A simple and economical method using graphene oxide for the fabrication of water/oil separation papers†
Hongyi Qin‡
a,
Tao Gong‡ a,
Yujin Chob,
Cheolmin Shinb,
Changgu Lee*ab and
Taesung Kim*ab
a,
Yujin Chob,
Cheolmin Shinb,
Changgu Lee*ab and
Taesung Kim*ab
aSKKU Advanced Institute of Nano Technology (SAINT), Sungkyunkwan University, Suwon 440-746, South Korea. E-mail: peterlee@skku.edu; tkim@skku.edu
bSchool of Mechanical Engineering, Sungkyunkwan University, Suwon 440-746, South Korea
First published on 29th June 2015
Abstract
We present a facile method using graphene oxide for the preparation of water/oil separation papers. We coated regular paper with graphene oxide by spraying or soaking, and performed thermal annealing at 180 °C for 1–3 hours. The thermal treatment transforms the paper from hydrophilic to hydrophobic due to the thermal reduction of graphene oxide, and thus water can hardly permeate through the separation paper. In contrast, low polarity organic solvents and oil can easily pass through the separation paper. The water/oil separation papers have very good reusable stability. The separation selectivity is above 99% for different mixtures of oily solvents and water.
1. Introduction
Water/oil separation has wide applications in a variety of fields, such as water pollution remediation,1 supply of clean water, and oil purification. With the increase in production of industrial oily wastewater and the stringent regulations on its discharge, the development of water/oil separation materials is important for scientific research, industrial communities and environmental protection.2 Many experimental techniques for water/oil separation have been reported, including the use of super-hydrophobic materials, super-hydrophilic coatings and porous materials.3–6 For example, Rohrbach et al. modified a hydrated regular cellulose paper filter with nano-fibrillated cellulose hydrogel to prepare a water/oil separation filter;7 Zang et al. utilized super-hydrophobic porous copper foams to separate and capture oils from water;8 Tai et al. prepared a carbon–silica nanofibrous membrane for ultrafast oil/water separation,9 etc. However, most of the methods need special materials, complicated equipment and experimental expertise, which are not conducive to cost reduction or scaling up production.In this work, we introduce a simple and low cost method to prepare water/oil separation paper, which can selectively separate oil and water. We coated the regular paper using graphene oxide (GO). And then, the GO coating was thermally reduced to be reduced graphene oxide (RGO). The RGO coating turns regular papers into hydrophobic functional materials. A detailed study on the microstructure and chemical constitution for the RGO separation paper was performed. The separation paper has a good reusable stability and can be recycled. The water/oil separation papers in this study can be used as a scalable and low cost material for water or oil purification.
2. Experimental
2.1 Synthesis of GO
All chemicals were obtained from Sigma-Aldrich. GO was synthesized from natural graphite flakes with the modified Hummer method.10 3 g of graphite powder (Alfa Aesar, 325 mesh, 99.8%), 2.5 g of NaNO3, and 185 g H2SO4 (purity 95%) were placed in a beaker with a stirrer. After 1 hour of cooling in an ice water bath, 12 g KMnO4 was added slowly into the mixture. The mixture was stirred at room temperature for 1 day. 300 mL of 5 wt% H2SO4 aqueous solution was added to the above solution and stirring was continued for 1 hour. Then, 30 wt% H2O2 aqueous solution was slowly added until the mixture became yellow and stirring was continued for one further hour. The products were then washed 20 times with centrifugation and DI water to obtain a GO aqueous solution. The concentration of the GO aqueous solution was diluted to 0.38–1.89 mg mL−1.2.2 Method for the preparation of water/oil separation paper
The paper is soaked in a GO aqueous solution for two minutes or a hand-held spray bottle is used (Fig. 1a and b). Next, the wet paper is placed in an oven with a temperature of 180 °C (Fig. 1c). After 1–3 h, the GO coating is thermally reduced to be RGO coating and the paper becomes hydrophobic. Thus, the fabrication of the water/oil separation paper is completed. If a hand-held spray bottle is used, the paper should be sprayed until fully wetted.3. Results and discussion
The development of low cost and simple techniques has practical significance for practical applications. After years of development, GO has realized commercialization and can be conveniently purchased from chemical companies. In addition, GO is also easily synthesized using the modified Hummer's method in the lab. In this study, only a small amount of GO aqueous solution is required, which is dependent on the size of the paper. The paper can be a filter paper or any tissue paper. Ovens are commonly used in the laboratory and in the kitchen. It is well known that GO can easily be reduced with reducing agents such as sodium borohydride, hydrazine and vitamin C.11,12 However, in order to further reduce the cost and simplify preparation process, thermal reduction was chosen as the means to obtain RGO in this experiment.13 The above factors effectively reduce the cost of water/oil separation papers.Paper is composed of cellulose fibers with hydroxyl and ether groups. GO also contains polar functional groups such as –COOH, –OH and epoxy. Hence, cellulose fibers interact strongly with GO sheets through hydrogen bonding. GO can be thermal reduced to be graphene. Graphene has a hydrophobic surface because of hydrocarbon contamination from the air, as reported by Li et al.14 Hydrophobic materials repel water and permit oil to flow through them.2 Therefore, if GO covering the surfaces of cellulose fibers is reduced by thermal treatment, the substrate paper will be endowed with hydrophobic ability.
As shown in Fig. 2a, the pure filter paper exhibits hydrophilic behavior. In contrast, the separation paper becomes grey because of the black RGO coating. Water cannot wet the separation paper as indicated by its contact angle of 89.2° (Fig. 2b, insert photo). Tissue paper can also be treated with this method and exhibits a contact angle of ∼90° (Fig. S1a and b†). The surface morphologies of pure paper and separation paper are shown in the scanning electron microscopy (SEM) images (Fig. 2c and d, S1c and d†). The cellulose fibers in the separation paper are covered with an RGO film due to hydrogen bonding. The paper has a rough texture and a porous structure because it consists of cellulose fibers. The cellulose fibers are hydrophilic material. Therefore, pure paper has a strong water-absorbing capacity (Fig. 2a). However, in the separation paper, the cellulose fibers are covered with hydrophobic RGO sheets, which endow the substrate paper with hydrophobicity. Note that the rough texture on the surface can enhance the intrinsic hydrophobicity due to the “lotus effect” or the “petal effect”.15 A rough surface with special micro- and nano-structures and a low surface energy can exhibit super-hydrophobicity. According to previous reports,16–18 the lotus leaf has a series of micro-size protrusions, which gives rise to a heterogeneous surface composed of both air and solid. As a result, the adhesive force between the water and the lotus leaf is extremely low, which means that water rolls off the leaf easily, the so-called “self-cleaning” phenomenon. The rose petal's micro- and nano-structures are larger in scale than those of the lotus leaf, so water can enter the larger grooves, but it cannot enter the smaller grooves. Therefore, the adhesive force between the water and the rose petal is high. As a result, the water droplet will not fall off even if the petal is turned upside down. In the surface of the separation paper prepared in this study, there are micro-size texture and grooves, so the water droplet can enter the larger grooves, but it cannot completely wet the hydrophobic surface of the RGO–cellulose fibers (Fig. 2e and f). Therefore, this separation paper also exhibits the “petal effect”, as shown in Fig. 2g and h, in which the water droplet still sticks to the surface of the separation paper even if it is rotated 180°. In the RGO separation paper, the petal structure prevents water from permeating through the paper, but oily solvents can penetrate due to the lipophilicity of the RGO surface.
Changes in the preparation conditions have significant influence on the water/oil separation performance. Some key factors in the preparation, such as the annealing time and the temperature, were varied and their influence was tested by measuring of the water contact angle. The temperature has a significant effect on the thermal reduction of GO, which directly influences the hydrophobicity of the surface of the separation paper. Theoretically, a higher temperature is expected to produce a greater reduction of GO, and thus to increase the hydrophobicity of the separation paper. As shown in Fig. 3, the contact angles of the filter papers depend on the temperatures of thermal reduction. The contact angles are 39.5°, 58.1°, and 89.4° after thermal treatment for 3 hours at 140 °C, 160 °C, and 180 °C, respectively. If the temperature of thermal reduction is less than 140 °C, the contact angle on the paper cannot be measured, as the water droplets are completely absorbed into the paper. However, if the temperature is more than 200 °C, the paper is oxidized and becomes brown (Fig. S2†). Therefore, 180 °C is a suitable treatment condition.
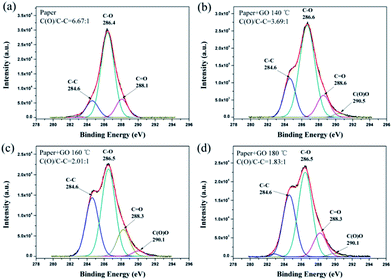
X-ray photoelectron spectroscopy (XPS) was used to further study the changes in the elemental composition of the paper after thermal treatment (Fig. 4). In the pure filter paper, there are three kinds of carbon bonding, C–C, C–O, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O. The intensity ratio of the carbon–oxygen bonds C(O) to other C–C bonds is 6.67. The water/oil separation paper contains new carbon bonds from GO, carboxyl C(O)O. Moreover, the intensity ratio of C(O)/C–C decreases with increase in the temperature: 3.69, 2.01, and 1.83 corresponding treatment temperatures: 140 °C, 160 °C, and 180 °C, respectively, which indicates that reduction of GO is greater at higher temperature. The Raman spectra measurements also show that the relative intensity of the D-band and the number of defects increase with temperature, as shown in Fig. S3a.†19 Attenuated total reflectance Fourier transform infrared spectroscopy (ATR-FTIR) (Fig. S3b†) produces two similar spectra, and only the COOH peak from RGO at 1728 cm−1 is slightly enhanced in the treated paper, which indicates that the thermal treatment at 180 °C does not alter the chemical structure of the paper. In addition, the influences of treatment time and the weight percentage (wt%) of the RGO were investigated with contact angle measurements, as shown in Fig. S4 and S5.† The time of thermal treatment was extended from 1 hour to 10 hours. The weight percentage of RGO in the paper was tuned from 0.063 wt% to 0.315 wt%. However, no significant variation with treatment duration and weight percentage was found in the contact angle. The contact angles of all the samples range from 82° to 88°. Therefore, the temperature is the most important parameter for the fabrication of this kind of water/oil separation paper.
O. The intensity ratio of the carbon–oxygen bonds C(O) to other C–C bonds is 6.67. The water/oil separation paper contains new carbon bonds from GO, carboxyl C(O)O. Moreover, the intensity ratio of C(O)/C–C decreases with increase in the temperature: 3.69, 2.01, and 1.83 corresponding treatment temperatures: 140 °C, 160 °C, and 180 °C, respectively, which indicates that reduction of GO is greater at higher temperature. The Raman spectra measurements also show that the relative intensity of the D-band and the number of defects increase with temperature, as shown in Fig. S3a.†19 Attenuated total reflectance Fourier transform infrared spectroscopy (ATR-FTIR) (Fig. S3b†) produces two similar spectra, and only the COOH peak from RGO at 1728 cm−1 is slightly enhanced in the treated paper, which indicates that the thermal treatment at 180 °C does not alter the chemical structure of the paper. In addition, the influences of treatment time and the weight percentage (wt%) of the RGO were investigated with contact angle measurements, as shown in Fig. S4 and S5.† The time of thermal treatment was extended from 1 hour to 10 hours. The weight percentage of RGO in the paper was tuned from 0.063 wt% to 0.315 wt%. However, no significant variation with treatment duration and weight percentage was found in the contact angle. The contact angles of all the samples range from 82° to 88°. Therefore, the temperature is the most important parameter for the fabrication of this kind of water/oil separation paper.
Water/oil separation through these papers is relatively fast due to their porous structures. Gravity can drive this water/oil separation process under normal atmospheric pressure. As shown in Fig. 5a and b, hexane is filtered easily by this separation paper, but water does not pass through it. Treated tissue paper can also be used as separation paper (Fig. S6†), but because it is soft and weak, at least two layers of paper are required for water/oil separation. Tissue paper is thinner and more porous, so separation is faster. The separation fluxes of separation paper for pure various organic solvents were measured using filter apparatus in Fig. 5c, the area of separation paper is ∼12.5 cm2, the volume of pure organic solvents is 20 mL, and the flux can be calculated by the filtration time. The average separation flux of filter paper is ∼358.2 L h−1 m−2. The average separation flux of tissue paper for organic solvents is ∼3821.6 L h−1 m−2, about one order higher. Generally, the viscosities of organic solvents at room temperature are relatively low (hexane ∼ 0.31 cP; chloroform ∼ 0.57 cP; dichloromethane ∼ 0.44 cP; toluene ∼ 0.59 cP). However, mineral oil has quite a high viscosity (∼16 cP),20 which greatly affects its separation flux. The separation flux of filter paper for mineral oil is ∼4.5 L h−1 m−2, and that of tissue paper for mineral oil is ∼81.9 L h−1 m−2. For the separation process of water/oil mixtures in the cone shaped separation paper, as shown in Fig. 5a and b, the separation flux can be measured by the volume of filtrate per unit time. The separation paper has similar average separation flux for the organic solvent–water mixtures (filter paper ∼ 0.54 L h−1, tissue paper ∼ 6.23 L h−1). The mineral oil–water mixture has a decreased separation flux (filter paper ∼ 0.008 L h−1, tissue paper ∼ 0.16 L h−1). It is found that the viscosity of liquid and porosity of separation paper have a huge influence on the separation flux.
To further investigate the separation performance of the prepared separation paper, the separation selectivity was determined by comparing the change in water weight in water/oil mixture before and after separation. 10 mL water and 10 mL oil were used for the test. As shown in Table S1,† the separation selectivity with respect to water and oil is quite high, above 99%, which indicates that the separation paper does not permit any appreciable amount of water to pass through its pores. Moreover, the use of different organic solvents and oil does not affect the separation selectivity. Due to lipophilicity of the separation paper, the adsorption is inevitable in the water/oil separation process. As shown in Table S1,† the loss ratio of low boiling point organic solvents is ranging from 5.24% to 8.41%, which is due to the combined effect of adsorption and evaporation. However, the viscous mineral oil has a higher loss ratio (19.51%), indicating the interaction and adsorption between mineral oil and lipophilic separation paper are quite strong. It is also a potential application for the separation paper in the clean-up of oil spill.21,22
The reusable stability of separation papers is also an important feature for reducing cost and environmental protection. In this study, the stability of separation paper was evaluated by elemental analysis (EA). The change in C/O atomic ratio of separation paper in water/oil separation process can be measured by EA, as shown in Table S2.† The C/O atomic ratio of pure filter paper is 1.093 and that of separation paper is increased a little, 1.166, which is due to the coated RGO sheets. However, there is almost no any change in C/O atomic ratio during 5 times water/oil separation tests (1.165–1.171), indicating the RGO coating and hydrophobicity of separation paper is very stable in separation process. Moreover, the selectivity of water and flux were not obviously affected by the multiple separation tests. Therefore, this is a reliable technique for the preparation of reusable water/oil separation papers.
Due to the hydrophobic properties of the porous paper, the water stays on top of the paper. However, if an external pressure (an intrusion pressure) is applied, the water will intrude into the pores of the paper. Intrusion pressure can arise from the gravitational force on the water and thus the maximum height of water that the separation paper can support can be calculated.23–26 As shown in Fig. 5c, the maximum height of water is approximately 9 cm, so the corresponding intrusion pressure is approximately 0.88 kPa. The theoretical intrusion pressure (Ptheor) can be calculated by using the Young–Laplace eqn (1):
Ptheor = 2γ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ/d θ/d
| (1) |
4. Conclusions
A facile and low cost method for the preparation of water/oil separation paper has been developed, using GO aqueous solution, paper, and an oven. After thermal reduction at 180 °C for 1–3 hours, the separation paper exhibits hydrophobic behavior and has a water contact angle of ∼90°. Low interfacial energy solvents and oil are filtered freely, but water is not. This separation is due to the hydrophobicity of the surface and the petal effect. Tissue paper was found to exhibit one order higher separation fluxes than filter paper because of its larger pore size. Hence, by controlling the size of pores or the paper structure, the separation efficiency can be improved. The separation papers have stable performance in recycling water/oil separation process. This method can be used in a variety of applications that require high separation rates and low costs. One example is the cleaning of ship oil spills in the ocean, in which GO coated papers could be used to draw out oil from contaminated seawater and then be recycled.Acknowledgements
This study was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF-2013R1A1A2058893) and National Research Foundation of Korea (NRF) grants funded by the Korean government Minisvtry of Science, ICT and Future Planning (MSIP) (No. 2011-0014209).Notes and references
- Q. Zhu, Q. Pan and F. Liu, J. Phys. Chem. C, 2011, 115, 17464–17470 CAS.
- Z. Xue, Y. Cao, N. Liu, L. Feng and L. Jiang, J. Mater. Chem. A, 2014, 2, 2445–2460 CAS.
- C. H. Lee, N. Johnson, J. Drelich and Y. K. Yap, Carbon, 2011, 49, 669–676 CrossRef CAS PubMed.
- L. Feng, Z. Zhang, Z. Mai, Y. Ma, B. Liu, L. Jiang and D. Zhu, Angew. Chem., Int. Ed., 2004, 43, 2012–2014 CrossRef CAS PubMed.
- G. Wang, Y. He, H. Wang, L. Zhang, Q. Yu, S. Peng, X. Wu, T. Ren, Z. Zeng and Q. Xue, Green Chem., 2015, 17, 3093–3099 RSC.
- Y. Si, Q. Fu, X. Wang, J. Zhu, J. Yu, G. Sun and B. Ding, ACS Nano, 2015, 9, 3791–3799 CrossRef CAS PubMed.
- K. Rohrbach, Y. Li, H. Zhu, Z. Liu, J. Dai, J. Andreasen and L. Hu, Chem. Commun., 2014, 50, 13296–13299 RSC.
- D. Zang, C. Wu, R. Zhu, W. Zhang, X. Yu and Y. Zhang, Chem. Commun., 2013, 49, 8410–8412 RSC.
- M. H. Tai, P. Gao, B. Y. L. Tan, D. D. Sun and J. O. Leckie, ACS Appl. Mater. Interfaces, 2014, 6, 9393–9401 CAS.
- H. Qin, T. Gong, Y. Cho, C. Lee and T. Kim, Polym. Chem., 2014, 5, 4466–4473 RSC.
- S. Pei and H.-M. Cheng, Carbon, 2012, 50, 3210–3228 CrossRef CAS PubMed.
- C. K. Chua and M. Pumera, Chem. Soc. Rev., 2014, 43, 291–312 RSC.
- W. Chen and L. Yan, Nanoscale, 2010, 2, 559–563 RSC.
- Z. Li, Y. Wang, A. Kozbial, G. Shenoy, F. Zhou, R. McGinley, P. Ireland, B. Morganstein, A. Kunkel and S. P. Surwade, Nat. Mater., 2013, 12, 925–931 CrossRef CAS PubMed.
- L. Jiang, Y. Zhao and J. Zhai, Angew. Chem., 2004, 116, 4438–4441 CrossRef PubMed.
- B. Bhushan and M. Nosonovsky, Philos. Trans. R. Soc., A, 2010, 368, 4713–4728 CrossRef CAS PubMed.
- L. Feng, Y. Zhang, J. Xi, Y. Zhu, N. Wang, F. Xia and L. Jiang, Langmuir, 2008, 24, 4114–4119 CrossRef CAS PubMed.
- K. Uchida, N. Nishikawa, N. Izumi, S. Yamazoe, H. Mayama, Y. Kojima, S. Yokojima, S. Nakamura, K. Tsujii and M. Irie, Angew. Chem., Int. Ed., 2010, 49, 5942–5944 CrossRef CAS PubMed.
- D. Zhan, Z. Ni, W. Chen, L. Sun, Z. Luo, L. Lai, T. Yu, A. T. S. Wee and Z. Shen, Carbon, 2011, 49, 1362–1366 CrossRef CAS PubMed.
- E. Piccin, D. Ferraro, P. Sartori, E. Chiarello, M. Pierno and G. Mistura, Sens. Actuators, B, 2014, 196, 525–531 CrossRef CAS PubMed.
- L. Kong, X. Chen, L. Yu, Z. Wu and P. Zhang, ACS Appl. Mater. Interfaces, 2015, 7, 2616–2625 CAS.
- L. Peng, S. Yuan, G. Yan, P. Yu and Y. Luo, J. Appl. Polym. Sci., 2014, 131, 40886 Search PubMed.
- W. Zhang, Z. Shi, F. Zhang, X. Liu, J. Jin and L. Jiang, Adv. Mater., 2013, 25, 2071–2076 CrossRef CAS PubMed.
- Z. Xue, S. Wang, L. Lin, L. Chen, M. Liu, L. Feng and L. Jiang, Adv. Mater., 2011, 23, 4270–4273 CrossRef CAS PubMed.
- Q. Wen, J. Di, L. Jiang, J. Yu and R. Xu, Chem. Sci., 2013, 4, 591–595 RSC.
- Z. Xue, Z. Sun, Y. Cao, Y. Chen, L. Tao, K. Li, L. Feng, Q. Fu and Y. Wei, RSC Adv., 2013, 3, 23432–23437 RSC.
- Y. Yao and D. Liu, Fuel, 2012, 95, 152–158 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: Fig. S1–S7 and Table S1 and S2. See DOI: 10.1039/c5ra08738d |
| ‡ These two authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2015 |