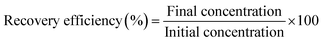Hollow fiber concentrator for water quality monitoring: role of surfactant based elution fluids
Saumyadeb Dasgupta,
Ravi Chavali,
Naga Siva Kumar Gunda and
Sushanta K. Mitra*
Department of Mechanical Engineering, Lassonde School of Engineering, York University, Toronto, Ontario M3J 1P3, Canada. E-mail: mitras@yorku.ca; Tel: + 1 416 736 5924
First published on 15th July 2015
Abstract
Simple and efficient concentrators are indispensable to the development of portable biosensor based detection platforms for the determination of microbial contamination in water. This requires the design of a concentrator which could operate for large liquid volume yet maintain the desired microbial concentration in the resultant concentrate. In the present study, we have utilized a commercially available hollow fiber filter modules to test for the concentration and recovery of Escherichia coli (E. coli) from water with the aid of a combination of surfactants and a chemical dispersant as the elution fluid. The process involves the tangential flow filtration of contaminated water through the hollow fiber filter to reduce the sample volume, followed by an elution step to facilitate the transfer of the concentrated mixture onto a portable hand-held water monitoring device. The effect of using two non-ionic surfactants (Tween-20 and Tween-80) and a chemical dispersant, sodium hexametaphosphate (SHMP), as elution fluids have been discussed in the present work. The lowest recovery efficiencies were associated with the case of concentration without elution. Recovery efficiencies using a mixture of SHMP and surfactants (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio) were found to be greater than when the surfactants were used individually as the elution fluid. A mixture of Tween 20 and Tween 80 (1
1 ratio) were found to be greater than when the surfactants were used individually as the elution fluid. A mixture of Tween 20 and Tween 80 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio) was even more efficient with greater than 80% recovery being observed. The best results were obtained using the elution fluid made up of a combination of Tween 20, Tween 80 and SHMP (1
1 ratio) was even more efficient with greater than 80% recovery being observed. The best results were obtained using the elution fluid made up of a combination of Tween 20, Tween 80 and SHMP (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio) with greater than 90% efficiency of bacteria recovery associated with it. Recovery efficiencies increased by 25% using this combination as elution fluid as compared to the case where no elution fluid was used.
1 ratio) with greater than 90% efficiency of bacteria recovery associated with it. Recovery efficiencies increased by 25% using this combination as elution fluid as compared to the case where no elution fluid was used.
1 Introduction
Microbial contamination of drinking water sources is one of the leading causes of waterborne diseases affecting people all over the world. Ingestion of water from polluted sources results in the outbreak of enteric diseases and poses a major public health concern. Even in low concentrations, the pathogens in water are detrimental to the well-being of the human population. Hence, there is an urgent need for developing efficient water monitoring systems that will improve global living standards. Conventional methods of pathogen detection1–4 are efficient and provide an accurate estimation of contaminants in water. However, these methods are limited by prolonged detection times (24 to 48 hours), requirement of trained personnel, along with expensive laboratory setup which hinder the effective monitoring of water quality at the point of use. A practical approach towards overcoming these shortcomings is the use of miniaturized systems such as microfluidics based detection platforms. Microfluidics based detection methods5,6 are rapid, portable and highly sensitive with the added advantage of being simple and cost-effective. However, the use of these devices is constrained by their inability to handle large sample volumes which is essential for the accurate estimation of microbial contaminants. Often, these microfluidics or miniaturized devices are able to handle only μL or picoliters of sample (water), which may not have enough pathogens (like Escherichia coli) to make it a meaningful water quality monitoring system. Thus, an important challenge lies in developing an efficient concentrator capable of reducing the sample volume to a few hundred microliters yet retaining viable concentration of target pathogens in water to accommodate the miniaturized detection platforms.Several concentration techniques like centrifugation,7,8 immunomagnetic separation,9,10 on-chip microfluidics based pre-concentration11,12 and membrane filtration13,14are available in literature. Concentration of microorganisms using centrifugation is not recommended due to the possibility of cell structure damage15 which would cause significant errors in their detection. Immunomagnetic separation is a very expensive as well as a complex process and hence, is not suitable for on-field applications. Among the other two concentration procedures, separation techniques using microfluidic channels like hydrodynamic filtration using pinched flow fractionation,11,16 ratchet based filtration using funnel shaped microfluidic ratchets,17 lateral displacement array based separation18,19 and electro-kinetic separation using electrophoresis12,20,21and di-electrophoresis22,23 have been found to be continuous, quick and efficient for the concentration of microorganisms. Nonetheless, such on-chip pre-concentrators and micro-mixers are still inadequate to handle large sample volumes to ensure proper estimation of microbial contamination.
The problem of large sample volume can be effectively addressed by the use of membrane filtration techniques. Membrane filters are polymer films with specific pore ratings. They retain particles and microorganisms larger than their pore sizes on the membrane surface by acting as a physical barrier. This filtration technique is efficient and finds its use in most conventional methods of pathogen detection from contaminated water.24,25 Many commercially available bacterial detection kits like Colilert®, Colibag® and others employ the membrane filtration technique for the concentration of microorganisms.3,26,27 Syringe filters are capable of concentrating pathogens from large volumes of water onto the filter surface but the transfer of the microorganisms from the filter surface to the microfluidics based platform poses a considerable challenge. Concentration procedure using membrane filtration is limited by filter fouling problems. Additionally, the entrapment of microorganisms within the filter pores poses a further challenge in their recovery and consequent detection. However, using a different configuration of the membrane filter alleviates the difficulty of such a transfer. One such configuration of membrane filters is the hollow fiber filter which has a very good potential to be effective as a field-deployable concentrating device for monitoring water quality.
Hollow fiber microfiltration and ultrafiltration techniques have gained popularity since the early 1970s when polio-I virus samples were first concentrated from water using this technique.28 Hollow fibers, as the name suggests, consist of a number of hollow cylindrical membranes, the walls of which are permeable to water but not to particles larger than the specified pore size. A hollow fiber filter module consists of a large number of these fibers bundled together, thereby increasing the effective surface area available for filtration. The modules usually consist of two end-ports for the sample input and a side port to collect the permeate as the sample passes through the filter. Ultrafiltration techniques have been used for the concentration and recovery of a wide range of microorganisms29–34 from a variety of water sources.35–37 These studies focus on the use of hollow fiber filters based on the tangential flow filtration process where the sample water is pumped tangentially across the hollow fibers through the end ports and recirculated under a set pressure until the desired volume of the concentrated mixture (retentate) is obtained. Size exclusion retains contaminants of sizes larger than the pore rating within the retentate which is consequently recovered. The hollow fibers can be used in a dead-end configuration as well, by closing one of the end-ports. The sample is forced to flow radially to the membrane surface resulting in the exit of the filtrate through the side port and the entrapment of the contaminants on the inner surface of the filter. Recovery of the contaminants in this case, is facilitated by a backwash step.38 Despite dead-end filtration being a clearer choice regarding process simplicity, there is an associated disadvantage of membrane fouling and reduced flow rate. The choice of the tangential flow configuration becomes appropriate when a subsequent elution step with suitable elution fluid leads to a significant increase in recovery efficiencies.39,40
The effect of non-ionic surfactants on the adhesion of bacteria to solid surfaces is well documented.41 Most of the linear ethoxylated surfactants have an anti-adhesive effect which helps to prevent bacterial attachment to surfaces. This property has been used to increase bacteria recovery efficiencies during hollow fiber concentration process.39 Surfactants like Tween 20 and Tween 80 reduce the hydrophobic interactions between microbes and filter surfaces. The surfactant molecules orient themselves on the filter surfaces and help to desorb microorganisms adhered to the surfaces.40,42
In our present study, we investigated the effects of a combination of surfactants (Tween-20 and Tween-80) and a chemical dispersant (SHMP) as elution fluid on the concentration and recovery efficiencies of bacteria using hollow fiber filter modules of different pore sizes. Initially, polystyrene latex particles were concentrated using this technique to obtain an estimate of the working conditions. Thereafter we investigated the effectiveness of two different hollow fibers under the same set of operating conditions for the recovery of E. coli from water using syringe operated tangential flow filtration. Finally we explored the effect of the different combination of surfactants on the recovery efficiencies using a wide range of bacteria test concentrations which will help to improve this concentrating technique. It is to be pointed out that one of our goals were to establish that one can directly use the commercial hollow fiber filters and an efficient recovery process can be governed solely by the introduction of elution fluids without pre-treating the hollow fiber surface, as carried out in the literature.39,40
2 Experimental section
2.1 Materials
Ultrapure water was obtained from Barnstead™ Nanopure™ water purifier (model no. D11971, Thermo Scientific, Waltham, Massachusetts). Blue polystyrene latex particles, 1 μm in diameter (catalogue no. PSB3788-0813) were procured from Magsphere Inc., Pasadena, CA. E. coli K-12 strain was obtained from New England Biolabs, Ipswich, Massachusetts, USA. Lauryl Tryptose Broth (catalogue no. DF0241-15-2) used as the medium for growing E. coli was obtained from Fisher Scientific, Canada. The concentration procedure was performed using two separate polyethersulfone membrane hollow fiber filter modules (MicroKros, C06-P20U-10-S) of 0.2 μm pore size and one modified polyethersulfone membrane hollow fiber module (MicroKros, C02-E750-10-N) of 750 kD pore size obtained from Spectrum Laboratories Inc., Compton, CA. The total surface area of the hollow fiber filters are 0.0041 m2 with the inner and outer diameters being 1 mm and 1.25 mm, respectively, as shown in Fig. 1. The concentration and recovery procedures were performed using four plastic, sterile 5 mL syringes with luer-lock tips (BD309695, BD Medicals, New Jersey, USA). Different combinations of non-ionic surfactants Tween 20 (catalogue no. P1379) and Tween 80 (catalogue no. P1754) and a chemical dispersant sodium hexametaphosphate (catalogue no. 305553) were used as the elution fluid. All the chemicals were obtained from Sigma Aldrich, Canada. A 60 mL syringe (BD309653, BD Medicals, New Jersey, USA) was used for the filter cleaning purposes in between experiments.2.2 Equipment
Absorbance values were measured using a spectrophotometer (Varian Cary 100 Bio, Agilent Technologies, Santa Clara, CA). Scanning Electron Microscopy (Quanta 3D FEG, FEI, Hillsboro, Oregon, USA) (SEM) was used to image the hollow fiber cross sections. Bacterial growth reagents were autoclaved (Primus Sterilizer Co., NE, USA) whenever required.2.3 Preparation of latex solutions
One milliliter of 10% latex particle solution was mixed with 40 mL of water and the solution was serially diluted to prepare different concentrations (expressed in particles per mL units) used in this study. Corresponding absorbance values were obtained from spectrophotometric readings taken at 570 nm and used to prepare the calibration curve.2.4 Preparation of bacteria concentrations
One hundred milliliters of Lauryl Tryptose Broth was prepared and autoclaved to be used as the growth medium. E. coli was grown in this media by incubating for 24 hours at 37 °C. Thereafter, bacteria solutions were prepared using the serial dilution technique. The resulting concentrations were determined using the plate counting method and expressed in Colony Forming Units (CFU) per mL. Absorbance values recorded at 600 nm were used to prepare the corresponding calibration curve.2.5 Experimental procedure
Prior to the start of the experiments the filter was rinsed with de-ionized water using the 60 mL syringe. The Concentration and recovery procedures were performed by attaching three 5 mL syringes to the hollow fiber module and closing one of the side ports (permeate ports). The attachments and step by step procedure are shown in Fig. 2. Sample syringe 1 (SS 1), containing the sample was attached to one of the end ports. The sample was subjected to a couple of passes through the hollow fibers until the final volume of approximately 0.5 mL (retentate without elution) was obtained in sample syringe 2 (SS 2) attached to the other end port of the module. The permeate was collected in a permeate syringe (PS) attached to the open permeate port. Four different combinations of the surfactants (0.01% Tween 20, vol/vol and 0.1% Tween 80, vol/vol) and chemical dispersant (0.01% SHMP, by weight) were used as the elution fluid. 0.5 mL of elution fluid was administered through the fibers using an elution fluid syringe attached to the end port by replacing SS 1. The elution fluid, after passing through the fiber lumen, was collected in SS 2, increasing the final volume to approximately 1 mL (retentate with elution). Similar protocols were followed for experiments with both bacteria and latex particles.For each of the test concentrations, several replicates were performed. Moreover, to maintain uniformity in measurements, the final concentrated volumes were increased to 5 mL by adding water and the corresponding absorbance values were recorded. A cleaning protocol, involving the flushing of the filter first with water and then by air, using a 60 mL syringe, was undertaken at the end of each experiment. Recovery efficiencies were calculated by dividing the final concentration by the initial concentration and multiplying the result by 100 to report the recovery in percentage.
To investigate filter surface characteristics and particle entrapment within the surface pore structures, a concentration procedure was undertaken without elution, using a latex particle solution. Once the final concentrate was obtained, the polypropylene housing was broken to remove the hollow fibers from the module. Small sections of the used hollow fibers were used to conduct Scanning Electron Microscopy (SEM) imaging studies. The samples were sputter-coated with gold and placed inside the base of the scanning electron microscope. Images were taken at high vacuum and high voltage (20 kV) with the beam current set at 7.5 pA and dwell time of 1 μs.
3 Results
3.1 Concentration and recovery of polystyrene latex particles in solution
Polystyrene latex microspheres were used as test particles to estimate the experimental conditions for the subsequent experiments with E. coli. The calibration curve, shown in Fig. 3, was used to calculate the final concentrations after the recovery experiments.The recovery efficiencies for latex particles with and without elution are shown in Fig. 4. The results indicate a distinct trend of increasing efficiency with the aid of elution using Tween 20 (0.01%, vol/vol) for all of the test concentrations. However, a steep drop in recovery efficiency is observed for the solution with a concentration of 4.5 × 106 particles per mL. Overall, the data is not very promising with only a single case portraying greater than 50% recovery with the aid of the elution step. On the other hand, these experiments provided an estimate of the working conditions like the number of passes to reach desired volume, time taken for each experiment and the optimum filter cleaning procedure, thereby supplying the necessary framework for the bacteria recovery experiments.
3.2 Concentration and recovery of E. coli (K-12 strain)
Experiments dealing with concentration and recovery of E. coli from water were conducted using filters of two different pore sizes of 0.2 μm and 750 kD. The calibration curve used for all relevant calculations regarding bacteria concentrations is shown in Fig. 5. The performances of the two filters under the same set of operating conditions were evaluated, as shown in Fig. 6. High initial sample concentrations, ranging between 107 and 108 CFU per mL, were used. Tween 20 (0.01%, vol/vol) was used as the elution fluid for these set of experiments. | ||
| Fig. 5 Calibration curve for bacteria (E. coli) concentrations in water. Correlation coefficient is 0.94365. Error bars indicate the standard errors obtained from ten replicate experiments. | ||
The filter with the lower pore size (750 kD) was found to be much more efficient as compared to the other (0.2 μm pore size) one. The results shown in Fig. 6 indicate an increase in recovery of nearly 20% for the 750 kD pore sized filter when no elution fluid was used. The introduction of the elution step resulted in significant improvements in recovery efficiencies for both the filters. In this case the 0.2 μm filter was associated with efficiencies greater than 80% while the 750 kD filter was more efficient and showed more than 90% recovery for every experiment performed in our study. This increased recovery of the 750 kD pore size filter can be attributed to the lower anisotropy (asymmetry) and pore size of the modified polyethersulfone membrane. Hence, along with the type of elution fluid, these parameters play a prominent role in the concentration and recovery of bacteria (E. coli) using hollow fiber filtration technique.
The effectiveness of different elution fluids on the recovery efficiency was investigated using the 750 kD pore sized filter. Sample concentrations ranging from 102 to 105 CFU per mL were used in this assessment. The calibration curve shown in Fig. 5 was used for all relevant calculations regarding bacteria concentrations. The corresponding experimental results are shown in Fig. 7. Among the individual surfactants, Tween 80 (0.1%, vol/vol) was found to be a better alternative to Tween 20 (0.01%, vol/vol) as an elution fluid. The recovery levels increased when each of the two surfactants were combined with SHMP (0.01%, by weight) in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio to constitute the elution fluid. Even higher recovery efficiencies were obtained with a mixture of Tween 20 and Tween 80 (in a 1
1 ratio to constitute the elution fluid. Even higher recovery efficiencies were obtained with a mixture of Tween 20 and Tween 80 (in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio) as the elution fluid, exhibiting greater than 85% recovery for all the test concentrations. However, the best elution fluid for concentration and recovery purposes was found to be a mixture of Tween 20, Tween 80 and SHMP (in a 1
1 ratio) as the elution fluid, exhibiting greater than 85% recovery for all the test concentrations. However, the best elution fluid for concentration and recovery purposes was found to be a mixture of Tween 20, Tween 80 and SHMP (in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio), yielding more than 90% recovery of E. coli from water. The recovery efficiency corresponding to 105 CFU per mL was found to be 98% using this mixture as the elution fluid. Recovery efficiencies were greater by 25% using this combination when compared to the case without the elution step.
1 ratio), yielding more than 90% recovery of E. coli from water. The recovery efficiency corresponding to 105 CFU per mL was found to be 98% using this mixture as the elution fluid. Recovery efficiencies were greater by 25% using this combination when compared to the case without the elution step.
4 Discussions
4.1 Hollow fiber SEM image analysis
The low recovery values observed for the latex particles in the concentration procedure indicate a distinct anisotropic nature of the 0.2 μm pore size hollow fiber membrane with most of the particles being captured on the inner surface pores. SEM images presented in Fig. 8 show the different cross sections of the hollow fiber after a concentration procedure using latex particles. Fig. 8A shows that particles were not only entrapped at the surface pores but rather a considerable number of these were embedded within the inner pore structures. There is a gradual increase in pore size from the outer to the inner wall as can be observed in Fig. 8C. This points out to the fact that inner surface pore sizes are larger than the specified 0.2 μm. Some surface pore sizes even exceed 1 μm resulting in the embedding of latex particles within the inner pore structures. The increase in recovery efficiency due to elution using Tween 20 (0.01%, vol/vol) can be attributed to the scouring effect of the surfactant which resulted in the recovery of the particles stuck on the filter surface. However, the elution fluid was unable to recover the particles embedded within the inner pore structure of the hollow fiber membrane. The embedded particles can be dislodged with the introduction of a subsequent back-flushing step. Fig. 8B shows the absence of any latex particle on the outer wall of the hollow fiber. Hence, we conclude that the low recovery of latex particles using this procedure is the result of the inherent asymmetry of the (0.2 μm pore size) polyethersulfone membrane.4.2 Analysis of bacteria recovery: role of surfactants
A comparison of the recovery rate obtained in this study is presented in Table 1 with some of the relevant studies in this field. The high recovery rates were obtained without any prior filter treatment or sample amendment techniques, thereby reducing the complexity and duration of the procedure.| Paper | Operating conditions | Tested microbes | Highest recorded recovery efficiency (%) | |||||
|---|---|---|---|---|---|---|---|---|
| Filter pre-treatment | Sample amendment | Backflushing/elution | Backflushing/elution agent | Bacteria | Virus | Protozoa | ||
| V. R. Hill et al., 2005 (ref. 40) | 5% FBS (Fetal Bovine Serum) and NaPP (0.1% and 0.01%) | NaPP (0.1% and 0.01%) and Tween 80 (0.01% and 0.002%) | Backflushing | Tween 80, Tween 20, NaPP | MS2, Echovirus, Salmonella E. faecalis, B. globigii, C. parvum, E. coli | 98 ± 8 (E. coli) | 97 ± 58 (Echovirus) | 98 ± 17 (C. parvum) |
| H. A. Morales-Morales et al., 2003 (ref. 39) | 5% FBS 1× PBS, 0.1% Tween 80 | — | — | — | E. coli, C. parvum, T1 phage, PP7 phage | 95 ± 7.8 (E. coli) | 62.8 ± 5.8 (PP7 phage) | 31.6 ± 11.9 (C. parvum) |
| Smith and Hill, 2009 (ref. 38) | — | — | Backflushing (dead end filtration) | Tween 80, NaPP, Y-30 antifoam | E. faecalis, C. perfringens, MS2, C. parvum | 94 ± 22 (C. perfringens) | 82 ± 14 (MS2) | 87 ± 18 (C. parvum) |
| Kuhn and Oshima, 2002 (ref. 34) | 5% FBS | 5% FBS | — | — | C. parvum oocysts | — | — | 74.1 ± 2.8 |
| J. Olszewski et al., 2005 (ref. 32) | 5% FBS | Glycine–NaOH | — | — | PP7 and T1 phage, Poliovirus 2 | — | 87.1 ± 24 (PP7) | — |
| L. J. Winona et al., 2001 (ref. 31) | 5% beef extract, 1% and 5% FBS | 10× PBS | Elution | 0.05 M glycine, 10% FBS | PP7 and T1 phage, Poliovirus 2 | — | 98 ± 7 (Poliovirus) | — |
| V. R. Hill et al., 2007 (ref. 30) | 5% FBS | NaPP (0.01% w/v) | Elution | 0.01% Tween 80, 0.01% NaPP, 0.001% Y-30 | PhiX174, MS2, E. faecalis, C. perfringens, C. parvum, G. intestinalis | 120 (E. faecalis, C. perfringens) | 120 (MS2) | 88 (C. parvum) |
| Present work | — | — | Elution | 0.01% Tween 20, 0.1% Tween 80, 0.01% SHMP (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio) 1 ratio) |
E. coli | 98.5 ± 0.63 | — | — |
E. coli recovery for all the cases discussed in this study are substantially high due to the deformable nature of bacterial cells and the hydrophilic nature of the polyethersulfone (0.2 μm) and modified polyethersulfone membranes (750 kD). Experiments with different combinations of surfactants and dispersant were conducted to evaluate concentration and recovery characteristics. Use of Tween 20 and Tween 80 as elution fluids led to better recoveries of E. coli as compared to the case where concentration was done without the additional elution step. The surfactants used in the study are characterized by a polyoxyethylene hydrophilic group and a long chain fatty acid hydrophobic group. Non-ionic surfactants aid in lowering hydrophobic interactions between bacteria and the filter surface by making the filter surfaces more hydrophilic.39,43 The hydrophobic and hydrophilic parts of the surfactant molecules congregate and orient themselves at the solid liquid interfaces preventing bacterial adhesion to the filter surface and helps keep the bacteria in suspension.44 The addition of 0.01% SHMP to the surfactant solution resulted in recovery efficiencies higher than that of the experiments with individual Tween 20 or Tween 80 as the elution fluid. SHMP falls in the category of sodium polyphosphates which are highly negatively charged chemical compounds. SHMP is a six unit compound which prevents bacterial adhesion to the filter surface by steric effects and surface charge alterations. The polyphosphate molecules are adsorbed onto the surfaces of E. coli. This imparts a high negative charge to the bacterial surface which, in turn, favours repulsion. The attractive forces between the bacteria are thereby reduced which facilitates the dispersion of E. coli in solution. The recovery levels corresponding to the low test concentration (100 CFU per mL) were surprisingly low for all other conditions except those two which resulted in 90% recovery. However, these levels gradually increased with the increasing bacterial concentration in the sample. Here we postulate that the increase in repulsion due to higher concentration of bacterial cells in water, facilitates the dispersion of bacteria rather than deposition onto the filter surface. The high recoveries obtained using a combination of Tween 20 and Tween 80 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 proportion) can be attributed to the augmentation of the hydrophilic characteristics of the filter surface by the surfactant molecules. Interestingly, the effect of SHMP (0.01% by weight) on bacteria dispersion is not masked by the presence of the two surfactants already in the solution as can be observed from the highest recoveries recorded using a combination of the surfactants and SHMP. The recovery efficiencies obtained using a combination of Tween 20 and Tween 80 and that using a combination of the surfactants and SHMP, do not exhibit any significant variance with respect to bacterial concentration in water sample. The reason can be attributed to the significant increase in membrane hydrophilicity and bacterial surface charge augmentation which prevents the attachment of the bacterial cells with he filter walls even at low cell concentrations. Tween 80 has potential membrane fouling characteristics as reported in previous hollow fiber ultrafiltration study.40 The surfactant, when used as a water amendment, resulted in lower flow rates on continuous recirculation. However, in the present work, the surfactants used as the elution fluids are only subjected to a single pass through the filter. Moreover, the device being hand operated with the aid of syringes, the change in flow rate could not be quantified to indicate the effects of filter fouling. Further studies based on the fouling effects and disadvantages of using the elution fluid would constitute a part of our future work in this area.
1 proportion) can be attributed to the augmentation of the hydrophilic characteristics of the filter surface by the surfactant molecules. Interestingly, the effect of SHMP (0.01% by weight) on bacteria dispersion is not masked by the presence of the two surfactants already in the solution as can be observed from the highest recoveries recorded using a combination of the surfactants and SHMP. The recovery efficiencies obtained using a combination of Tween 20 and Tween 80 and that using a combination of the surfactants and SHMP, do not exhibit any significant variance with respect to bacterial concentration in water sample. The reason can be attributed to the significant increase in membrane hydrophilicity and bacterial surface charge augmentation which prevents the attachment of the bacterial cells with he filter walls even at low cell concentrations. Tween 80 has potential membrane fouling characteristics as reported in previous hollow fiber ultrafiltration study.40 The surfactant, when used as a water amendment, resulted in lower flow rates on continuous recirculation. However, in the present work, the surfactants used as the elution fluids are only subjected to a single pass through the filter. Moreover, the device being hand operated with the aid of syringes, the change in flow rate could not be quantified to indicate the effects of filter fouling. Further studies based on the fouling effects and disadvantages of using the elution fluid would constitute a part of our future work in this area.
5 Conclusions
In the present study, we developed a combination of surfactants and chemical dispersant as elution fluid for very high bacteria recovery efficiencies. Using a hand-held hollow fiber filtration device, E. coli was efficiently concentrated and recovered from 5 mL water samples. This study indicates that along with the type of elution fluid, filter anisotropy and pore size are the principle parameters dictating bacteria recovery efficiencies using hollow fiber filter concentration technique. There is a great potential for the integration of the procedure discussed here with portable microfluidics based biosensor devices for pathogen detection in contaminated water. The high recovery rates would ensure accurate estimation of pathological contaminants and with further modifications, the process can be integrated with the portable biosensor platforms for effective water quality monitoring at the point of use.Acknowledgements
We would like to thank Dr Dasantila Golemi-Kotra, Department of Chemistry, York University and Dr John C. McDermott, Department of Biology, York University, Ontario, Canada for their help in providing us access to the spectrophotometer used in this study. Financial support from the Natural Science and Engineering Council (NSERC), through the Grant No. NSERC - CRDPJ: 451554-13, is highly acknowledged here.References
- P. M. Fratamico, S. K. Sackitey, M. Wiedmann and M. Y. Deng, J. Clin. Microbiol., 1995, 33, 2188–2191 CAS.
- O. Lazcka, F. J. Del Campo and F. X. Munoz, Biosens. Bioelectron., 2007, 22, 1205–1217 CrossRef CAS PubMed.
- J. Olstadt, J. Schauer, J. Standridge and S. Kluender, J. Water Health, 2007, 5, 267–282 Search PubMed.
- A. K. Bej, M. H. Mahbubani, R. Miller, J. L. DiCesare, L. Haff and R. M. Atlas, Mol. Cell. Probes, 1990, 4, 353–365 CrossRef CAS.
- D. A. Boehm, P. A. Gottlieb and S. Z. Hua, Sens. Actuators, B, 2007, 126, 508–514 CrossRef CAS PubMed.
- C. Ruan, L. Yang and Y. Li, Anal. Chem., 2002, 74, 4814–4820 CrossRef CAS.
- I. Fliss, E. Emond, R. Simard and S. Pandian, BioTechniques, 1991, 11, 453–456 CAS.
- J. Tjhie, F. van Kuppeveld, R. Roosendaal, W. Melchers, R. Gordijn, D. M. MacLaren, J. Walboomers, C. Meijer and A. van Den Brule, J. Clin. Microbiol., 1994, 32, 11–16 CAS.
- Y. J. Sung, H.-J. Suk, H. Y. Sung, T. Li, H. Poo and M.-G. Kim, Biosens. Bioelectron., 2013, 43, 432–439 CrossRef CAS PubMed.
- X.-L. Su and Y. Li, Anal. Chem., 2004, 76, 4806–4810 CrossRef CAS PubMed.
- E. Chmela, R. Tijssen, M. T. Blom, H. J. Gardeniers and A. van den Berg, Anal. Chem., 2002, 74, 3470–3475 CrossRef CAS.
- D. E. Raymond, A. Manz and H. M. Widmer, Anal. Chem., 1994, 66, 2858–2865 CrossRef CAS.
- W. Hijnen, D. van Veenendaal, W. van der Speld, A. Visser, W. Hoogenboezem and D. van der Kooij, Water Res., 2000, 34, 1659–1665 CrossRef CAS.
- G. L. Pettipher, R. Mansell, C. H. McKinnon and C. M. Cousins, Appl. Environ. Microbiol., 1980, 39, 423–429 CAS.
- B. W. Peterson, P. K. Sharma, H. C. van der Mei and H. J. Busscher, Appl. Environ. Microbiol., 2012, 78, 120–125 CrossRef CAS PubMed.
- M. Yamada, M. Nakashima and M. Seki, Anal. Chem., 2004, 76, 5465–5471 CrossRef CAS PubMed.
- S. M. McFaul, B. K. Lin and H. Ma, Lab Chip, 2012, 12, 2369–2376 RSC.
- K. Loutherback, K. S. Chou, J. Newman, J. Puchalla, R. H. Austin and J. C. Sturm, Microfluid. Nanofluid., 2010, 9, 1143–1149 CrossRef.
- L. R. Huang, E. C. Cox, R. H. Austin and J. C. Sturm, Science, 2004, 304, 987–990 CrossRef CAS PubMed.
- V. Dolnik and S. Liu, J. Sep. Sci., 2005, 28, 1994–2009 CrossRef CAS PubMed.
- A. T. Aranas, A. M. Guidote Jr. and J. P. Quirino, Anal. Bioanal. Chem., 2009, 394, 175–185 CrossRef CAS PubMed.
- C. D. James, M. Okandan, S. S. Mani, P. C. Galambos and R. Shul, J. Micromech. Microeng., 2006, 16, 1909 CrossRef CAS.
- N. S. K. Gunda, S. Bhattacharjee and S. K. Mitra, Biomicrofluidics, 2012, 6, 034118 CrossRef PubMed.
- N. S. K. Gunda, S. Naicker, S. Shinde, S. Kimbahune, S. Shrivastava and S. Mitra, Anal. Methods, 2014, 6, 6236–6246 RSC.
- J. de Jong, R. Lammertink and M. Wessling, Lab Chip, 2006, 6, 1125–1139 RSC.
- W. P. Hamilton, M. Kim and E. L. Thackston, Water Res., 2005, 39, 4869–4878 CrossRef CAS PubMed.
- M. Manafi, Int. J. Food Microbiol., 2000, 60, 205–218 CrossRef CAS.
- G. Belfort, Y. Rotem and E. Katzenelson, Water Res., 1975, 9, 79–85 CrossRef.
- P. Liu, V. R. Hill, D. Hahn, T. B. Johnson, Y. Pan, N. Jothikumar and C. L. Moe, J. Microbiol. Methods, 2012, 88, 155–161 CrossRef PubMed.
- V. R. Hill, A. M. Kahler, N. Jothikumar, T. B. Johnson, D. Hahn and T. L. Cromeans, Appl. Environ. Microbiol., 2007, 73, 4218–4225 CrossRef CAS PubMed.
- L. Winona, A. Ommani, J. Olszewski, J. Nuzzo and K. Oshima, Can. J. Microbiol., 2001, 47, 1033–1041 CrossRef CAS.
- J. Olszewski, L. Winona and K. H. Oshima, Can. J. Microbiol., 2005, 51, 295–303 CrossRef CAS PubMed.
- S. D. Leskinen and D. V. Lim, Appl. Environ. Microbiol., 2008, 74, 4792–4798 CrossRef CAS PubMed.
- R. C. Kuhn and K. H. Oshima, Can. J. Microbiol., 2002, 48, 542–549 CrossRef CAS.
- P. S. Knappett, A. Layton, L. D. McKay, D. Williams, B. J. Mailloux, M. R. Huq, M. J. Alam, K. M. Ahmed, Y. Akita and M. L. Serre, et al., Groundwater, 2011, 49, 53–65 CrossRef CAS PubMed.
- K. Shapiro, J. A. Mazet, A. Schriewer, S. Wuertz, H. Fritz, W. A. Miller, J. Largier and P. A. Conrad, Water Res., 2010, 44, 893–903 CrossRef CAS PubMed.
- H. A. Lindquist, S. Harris, S. Lucas, M. Hartzel, D. Riner, P. Rochele and R. DeLeon, J. Microbiol. Methods, 2007, 70, 484–492 CrossRef CAS PubMed.
- C. M. Smith and V. R. Hill, Appl. Environ. Microbiol., 2009, 75, 5284–5289 CrossRef PubMed.
- H. A. Morales-Morales, G. Vidal, J. Olszewski, C. M. Rock, D. Dasgupta, K. H. Oshima and G. B. Smith, Appl. Environ. Microbiol., 2003, 69, 4098–4102 CrossRef CAS.
- V. R. Hill, A. L. Polaczyk, D. Hahn, J. Narayanan, T. L. Cromeans, J. M. Roberts and J. E. Amburgey, Appl. Environ. Microbiol., 2005, 71, 6878–6884 CrossRef CAS PubMed.
- M. Humphries, J. F. Jaworzyn, J. B. Cantwell and A. Eakin, FEMS Microbiol. Lett., 1987, 42, 91–101 CrossRef CAS PubMed.
- J. Méndez, A. Audicana, A. Isern, J. Llaneza, B. Moreno, M. L. Tarancón, J. Jofre and F. Lucena, J. Virol. Methods, 2004, 117, 19–25 CrossRef PubMed.
- J. H. Paul and W. H. Jeffrey, Can. J. Microbiol., 1985, 31, 224–228 CrossRef CAS.
- M. Lepesteur, S. Blasdall and N. Ashbolt, Lett. Appl. Microbiol., 2003, 37, 218–229 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2015 |