used as the eluent and the flow rate was 0.3 mL min−1. The number-average molecular weight (Mn) and polydispersity (Mw/Mn) data are reported relative to polystyrene standards. Field emission electron microscopy (FE-SEM) and energy-dispersive X-ray spectra (EDX) observations were performed on a FEI Sirion SU8020 system with an energy-dispersive X-ray elemental composition analyzer at an accelerating voltage of 3 kV. Before measurements, the specimens were exposed in a vacuum oven and a thin layer of gold was sputter-sprayed on their surface. Dynamic mechanical analysis (DMA) was performed on a DMA Q800 instrument. The cured specimens (length 8.8 mm, width 4.8 mm and thickness 1.8 mm) were investigated in a tensile deformation mode from 35 to 300 °C at a heating rate of 5 K min−1 and also subjected to multi-frequency strain at room temperature. The burning behaviour by the limited oxygen index was recorded according to the GB/T 2406 standard with a test specimen bar of 90 mm in length, 7 mm in width and about 2.5 mm in thickness, which was conducted on XZT-100A (ChengDe Testing Machine Co., Ltd, China) apparatus for limited oxygen index testing. Each compound was prepared with two test specimens. The flame retardance experiment was conducted by adjusting the proportion of nitrogen and oxygen in a vertical glass bottle.Thermogravimetric Analysis (TGA) was performed on NETZSCH STA 449F3 at a heating rate of 10 K min−1 up to 800 °C in a nitrogen atmosphere. All cured specimens of 5–8 mg were put in a platinum pan. The gas flow rate was 50 mL min−1.
3. Results and discussion
3.1 Synthesis and characterizations
Mo-CPF/P1B has been prepared by blending P1B with Mo-CPF. The structure of CPF and Mo-CPF was confirmed by 1H-NMR19 and SEC. EDX layered images indicated that molybdenum and phosphorus were randomly distributed in Mo-CPF/P1B. Details can be found in the following discussion. Table 1 lists the thermal properties of Mo-CPF/P1B.| Specimens | CPF | Mo-CPF | Mo-CPF/P1B |
|---|---|---|---|
| T5 (°C) | 120 | 306 | 288 |
| T10 (°C) | 306 | 374 | 359 |
| Residues at 800 °C (%) | 15 | 27 | 34 |
| Weight content of P (%) | 0 | 0 | 1.78 |
| Weight content of Mo (%) | 0 | 5.43 | 0.91 |
| LOI (%) | 19.2 | 23 | 26.1 |
| Tg (°C) | 98 | 170 | 131 |
| Flexural modulus (MPa) | 123.7 | 167.7 | 1019 |
CPF is a phenolic resin prepared from cardanol. Fig.1 and 2 show the results of the 1H-NMR spectra of CPF and cardanol. These spectra were all recorded in DMSO, and the chemical shift of DMSO and water was set as 2.46 ppm and 3.3 ppm, respectively. By comparing Fig. 1 and 2, it could be found that there are another three peaks that appeared in the 1H-NMR spectrum of CPF than that of cardanol. Firstly, there is a peak at 3.14 ppm, which corresponds to the proton in the CH2 groups between the two benzenes in CPF. Secondly, four peaks appear among 4.3–4.6 ppm. These are the characteristic protons of active hydroxymethyl or methyl ether groups (–CH2OH– or –CH2OCH2–), which show that the benzene rings in CPF would be connected with the methyl ether bridge or that there exists many active hydroxymethyl groups in CPF. CPF could be soluble in ethanol, so this kind of phenolic resin from cardanol has a low molecular weight. The peaks appearing at 3.14 and among 4.3–4.6 ppm would due to the many oligomers formed and a variety of substituents existed between the benzene rings in CPF. Thirdly, the aromatic protons appear as a multiplet at 7.2, 7.1, and 7.05 ppm, which also demonstrates that many kinds of substituents are formed between the benzene rings. The other characteristic protons of CPF are almost the same as those in the 1H-NMR spectrum of cardanol, except for one peak at 5.97 ppm, which disappeared and showed that one aromatic proton in cardanol reacted and be substituted.18 All these data confirm that CPF was successfully synthesized from cardanol.5
The 1H-NMR spectrum of Mo-CPF is shown in Fig. 3. By comparing Fig. 3 with Fig. 1 and 2, it could be found that three pronounced differences exist in Fig. 3. Firstly, a peak at 3.14 ppm corresponding to the methylene bridge between the benzene rings disappeared in the 1H-NMR spectrum of Mo-CPF, which shows that the methylene bridge broke after CPF was modified by molybdenum, so there was no methylene bridge between the benzene rings in Mo-CPF. Secondly, the peaks at 4.47–4.3 ppm corresponding to the characteristic protons of active hydroxymethyl or methyl ether groups (–CH2OH– or –CH2OCH2–) also appear in Fig. 3, however, the intensity and quantity of the peaks all decreased, which confirms that the –Mo–O–CH2– group was generated between the benzene rings after CPF was modified by molybdenum. Thirdly, a peak at 9.15 ppm appeared, which would be due to the characteristic protons of active phenolic protons of –OH, which are not found in the 1H-NMR spectrum of CPF.
The SEC chromatograms of CPF and Mo-CPF are presented in Fig. 4 and 5, which both indicate the formation of a broad dispersed polymer. However, Fig. 4 exhibits that the molecular distribution of CPF is too broad to predict the molecular weight, the oligomers elute at about 27 minutes, then the monomer of CPF elutes after 30 minutes. Fig. 5 shows the part of the molecular distribution of Mo-CPF that could be calculated, and the values of the number-average molecular weight (Mn), the weight-average molecular weight (Mw) and the molecular weight distribution (MWD = Mw/Mn) of Mo-CPF are given in Table 2. Long chain alkyl resins exhibit in general low Mn, Mw, and Mw/Mn values. According to all these data, the structure of CPF and Mo-CPF is given in Scheme 2.
In order to confirm that the element distribution on the surface was changed, energy-dispersive X-ray spectra (EDX) of Mo-CPF and Mo-CPF/P1B were examined and the result is shown in Fig. 6 and 7.3 The images display that there are carbon, oxygen and molybdenum elements existing on the surface of Mo-CPF, and the green spots on the top of Fig. 6 is the molybdenum element which distributes uniformly in Mo-CPF. At the bottom of Fig. 6 is the mapping of each element, and the mapping of the carbon element is referred to as a controlled image. Comparing the mapping of the molybdenum element with that of carbon, it can be seen that the molybdenum is dispersed in the CPF matrix uniformly and exhibits a tougher surface.
The energy-dispersive X-ray spectra (EDX) of Mo-CPF/P1B are shown in Fig. 7. By comparing the images, not only carbon, oxygen and molybdenum elements exist on the surface of Mo-CPF/P1B, but also the phosphorus element exists in Mo-CPF/P1B. At the bottom of Fig. 7 is the mapping of each element, and the mapping of the carbon element is referred to as a controlled image. It could be found that molybdenum and phosphorus are dispersed in composite uniformly. The irregular granules are the molybdenum element.
Field emission electron microscopy (FE-SEM) was applied to study the surface morphologies of cured CPF, Mo-CPF and Mo-CPF/P1B, and typical images are shown in Fig. 8–10. Compared with the relatively smooth surface of CPF and Mo-CPF, the surface of Mo-CPF/P1B is much more complicated. CPF shows a relatively smooth glassy fracture surface with cracks in different cross-sections, and this suggested that the phenolic resin is brittle.20
Cured Mo-CPF showed many spherical-like protrusions on the surface (Fig. 9) and these particles may be molybdenum particles.21 The surface was tougher than CPF, suggesting the successful modification of hexaammonium molybdate tetrahydrate. The surface of Mo-CPF/P1B was much more complicated: more wrinkles and particles appeared. The compatibility between Mo and P with CPF was very good after the copolymerization, as no phase separation happened (refer to the high magnification image, Fig. 10b). It seemed that the similar alkyl chain of Mo-CPF and P1B contributed to the improvement of compatibility of the final product.
3.2 Properties study
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ curve of the cured CPF, Mo-CPF and Mo-CPF/P1B films has one peak centered at about 98, 170 and 131 °C, respectively. The cured Mo-CPF and Mo-CPF/P1B showed a more drastic increase in their Tg than neat CPF did, which might be due to the incorporation of molybdenum resulting in the formation of a new network linked by –Mo–O bonds.23 When introducing phosphorus into Mo-CPF/P1B, the incorporation of P1B decreased the density of the link –Mo–O group, which resulted in the decrease of the Tg.24
δ curve of the cured CPF, Mo-CPF and Mo-CPF/P1B films has one peak centered at about 98, 170 and 131 °C, respectively. The cured Mo-CPF and Mo-CPF/P1B showed a more drastic increase in their Tg than neat CPF did, which might be due to the incorporation of molybdenum resulting in the formation of a new network linked by –Mo–O bonds.23 When introducing phosphorus into Mo-CPF/P1B, the incorporation of P1B decreased the density of the link –Mo–O group, which resulted in the decrease of the Tg.24
Our measurements indicated that the cured CPF resin had a broad Tg transition area ranging from 98 °C to 200 °C, which may be due to the complicated structures formed after the condensation between functional groups such as CH2OH or –OH in CPF. However, the cured Mo-CPF/P1B resin showed a step change of the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ curve after the temperature increased to 200 °C. As the glass transition is normally affected by the molecular mobility of the material’s cooperative units, in our system, the frequency of the cooperative rearrangement of copolymerized Mo-CPF and P1B could correspond to the measurement frequency, resulting in a step. The present results indicated that the Tg could be manipulated to a high temperature by introducing molybdenum in CPF.25
δ curve after the temperature increased to 200 °C. As the glass transition is normally affected by the molecular mobility of the material’s cooperative units, in our system, the frequency of the cooperative rearrangement of copolymerized Mo-CPF and P1B could correspond to the measurement frequency, resulting in a step. The present results indicated that the Tg could be manipulated to a high temperature by introducing molybdenum in CPF.25
The cured specimens (length 17.5 mm, width 10.5 mm and thickness 3.92 mm) were investigated with controlled force DMA in a single cantilever mode at room temperature. The stress–strain curves of CPF, Mo-CPF and Mo-CPF/P1B are plotted in Fig. 12. The bottom curve with dot signs is the stress–strain curve for cured CPF, which is a typical stress–strain curve for a phenolic resin and the curve is close to linear. The middle curve with dot signs is the stress–strain curve for cured Mo-CPF. The top curve with triangle signs is the stress–strain curve for cured Mo-CPF/P1B. As shown in Fig. 12, all resins exhibited a plastic behaviour. For comparison, Mo-CPF/P1B showed the best mechanical properties, highest flexural modulus with 1019 MPa compared to the others (123.7 MPa for CPF and 167.7 MPa for Mo-CPF), which may be due to the lightly crosslinked network formed after conjugating CPF with Mo and P. Therefore the mechanical properties could be raised by treatment with molybdenum and phosphorus.26
The bottom curve is the thermogravimetric curve of CPF, which shows two mass loss steps. The main decomposition stage happened at 300–500 °C, and there was around 15% mass residual finally. After the reaction between hexaammonium molybdate tetrahydrate and CPF, which linked the CPF polymer chain through –Mo–O– bonds, Mo-CPF showed better thermal stability: 27% mass residual finally. The main loss of Mo-CPF/P1B happened at the temperature range of 400 °C to 482 °C and 34% mass residue was observed finally with a significant difference from CPF and Mo-CPF. Significant differences in the mass residue suggested that the incorporation of a little amount of molybdenum and phosphorus into Mo-CPF/P1B not only formed a structure linked with a lot of –Mo–O– and –O– groups between composites, but also meant that P1B could release phosphorus, which then worked in the gas phase and blocked the rate of decomposition. So the thermal stability of Mo-CPF/P1B was further improved. Details can be found in Table 1.
4. Conclusions
A Mo-CPF/P1B hybrid with raw materials prepared from cardanol has been synthesized successfully. FESEM images suggested that the surface of the hybrid was much tougher than CPF and Mo-CPF. DMA, flame retardancy and TGA measurements all revealed that the Mo-CPF/P1B hybrid exhibited improved mechanical properties, perfect performance of flame retardancy and good thermal stability, which confirmed that Mo-CPF and P1B reacted and formed a crosslinked structure during hybridization. The DMA study indicated that the Tg of the hybrid is 131 °C which is different from that of CPF and Mo-CPF, and the flexural modulus is 1019 MPa. The flame retardant property is also perfect and the LOI value could reach 26.1. It had better thermal stability in air and 34% char yield at 800 °C.Acknowledgements
Authors acknowledge the support from the National Natural Science Foundation of China (grant no. 51273054); authors also thank the kind help from Science and Technology Project of Anhui Province (grant no. 1206c0805031 and 1406c085021); China and Engineering Technology Center of Fine Chemicals Engineering of AnHui, China; authors also thank the kind help from professor Tao He of Hefei University of Technology.References
- P. W. Kopf and A. D. Little, Phenolic Resins in ECT (online), in: Kirk-Othmer Encyclopedia of Chemical Technology, John Wiley & Sons, Inc., 2000 Search PubMed.
- G. Rivero, S. Villanueva and L. B. Manfredi, Fire Mater., 2014, 6, 683–694 CrossRef PubMed.
- C.-T. Lin, H.-T. Lee and J.-K. Chen, Appl. Surf. Sci., 2013, 297–307 CrossRef PubMed.
- B. Lochab, I. K. Varma and J. Bijwe, J. Therm. Anal. Calorim., 2012, 2, 1357–1364 Search PubMed.
- T. Periyasamy, S. Asrafali and S. Muthusamy, New J. Chem., 2014 10.1039/c4nj02047b.
- L. Jin, T. Agag and H. Ishida, Eur. Polym. J., 2010, 2, 354–363 CrossRef PubMed.
- D. Wang, B. Li, Y. Zhang and Z. Lu, J. Appl. Polym. Sci., 2013, 516–522 CrossRef CAS PubMed.
- B. Lochab, I. K. Varma and J. Bijwe, J. Therm. Anal. Calorim., 2011, 2, 661–668 Search PubMed.
- D. Balgude and A. S. Sabnis, J. Coat. Technol. Res., 2013, 2, 169–183 Search PubMed.
- C. Voirin, S. Caillol, N. V. Sadavarte, B. V. Tawade, B. Boutevin and P. P. Wadgaonkar, Polym. Chem., 2014, 9, 3142–3162 RSC.
- V. S. Balachandran, S. R. Jadhav, P. K. Vemula and G. John, Chem. Soc. Rev., 2013, 2, 427–438 RSC.
- E. Calò, A. Maffezzoli, G. Mele, F. Martina, S. E. Mazzetto, A. Tarzia and C. Stifani, Green Chem., 2007, 7, 754 RSC.
- S. K. Shukla, A. Maithani and D. Srivastava, Int. J. Chem. Kinet., 2013, 7, 469–476 CrossRef PubMed.
- W. Bai, X. Xiao, Q. Chen, Y. Xu, S. Zheng and J. Lin, Prog. Org. Coat., 2012, 3, 184–189 CrossRef PubMed.
- G. Vasapollo, G. Mele and R. Del Sole, Molecules, 2011, 12, 6871–6882 CrossRef PubMed.
- M. Kathalewar and A. Sabnis, J. Coat. Technol. Res., 2014, 4, 601–618 CrossRef.
- Y. Ma, J. Wang, Y. Xu, C. Wang and F. Chu, J. Therm. Anal. Calorim., 2013, 3, 1143–1151 CrossRef.
- G. Xu, T. Shi, Q. Wang, J. Liu and Y. Yi, J. Appl. Polym. Sci., 2015, 11, 3534–3545 Search PubMed.
- C. Li, H. Fan, D.-Y. Wang, J. Hu, J. Wan and B. Li, Compos. Sci. Technol., 2013, 189–195 CrossRef CAS PubMed.
- K. Huang, Y. Zhang, M. Li, J. Lian, X. Yang and J. Xia, Prog. Org. Coat., 2012, 74, 240–247 CrossRef CAS PubMed.
- P. Zhao, Q. Zhou, Y. Y. Deng, R. Q. Zhu and Y. Gu, RSC Adv., 2014, 4, 61634–61642 RSC.
- F. Taheri-Behrooz, B. Memar Maher and M. M. Shokrieh, Comput. Mater. Sci., 2015, 411–415 CrossRef CAS PubMed.
- Z. Bu, J. Hu and B. Li, Thermochim. Acta, 2014, 244–253 CrossRef CAS PubMed.
- J. Zhang, C. Zhu, P. Geng, Y. Wei and Z. Lu, J. Appl. Polym. Sci., 2014, 131, 41072–41079 Search PubMed.
- Z. Chen, B. J. Chisholm, D. C. Webster, Y. Zhang and S. Patel, Prog. Org. Coat., 2009, 2, 246–250 CrossRef PubMed.
- E. F. Assanvo, P. Gogoi, S. K. Dolui and S. D. Baruah, Ind. Crops Prod., 2015, 293–302 CrossRef CAS PubMed.
- H. Yu, J. Liu, X. Wen, Z. Jiang, Y. Wang, L. Wang, J. Zheng, S. Fu and T. Tang, Polymer, 2011, 21, 4891–4898 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2015 |