Supramolecular solvent based liquid–liquid microextraction of aluminum from water and hair samples prior to UV-visible spectrophotometric detection
Mansoor Khanab and
Mustafa Soylak*a
aErciyes University, Faculty of Sciences, Department of Chemistry, 38039 Kayseri, Turkey. E-mail: soylak@erciyes.edu.tr; Fax: +90-3524374933
bInstitute of Chemical Sciences, University of Peshawar, Peshawar, Pakistan
First published on 10th July 2015
Abstract
A new method for the microextraction of aluminum from water and hair samples has been developed using a supramolecular solvent (undecanol–tetrahydrofuran) prior to UV-visible spectrophotometric determination. 8-Hydroxyquinoline was used as chelating agent which forms a chelate with Al(III) at pH 8.0. The Al(III)-8-hydroxyquinoline chelate was quantitatively extracted into the supramolecular solvent (Ss) phase. The molar absorptivity calculated for the complex was 1.8 × 103 L mol cm−1 at 380 nm. Factors effecting extraction efficiency of the method including pH, amount of chelating agent, sample volume, type and volume of supramolecular solvent (Ss) and matrix effect were optimized. A pre-concentration factor of 30 was achieved with a limit of quantification of 0.47 μg L−1 with relative standard deviation of 0.3%. The accuracy of the developed method was evaluated by the analysis of the certified reference materials (SPS WW2 waste water, TMDA-53.3 fortified water and NCS ZC81002B human hair) and by addition-recovery studies for water and hair samples.
1. Introduction
Aluminum is the third most abundant element in the earth’s crust and due to its low density has wide spread applications in automotive, aerospace and automated instrument industries.1,2 The maximum permissible level of aluminum given by the US Environmental Protection Agency (EPA) in drinking water is 0.2 mg L−1.3 Due to natural and human activities like industry and traffic, the amount of total dissolved aluminum in environmental samples including water and food is increasing. It may cause some serious complications in the nervous system, parathyroid gland, chromosomes and is also studied as a possible factor in the formation of Alzheimer’s disease, renal osteodystrophy, and Parkinson’s disease.4,5 Aluminum is also used for coagulation and flocculation in the treatment of colloidal and suspended particles in water effluents.6–8 Due to both negative and positive aspects of aluminum, it is very important to develop analytical methods for the determination of aluminum at trace levels both in environmental and biological samples.9,10Analytical methods which are widely used for the determination of aluminum are chemiluminescence analysis,11 electrothermal atomic absorption spectrometry (ETAAS),12 inductively coupled plasma optical emission spectrometry (ICP-OES),13 electroanalytical techniques14 and high performance liquid chromatography (HPLC).15 However, due to a lack of selectivity and sensitivity and interference effects of the matrix, the direct determination of Al(III) using these methods are not possible.12 Other important factors related to these methods are highly expensive and expert analysts are required for their operation.
UV-visible spectrophotometric methods are preferable as compared to the above mentioned analytical methods and are simpler and cheaper. However due to a lack of selectivity and sensitivity, the direct determination of Al(III) by UV-visible spectrophotometry is problematic.16 These problems for aluminum and other metal ions can be overcome by using specific separation–preconcentration methods like cloud point extraction,17,18 solid phase extraction,19 and liquid–liquid extraction.20
The excessive discharge of organic solvents in laboratories during liquid–liquid microextraction experiments causes some serious environmental hazards. To reduce the amount of toxic organic solvents discharged, supramolecular solvent based liquid–liquid microextraction has been investigated recently.24 Some unique properties of supramolecular solvents, such as their hydrophobic nature and hydrogen bonding interactions with chelates, make supramolecular solvent based liquid–liquid microextraction far superior in extraction efficiency than ordinary other kinds of liquid–liquid microextraction.25,26 The concept of a supramolecular solvent-based extraction technique was reported for first time by Ballesteros-Gómez and co-workers.27 Different supramolecular solvent systems like 1-decanol, undecanol and decanoic acid have been used for liquid–liquid microextraction of heavy metals.28
Previous research has used different chromogenic reagents like chrome azurol S (CAS),21 8-hydroxyquinoline16, eriochrome cyanine R (ECR)22 and pyrocatechol violet23 that form colored complexes with Al(III) for UV-visible spectrophotometric determination. 8-Hydroxyquinoline was selected as the chromogenic reagent in the presented work.
The aim of the present work is to develop a supramolecular solvent based liquid–liquid microextraction method for aluminum from water and biological samples. A complexing reagent, 8-hydroxyquinoline, formed a complex with Al(III) at pH 8.0,29 which was then quantitatively extracted by supramolecular solvent based liquid–liquid microextraction. The concentration of Al(III) in the supramolecular solvent phase was then determined by using a UV-visible spectrophotometer.
2. Experimental
2.1. Apparatus
Ultrapure water obtained from a MilliQ Direct-16 purification system (18.2 MΩ cm, Millipore) was used in all experiments. A UV-visible spectrophotometer (Hitachi 150-20) with a quartz micro-cell with a path length of 10 mm and a volume of 0.7 mL was used for the absorbance measurements. pH measurements were made using a Nel pH 900 model (Ankara-Turkey) pH meter with a glass electrode. A centrifuge (ALC PK 120 Model, Buckinghamshire, UK) was used for phase separation. A vortex mixer (Wiggen Hauser, Malaysia) was used for thorough mixing of solutions.2.2. Chemicals and solutions
Stock solutions of aluminum(III) as a Al(NO3)3 salt was produced by diluting a stock solution of 1000 mg L−1 of the given elements supplied by Sigma-Aldrich (St. Louis, MO, USA). This was diluted for the preparation of standard solutions of Al(III). 0.1% (w/v) solution of complexing agent was prepared by dissolving 0.1 g of 8-hydroxyquinoline (Sigma-Aldrich, St. Louis, MO, USA) in 100 mL of ethanol. The extraction solutions undecanol, 1-decanol and decanoic acid were provided by E. Merck (Darmstadt, Germany) and Sigma-Aldrich (St. Louis, MO, USA) respectively. Tetrahydrofuran (THF) was provided by Lab-Scan (Ireland). Buffer solutions were used as given in the literature.162.3. Supramolecular solvent based liquid–liquid microextraction (Ss-LLME) of Al(III)
The model studies for method development were performed using 10 mL ultrapure distilled water. 100 μL of Al(III) was taken from a stock solution of 20 mg L−1 in a 50 mL centrifuge tube. To this 2 mL of ammonia/ammonium chloride buffer solution was added and the pH was adjusted to 8.0 with 0.2 M NaOH and 0.2 M HCl. The solution was diluted to 10 mL with ultrapure distilled water. After the addition of 0.3 mg of 8-hydroxyquinoline, the solution became yellowish in color which showed that the complex formation had taken place. The solution was allowed to stand for 5 min to ensure that the complex formation was complete. For the extraction of the complex, 0.2 mL of the extraction solvent consisting of 0.1 mL of THF and 0.1 mL of undecanol was injected in to the sample solution. The supramolecular solvent spontaneously formed in this solution. The solution was subjected to a vortex for 2 minutes at vortex speed of 40 × 100 rpm) to ensure extraction of the Al(III)-8-hydroxyquinoline complex. The test tube was centrifuged at 4000 rpm for 10 min to achieve phase separation. The water phase was separated from the bottom of the solution and discarded. The extraction phase was diluted to 1 mL with ethanol. The concentration of aluminum in the last volume was measured at 380 nm using a UV-Vis double beam spectrophotometer.2.4. Application to actual environmental and human samples
The presented method was applied to water samples including tap water from Kayseri City-Turkey, sea water from Marmara Sea-Turkey, underground water samples from Kayseri City-Turkey and hair samples taken from a male living in Kayseri, Turkey. The method was also applied to certified reference materials (SPS-WW2 waste water, TMDA-53.3 fortified water and NCS ZC81002B human hair). All water samples were filtered through a cellulose membrane filter of 0.45 μm (Millipore) prior to use. In the natural water analysis, 30 mL water samples were used.The NCS ZC81002B human hair certified reference material and hair samples were subjected to a wet digestion method prior to use, in which 0.04 g of NCS ZC81002B human hair certified reference material and 0.1 g of hair sample were weighed into beakers and digested with 10 mL of concentrated HNO3 at room temperature for 30 min, and then kept at 100 °C on a hot plate until a dry residue was obtained. After cooling, the residues were again digested with 15 mL mixtures of HNO3 and H2O2 (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) following the same procedure described above. The final residue in each beaker was dissolved in10–15 mL of distilled water and filtered to obtain a clear solution. The resulting solutions were used for supramolecular solvent based liquid–liquid microextraction (Ss-LLME) of Al(III) which is given in Section 2.3.
1 v/v) following the same procedure described above. The final residue in each beaker was dissolved in10–15 mL of distilled water and filtered to obtain a clear solution. The resulting solutions were used for supramolecular solvent based liquid–liquid microextraction (Ss-LLME) of Al(III) which is given in Section 2.3.
3. Results and discussion
3.1. Effect of pH
In order to investigate the effect of pH on % recovery of Al(III), supramolecular solvent based liquid–liquid microextraction studies of Al(III) were carried out in a pH range of 2.0 to 10.0. The results are shown in Fig. 1. The % recovery of Al(III) increases with an increase in pH in the range 2.0 to 8.5 and achieved quantitative recoveries between pH 7.5 and 8.5. It can be explained that the hydrophobic complex formation and the best formation of supramolecular solvent were achieved at these pH values.29 Hence, pH 8.0 was used in subsequent work. | ||
| Fig. 1 Influence of pH on extraction efficiency of Al(III) (volume of sample: 10 mL, volume of undecanol: 0.1 mL, volume of THF: 0.1 mL, amount of 8-hydroxyquinoline: 0.3 mg, N = 3). | ||
3.2. Effect of the amount of 8-hydroxyquinoline
The effect of the amount of 8-hydroxyquinoline was examined in the range of 0.1–0.7 mg. The results are shown in Fig. 2. The obtained results indicate that the quantitative recoveries for Al(III) were obtained after addition of 0.3 mg of complexing agent and further increase in the amount of complexing agent has no significant change on % recovery of Al(III). Therefore 0.3 mg of 8-hydroxyquinoline was used in further work.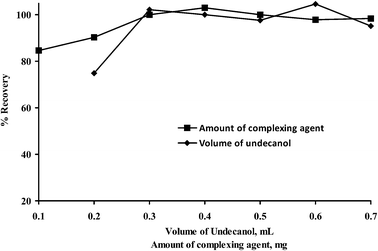 | ||
| Fig. 2 Influence of volume of undecanol and amount of complexing agent on the extraction efficiency of Al(III) (pH: 8.0, volume of sample: 10 mL, volume of THF: 0.1 mL, N = 3). | ||
3.3. Effect of the type and volume of supramolecular solvent (Ss)
In order to achieve the quantitative extraction of Al(III), the choice of the best supramolecular solvent is important. Three supramolecular solvents including 1-decanol–THF, undecanol–THF and decanoic acid–THF were tested. The results are given in Table 1. Undecanol–THF was a better choice having high extraction efficiency and was used for formation of the supramolecular solvent in the rest of the proposed method.The volume ratio of undecanol and THF was also optimized. For this purpose the Ss-LLME of Al(III) study was carried out using different volumes of undecanol in the range of (0.075–0.5 mL) keeping the volume of THF constant. The results are shown in Fig. 2. Quantitative recoveries of Al(III) were obtained in the range of 0.1 and 0.5 mL of undecanol. In order to use the minimum volume of undecanol, 0.1 mL of undecanol was selected for further experiments.
In the second step, the volume of undecanol was kept constant and the Ss-LLME method was carried out using different volumes of THF (0.075–0.5 mL). The results are given in Fig. 3. It can be concluded from the graph that 0.1 mL of THF is enough to obtain quantitative recovery of Al(III). Therefore 0.2 mL of supramolecular solvent (undecanol–THF) was used in further microextraction of Al(III) experiments.
 | ||
| Fig. 3 Influence of the THF volume on extraction efficiency of Al(III) (pH: 8.0, volume of sample: 10 mL, volume of undecanol: 0.1 mL, amount of 8-hydroxyquinoline: 0.3 mg, N = 3). | ||
3.4. Matrix effects
In order to check the selectivity of the method,30–34 the proposed supramolecular solvent based liquid–liquid microextraction method was carried out in the presence of coexisting ions given in Table 2. The effects of coexistent ions found in water and acid digested hair samples on Al(III) determination were studied. Quantitative recoveries of Al(III) even in the presence of high concentrations of these interfering ions show that the method is highly selective and free of interferences.| Interfering ion | Added as | Concentration, mg L−1 | Recovery, % |
|---|---|---|---|
| a Mean ± standard deviation. | |||
| Na+ | NaNO3 | 5000 | 102 ± 2a |
| K+ | KCl | 2500 | 103 ± 0 |
| Ca2+ | Ca(NO3)2·4H2O | 1000 | 99 ± 1 |
| Mg2+ | Mg(NO3)2·6H2O | 1000 | 96 ± 2 |
| Cu2+ | Cu(NO3)2·6H2O | 10 | 100 ± 3 |
| Zn2+ | Zn(NO3)2·6H2O | 10 | 99 ± 4 |
| Co2+ | Co(NO3)2·6H2O | 5 | 97 ± 4 |
| Ni2+ | Ni(NO3)2·6H2O | 5 | 96 ± 4 |
| Mn2+ | Mn(NO3)2·4H2O | 5 | 97 ± 1 |
| SO42− | Na2SO4 | 10 | 103 ± 0 |
| CO32− | Na2CO3 | 10 | 102 ± 1 |
| F− | NaF | 5 | 99 ± 2 |
3.5. Analytical parameters
Information about the extraction efficiency of the proposed method can be determined by calculating various analytical parameters such as the limit of detection (LOD), relative standard deviation (RSD), enhancement factor (EF), preconcentration factor (PF) and consumptive index (CIn) under the optimized experimental conditions. The value of the LOD and LOQ are 0.16 μg L−1 and 0.47 μg L−1, which were calculated as the ratio of three times standard deviation of ten blank absorbances to the slope of the regression equation and ten times the standard deviation of the ten blank solutions to the slope of the regression equation respectively. The values of (RSD), PF, EF and CIn are 0.3%, 30, 29.6 and 2 which were calculated as given in the literature.35The quantitative recoveries of Al(III) were obtained in a sample volume of 30 mL. Therefore, a high preconcentration factor 30 of Al(III) was achieved using 30 mL sample volume. The molar absorptivity calculated for a complex was 1.8 × 103 L mol cm−1 at 380 nm. The straight line equation, based on the relationship between the absorbance (A) of Al(III) in the UV-visible spectrophotometric measurement and the concentration of Al(III) (C) was A = 0.09 + 0.07C with a correlation coefficient (r2 = 0.991).
3.6. Applications
The accuracy of the proposed Ss-LLME method was checked by applying this method to three certified reference materials (SPS-WW2 waste water, TMDA-53.3 fortified water and NCS ZC81002B human hair). The obtained recovery results for Al(III) given in Table 3 indicate that the method is highly accurate and valid for the determination of aluminum at trace level.| Certified reference material | Certified value, μg g−1 | Found value, μg g−1 | Recovery, % |
|---|---|---|---|
| a Mean ± standard deviation. | |||
| NCS ZC81002B hair | 25 | 25.4 ± 0.2 | 101 |
| Certified value, mg L−1 | Found value, mg L−1 | Recovery, % | |
|---|---|---|---|
| SPS WW2 water | 10 | 10.1 ± 0.1 | 100 |
| TMDA 53.3 water | 0.36 | 0.37 ± 0.04 | 102 |
The method was also applied to a tap water sample from Kayseri City-Turkey, a sea water sample from Marmara Sea-Turkey and an underground water sample from Kayseri City-Turkey and a hair sample taken from a male living in Kayseri, Turkey to prove the accuracy of the developed Ss-LLME method. A known amount of Al(III) was added to these samples and recovery studies were performed. The results given in Table 4 show that the method is valid for the determination of aluminum in hair and water samples. The addition-recovery tests to hair and water samples given in Table 4 show that the organic and inorganic matrices of real samples did not affect our microextraction system.
| Sample | Added, μg | Found, μg | Recovery, % |
|---|---|---|---|
| a Mean ± standard deviation.b BDL = below the detection limit. | |||
| Hair | 0 | 1.8 ± 0.01a | — |
| 3 | 4.7 ± 0.06 | 95 | |
| 5 | 6.6 ± 0.03 | 96 | |
| Tap water | 0 | BDLb | — |
| 3 | 3.0 ± 0.01 | 100 | |
| 6 | 6.1 ± 0.06 | 101 | |
| Sea water | 0 | 2.2 ± 0.06 | — |
| 4 | 6.2 ± 0.05 | 99 | |
| 8 | 9.7 ± 0.01 | 94 | |
| Underground water | 0 | 3.8 ± 0.03 | — |
| 4 | 7.6 ± 0.01 | 96 | |
| 6 | 9.4 ± 0.00 | 94 | |
The described Ss-LLME method was compared with other preconcentration methods for Al(III) in the literature based on analytical parameters including LOD and PF (Table 5). Low LOD values and high PF values confirm that the proposed Ss-LLME is comparable to or more efficient than other preconcentration methods in the literature.
| Method | Analysis | LOD, μg L−1 | Ref. |
|---|---|---|---|
| Cloud point extraction | GFAAS | 0.79 | 36 |
| Cloud point extraction | ICP-OES | 0.25 | 37 |
| Solid phase extraction | ICP-AES | 0.19 | 38 |
| Solid phase extraction | AAS | 2 | 39 |
| Dispersive liquid–liquid microextraction | ICP-OES | 0.8 | 40 |
| Supramolecular solvent based liquid–liquid microextraction (Ss-LLME) | UV-visible spectrophotometry | 0.16 | This work |
4. Conclusion
The objective of this work was to develop a new supramolecular solvent (Ss) based liquid–liquid microextraction method for the separation and preconcentration of Al(III) from water and biological samples prior to its determination by UV-visible spectrophotometry. The presented method is highly sensitive with a low LOD value of 0.16 μg L−1. High extraction efficiencies were obtained with PF and EF values of 30 and 29.6 respectively. The method is cost effective because there is no need for special laboratory equipment. The proposed method is comparable to or more efficient than the other preconcentration methods with regard to the LOD. The method was successfully applied to water and hair samples.Acknowledgements
Mansoor Khan is thankful to the Scientific and Technological Research Council of Turkey (TUBITAK) under “2216 Research Fellowship Programme for Foreign Citizens” for financial support. Authors also thank Erkan Yilmaz for his help.References
- V. Andruch, I. S. Balogh, L. Kocúrová and J. Šandrejová, Appl. Spectrosc. Rev., 2013, 48, 161–259 CrossRef CAS PubMed.
- O. Yalcinkaya, H. Erdoğan, H. Çiftçi and A. R. Türker, Spectrosc. Lett., 2012, 45, 344–351 CrossRef CAS PubMed.
- http://water.epa.gov/drink/contaminants, (accessed on June 23th, 2015).
- G. L. Klein, Curr. Opin. Pharmacol., 2005, 5, 637–640 CrossRef CAS PubMed.
- M. Sun and Q. Wu, J. Hazard. Mater., 2010, 176, 901–905 CrossRef CAS PubMed.
- Z. L. Yang, B. Y. Gao, Q. Y. Yue and Y. Wang, J. Hazard. Mater., 2010, 178, 596–603 CrossRef CAS PubMed.
- M. B. Baskan and A. Pala, Desalination, 2010, 254, 42–48 CrossRef PubMed.
- S. Ghafari, H. A. Aziz, M. H. Isa and A. A. Zinatizadeh, J. Hazard. Mater., 2009, 163, 650–656 CrossRef CAS PubMed.
- O. Oter and S. Aydogdu, J. Fluoresc., 2011, 21, 43–50 CrossRef CAS PubMed.
- P. Vanloot, J.-L. Boudenne, L. Vassalo, M. Sergent and B. Coulomb, Talanta, 2007, 73, 237–245 CrossRef CAS PubMed.
- X.-H. Xu, H. C. Yang, T. E. Mallouk and A. J. Bard, J. Am. Chem. Soc., 1994, 116, 8386–8387 CrossRef CAS.
- I. Narin, M. Tuzen and M. Soylak, Talanta, 2004, 63, 411–418 CrossRef CAS PubMed.
- M. Rezaee, Y. Yamini, A. Khanchi, M. Faraji and A. Saleh, J. Hazard. Mater., 2010, 178, 766–770 CrossRef CAS PubMed.
- X. Wang, J. Lei, S. Bi, N. Gan and Z. Wei, Anal. Chim. Acta, 2001, 449, 35–44 CrossRef CAS.
- H. Matsumiya, N. Iki and S. Miyano, Talanta, 2004, 62, 337–342 CrossRef CAS PubMed.
- V. N. Bulut, D. Arslan, D. Ozdes, M. Soylak and M. Tufekci, J. Hazard. Mater., 2010, 182, 331–336 CrossRef CAS PubMed.
- L. Sombra, M. Luconi, M. F. Silva, R. A. Olsina and L. Fernandez, Analyst, 2001, 126, 1172–1176 RSC.
- H. Sang, P. Liang and D. Du, J. Hazard. Mater., 2008, 154, 1127–1132 CrossRef CAS PubMed.
- M. Tuzen and M. Soylak, J. Hazard. Mater., 2008, 154, 519–525 CrossRef CAS PubMed.
- M. Soylak and E. Yilmaz, Desalination, 2011, 275, 297–301 CrossRef CAS PubMed.
- M. Bahram, T. Madrakian, E. Bozorgzadeh and A. Afkhami, Talanta, 2007, 72, 408–414 CrossRef CAS PubMed.
- M. Soylak, U. Sahin, A. Ulgen and M. Dogan, Anal. Sci., 1997, 13, 287–289 CrossRef CAS.
- M. C. Vargas, O. D. Renedo and M. J. Arcos Martinez, Helv. Chim. Acta, 2003, 86, 2434–2440 CrossRef CAS PubMed.
- F. Rezaei, Y. Yamini and M. Moradi, J. Chromatogr. A, 2014, 1327, 155–159 CrossRef CAS PubMed.
- F. Rezaei, Y. Yamini, M. Moradi and B. Daraei, Anal. Chim. Acta, 2013, 804, 135–142 CrossRef CAS PubMed.
- A. Moral, M. D. Sicilia and S. Rubio, Anal. Chim. Acta, 2009, 650, 207–213 CrossRef CAS PubMed.
- A. Ballesteros-Gómez, S. Rubio and D. Pérez-Bendito, J. Chromatogr. A, 2009, 1216, 530–539 CrossRef PubMed.
- F. Aydin, E. Yilmaz and M. Soylak, RSC Adv., 2015, 5, 40422–40428 RSC.
- R. B. Barnes, Chem. Geol., 1975, 15, 177–191 CrossRef CAS.
- M. Soylak and N. D. Erdogan, J. Hazard. Mater., 2006, 137, 1035–1041 CrossRef CAS PubMed.
- A. Huseyinli, R. Alieva, S. Haciyeva and T. Güray, J. Hazard. Mater., 2009, 163, 1001–1007 CrossRef CAS PubMed.
- V. N. Bulut, A. Gundogdu, C. Duran, H. B. Senturk, M. Soylak, L. Elci and M. Tufekci, J. Hazard. Mater., 2007, 146, 155–163 CrossRef CAS PubMed.
- M. Soylak, L. Elci and M. Dogan, Talanta, 1995, 42, 1513–1517 CrossRef CAS.
- M. Ghaedi, K. Niknam, K. Taheri, H. Hosseinian and M. Soylak, Food Chem. Toxicol., 2010, 48, 891–897 CrossRef CAS PubMed.
- Z. A. Alothman, E. Yilmaz, M. Habila and M. Soylak, Ecotoxicol. Environ. Saf., 2015, 112, 74–79 CrossRef CAS PubMed.
- A. Bavili Tabrizi, Food Chem., 2007, 100, 1698–1703 CrossRef CAS PubMed.
- L. L. Sombra, M. O. Luconi, L. P. Fernández, R. A. Olsina, M. F. Silva and L. D. Martínez, J. Pharm. Biomed. Anal., 2003, 30, 1451–1458 CrossRef CAS.
- P. Liang, L. Yang, B. Hu and Z. Jiang, Anal. Sci., 2003, 19, 1167–1171 CrossRef CAS.
- A. Safavi, S. Momeni and N. Saghir, J. Hazard. Mater., 2009, 162, 333–337 CrossRef CAS PubMed.
- M. Rezaee, Y. Yamini, A. Khanchi, M. Faraji and A. Saleh, J. Hazard. Mater., 2010, 178, 766–770 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |
