Structural characteristics of (−)-epigallocatechin-3-gallate inhibiting amyloid Aβ42 aggregation and remodeling amyloid fibers†
Yun Liu*a,
Yang Liu‡
a,
Shihui Wang‡a,
Shengzhao Dong‡a,
Ping Changb and
Zhaofeng Jiangb
aBeijing Key Laboratory of Bioprocess, College of Life Science and Technology, Beijing University of Chemical Technology, Beijing 100029, China. E-mail: liuyun@mail.buct.edu.cn; liuyunprivate@sina.com; Fax: +86-10-64416428; Tel: +86-10-64421335
bCollege of Applied Arts and Science, Beijing Union University, Beijing 100101, China
First published on 15th July 2015
Abstract
To elucidate the structural requirements by which EGCG analogs inhibit Aβ42 protein aggregation and remodel amyloid fibers, the molecular interactions between Aβ42 and four EGCG analogs, epigallocatechin-3-gallate (EGCG), (−)-gallocatechin gallate (GCG), (−)-epicatechin-3-gallate (ECG) and (−)-epigallocatechin (EGC), were investigated by thioflavin T fluorescence (ThT), circular dichroism (CD), atomic force microscopy (AFM), differential scanning calorimetry (DSC) and BCA protein assay. Results revealed that the four EGCG analogs had the ability to prevent the increase of β-sheet content and inhibit Aβ42 fibrillation when added in the lag and growth phases of Aβ42 fibrillation process. When added in the equilibrium phase, the four EGCG analogs can disaggregate the preformed protofibrils/fibrils to oligomers and unfold or partially unfold oligomers. It was also observed that EGCG showed the highest inhibitory effect on Aβ42 fibrillation, followed by GCG, ECG and EGC. From the values of IC50, kinetic parameters, secondary structures, thermo-stability and solubility measurement, a reasonable conclusion can be preliminarily drawn that the structural contribution efficiency of EGCG to inhibit Aβ42 aggregation and remodel Aβ42 amyloid fibrils decreases by the order of 3′-hydroxyl group of trihydroxyphenyl ring > gallol ester moiety > stereoisomer. The findings in this work provide the structure based molecular interaction mechanism between EGCG analogs and Aβ42 amyloid protein.
Introduction
The typical feature of Alzheimer's disease (AD) is the aggregation of amyloid β-protein (Aβ) from soluble random-coil into β-sheet rich fibrils.1 Aβ protein contains several different species (Aβ39-43) according to the numbers of amino acids in the backbone structure. Among them, amyloid β-protein 42 (Aβ42) has been considered as the most crucial factor for the onset of AD due to its extremely severe neurotoxicity and strong aggregation capability.2,3 Hence, prevention of Aβ42 aggregation has a key point to AD pathology.4 Researchers have revealed that (−)-epigallocatechin 3-gallate (EGCG) shows excellent inhibitory effect for the aggregation/fibrillation of Aβ protein both in vivo and in vitro.5–7 For instance, Porat and co-workers addressed that EGCG was a potent inhibitor of Aβ40 aggregation and its IC50 was 3.0 μM.8 EGCG is thought to bind to unaggregated polypeptides and redirect the pathway of amyloid formation to off-pathway nontoxic oligomers,9 and it is a promising new drug-delivery system to the special position.10 Wang and co-workers11 reported that there were no specific interactions and binding sites in the Aβ42 and EGCG binding from the data of isothermal titration calorimetry (ITC).EGCG and its analogs, (−)-epicatechin gallate (ECG), (−)-epigallocatechin (EGC) and (−)-gallocatechin gallate (GCG), are natural polyphenolic compounds extracted from green tea, and show little toxicity to human.12,13 The chemical structures of EGCG analogs are depicted in Fig. 1. It can be obviously found that GCG is the stereoisomer of EGCG. The structure of ECG is similar to EGCG but lacks the 3′-hydroxyl group on the trihydroxyl ring (B ring). The structure of EGC includes the 3′-hydroxyl group but lacks the gallate ester (D ring). Comparative analysis of EGC and EGCG allows an assessment of the gallate moiety role in inhibiting Aβ42 aggregation and remodeling Aβ42 mature fibrils. Similarly, comparative analysis of ECG and EGCG can address the role of 5-hydroxyl group on the trihydroxyl ring. And comparative analysis of GCG and EGCG can show the role of stereoisomer in the polyphenolic structure.
It has been demonstrated that the structural constraints and specific aromatic interactions directed polyphenol inhibitors to the amyloidogenic core.8 Churches and co-workers14 suggested that both the number of hydroxyl groups and the positioning of these groups on the polyphenolic structure are important against amyloid protein aggregation. Akaishi and co-workers15 also demonstrated that phenolic hydroxyl groups were important for the inhibition of EGCG against Aβ42 aggregation. Many other researchers confirmed that galloyl moiety (D ring in Fig. 1) in polyphenols structure played an essential role of binding to many proteins.16–18 In our previous work, we elucidated that epimers EGCG and GCG showed discrepancy inhibitory effects on lipase activity, indicating spatial conformation might have an important role of binding to protein.18
These findings raise a challenge question of what link between the hydroxyl group, gallol moiety, and epimer in the polyphenolic structural analogs and corresponding anti-aggregatory activity. To meet the challenge, using EGCG, GCG, ECG, and EGC analogs as model inhibitors, we would like to characterize the structure and activity between EGCG analogs and β-sheet linked polymerization and depolymerization of Aβ42 protein fibrils. Although EGCG and its structural analogs are well known to inhibit Aβ42 aggregation, however, it is not known the structural requirements by which EGCG analogs inhibit Aβ42 protein aggregation and remodel amyloid fibrils. Thus, we evaluate the effects of adding four polyphenolic structural analogs in each specific phase of fibrillation on Aβ42 aggregation and disaggregation. Therefore, three main purposes of this work are focused on: (1) to explore the influences of EGCG structural analogs on the Aβ42 aggregation/fibrillation when added in the lag and growth phases of Aβ42 fibrillation; (2) to address the effects of EGCG structural analogs on the Aβ42 disaggregation when adding in the equilibrium phase of Aβ42 fibrillation; (3) to evaluate the contribution efficiency of 3′-hydroxyl group, galloyl moiety, and epimer in EGCG structure on the redirection of Aβ42 amyloid formation. The findings of our work will shed light on the inhibitory mechanisms of EGCG analogs against Aβ42 aggregation, and provide the structured based mechanism between polyphenols inhibitors and Aβ42 amyloid protein at molecular level.
Materials and methods
Materials
Aβ42 with a purity of more than 95% was purchased from GL Biochem Ltd (Shanghai, China). Aβ42 protein was kept in −80 °C fringe before experimental usage. The polyphenols analogs of EGCG, ECG, EGC, and GCG with the purity of 98% were bought from Shanghai Yuanye Biological Technology Co., Ltd (Shanghai, China). The molecular chemical structures of four EGCG analogs are shown in Fig. 1. Hexafluoroisopropanol (HFIP) with the purity of more than 99.5% was available from Sigma (St. Louis, MO, USA). All other chemicals and agents were of the analytic grades and bought from local sources in China.r parameter determination
To facilitate the comparison of different EGCG analogs under various conditions, r representing the molar ratio of a certain EGCG analogs to Aβ42 was introduced in this work, which was calculated with eqn (1):
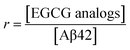 | (1) |
The final Aβ42 concentration in PBS was 25 μmol L−1, and the final concentrations of each EGCG analogs in PBS were 25, 125, and 250 μmol L−1, respectively. Hence, in this work, the values of r were 1, 5, and 10, respectively. To maintain low oxygen partial pressure and suppress the oxidation of EGCG analogs, PBS was degassed for 20 min before use and the solutions were flushed with nitrogen during operations.
Aβ42 sample solution preparation
Lyophilized Aβ42 in the vial was stored at −80 °C fringe before use. The peptide was allowed to stand at room temperature for 30 min to avoid condensation upon opening the vial cap. Aβ42 stock solution was prepared by dissolving the Aβ42 peptide in HFIP with the concentration of 1 mg mL−1. The solution was subjected to ultrasonic concussion for 10 min and incubated at 25 °C for 30 min in order to eliminate all the secondary structures. Then the volatile solvent was removed off by vacuum freeze-drying overnight. The treated Aβ42 peptide was dissolved in 20 mmol L−1 NaOH solution with a concentration of 1 mg mL−1. After ultrasonic concussion for 5 min, Aβ42 solution was centrifuged with the rate of 5000 rpm for 5 min at 5 °C. 1 mL of supernatant was drawn and diluted with 100 mmol L−1 phosphate buffer solutions (PBS) (pH 7.4) to the final concentration of 25 μM Aβ42 for experiments.Fibrillation kinetics monitoring by thioflavin T (ThT) fluorescence
ThT fluorescence method was employed to monitor the fibrillation kinetics of Aβ42 in the absence and presence of EGCG analogs at different concentrations at 37 °C with the agitation of 200 rpm.19 At appropriate intervals, aliquot of 200 μL samples was removed from the bulk solution and mixed with 2 mL of 20 μmol L−1 ThT. The solution was injected into a 1 cm-path length quartz cuvette and assayed on a Varian Cary Eclipse Fluorescence Spectrometer (Varian Inc., Palo Alto, California, USA). The excitation wavelength was 400 nm, and emission wavelength was 480 nm. The excitation and emission slits were both 5 nm. The scanning rate was 600 nm min−1 and the resolution was 1.0 nm.Analysis of kinetic parameters
Aβ42 fibrillation kinetic parameters were analyzed according to the method reported by Wang and co-workers.20 The fibrillation of Aβ42 could be described as a sigmoidal time-dependent curve which sequentially involves three stages, an initial lag phase where the ThT fluorescence intensity at 480 nm (I480) shows no changes, a subsequent fast growth phase where I480 increases exponentially with time, and a final equilibrium phase where I480 reaches a plateau indicating the end of fibril formation.21 Hence, the I480 values were plotted as a function of incubation time and regressed by a sigmoidal curve described by eqn (2):
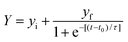 | (2) |
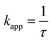 | (3) |
| Tlag = t0 − 2τ | (4) |
| Ymax = yf + yi | (5) |
Conformation analysis by circular dichroism (CD) spectroscopy
The secondary structure changes of Aβ42 protein during the fibrillation in the absence and presence of EGCG analogs were detected by a Jasco 810 circular dichroism spectrophotometer (Jasco Inc., Tokyo, Japan) according to the method in our previous work.22 Specifically, an aliquot of 500 μL sample was taken out from the bulk solution at different incubation times and centrifuged at 5000 rpm for 5 min. The supernatant was injected into a 1 mm path length quartz cuvette. A background CD spectrum of buffer solution was subtracted from the sample spectrum for baseline correction. The conditions of CD analysis were: a resolution of 0.5 nm, scanning rate of 100 nm min−1, response time of 1 s, bandwidth of 2 nm, room temperature and the wavelength ranges from 190 to 250 nm. The Aβ42 secondary elements of α-helix, β-sheet, turn, and unordered coil were calculated from the spectra data using the public database of DichroWeb (http://dichroweb.cryst.bbk.ac.uk/html/home.shtml).23 Deconvolution protocol: SELCON3. Specific parameters: file format of JASCO 1.50 (with preview); input and output units of theta; initial wavelength of 250 nm; final wavelength of 190 nm; optional scaling factor of 1.0; mean residue weigh of 110.1 daltons (molecular weight/(number of residues −1)).Morphology detection by atomic force microscope (AFM)
10 μL samples were pipetted onto freshly cleaved mica plate (1 cm × 1 cm) fixed onto a glass slide and incubated at room temperature for 3 min. The remaining salts and loose deposits in the suspension were triplicate rinsed with ultrapure water (50 μL, Millipore) and then air-dried for a whole night. AFM images were obtained on a dimension FastScan AFM (Bruker, German) with FASTSCAN-A probe in ScanAsyst mode under ambient conditions. Scanning frequency was from 0.5 to 2.0 Hz. At least three different regions of the surface were examined to verify that morphology was similar throughout the sample.24Thermodynamic parameter (Tm) assessment by differential scanning calorimetry (DSC)
Thermodynamic parameter Tm, the transition midpoint temperature, was determined by a VP differential scanning calorimetry (MicroCal, Northampton, MA).18 Temperature scans were set from 30 to 90 °C at a scan rate of 1 °C min−1 (i.e., ca. 60 min for a round). The sample cell was loaded with Aβ42 solution in the absence or presence of a certain EGCG analogs, and the same concentration of PBS was loaded into the reference cell as the blank control. Samples were removed from the bulk solutions at a certain incubation time, which was set as the sampling time in Fig. 2D and 5. The samples and control were degassed for 15 min at 4 °C immediately before DSC scanning using the included degassing system. A buffer–buffer reference scan was subtracted from each sample scan prior to concentration normalization. DSC data were analyzed by MicroCal Origin Version 7.0.18Inhibition and disaggregation of protofibrils/fibrils
Aβ42 solution alone was first pre-incubated at 37 °C with an agitation of 200 rpm for a certain time. At 10 h (the growth phase) and 24 h (the equilibrium phase), EGCG analogs were added into the preformed Aβ42 protofibril or fibril solutions with a final concentration of 125 μmol L−1 (r = 5) and further incubated to 40 h. At certain incubation times, aliquots of samples were removed from the bulk solution and examined by ThT fluorescence, CD spectroscopy, AFM, and DSC to evaluate the effect of EGCG analogs on the inhibition or disaggregation of Aβ42 protofibrils and fibrils.Solubility measurement by bicinchoninic acid (BCA) protein assay
The inhibitory efficacy of EGCG analogs on Aβ42 aggregation and disaggregation was further studied by measuring the concentration of soluble Aβ42 in the supernatant with BCA protein assay.25 Briefly, 20 μL of protein standards or Aβ42 samples were centrifuged at 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 30 min, and then added to 220 μL of the mixture solutions of BCA reagent A and B (50
000 rpm for 30 min, and then added to 220 μL of the mixture solutions of BCA reagent A and B (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v, Beyotime Biotechnology Co., Shanghai, China). The mixed samples were subjected to a 96 well plate and incubated for 30 min at 37 °C. After that, the plate was cooling for 15 min at room temperature. The Aβ42 protein solubility was calculated by measuring the absorbance of each well on a Multiskan spectrum (Thermo Fisher Scientific Inc., MA, USA) at a wavelength of 562 nm.
1, v/v, Beyotime Biotechnology Co., Shanghai, China). The mixed samples were subjected to a 96 well plate and incubated for 30 min at 37 °C. After that, the plate was cooling for 15 min at room temperature. The Aβ42 protein solubility was calculated by measuring the absorbance of each well on a Multiskan spectrum (Thermo Fisher Scientific Inc., MA, USA) at a wavelength of 562 nm.
Results and discussion
Fibrillation kinetics of Aβ42 alone
We first evaluated the fibrillation characteristics of Aβ42 alone from the aspects of kinetics, structure, morphology, and thermodynamics. The data were summarized in Fig. 2.The Aβ42 fibrillation kinetics were assayed using ThT fluorescence due to the fact that the fibril contents can be quantified by ThT fluorescence intensity I480.26 As shown in Fig. 2A, Aβ42 fibrillation formation displayed a typical sigmoidal curve at physiological condition (37 °C and 100 mmol L−1 PBS). To obtain the kinetic parameters of Aβ42 fibrillation, the curve was fitted by eqn (2) described by Nielsen and co-workers,21 and Tlag value of 3.2 ± 0.2 h, kapp value of 102.3 ± 8.8 h−1, and Ymax value of 931.1 ± 9.8 were achieved (Table 1). Accordingly, the sigmoidal time-course curve can be sequentially divided into three stages: a rate-limiting lag phase of 0–3.2 h (equal to the Tlag value) where an ordered oligomeric nucleus formed, a growth phase of 3.2–15 h (at 15 h, the ThT fluorescence intensity reached the maximum of 931.1 ± 9.8) where Aβ42 protofibrils and fibrils formed/elongated with an apparent rate of 102.3 ± 8.8 h−1, an equilibrium phase of >15 h where the mature fibrils formed and the fibril-mass concentration no longer changed.27–29
| r = [EGCG analogs]/[Aβ42] | Tlag (h) | kapp (h−1) | Ymax | |
|---|---|---|---|---|
| a NA: data were unavailable due to the low Ymax. | ||||
| 0 (Aβ42 alone) | 3.2 ± 0.2 | 102.3 ± 8.8 | 931.1 ± 9.8 | |
| EGCG | 1 | 4.8 ± 0.4 | 27.4 ± 1.0 | 263.9 ± 3.5 |
| 5 | NAa | NAa | 75.9 ± 6.0 | |
| 10 | NAa | NAa | 64.0 ± 5.9 | |
| GCG | 1 | 9.4 ± 0.3 | 49.3 ± 2.1 | 277.9 ± 3.2 |
| 5 | NAa | NAa | 72.7 ± 5.2 | |
| 10 | NAa | NAa | 55.4 ± 5.4 | |
| ECG | 1 | 3.7 ± 0.4 | 19.8 ± 0.7 | 282.7 ± 5.8 |
| 5 | 19.9 ± 1.0 | 10.6 ± 0.2 | 175.8 ± 12.3 | |
| 10 | 26.3 ± 1.6 | 7.2 ± 0.4 | 159.1 ± 9.3 | |
| EGC | 1 | 5.6 ± 0.5 | 50.2 ± 1.6 | 441.6 ± 23.3 |
| 5 | 6.2 ± 0.3 | 34.3 ± 2.0 | 291.5 ± 6.4 | |
| 10 | 6.4 ± 0.6 | 30.9 ± 2.4 | 259.7 ± 3.8 | |
The secondary structure changes of Aβ42 during fibrillation were detected by CD spectroscopy, and the spectra were fitted by the public database of DichroWeb.23 The resulting Aβ42 secondary element percentages of α-helix, β-sheet, turn, and unordered coil were listed in Table S1.† Since the fibrillation of Aβ42 mainly involves the conversion of β-sheet,27,30 we presented the changing trend of β-sheet content during the fibrillation in Fig. 2B to facilitate the comprehensive analysis of different results. As listed in Table S1,† the freshly prepared Aβ42 molecules (0 h) contained a random-coil content of 52.7% and a β-sheet content of 29.5%. The β-sheet contents increased rapidly at 0–3.2 h (the lag phase), indicating that most of the Aβ42 molecules converted into β-sheet rich nuclei and oligomers at this stage (Fig. 2B). With the further increase of incubation time at 3.2–15 h (the growth phase), the β-sheet structures gradually increased with time, which suggested the continued conversion of β-sheet rich oligomers from unfolded Aβ42 molecules and the formation/elongation of protofibrils and fibrils from oligomers. However, after 15 h, the β-sheet structures started to decrease with incubation time. The same phenomenon has also been observed by Ruggeri et al.27 during the study of impact of β-sheet content on the mechanical properties of Aβ42 fibrils. They suggested that the decrease of β-sheet content mainly stemmed from the sedimentation of insoluble aggregates during the measurement. In this study, since the kinetics of Aβ42 fibril formation have already reached the stationary equilibrium phase after incubation for 15 h according to the ThT signals (Fig. 2A), we also proposed that the reduction of β-sheet content resulted from the precipitation of insoluble aggregates during centrifugation before the CD assay (see the method section for detail information).
Fig. 2C shows the morphology images of Aβ42 fibrillation assayed by AFM. At 0 h, only a few early oligomers were observed due to the fact that Aβ42 mainly displayed as a random-coil monomer, which agreed well with the data of CD and ThT analysis. After 10 h incubation, abundant oligomers and protofibrils appeared in the image. At the end of growth phase (15 h), mature and un-branched Aβ42 fibrils were seen in the image. At an equilibrium phase of 24 h, branched fibrils with larger cross-sectional diameters were obviously found in AFM image. Although the fibril-mass concentration no longer changes at the equilibrium phase, the Aβ42 molecules were in a dynamic equilibrium, which led to secondary fibrillation on the basis of readily formed fibrils and therefore wider branched fibrils were formed.27,28 Our results were in well agreement with those reported in the literatures that the surfaces of Aβ42 fibrils could serve as templates for the replication of the parent structure, which resulted in the form of wider branched fibrils.31,32
Furthermore, DSC was employed to investigate the transition midpoint temperature (Tm) of Aβ42 during the fibrillation process. Fig. 2D showed the Tm values picked from the DSC spectra. It showed that Aβ42 fibrillation could be divided into three distinct phases according to Tm values. At 0–8 h, the 1st phase, Tm1 value decreased with the incubation time from 55 to 50 °C, indicating that the thermostability of Aβ42 declined. Within this period, Aβ42 formed nuclei and oligomers according to the ThT intensity and AFM image (Fig. 2A and C). Since the formation of nuclei and oligomers were reversed and unstable, the Tm1 value declined. At 10–18 h (the 2nd phase), another transition midpoint temperature, Tm2, showed up and remained a constant value of 70 °C within this stage. It suggested that more stable species were formed in the solution. According to the AFM image in Fig. 2C, these stable species might be long and un-branched Aβ42 protofibrils/fibrils. At 20–30 h, Tm1 and Tm2 merged into Tm3 and gradually increased from 60 to 67 °C. The merge of Tm1 and Tm2 indicated that most of the nuclei, oligomers, and protofibrils have formed relatively homogeneous fibrils at this stage. Moreover, the increase of Tm3 value at 20–24 h were assigned to the secondary fibrillation and the formation of more stable and wider branched fibrils described in AFM images (Fig. 2C, 24 h).
Effects of EGCG analogs on Aβ42 fibrillation when added in the lag phase
As above mentioned, the Aβ42 alone fibrillation kinetics displayed a typical sigmoidal curve, which can be sequentially divided into three stages: lag phase, fast growth phase and equilibrium phase. By adding EGCG analogs in the lag phase, the influences of four EGCG analogs on Aβ42 fibrillation were investigated and the results were depicted in Fig. 3. It can be obviously found that ThT intensity decreased with the increasing of concentrations from 25, 125, to 250 μmol L−1 (r = 1, 5, and 10) for each EGCG analogs. The 50% inhibition concentration (IC50) values for EGCG, GCG, ECG and EGC were 1.70 ± 0.17, 2.92 ± 0.22, 4.27 ± 0.26, 18.37 ± 2.17 μmol L−1, respectively (seen Fig. S1†). It suggested that EGCG analogs show capabilities of inhibiting Aβ42 fibrillation. The ThT experimental data were regressed by eqn (2) and the kinetic parameters were summarized in Table 1. Tlag values were prolonged while kapp and Ymax values decreased in a concentration-dependent manner through adding the four EGCG analogs in the lag phase. Therefore, from Table 1 and Fig. S1,† we confirmed that the inhibitory effects of the four EGCG analogs decreased by the order of EGCG ≈ GCG > ECG > EGC.The β-sheet contents of Aβ42 alone increased firstly at 0–15 h and then decreased at 15–36 h (Fig. 4A, black line). When EGCG analogs were added into the Aβ42 solution in the initial lag phase, the contents of β-sheet changed to different extents for each analog (Fig. 4A and Table S1†). In view of EGCG and GCG, the β-sheet contents firstly increased before 3.2 h, and then dramatically decreased to 15.5 and 17.2% after 24 h incubation, respectively. By comparing the profiles for EGCG and GCG in Fig. 4A, it can also be found that the inhibitory effect of GCG was better than EGCG before ca. 10 h, indicating that GCG had better capability for prolonging the lag time of Aβ42. This conclusion was in consistence with Tlag values in Table 1, where Tlag value for GCG at r = 1 (9.4 h) was much longer than those for EGCG (4.8 h), ECG (3.7 h), and EGC (5.6 h). As for ECG and EGC, the turning points of the profiles were about 11 and 15 h, and the β-sheet contents at 36 h were 26.1 and 32.1%, respectively. Summarily, the inhibitory effects of the four molecules decreased in the order of EGCG > GCG > ECG > EGC.
To address the thermostability of EGCG analogs and Aβ42 combination, the effects of EGCG analogs on the DSC curves of Aβ42 were investigated when EGCG analogs were added in the lag phase. Fig. 5A showed that the addition of EGCG analogs broadened the peak width of Aβ42 by the order of GCG > EGCG > ECG > EGC, indicating that they bound to Aβ42 nonspecifically and broke its homogeneity to some extents. Noticeably, the Tm value for GCG was the highest, which suggested that GCG and Aβ42 nuclei formed more stable complexes in the lag phase, prolonging the lag time and preventing the oligomerization and fibrillation. This phenomenon was in well agreement with the Tlag value in Table 1 and CD results in Fig. 4A.
The morphology of Aβ42 aggregates with EGCG analogs added in the lag phase were detected through AFM after 24 h co-incubation (Fig. 6). Compared with the AFM images of Aβ42 alone in Fig. 2C (24 h), amorphous aggregates, instead of fibrils, appeared at the equilibrium stage with the addition of the four EGCG analogs, revealing that the four EGCG analogs could prevent the formation of fibrils. However, the size of the aggregates for EGCG and GCG were much larger than those for EGC and ECG, which was in consistence with Ymax values in Table 1. This phenomenon might stem from the fact that ThT mainly binds to the cavities running parallel to the fibril axis of Aβ42 which is rich in β-sheet structure, rather than amorphous aggregates or monomers whose β-sheet contents were low.24,28 The results, however, implied that EGCG and GCG could remodel Aβ42 into ThT undetectable, off-pathway aggregates, which were also found by other researchers.5,33
Effects of EGCG analogs on Aβ42 inhibition when added in the growth phase
To evaluate whether EGCG analogs can inhibit the aggregation of Aβ42 when added in the growth phase, Aβ42 was incubated alone for 10 h and then EGCG analogs were added to the solution with a final concentration of 125 μmol L−1 (r = 5). As can be seen in Fig. 7A, after the addition of the four analogs, the ThT intensity decreased by the order of EGCG ≈ GCG > ECG > EGC, indicating that the four EGCG analogs inhibited the Aβ42 oligomers and protofibrils and the inhibitory effects decreased by the order of EGCG ≈ GCG > ECG > EGC. AFM images in Fig. 7B confirmed that average size of the aggregates for EGCG and GCG was smaller than that of Aβ42 alone (Fig. 2C, 10 h), suggesting that EGCG and GCG could inhibit the formation of oligomers and protofibrils. However, there were also some amorphous aggregates with sizes larger than those of Aβ42 alone (Fig. 2C, 10 h). Since the ThT intensity and CD signal were both weak at this point (Fig. 7A and 4B), it could be inferred that the large amorphous aggregates resulted from the remodel of unfolded or partially unfolded oligomers by EGCG or GCG.5,33 As for ECG and EGC, the remodel were not observed since the size of the aggregates in Fig. 7B were smaller than those of Aβ42 alone (Fig. 2C, 10 h).Furthermore, as can be observed from Fig. 4B, when adding four EGCG analogs in the growth phase of fibrillation, the β-sheet contents of the Aβ42 peptide started to decrease instead of increase, indicating that the four EGCG analogs could bind to oligomers and protofibrils, unfold them, and prevent the fibrillation process (Fig. 4B). At the fast growth phase, Aβ42 alone showed broad peak width and two Tm values (Fig. 5B and 2), indicating that oligomers and protofibrils co-existed. The addition of EGCG analogs merged the two Tm values and narrow the peak width (Fig. 5B), suggesting that the homogeneity was increased by the analogs. Taken together, it implied that the EGCG analogs inhibited the conversion of protofibrils from oligomers and therefore increased the homogeneity of the solution.
Effects of EGCG analogs on remodeling Aβ42 fibrillation when added in the equilibrium phase
In order to investigate whether EGCG analogs remodeled mature Aβ42 fibrils, Aβ42 was incubated alone for 24 h and then EGCG analogs were added to the solution with a final concentration of 125 μmol L−1 (r = 5). As shown in Fig. 8A, ThT intensity decreased with the addition of EGCG analogs by the order of EGCG > GCG > ECG > EGC. At about 35 h, ThT intensity became unchanged. AFM images in Fig. 8B showed that the morphology of Aβ42 converted from fibrils to small oligomers, indicating that all the four EGCG analogs can disaggregate Aβ42 fibrils. Specifically, the sizes of aggregates for EGCG and GCG were larger than those for ECG an EGC, again indicating that EGCG and GCG can remodel the small oligomers to amorphous aggregates.5,33From the β-sheet contents variances of Aβ42 in the absence and presence of EGCG analogs (Fig. 4C), when EGCG analogs were added in the equilibrium phase, as shown in Fig. 4C, the β-sheet contents started to increase and then decrease with increasing of incubation time. It suggested that EGCG analogs first disaggregated fibrils and released soluble β-sheet-containing monomers/oligomers in the solution (resulting in the increase of β-sheet contents), and then unfolded or partially unfolded these monomers/oligomers (resulting in the decrease of β-sheet contents). The Tm3 value of Aβ42 alone at equilibrium phase of 24 h was 67 °C (Fig. 2D). The addition of EGCG, GCG, ECG, and EGC decreased the Tm3 value to 56.51 ± 0.10, 57.37 ± 0.12, 58.25 ± 0.08, and 61.27 ± 0.19 °C, respectively (Fig. 5C). Since the typical temperature of Aβ42 alone at lag phase was about 50–55 °C (Fig. 2D), close to the above four temperatures, it can be inferred that the four EGCG analogs remodeled the fibrils into oligomers and the disaggregation effects were by the order of EGCG > GCG > ECG > EGC.
Molecular interaction mechanism between EGCG analogs and Aβ42 protein
In this study, molecular interactions between four EGCG analogs and Aβ42 were investigated by adding the EGCG analogs at the lag phase, growth phase, and equilibrium phase of Aβ42 fibrillation. EGCG analogs could inhibit the fibrillation of Aβ42 and disaggregate the preformed fibrils and protofibrils in dependence on structural characteristics. Among the four EGCG analogs, EGCG and GCG could remodel the Aβ42 fibrils into ThT undetectable, off-pathway amorphous aggregates no matter added in each phase. To further confirm the results, we detected the solubility of Aβ42 in the absence or presence of EGCG analogs when added in different phases. As shown in Fig. 9, the solubility of Aβ42 in the presence of EGCG analogs was higher than Aβ42 alone for the three fibrillation phases with or without ultrasonic treatment, and the Aβ42 solubility increased by the order of EGC < GCG ≈ EGCG < ECG. Obviously, EGC showed the least Aβ42 solubility, which indicated the worst inhibitory effect. As for EGCG and GCG, the solubility was lower than ECG, it was probably that the two molecules remodeled the disaggregated oligomers into amorphous aggregates. Similarly, Ehrnhoefer and co-workers5 found that EGCG directly bound to unfolded Aβ42 molecules and redirected them into unstructured, off-pathway oligomers. Palhano and co-workers9 observed that EGCG remodeled the fibrils of Aβ40 into amorphous aggregates. Wang and co-workers34 also demonstrated that EGCG can bind to insulin, another amyloid protein, change its secondary structure, and induce it into amorphous aggregates.In order to probe the molecular interaction mechanisms between the four EGCG analogs and Aβ42, we further determined the half maximal inhibitory concentration (IC50) of the four EGCG analogs by varying the addition concentration from 0 to 250 μmol L−1. It was found that the IC50 value for EGCG, GCG, ECG, and EGC were 1.70 ± 0.17, 2.92 ± 0.22, 4.27 ± 0.26, and 18.37 ± 2.17 μmol L−1, respectively (Fig. S1†). Taken together, we found that EGCG showed the best inhibitory effect, followed by GCG, ECG, and EGC (Fig. 3, 6, 4B and Table 1). In view of the structural characteristics of four molecules (Fig. 1), EGCG and GCG are epimers, comparative analysis of GCG and EGCG can discover the role of stereoisomer on Aβ42 fibrillation in the polyphenolic structure. EGCG was better than GCG in the aspects of IC50, kapp, and Ymax, while GCG was better than EGCG according to Tlag (Table 1). This phenomenon might stem from the difference ways EGCG and GCG bound to Aβ42 molecules. Similar results were observed our previous work.18 We found that epimers EGCG and GCG bound with protein in different way. For instance, in the solution state, some EGCG molecules directly bound to proteins and others just encountered proteins collisionally, while all GCG molecules only bound to proteins. Moreover, the binding constant of EGCG to proteins was much higher than that of GCG.18 These results indicated that spatial conformation of EGCG and GCG caused different binding mode with protein, leading to different inhibitory effects. Nevertheless, spatial conformation of EGCG and GCG did not change the remodel ability from disaggregated Aβ42 oligomers into amorphous aggregates (Fig. 6–8).
The structural difference between EGCG and ECG lies on 3′-hydroxyl group. Comparative analysis of ECG and EGCG can address the role of 3′-hydroxyl group on the trihydroxyl ring (B ring). From IC50 values in Fig. S1† and kinetic parameters in Table 1, it indicated that 3′-hydroxyl group was an important functional group for the inhibition of Aβ42 fibrillation. This phenomenon was also confirmed by Akaishi and co-workers, who studied structural requirements for the flavonoid fisetin in inhibiting fibril formation of Aβ protein, and suggested that 3′,4′-dihydroxyl group, but not 3- or 7-hydroxyl group, is essential for the inhibitory effect of fisetin on Aβ1-42 fibril formation.15 According to our previous work and literature, the interaction between hydroxyl group and Aβ42 is supposed to be hydrogen bonding.5,11,35 Lacking 3′-hydroxyl group would decline the binding affinity of ECG to Aβ42, and suppress the remodel ability of Aβ42 (Fig. 6–8).
The structure of EGC lacks the gallate ester (D ring) in comparison with EGCG. Comparative analysis of EGC and EGCG allowed an assessment of the gallate moiety role in inhibiting Aβ42 aggregation and remodeling Aβ42 mature fibrils. From IC50 in Fig. S1† and kinetic parameters in Table 1, it is reasonably speculated that the galloyl moiety played an essential role in the inhibition of Aβ42 fibrillation. The galloyl moiety contains one phenyl group and three hydroxyl groups, so it could bind to Aβ42 through hydrophobic interactions and hydrogen bonding. Lacking galloyl moiety greatly lowered the binding affinity to Aβ42, EGC showed the least influence on the conformation and thermostability of Aβ42 (Fig. 4 and 5). Ishii and co-workers36 compared the interactions between human serum albumin (HSA) and EGCG as well as EGC. The authors pointed out that the galloyl moiety was of critical importance in the interaction between EGCG and HAS.33
Therefore, it was first demonstrated the structural characteristics of EGCG to inhibit amyloid Aβ42 aggregation and remodel amyloid fibers. In other words, 3′-hydroxyl group, gallol moiety and epimer of these functional groups in the EGCG structure were all important to inhibit amyloid fibrillation, and the inhibitory effect decreased by the order of galloyl moiety > 3′-hydroxyl group > epimer.
Conclusions
To elucidate the structural characteristics of EGCG inhibiting amyloid Aβ42 aggregation and remodeling amyloid fibers, molecular interactions between four EGCG analogs and Aβ42 were investigated by ThT fluorescence, CD spectroscopy, AFM, DSC and BCA protein assay. Results showed that four EGCG analogs could prevent the increase of β-sheet structure of Aβ42 and inhibit Aβ42 fibrillation when added at the lag and growth phases. When added at the equilibrium phase, EGCG analogs remodeled the preformed protofibrils and fibrils to oligomers and unfolded or partially unfolded the oligomers from β-sheet. From the Aβ42 solubility measurement, EGCG and GCG could remodel the fibrils into ThT undetectable, off-pathway amorphous aggregates. The inhibitory effects of the EGCG analogs were EGCG > GCG > ECG > EGC. Comprehensive analysis of functional groups of EGCG analogs, the contribution efficiency of those main groups decreased by the order of galloyl moiety > 3′-hydroxyl group > epimer to inhibit Aβ42 fibrillation. In conclusion, this work provided the structural characteristics of EGCG analogs on inhibiting Aβ42 fibrillation at molecular level.Acknowledgements
This work was financially supported by the project of Beijing Municipal Natural Science Foundation (5142013). AFM experiments are supporting by the long-term subsidy mechanism from the Ministry of Finance and the Ministry of Education of China.References
- M. Goedert and M. G. Spillantini, Science, 2006, 314, 777–781 CrossRef CAS PubMed.
- T.-I. Kam, Y. Gwon and Y.-K. Jung, Cell. Mol. Life Sci., 2014, 71, 4803–4813 CrossRef CAS PubMed.
- P. Joshi, E. Turola, A. Ruiz, A. Bergami, D. Libera, L. Benussi, P. Giussani, G. Magnani, G. Comi and G. Legname, Cell Death Differ., 2014, 21, 582–593 CrossRef CAS PubMed.
- N. J. Izzo, J. Xu, C. Zeng, M. J. Kirk, K. Mozzoni, C. Silky, C. Rehak, R. Yurko, G. Look and G. Rishton, PLoS One, 2014, 9, e111899 Search PubMed.
- D. E. Ehrnhoefer, J. Bieschke, A. Boeddrich, M. Herbst, L. Masino, R. Lurz, S. Engemann, A. Pastore and E. E. Wanker, Nat. Struct. Mol. Biol., 2008, 15, 558–566 CAS.
- K. Rezai-Zadeh, G. W. Arendash, H. Y. Hou, F. Fernandez, M. Jensen, M. Runfeldt, R. D. Shytle and J. Tan, Brain Res., 2008, 1214, 177–187 CrossRef CAS PubMed.
- K. Rezai-Zadeh, D. Shytle, G. Arendash, N. Sun, H. Hou, J. Zeng, T. Mori, D. Morgan and J. Tan, Cell Transplant., 2007, 16, 342 Search PubMed.
- Y. Porat, A. Abramowitz and E. Gazit, Chem. Biol. Drug Des., 2006, 67, 27–37 CAS.
- F. L. Palhano, J. Lee, N. P. Grimster and J. W. Kelly, J. Am. Chem. Soc., 2013, 135, 7503–7510 CrossRef CAS PubMed.
- A. Smith, B. Giunta, P. C. Bickford, M. Fountain, J. Tan and R. D. Shytle, Int. J. Pharm., 2010, 389, 207–212 CrossRef CAS PubMed.
- S. Wang, X.-Y. Dong and Y. Sun, J. Phys. Chem. B, 2012, 116, 5803–5809 CrossRef CAS PubMed.
- S. Mandel, O. Weinreb, T. Amit and M. B. H. Youdim, J. Neurochem., 2004, 88, 1555–1569 CrossRef CAS.
- Y. Levites, T. Amit, M. B. Youdim and S. Mandel, J. Biol. Chem., 2002, 277, 30574–30580 CrossRef CAS PubMed.
- Q. I. Churches, J. Caine, K. Cavanagh, V. C. Epa, L. Waddington, C. E. Tranberg, A. G. Meyer, J. N. Varghese, V. Streltsov and P. J. Duggan, Bioorg. Med. Chem. Lett., 2014, 24, 3108–3112 CrossRef CAS PubMed.
- T. Akaishi, T. Morimoto, M. Shibao, S. Watanabe, K. Sakai-Kato, N. Utsunomiya-Tate and K. Abe, Neurosci. Lett., 2008, 444, 280–285 CrossRef CAS PubMed.
- M. Nakai, Y. Fukui, S. Asami, Y. Toyoda-Ono, T. Iwashita, H. Shibata, T. Mitsunaga, F. Hashimoto and Y. Kiso, J. Agric. Food Chem., 2005, 53, 4593–4598 CrossRef CAS PubMed.
- N. Popovych, J. R. Brender, R. Soong, S. Vivekanandan, K. Hartman, V. Basrur, P. M. Macdonald and A. Ramamoorthy, J. Phys. Chem. B, 2012, 116, 3650–3658 CrossRef CAS PubMed.
- S. Wang, Z. Sun, S. Dong, Y. Liu and Y. Liu, PLoS One, 2014, 9, e111143 Search PubMed.
- K. J. Barnham, V. B. Kenche, G. D. Ciccotosto, D. P. Smith, D. J. Tew, X. Liu, K. Perez, G. A. Cranston, T. J. Johanssen and I. Volitakis, Proc. Natl. Acad. Sci. U. S. A., 2008, 105, 6813–6818 CrossRef CAS PubMed.
- S. Wang, X.-Y. Dong and Y. Sun, Biochem. Eng. J., 2012, 63, 38–49 CrossRef CAS PubMed.
- L. Nielsen, R. Khurana, A. Coats, S. Frokjaer, J. Brange, S. Vyas, V. N. Uversky and A. L. Fink, Biochemistry, 2001, 40, 6036–6046 CrossRef CAS PubMed.
- S. Wang, S. Dong, R. Zhang, H. Shao and Y. Liu, Process Biochem., 2014, 49, 237–243 CrossRef CAS PubMed.
- DichroWeb, available: http://dichroweb.cryst.bbk.ac.uk/html/home.shtml, accessed 15 June 2015.
- J. Zhang, X. Zhou, Q. Yu, L. Yang, D. Sun, Y. Zhou and J. Liu, ACS Appl. Mater. Interfaces, 2014, 6, 8475–8487 CAS.
- X. Wang, X. Wang, C. Zhang, Y. Jiao and Z. Guo, Chem. Sci., 2012, 3, 1304–1312 RSC.
- H. LeVine, Methods Enzymol., 1999, 309, 274–284 CAS.
- F. S. Ruggeri, J. Adamcik, J. S. Jeong, H. A. Lashuel, R. Mezzenga and G. Dietler, Angew. Chem., Int. Ed., 2015, 26, 4569–4574 Search PubMed.
- R. Sabaté, M. Gallardo and J. Estelrich, Int. J. Biol. Macromol., 2005, 35, 9–13 CrossRef PubMed.
- A. Lomakin, D. S. Chung, G. B. Benedek, D. A. Kirschner and D. B. Teplow, Proc. Natl. Acad. Sci. U. S. A., 1996, 93, 1125–1129 CrossRef CAS.
- T. Lührs, C. Ritter, M. Adrian, D. Riek-Loher, B. Bohrmann, H. Döbeli, D. Schubert and R. Riek, Proc. Natl. Acad. Sci. U. S. A., 2005, 102, 17342–17347 CrossRef PubMed.
- J. S. Jeong, A. Ansaloni, R. Mezzenga, H. A. Lashuel and G. Dietler, J. Mol. Biol., 2013, 425, 1765–1781 CrossRef CAS PubMed.
- M. Žganec and E. Žerovnik, BBA, Biochim. Biophys. Acta, Gen. Subj., 2014, 1840, 2944–2952 CrossRef PubMed.
- J. Bieschke, J. Russ, R. P. Friedrich, D. E. Ehrnhoefer, H. Wobst, K. Neugebauer and E. E. Wanker, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 7710–7715 CrossRef CAS PubMed.
- S. Wang, X.-Y. Dong and Y. Sun, Int. J. Biol. Macromol., 2012, 50, 1229–1237 CrossRef CAS PubMed.
- S. Wang, F.-F. Liu, X.-Y. Dong and Y. Sun, J. Phys. Chem. B, 2010, 114, 11576–11583 CrossRef CAS PubMed.
- T. Ishii, T. Ichikawa, K. Minoda, K. Kusaka, S. Ito, Y. Suzuki, M. Akagawa, K. Mochizuki, T. Goda and T. Nakayama, Biosci., Biotechnol., Biochem., 2010, 75, 100–106 CrossRef PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra09608a |
| ‡ Equal contributor. |
| This journal is © The Royal Society of Chemistry 2015 |









