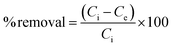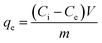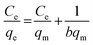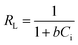Microwave assisted decoration of titanium oxide nanotubes with CuFe2O4 quantum dots for solid phase extraction of uranium
W. I. Mortada*a,
A. F. Moustafab,
A. M. Ismailc,
M. M. Hassaniend and
A. A. Aboudef
aUrology and Nephrology Center, Mansoura University, Mansoura, Egypt. E-mail: w.mortada@yahoo.com; Fax: +20 502202717; Tel: +20 1022772144
bEnvironmental Impact Assessment Unit, Environmental monitoring laboratory Unit, General Administration of Environmental Affairs, Beni-Suef Governorate, Egypt
cTechnological & Industrial Development Sector, Ministry of Industry, Trade & S&M Projects, Cairo, Egypt
dChemistry Department, Industrial Education College, Beni-Suef University, Beni-Suef, Egypt
ePhysical Department, Faculty of Science, Beni-Suef University, Beni-Suef, Egypt
fPhysical Department, Faculty of Science, El-Baha University, Saudi Arabia
First published on 14th July 2015
Abstract
Titanium oxide nanotubes (TiOxNTs) were prepared using a hydrothermal method followed by ion exchange and phase transformation. The obtained TiOxNTs were decorated with CuFe2O4 quantum dots by assisted microwave procedures. The prepared nanomaterials were characterized by XRD, TEM, and IR spectroscopy. The decorated nanotubes (DTiOxNTs) were used as adsorbents for the removal of U(VI) from aqueous solutions prior to its determination by inductively coupled plasma-optical emission spectrometry. Experimental parameters including pH, contact time and amount of adsorbent were investigated by batch mode. Optimum sorption of U(VI) ions obtained at pH 6–8. The maximum adsorption capacity of DTiOxNTs towards U(VI) was found to be 366 mg g−1 which is better than that obtained by using TiOxNTs (277 mg g−1). The equilibrium adsorption isotherm of U(VI) was fitted with the Langmuir adsorption model. Moreover, a mini-column packed with DTiOxNTs was used for column-mode extraction and preconcentration of U(VI). The effects of layer thickness, sample volume, sample flow rate and eluent conditions were studied. Under the optimized column procedures, the preconcentration factor for U(VI) was 200. The 3σ detection limit and 10σ quantification limit were found to be 0.12 and 0.40 ng mL−1, respectively. The calibration curve was linear up to 1500 ng mL−1. The proposed method showed good performance in analyzing water samples of different sources and soil sample digests collected from agriculture land near an industrial area.
1. Introduction
Uranium is a strategic element because of its industrial applications especially in the production of nuclear energy.1 It can be found naturally in very small amount in soils, rocks, air and water. However, significant quantities of uranium have been released into the environment through industrial activities. Additionally, contamination may be caused by catalysts, staining pigments, burning of fossil fuels and the manufacture and use of phosphate fertilizers that contain uranium.2–4Uranium and its compounds are highly toxic and can cause adverse health effects to human depending on the way of exposure.5 Ingestion of uranium can attack the kidneys. The major target is the renal proximal tubule,6 but the glomeruli may also be affected.7 The most serious health hazard associated with the uranium is lung cancer, which is due to the inhalation of radon, a product of uranium decay.8 Chronic uranium exposure may also result in neurotoxicity.9
Several techniques have been used for the determination of uranium such as alpha-spectrometry,10 inductively coupled plasma optical emission spectrometry (ICP-OES),11–14 inductively coupled plasma mass spectrometry (ICP-MS),15,16 X-ray fluorescence spectrophotometry (XRF),17 neutron activation18 and spectrophotometery.19–22 The low concentrations of uranium in the real samples as well as the matrix interference hinder the determination of uranium by these techniques, so a separation/preconcentration step is required before the determination. For this purpose, various procedures have been used, such as solid-phase extraction (SPE),11–22 liquid–liquid extraction,23 cloud point extraction,24 ion exchange,25 flotation26 and extraction chromatography.27 Solid phase extraction (SPE) has many advantages over the other separation procedures, including simplicity, higher enrichment factors, low cost, lower consumption of toxic chemicals.28
Nano-adsorbents have significant interest within the scientific community owing to their special properties, such as their large surface areas and high adsorption capacity.29,30 Among these adsorbents, TiO2 nanostructures has great advantages due to its physical and chemical stability, comparatively lower cost, non-toxicity and large surface area thence nano-TiO2 is a good choice for SPE and modification of its surface sometimes results in enhancements of its properties.31,32 Decoration of TiO2 nanotubes with nanoparticles is considerably used to improve their properties such as changing the surface band bending, creation of suitable surface states that promote charge transfer with surroundings, enhancement of catalytic effects for chemical reactions.33
In the present study, decorated titanium oxides nanotubes with CuFe2O4 quantum dots (DTiOxNTs) were prepared and characterized. The sorption behavior of DTiOxNTs toward U(VI) was investigated in both batch and column procedures. The decorated nanotubes were used as sorbents in the SPE of U(VI) from water and soil samples.
2. Experimental
2.1. Reagents and chemicals
Unless stated, all chemicals used were analytical reagent grade and purchased from Merck (Darmstadt, Germany). All solutions were prepared in deionized water that had been obtained by Milli-Q water purification system (Millipore, Billerica, MA, USA). The laboratory glassware was kept overnight in 10% v/v HNO3 solution. Before the use, the glassware was washed with deionized water and dried in a dust free environment. Stock standard solution of U(VI) was prepared by dissolving the appropriate amounts of UO2(NO3)2·6H2O in 0.5 mol L−1 HNO3, and then diluted to the desired volume. The solution was standardized gravimetrically using 8-hydroxyquinoline.34 Working solutions were prepared daily from the stock solution by dilutions with deionized water.2.2. Instrumentation
Determination of U(VI) was performed using inductively coupled plasma optimal emission spectrometer (ICP-OES) Perkin Elmer Model Optima 8300. The selected operating conditions of ICP-OES is presented in Table 1. The solution pH was adjusted using digital pH meter (Hanna instrument model HI 8519, Italy). Digestion of soil samples was carried out in a CEM MDS 2000 microwave digestion system (Matthews, NC, USA).| RF power | 1350 W |
| Nebulizer gas flow rate | 0.45 L min−1 |
| Auxiliary gas flow rate | 0.2 L min−1 |
| Plasma gas flow rate | 15 L min−1 |
| Pump rate | 2.5 mL min−1 |
| Sample uptake rate | 2.5 mL min−1 |
| Plasma viewing | Axial |
| Peak processing mode | Peak area |
| Delay time | 7 s |
| Integration time | 3 s |
| Replicates | 3 |
| Wavelength | 385.958 nm |
2.3. Synthesis of DTiOxNTs
TiOxNTs were prepared as previously described.35 Five grams of commercial TiO2 nanopowder (rutile phase) was dispersed in 150 mL of 10 mol L−1 NaOH and stirred for 15 min till the milk like suspension was obtained. The dispersion was then transferred to a Teflon lined autoclave kept at 150 °C for 144 h. A white precipitate (Na2Ti3O7) was obtained; this precipitate was washed with 1 mol L−1 HCl to neutralize the excess NaOH. The obtained TiOxNTs where immersed in mixture of CuCl2·2H2O and FeCl3·6H2O solution with molar ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, respectively. The mixture then turned into microwave operating at a power of 1200 W for 18 min.
2, respectively. The mixture then turned into microwave operating at a power of 1200 W for 18 min.
2.4. Characterization of the nano sorbent
Fourier transform infrared (FTIR) spectrum was measured on SHIMADZU FTIR-8101 system (Shimadzu, Kyoto, Japan) in the range of 4000 to 400 cm−1. Calibration of the frequency reading was made with polystyrene film. The X-ray diffraction (XRD) spectrum (Bruker 8 advance, CuKα radiation, target with secondary monochromator, Germany) was applied to identify the phase formation and crystal size. The size and morphology of the synthesized nanomaterial were investigated using JEOL transmission electron microscope (TEM) 2100 high resolution transmission electron microscope (JEOL Ltd, Tokyo, Japan).2.5. Sorption experiments
where Ci and Ce are the initial and equilibrium concentration of U(VI) (mg L−1), respectively.
All tests were conducted in triplicate at room temperature and their mean values were used in the calculations.
where qe represents the adsorption capacity (mg g−1), Ci and Ce are the initial and equilibrium concentrations of the metal ions (mg L−1), m is the mass of HAPNR (g), and V is the volume of solution (L).
2.6. Real samples
Water samples were filtered through a 0.45 μm pore sized Millipore cellulose nitrate membrane, acidified to pH 1 with HNO3 and stored in a refrigerator in a dark polyethylene bottle until analysis. Twelve soil samples were collected from agricultural land near Abu-Zaabal fertilizer's factory at 20–50 cm from the surface. The samples were dried at 90 °C for 2 h, ground, passed through a sieve of 120 meshes and homogenized. Five milliliters of concentrated HNO3, 2 mL of concentrated HF and 2 mL of deionized water were added to 0.5 g of the sample in a Teflon vessel. The microwave system was operated as follow: 2 min at 8 atm and 600 W; 3 min at 12 atm and 800 W; 8 min at 16 atm and 800 W.36 Finally, 20 mL of 5% w/v boric acid was added, to neutralize excess HF, and the solution was filtered and brought to a final volume of 50 mL with deionized water.3. Results and discussion
3.1. Characterization of raw and TiOxNTs
Fig. 1a and b show the XRD pattern of TiO2 powder and prepared TiOxNTs, respectively. It was obtained from Fig. 1a that the raw material is pure rutile phase of TiO2 powder with crystal size of 93 nm and high crystallinity. As a result of thermal treatment process of TiO2 powder different oxides of sodium titanium and titanium oxide nanotubes were formed with low crystallinity (as illustrated in Fig. 1b). | ||
| Fig. 1 XRD patterns of (a) commercial rutile phase TiO2 nano-powder (b) titanium oxides nanotubes where, (1) Na2Ti3O7, (2) Na2Ti6O16, (3) Na2Ti9O19, (4) Ti3O5. | ||
TEM analysis was used for particles size determination and to study the morphology of the obtained nanotubes. Fig. 2 illustrates TEM of prepared TiOx nanotubes, it can be deduced that the obtained TiOx nanotubes have uniform, hollow and layered structures. The geometry of the obtained nanotubes was measured to be 5 nm inner diameters, 9 nm outer diameter and 141 nm for the length.
3.2. Characterization of DTiOxNTs
Fig. 3 shows the XRD patterns of DTiOxNTs. It can be illustrated that; the formed phases are sodium titanium oxide (Na2Ti3O7), sodium iron oxide (Na4Fe2O5) and Landauite phase of titanium iron oxide (Ti2Fe2O7) with no evidence for presence of CuFe2O4 or CuOx nanoparticles.The absence of CuFe2O4 or CuOx peaks may be attributed to high dispersion of CuFe2O4 or CuOx on the surface and/or lattice of titanium nanotubes and/or the small portion of the formed CuFe2O4 quantum dots.37 Semi-quantitative (SQ) analysis of XRD showed that Landauite phase of titanium iron oxide (Ti2Fe2O7) has the highest percentage (37.2%) then sodium iron oxide (Na4Fe2O5) (33.5%) finally sodium titanium oxide (Na2Ti3O7) has the smallest percentage (29.4%). The high SQ percentage of Ti2Fe2O7 and Na4Fe2O5 indicate high loading percentage of iron oxides on the surface of TiOx nanotubes. Low crystallinity may be attributed to the small crystal size and high dispersion of Q.Ds on the surface of the TiOxNTs.37
Fig. 4 shows TEM image of DTiOxNTs, as shown the decoration process take place on the outer surface of the nanotubes which suggesting the successful loading process. The decorating nanoparticles have cubic shape with quantum size of 6 nm. Also it can be observed that the nanotubes still save their tubular form which indicating that microwave assisted the decoration of the nanotubes without distorting.
The vibrational spectrum of a molecule is considered to be a unique physical property and is characteristic of the molecule. As such, the infrared spectrum can be used as a fingerprint for identification by comparing the unknown spectrum with previously recorded reference spectra. The exact band position depending on the chemical composition of the sample as well as calibration and resolution of the instrument. FT-IR spectrum is useful to study the surface chemical structure of the sample. Fig. 5 shows FT-IR spectrum of DTiOxNTs recorded at the range of 4000 to 400 cm−1. The two main metal oxygen bands appears between 600–400 cm−1 is observed which arises the vibration of ions in the crystal lattices of ferrites compounds.38 The absorption band at 462.8 cm−1 attributed to octahedral sites of Cu cations in CuFe2O4 Q.Ds (Cu–O), while their vibration band appears at 440 cm−1.39 On the other hand the observed absorption band at 544.8 cm−1 may be attributed to the intrinsic vibrations of the tetrahedral metal complexes (Fe–O).38,40 It was concluded that the disappearance of absorption band 510 cm−1 in the Fe/Ti mixed oxide is due to the formation of pseudobrookite phase (Fe2TiO5) where Fe3+ is dispersed in to the lattice of TiOxNTs which confirm the low crystallinity obtained from XRD.41 The band at 700–500 cm−1 corresponding to the vibration of the Ti–O bonds in the TiOx lattice.42
The shift in the lower of the O–Ti–O band width increase may be assumed to the decreasing in size of the nanoparticles. The broad band at 3359 cm−1 due to the presence of surface OH group (adsorbed water) which corresponding to stretching vibrations of OH groups.
3.3. Batch experiments
3.4. Sorption isotherm
The sorption isotherm of U(VI) ions on DTiOxNTs is shown in Fig. 7a. As shown, with the increase of U(VI) concentration, the sorption capacities first increase sharply, then increase slowly, and finally reach saturation. To gain a better understanding of sorption mechanism, Langmuir model is used to express the experimental data. Langmuir equation is based on the assumption of ideal and uniform monolayer adsorption.45 The general form of the Langmuir isotherm iswhere Ce (mg L−1) is the equilibrium metal concentration, qe (mg g−1) denotes the amount of metal ion sorbed per unit weight of DTiOxNTs, qm (mg g−1) means the maximum sorption capacity of the surface and b (L mg−1) indicates the adsorption constant related to bonding-energy of the adsorbate to the adsorbent. The constants b and qm can be determined from the slope and intercept of the linear plot Ce versus Ce/qe. The results (Fig. 7b) showed that the value of correlation coefficient for the adsorption of U(VI) onto DTiOxNTs was 0.9972, which demonstrated the good fitting of experimental data by this model. The Langmuir constant for U(VI) was 0.164 L mg−1. The qm value for the adsorption of U(VI) by DTiOxNTs was 366 mg g−1. Table 2 displays a comparison of the maximum sorption capacity of DTiOxNTs with that of other sorbents,46–52 indicating the excellent sorption abilities of DTiOxNTs toward U(VI). The results presented in Table 2 also illustrated that DTiOxNTs has better U(VI) adsorption capacity than TiOxNTs. This denotes that the decoration of TiOxNTs with CuFe2O4 enhances the adsorption activity to U(IV).
| Sorbents | pH | qm (mg g−1) | Ref. |
|---|---|---|---|
| Graphene oxide–polypyrrole composites | 5 | 147.06 | 46 |
| Carboxymethyl cellulose grafted on multiwalled carbon nanotubes | 5 | 126.00 | 47 |
| Graphene oxide nanosheets | 5 | 97.50 | 48 |
| Magnetic yolk–shell iron oxide@magnesium silicate microspheres | 5.5 | 242.50 | 49 |
| Amidoximated magnetite/graphene oxide composites | 5 | 284.90 | 50 |
| Magnetic ion-imprinted composite | 4 | 354.85 | 51 |
| Manganese dioxide–iron oxide–graphene magnetic nanocomposite | 6 | 108.7 | 52 |
| TiO2 nanotubes | 6 | 277 | The present study |
| TiO2 nanotubes decorated with CuFe2O4 | 6 | 366 |
The Langmuir isotherm can be expressed in terms of a dimensionless constant named separation factor or equilibrium parameter, RL which is defined as:
3.5. Column experiments
| Eluent | Volume | % removal of U(VI) |
|---|---|---|
| 0.1 mol L−1 HNO3 | 2 | 62.4 ± 3.6 |
| 5 | 68.6 ± 4.7 | |
| 0.2 mol L−1 HNO3 | 2 | 71.0 ± 5.5 |
| 5 | 78.3 ± 3.2 | |
| 0.5 mol L−1 HNO3 | 2 | 82.5 ± 4.7 |
| 5 | 84.9 ± 4.5 | |
| 0.1 mol L−1 HCl | 2 | 74.2 ± 3.3 |
| 5 | 83.0 ± 4.2 | |
| 0.2 mol L−1 HCl | 2 | 85.4 ± 3.9 |
| 5 | 90.2 ± 3.7 | |
| 0.5 mol L−1 HCl | 2 | 91.8 ± 4.1 |
| 5 | 97.2 ± 2.5 |
| Ion | Tolerable ratio ion/U(VI) | Extraction (%) | RSDa (%) |
|---|---|---|---|
| a RSD of three replicate experiments. | |||
| Li+ | 1000 | 96.5 ± 3.3 | 2.7 |
| Na+ | 1000 | 97.1 ± 3.6 | 3.0 |
| K+ | 1000 | 99.0 ± 2.0 | 1.8 |
| Mg2+ | 500 | 98.6 ± 2.6 | 2.2 |
| Ca2+ | 500 | 101.3 ± 2.8 | 2.8 |
| Co2+ | 200 | 98.5 ± 2.3 | 1.5 |
| Zn2+ | 200 | 95.0 ± 3.8 | 2.1 |
| Pb2+ | 200 | 96.4 ± 3.5 | 2.8 |
| Cd2+ | 200 | 98.0 ± 2.9 | 1.7 |
| Cu2+ | 200 | 100.1 ± 2.8 | 2.0 |
| Fe2+ | 200 | 99.0 ± 2.7 | 2.7 |
| Fe3+ | 200 | 99.5 ± 1.9 | 3.1 |
| Al3+ | 200 | 95.5 ± 3.3 | 1.8 |
| Cr3+ | 200 | 97.1 ± 2.6 | 1.5 |
| F− | 1000 | 98.2 ± 2.2 | 3.0 |
| Cl− | 1000 | 96.5 ± 3.0 | 2.2 |
| Br− | 1000 | 99.0 ± 2.0 | 2.6 |
| NO2− | 1000 | 95.9 ± 3.2 | 1.9 |
| NO3− | 1000 | 96.3 ± 3.4 | 3.2 |
| SO42− | 1000 | 96.8 ± 2.9 | 2.4 |
| PO43− | 500 | 102.0 ± 2.0 | 1.4 |
| Citric acid | 50 | 95.6 ± 3.3 | 3.0 |
| Oxalic acid | 50 | 97.6 ± 2.5 | 2.2 |
| Humic acid | 20 | 95.8 ± 3.1 | 2.9 |
| Fulvic acid | 20 | 98.0 ± 2.7 | 2.6 |
3.6. Analytical performance
The limit of detection (LOD), limit of quantitation (LOQ), precision, accuracy, linear range and regression equation were used to evaluate the presented method for quantification of U(VI). LOD and LOQ for U(VI) were determined using the column procedure by passing a blank solution through DTiOxNTs under the optimal experimental conditions. According to the definition of IUPAC, the LODs (3Sb) of this procedure was 0.12 ng mL−1, where “Sb” is standard deviation of blank solution. The LOQs, defined as ten times of the Sb values, were calculated as 0.40 ng mL−1. To monitor precision (in terms of RSD), 9 replicate standards samples containing 25 ng mL−1 of U(VI) were measured. The U(VI) mean concentration was found to be 24.1 ng mL−1 with RSD values of 2.2%. The calibration curve was linear (r2 = 0.9964) from LOQ value up to 1500 ng mL−1 with the equation of I = 4.6 × 10−3C + 0.028 (where I is the intensity, and C is the concentration of U(VI) in ng mL−1). The accuracy of the proposed preconcentration method was confirmed by analysis of spiked water samples. Results were tabulated in Table 5, which show the applicability of the suggested method for determination of U(VI) in real samples.| Sample | Added (ng mL−1) | Found (ng mL−1) | Recovery (%) |
|---|---|---|---|
| River Nile water (Mansoura, Nile Delta) | 0 | 5.70 ± 0.62 | — |
| 2.5 | 8.13 ± 1.10 | 97.2 | |
| 5 | 10.60 ± 1.37 | 98.0 | |
| Mediterranean Sea water (Port Said) | 0 | 2.94 ± 0.41 | — |
| 2.5 | 5.40 ± 1.21 | 98.4 | |
| 5 | 8.06 ± 1.42 | 102.4 | |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||
| Added (μg g−1) | Found (μg g−1) | Recovery (%) | |
| Soil samples (Abu Zaabal) | 0 | 1.97 ± 0.46 | — |
| 0.5 | 2.45 ± 0.65 | 96.0 | |
| 1.0 | 2.96 ± 0.84 | 99.0 | |
3.7. Comparison of the proposed method with other methods
A comparison of figures of merit obtained for our study and other studies using various sorbents for the preconcentration of U(VI) is given in Table 6. As seen, the limit of detection by the present method is superior or comparable to the other methods except those used ICP-MS as detection technique. The procedure is linear over a wide range of U(VI) concentrations. The feasibility of the repeated use of DTiOxNTs as well as the simple way of preparation are additional advantages.| Sorbent | Analytical technique | PFa | LOD (ng mL−1) | Linearity (ng mL−1) | Ref. |
|---|---|---|---|---|---|
| a PF = Preconcentration factor. | |||||
| Mesoporous silica modified with 5-nitro-2-furaldehyde | ICP-OES | 100 | 0.30 | 1–1500 | 11 |
| Functionalized Fe3O4 magnetic nanoparticles with salicylaldehyde groups | ICP-OES | 1000 | 0.24 | 1–5000 | 12 |
| Salicylaldehyde-modified mesoporous silica | ICP-OES | 100 | 0.40 | 2–1000 | 13 |
| Phenylarsonic acid-type chitosan resin | ICP-OES | 25 | 0.1 | 1–10 | 14 |
| Merrifield polymer-octyl(phenyl)-N,N-diisobutylcarbamoyl-methylphosphine oxide | Fluorescence spectrophotometer | 400 | 20 | — | 19 |
| Chloromethylated Calix[4]arene anchored chloromethylated polystyrene | UV-visible | 143 | 6.14 | 0.1–15 | 20 |
| Grafting merrifield chloromethylated resin | UV-visible | 100 | 10 | — | 21 |
| Quinoline-8-ol anchored chloromethylated polymeric resin | UV-visible | 100 | 5 | 5–200 | 22 |
| Duolite XAD761 | ICP-MS | 30 | 4.5 × 10−3 | — | 16 |
| TiOx nanotubes decorated with CuFe2O4 | ICP-OES | 200 | 0.12 | 0.4–1500 | The present study |
3.8. Application
As our procedure is precise and accurate, it has been used to determine U(VI) in soil samples collected from farmland near Abu Zaabal industrial area. This area is located in the north of Cairo city and involves more than 700 industrial facilities beside Abu Zaabal for special chemicals and Abu Zaabal for fertilizers and chemicals industries. There are many studies have confirmed the adverse impact of pollutants on the environment in this region.54–58 One of these pollutants is uranium. 12 samples were collected from agricultural soils of different places in this region. All samples were treated as described in the experimental part and analyzed by the proposed procedure. As shown in Table 5, the concentration of U in the collected soil samples were ranged from 1.25 to 2.41 μg g−1 with a mean value of 1.97 μg g−1. According to the Canadian Council of Ministers of the Environment (CCME) soil guidelines,59 concentrations below 23 μg g−1 of U were suggested for agricultural and residential purposes. United Nations Scientific Committee on the Effects of Atomic Radiation60 defined the normal concentration of U in soil as below 11.7 μg g−1. Based on our results, none of the soil samples had exceeded the limits for agricultural activities. Our results confirmed that the concentration of U in the soil in this region is within the allowable level.4. Conclusion
In the present study, DTiOxNTs showed a high adsorption capacity towards U(VI). This nano-sorbent was successfully applied to the separative preconcentration of trace amounts of U(VI) from aqueous solution without matrix effects. The proposed method is simple, rapid and of low analysis cost. The good analytical features of the presented method showed that it is a appropriate and promised one. The method provides good precision and high accuracy indicated by the quantitative recovery of spiked U(VI) samples. The results of this study clearly showed the importance of this method, which could apply to determination of U(VI) in real samples.References
- J. S. Santos, L. S. G. Teixeira, W. N. L. d. Santos, V. A. Lemos, J. M. Godoy and S. L. C. Ferreira, Anal. Chim. Acta, 2010, 674, 143 CrossRef CAS PubMed.
- J. D. Wall and L. R. Krumholz, Annu. Rev. Microbiol., 2006, 60, 149 CrossRef CAS PubMed.
- E. R. Landa and T. B. Councell, Health Phys., 1992, 63, 343 CrossRef CAS.
- M. Betti and L. A. Delasheras, Spectrochim. Acta, Part B, 2004, 59, 1359 CrossRef PubMed.
- J. Liu, R. A. Goyer and M. P. Waalkes, Toxic effects of metals, in Casarett and Doull's Toxicology, The Basic Science of Poisons, ed. C. D. Klaassen, McGraw-Hill, New York, 7th edn, 2008, pp. 931–979 Search PubMed.
- P. Kurttio, A. Auvinen, L. Salonen, H. Saha, J. Pekkanen, I. Mäkeläinen, S. B. Väisänen, I. M. Penttilä and H. Komulainen, Environ. Health Perspect., 2002, 110, 337 CrossRef CAS.
- S. M. Pinney, R. W. Freyberg, G. E. Levine, D. E. Brannen, L. S. Mark, J. M. Nasuta, C. D. Tebbe, J. M. Buckholz and R. Wones, Int. J. Occup. Med. Environ. Health., 2003, 16, 139 Search PubMed.
- H. Laborde-Castérot, D. Laurier, S. Caër-Lorho, C. Etard, A. Acker and E. Rage, Occup. Environ. Med., 2014, 71, 611 CrossRef PubMed.
- G. C. Jiang and B. Aschner, Biol. Trace Elem. Res., 2006, 110, 1 CrossRef CAS.
- E. Manickam, S. Sdraulig and R. A. Tinker, J. Environ. Radioact., 2008, 99, 491 CrossRef CAS PubMed.
- S. R. Yousefi, S. J. Ahmadi, F. Shemirani, M. R. Jamali and M. S. Niasari, Talanta, 2009, 80, 212 CrossRef CAS PubMed.
- A. Rezaei, H. Khani, M. M. Farahani and M. K. Rofouei, Anal. Methods, 2012, 4, 4107 RSC.
- M. R. Jamali, Y. Assadi, F. Shemirani, M. R. Milani Hosseini, R. R. Kozani, M. Masteri-Farahani and M. Salavati-Niasari, Anal. Chim. Acta, 2006, 579, 68 CrossRef CAS PubMed.
- K. Oshita, K. Seo, A. Sabarudin, M. Oshima, T. Takayanagi and S. Motomizu, Anal. Bioanal. Chem., 2008, 390, 1927 CrossRef CAS PubMed.
- K. Benkhedda, V. N. Epov and R. D. Evans, Anal. Bioanal. Chem., 2005, 381, 1596 CrossRef CAS PubMed.
- F. A. Aydin and M. Soylak, Talanta, 2007, 72, 187 CrossRef CAS PubMed.
- N. L. Misra, S. Dhara and K. D. Singh Mudher, Spectrochim. Acta, Part B, 2006, 61, 1166 CrossRef PubMed.
- S. Landsberger and R. Kapsimalis, J. Environ. Radioact., 2013, 117, 41 CrossRef CAS PubMed.
- Ch. Siva Kesava Raju and M. S. Subramanian, J. Hazard. Mater., 2007, 145, 315 CrossRef PubMed.
- V. K. Jain, R. A. Pandya, S. G. Pillai and P. S. Shrivastav, Talanta, 2006, 70, 257 CrossRef CAS PubMed.
- C. Siva Kesava Raju and M. S. Subramanian, Talanta, 2005, 67, 81 CrossRef PubMed.
- R. S. Praveen, P. Metilda, S. Daniel and T. Prasada Rao, Talanta, 2005, 67, 960 CrossRef CAS PubMed.
- A. Takahashi, Y. Ueki and S. Igarashi, Anal. Chim. Acta, 1999, 387, 71 CrossRef CAS.
- C. Labrecque, S. Potvin, L. Whitty-Léveillé and D. Larivière, Talanta, 2013, 107, 284 CrossRef CAS PubMed.
- A. Aziz, S. Jan, F. Waqar, B. Mohammad, M. Hakim and W. Yawar, J. Radioanal. Nucl. Chem., 2010, 284, 117 CrossRef CAS.
- M. E. Khalifa, Sep. Sci. Technol., 1998, 33, 2123 CrossRef CAS PubMed.
- M. L. Dietz, E. P. Horwitz, L. R. Sajdak and R. Chiarizia, Talanta, 2001, 54, 1173 CrossRef CAS.
- V. Camel, Spectrochim. Acta, Part B, 2003, 58, 1177 CrossRef.
- Y. Zhang, C. Zhong, Q. Zhang, B. Chen, M. He and B. Hu, RSC Adv., 2015, 5, 5996 RSC.
- W. I. Mortada, I. M. M. Kenawy, A. M. Abdelghany, A. M. Ismail, A. F. Donia and K. A. Nabieh, Mater. Sci. Eng., C, 2015, 52, 288 CrossRef CAS PubMed.
- N. Pourreza, S. Rastegarzadeh and A. Larki, J. Ind. Eng. Chem., 2014, 20, 2680 CrossRef CAS PubMed.
- H. Zhang, S. Shuang, G. Wang, Y. Guo, X. Tong, P. Yang, A. Chen, C. Dong and Y. Qin, RSC Adv., 2015, 5, 4343 RSC.
- K. Lee, A. Mazare and P. Schmuki, Chem. Rev., 2014, 114, 9385 CrossRef CAS PubMed.
- G. H. Jeffery, J. Bassett, J. Mendham and R. C. Denney, Vogel's Textbook of Quantitative Chemical Analysis, Longman Scientific and Technical, New York, 5th edn, 1989, p. 474 Search PubMed.
- M. Bahgat, A. A. Farghali, A. F. Moustafa, M. H. Khedr and M. Y. Mohassab-Ahmed, Appl. Nanosci., 2013, 3, 241 CrossRef CAS.
- L. Moberg, K. Pettersson and I. Gustavssonb, J. Anal. At. Spectrom., 1999, 14, 1055 RSC.
- Y. Yu, H.-H. Wu, B.-L. Zhu, S.-R. Wang, W.-P. Huang, S.-H. Wu and S.-M. Zhang, Catal. Lett., 2008, 121, 165 CrossRef CAS.
- V. A. M. Brabers, Phys. Status Solidi, 1969, 33, 563 CrossRef CAS PubMed.
- M. Kanagaraj, P. Sathishkumar, G. K. Selvan, I. P. Kokila and S. Arumugam, Indian J. Pure Appl. Phys., 2014, 52, 124 CAS.
- R. D. Waldron, Phys. Rev., 1955, 99, 1727 CrossRef CAS.
- J. Rouquerol, D. Avnir, C. W. Fairbridge, D. H. Everett, J. M. Haynes, N. Pernicone, J. D. F. Ramsay, K. S. W. Sing and K. K. Unger, Pure Appl. Chem., 1994, 66, 1739 CrossRef CAS.
- X. Li, D. Wang, Q. Luo, J. An, Y. Wang and G. Cheng, J. Chem. Technol. Biotechnol., 2008, 83, 1558 CrossRef CAS PubMed.
- J. Song, H. Kong and J. Jang, J. Colloid Interface Sci., 2011, 359, 505 CrossRef CAS PubMed.
- L. Tan, J. Wang, Q. Liu, Y. Sun, X. Jing, L. Liu, J. Liu and D. Song, New J. Chem., 2015, 39, 868 RSC.
- K. Y. Foo and B. H. Hameed, Chem. Eng. J., 2010, 156, 2 CrossRef CAS PubMed.
- R. Hu, D. Shao and X. Wang, Polym. Chem., 2014, 5, 6207 RSC.
- D. D. Shao, Z. Q. Jiang, X. K. Wang, J. X. Li and Y. D. Meng, J. Phys. Chem. B, 2009, 113, 860 CrossRef CAS PubMed.
- G. X. Zhao, T. Wen, X. Yang, S. B. Yang, J. L. Liao, J. Hu, D. D. Shao and X. K. Wang, Dalton Trans., 2012, 41, 6182 RSC.
- M. Zeng, Y. Huang, S. Zhang, S. Qin, J. Li and J. Xu, RSC Adv., 2014, 4, 5021 RSC.
- Y. G. Zhao, J. X. Li, S. W. Zhang, H. Chen and D. D. Shao, RSC. Adv., 2013, 3, 18952 RSC.
- M. Liu, C. Chen, T. Wen and X. Wang, Dalton Trans., 2014, 7050 RSC.
- L. Tan, J. Wang, Q. Liu, Y. Sun, X. Jing, L. Liu, J. Liu and D. Song, New J. Chem., 2015, 39, 868 RSC.
- T. W. Weber and R. K. Chakkravorti, AIChE J., 1974, 20, 228 CrossRef CAS PubMed.
- A. A. S. Ali, N. M. El Taieb, A. M. A. Hassan, Y. H. Ibrahim and S. G. Abd El Wahab, Journal of American Science, 2011, 7, 347 Search PubMed.
- M. E. Goher, A. M. Hassan, I. A. Abdel-Moniem, A. H. Fahmy and S. M. El-sayed, Egypt. J. Aquat. Res., 2014, 40, 225 CrossRef PubMed.
- W. M. Arafa, W. M. Badawy, N. M. Fahmi, K. Ali, M. S. Gad, O. G. Duliu, M. V. Frontasyeva and E. Steinnes, J. Afr. Earth Sci., 2015, 107, 57 CrossRef CAS PubMed.
- H. S. Ibrahim, M. A. Ibrahim and F. A. Samhan, J. Hazard. Mater., 2009, 168, 1012–1016 CrossRef CAS PubMed.
- A. H. Orabi, E. M. El-Sheikh, A. R. Mowafy, M. Abdel-Khalek and M. Y. El Kady, Int. J. Miner. Process., 2015, 137, 26 CrossRef CAS PubMed.
- CCME, Canadian soil quality guidelines for the protection of environmental and human health: summary tables, in Canadian Environmental Quality Guidelines, CCME, Winnipeg, 2007 Search PubMed.
- United Nations Scientific Committee on the Effects of Atomic Radiation, Sources and Effects of Ionizing Radiation: UNSCEAR 1993 Report to the General Assembly, with Scientific Annexes, United Nations, New York, 1993 Search PubMed.
| This journal is © The Royal Society of Chemistry 2015 |