Optimizing the photocatalysis in ferromagnetic Bi6Fe1.9Co0.1Ti3O18 nanocrystal by morphology control†
Wen
Ge
a,
Zhengping
Fu
ac,
Xiaoning
Li
a,
Jianlin
Wang
a,
Zhu
Zhu
a,
Min
Liu
*ac,
Ranran
Peng
ac and
Yalin
Lu
*abcd
aCAS Key Laboratory of Materials for Energy Conversion, Department of Materials Science and Engineering, University of Science and Technology of China, Hefei 230026, P. R. China. E-mail: liumin1106@ustc.edu.cn; yllu@ustc.edu.cn
bHefei National Laboratory for Physical Sciences at the Microscale, University of Science and Technology of China, Hefei 230026, P. R. China
cSynergetic Innovation Center of Quantum Information & Quantum Physics, University of Science and Technology of China, Hefei 230026, P. R. China
dLaser Optics Research Center, Physics Department, United States Air Force Academy, Colorado 80840, USA
First published on 15th June 2015
Abstract
Size and morphology are critical to monitoring the properties of nanocrystals. In this work, Bi6Fe1.9Co0.1Ti3O18 (BFCTO) nanocrystals, which are a visible-light active photocatalyst with room temperature ferromagnetism, were successfully synthesized through a hydrothermal process. Furthermore, the morphology dependent magnetism and band gap of the BFCTO-1.00/BFCTO-1.50/BFCTO-2.00 nanocrystals are studied by adjusting the NaOH concentration to 1.00 M/1.50 M/2.00 M in the hydrothermal process, respectively. As the {117} facet ratio increases, the absorption edge has an obvious red-shift by 32 nm and the corresponding bandgap is reduced from 2.58 to 2.42 eV. Remarkably, the specific surface area normalized degradation rate of BFCTO-1.50 was considerably higher than those of BFCTO-1.00 and BFCTO-2.00 due to the optimal ratio of {001} and {117} facets.
1. Introduction
The problem of environment pollution is getting worse with the increase of the human population and the abuse of natural resources.1–5 A possible way to treat this problem is to decompose the contaminants by utilizing solar energy with effective photocatalysts; in this case, nano-sized particles are usually adopted to improve the efficiency.6–8 It has been well known that the properties of the nanoparticles are closely related to not only the particle size, but also the morphology.9–12 The morphology can improve the photocatalysis properties of nanoparticles by two mechanisms: the first is to expose the crystal with more reactive facets;13–15 the second mechanism is the synergistic effect between the separated reduction and oxidation facets. For example, T. Ohno et al. found that the rutile TiO2 {110} and {011} facets act as efficient reduction and oxidation sites, respectively. Because of the synergistic effect between the {110} and {011} facets, the rutile TiO2 particles are considered to be efficient for some photocatalytic reactions.16 Though the synergistic effect between the separated reduction and oxidation facets has been proved to be successful in modulating the photocatalysis properties of some oxide,17–21 much works are to be done to extend this concept to more materials, in order to explore more efficient photocatalysts.Layer-structured bismuth-containing Aurivillius complex oxides, with a general formula Bin+1Fen−3Ti3O3n+3 (n denotes the number of perovskite layers), are alternately stacked with fluorite-type (Bi2O2)2+ layers and perovskit-etype (Bin−1Fen−3Ti3O3n+1)2− slabs. By doping ions or changing number of perovskite layers, we can modulate their ferroelectric and ferromagnetic properties.22–24 Recent research have found that the Aurivillius phase oxide are possible to integrate the visible light photocatalysis and multiferroic properties in single phase material, which is important to recycle usage of the nano-sized photocatalysts.25–28 Besides the photocatalysis performance, the multiferroic properties of the Aurivillius nanoparticles can also be improved by changing the size and morphology.21,29 Therefore, it is desirable to modulate the morphology of Aurivillius nanoparticles, in order to optimize their performance in photocatalysis and multiferroic. Presently, various methods, including sol–gel,30 Pechini's31 and hydro(solvo)thermal method27 for the preparation of nanosized Aurivillius phase particles have been employed. Remarkable, the hydro(solvo)thermal method is a typical solution-based chemical synthesis approach utilized solvent under pressure and temperature above its critical point to increase the solubility and reactivity of reactants and excellent controls over particle size and morphology.32 In hydro(solvo)thermal process, the size and morphology of nanoparticles can be effectively controlled by several factors, such as pH value, temperature, reaction time, precursor, surfactants and mineralizers, especially, the molar concentration of NaOH has attracted considerable attention due to OH− ions can affect the nucleation and growth behaviors of crystal, namely OH− ions can serve as capping agents and adsorb on certain faces of oxide crystals, which may create additional growth anisotropy and direct the crystal growth as well.33,34
In this manuscript, we report a facile hydrothermal method for synthesis of single-crystalline Bi6Fe1.9Co0.1Ti3O18 (BFCTO) nanocrystals with various morphology and size by adjusting alkaline concentration. Remarkably, the specific surface area normalization of degradation rate of BFCTO-1.50 showed considerably higher than BFCTO-1.00 and BFCTO-2.00. This could be explained as follows: (i) with the increase of NaOH concentration, the {117} facets increase remarkably, and the corresponding band gap is shorten, suggesting that the conducting band edge of {117} facets is lower than that of {001} facets. Thereby the photogenerated electrons may preferably diffuse to the {117} facets and the photogenerated holes will diffuse to the {001} facets, leading to the separation of electrons and holes; (ii) in BFCTO-1.50 sample, a similar oxidation and reduction reaction ratio on these two facets may appear due to more appropriate area ratio between the {001} facets and the {117} facets. Therefore, the synergistic effect of {001} and {117} facets in the separation of electrons and holes and oxidation/reduction reaction efficiently inhibit the recombination of the charge carrier, and thereby the BFCTO-1.50 sample shows the highest photocatalysis efficiency.
2. Materials and methods
2.1 Materials
All the chemicals were purchased from Sinopharm Chemical Reagent Co., Ltd. without further purification. Ti(OC4H9)4 (≥99.7%), Bi(NO3)3·5H2O (≥99%), Fe(NO3)3·9H2O (≥98.5%), Co(NO3)3·6H2O (≥98.5%), HNO3 (65.0–68.0%), NaOH (≥96.0%), ethanol (99.7%).2.2 Synthesis of different morphologies BFCTO nanoparticles
The different morphologies BFCTO nanoparticles were synthesized by the hydrothermal method. The detailed process is as follows: 0.483 g of Ti(OC4H9)4, 1.377 g of Bi(NO3)3·5H2O, 0.363 g of Fe(NO3)3·9H2O and 0.014 g of Co(NO3)3·6H2O were dissolved in 5 mL HNO3 (4 M) and stirred 20 min to form a transparent and uniform solution. Then NaOH solutions were added to the above solution and the alkaline concentration were adjusted to 1.00 M, 1.50 M and 2.00 M. After stirring for 30 min, the uniform solution was transferred to a 50 mL Teflon-lined stainless steel autoclave up to 80% of the total volume. The autoclave was sealed and heated at 200 °C for 48 h and then cool to room temperature. The BFCTO nanoparticles were obtained by centrifugation and washing three times with deionized water and ethanol, respectively. The final product was obtained after dried under vacuum at 60 °C for 12 h.2.3 Photocatalytic activity evaluation
Photocatalytic activities of the as-prepared samples were evaluated by the photodecomposition of RhB under visible-light irradiation of a 20 W fluorescent lamp with wavelength of 400–720 nm. The irradiation distance between the lamp and the sample was 10 cm. The BFCTO sample (50 mg) was dispersed uniformly into 50 mL of RhB solution (5 mg L−1). Before irradiation, the suspension was stirred for 30 min in the dark to ensure the establishment of an adsorption/desorption equilibrium. At a certain time interval, 3 mL of the reaction solution was taken and centrifuged. The filtrates were measured on a UV-Visible spectrometer at a maximum absorption wavelength of 554 nm.2.4 Characterizations
Phase structure and crystallinity of the samples were characterized by X-ray diffraction (XRD), Philips X'pert diffractometer employing the Cu Kα radiation (λ = 1.5405 Å). Morphologies of samples were observed by scanning electron microscopy (SEM, JEOL, JSM-6700F). Low-resolution, high-resolution transmission electron microscopy (TEM, HRTEM) and selected-area electron diffraction (SAED) measurements were carried out on a JEOL-JEM 2010 transmission electron microscope operating at an acceleration voltage of 200 KV. Ultraviolet-visible-near infrared (UV-Visible-NIR) diffuse reflectance spectra were recorded with a Shimadzu SolidSpec-3700 equipped with an integrating sphere, and BaSO4 was used as the reference. Magnetic properties of the resulting samples were characterized using a physical property measurement system (PPMS DynaCool, Quantum Design). The Brunauer–Emmett–Teller (BET) surface area was determined by nitrogen sorption using a Micromeritics TriStar II 3020 V1.03.3. Results and discussion
The purity and crystallinity of the as-prepared BFCTO samples obtained by adjusting NaOH concentrations in precursor solutions were examined by XRD and shown in Fig. 1(a). All the diffraction peaks of the samples can be indexed by BFCTO with an orthorhombic lattice, which was consistent with the previous reported results in five-layered Aurivillius phase (Fig. 1(b)).22 The XRD patterns indicated that the samples are mainly of BFCTO with the secondary phases under the detecting limits. We refined the structural parameters by using the Pawley method. As an example, the experimental and calculated XRD patterns of BFCTO-2.00 were showed in Fig. S1.† The fit between the experimental and calculated XRD patterns was relatively good based on the consideration of the value of Rwp and Rp were 13.28% and 21.74%, respectively. Meanwhile, as the NaOH concentration increase, the crystal crystallization get better, while the main diffraction peaks (11![[1 with combining low line]](https://www.rsc.org/images/entities/char_0031_0332.gif)
![[1 with combining low line]](https://www.rsc.org/images/entities/char_0031_0332.gif) ) narrowed, indicating an increase of the average crystallite size, and this conclusion can be demonstrated by the corresponding SEM images.
) narrowed, indicating an increase of the average crystallite size, and this conclusion can be demonstrated by the corresponding SEM images.
 | ||
| Fig. 1 (a) The XRD patterns of BFCTO-1.00, BFCTO-1.50 and BFCTO-2.00 samples; (b) the schematic illustration of BFCTO crystal structure along the [010] direction. | ||
The representative SEM images of BFCTO-1.00, BFCTO-1.50 and BFCTO-2.00 (Fig. 2(a)–(c)) displayed various well-defined shapes of nanosheet, nanoplate and truncated tetragonal bipyramid, respectively. At low NaOH concentration of 1.00 M, we observed nanosheets with irregular shapes (Fig. 2(a)). The thickness had an average size of 40 nm, with the edge length ranging from 100 nm to 1 μm. As the NaOH concentration increase from 1.50 M to 2.00 M, the main morphology changed from regular nanoplates to truncated tetragonal bipyramid particles with the thickness and edge length ranging from 100 nm to 1 μm and 1 μm to 2 μm, respectively (Fig. 2(b) and (c)). As shown in Fig. S2,† the specific surface area obtained with BET method decrease from the 13.98 m2 g−1 for BFCTO-1.00 to 2.26 m2 g−1 for BFCTO-1.50, and then to 1.92 m2 g−1 for BFCTO-2.00 sample, in agreement with the size evolution of the samples. Therefore, the NaOH concentration in precursor solutions had been found to be important for morphological control. In addition, the low-magnification TEM images (Fig. 3(a)–(c)) demonstrated the typical profiles of above-mentioned BFCTO samples. To investigate the morphology transformation process from nanosheets to truncated tetragonal bipyramid particles, more detailed structural information were revealed by HRTEM and SAED. The HRTEM and SAED images were showed in Fig. 3(d)–(i) taken from their corresponding TEM images indicated by red rectangle with number 1 and number 2. The HRTEM image in Fig. 3(d) recorded along the [001] zone axis of a nanosheet shows the lattice fringes with spacings of 0.273, 0.273 and 0.383 nm can be attributed to the (200), (020) and (110) plane of BFCTO orthorhombic phase, respectively. The corresponding SAED pattern confirmed the formation of well-developed single-crystalline of BFCTO. By measuring the HRTEM image of laterally-viewed of nanosheet in Fig. 3(g), it displayed five perovskite layers between two bismuth oxide layers. And the inserted SAED pattern can be ascribed to the [110] zone axis. Meanwhile, we found that the BFCTO-1.50 nanoplates (Fig. 3(b)) had the similar structural character with that of BFCTO-1.00 nanosheets displayed in Fig. 3(e) and (h). Besides, there is an interfacial angle between the lateral surface and the (001) facet (Fig. 2(b). When NaOH concentration reaching to 2.00 M, we obtained the BFCTO-2.00 product with truncated tetragonal bipyramid morphology (Fig. 2(c)). The TEM image indicated the laterally-viewed hexagonal shape and top-viewed square shape (Fig. 3(c)). Statistically, from the laterally-viewed TEM image, the average interfacial angle between the top parallel surface and the lateral surface was about 61.5° as shown in Fig. 3(c), which matches well with the theoretical value of the angle between the {001} and {117} facets of orthorhombic BFCTO. Furthermore, we showed the method determining the {117} facets in ESI.† The HRTEM images of the two opposite corners were displayed in Fig. 3(f) and (i), indicating the truncated tetragonal bipyramid particles grew along the [001] direction and gradually formed {117} facets. And the corresponding SAED pattern in the inset of Fig. 3(f) showed sharp diffraction spots, indicating the good crystallinity.
 | ||
| Fig. 2 The representative SEM images of (a) BFCTO-1.00 (b) BFCTO-1.50 and (c) BFCTO-2.00 (insert: the corresponding schematic drawing morphology). | ||
In the hydrothermal synthesis of oxide crystals, the nucleation rate and crystal growth speed have great effect on the size and morphology of product during the hydrothermal ripening process.35,36 If the rate of crystal nucleation is greater than that of crystal growth, the crystal will be small and the aspect ratio will be low; otherwise they will be large with a high aspect ratio along preferential directions. In this experiment, a low NaOH concentration facilitated a fast nucleation and formation of a large number of crystal nuclei, which resulted in crystals with a low aspect ratio and small size; a high NaOH concentration led to crystals with a high aspect ratio and large size because of fast crystal growth. Importantly, we obtained the well-defined truncated tetragonal bipyramid BFCTO-2.00 particles enclosed by eight predominantly exposed isosceles trapezoidal high-index {117} facets and two square {001} facets. Herein, the formation of high-index orthorhombic {117} facets might be due to the synergistic effects of thermodynamic and kinetic factors that control the crystal nucleation and subsequent growth.37
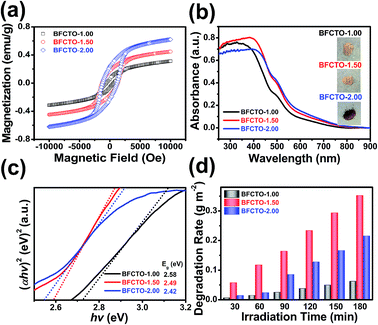
To study the magnetic behavior of BFCTO samples with different morphologies at room temperature, we performed the room temperature M − H hysteresis loops and temperature dependent ZFC and FC magnetization curves under a magnetic field of 500 Oe in Fig. 4(a). In Fig. 4(a), it showed that all samples have spontaneous magnetic moments, indicating the ferromagnetic nature. The Mr enhanced with the increase of NaOH concentration, and reached a peak value of ∼0.23 emu g−1 with a coercive field (Hc) of 1189 Oe for the BFCTO-2.00 sample. This phenomenon can be attributed to the enhancement of crystal crystallization and crystal growth completeness along the [001] direction with NaOH concentration increase. Meanwhile, we observed a divergence between the ZFC and FC magnetization curves in Fig. S3,† indicating a typical spin glass-like behavior.24
The UV-Vis diffuse reflectance spectra in the wavelength range of 250–900 nm for the BFCTO products with various morphologies are displayed in Fig. 4(b). The BFCTO products have a broad absorbance in the visible-light range because of Fe and Co ions doping, which is consistent with the color of the samples (insert of the Fig. 4(b)). The band gap values were calculated using the UV-Vis absorption spectra according to the following equation: αhv = A(hv − Eg)n/2, where α, hv, A, and Eg signify the absorption coefficient, photo energy, proportionality constant and gap band, respectively. The value of n depends on whether the electron transition is direct (n = 1) or indirect (n = 4). The BFCTO products possess a direct transition between bands, then n = 1. The energy of the band gap is calculated by extrapolating a straight line to the abscissa axis. Fig. 4(c) showed the plot of the (αhv)2 verse Eg.27,38 The band gaps of products were estimated to be 2.58 eV for BFCTO-1.00, 2.49 eV for BFCTO-1.50 and 2.42 eV for BFCTO-2.00. With a remarkable increase in {117} facet, the absorption edge has an obvious red-shift by 32 nm and the corresponding bandgap decrease from 2.58 to 2.42 eV.
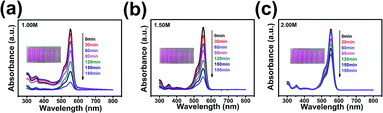
The photocatalytic activities of the prepared BFCTO samples were evaluated by photodegradation of RhB under visible-light illumination. The temporal evolution of absorption spectra changes taking place during the photodegradation of RhB over various morphology BFCTO particles were displayed in Fig. 5. To identify the morphology dependence of the photocatalysis performance, the degradation rate of all the samples were normalized by the specific surface area and shown in Fig. 4(d). It can be seen that BFCTO-1.50 showed considerably higher photocatalysis efficiency than BFCTO-1.00 and BFCTO-2.00. Based on the bandgap evolution for the samples and similar to the case of anatase,20 we infer that conducting band edge of {117} facets is lower than that of {001} facets (Fig. 6), the photogenerated electrons may preferably diffuse to the {117} facets and react with O2 in the solution to form the strong superoxide anion radical O2−˙, while the photogenerated holes will diffuse to the {001} facets and react with H2O to form hydroxyl radical OH˙, leading to the separation of electrons and holes. On the other hand, with the increase of {117} facets in BFCTO-1.50 sample, appropriate area ratio between the {001} facets and the {117} facets may appear, which results in a similar oxidation and reduction reaction ratio on these two facets. Therefore, the synergistic effect of {001} and {117} facets in the separation of electrons and holes and oxidation/reduction reaction efficiently inhibit the recombination of the charge carrier, and thereby the BFCTO-1.50 sample shows the highest photocatalysis efficiency.
 | ||
| Fig. 6 The BFCTO-1.50 sample schematic representation of charge separation between {001} and {117} facets. | ||
In order to evaluate the photocatalytic efficiency of BFCTO samples, it is very important to get the information about the turnover number (TON) and the turnover frequency (TOF) of the catalyst, which are usually calculated by formulas (1) and (2), respectively. In this work, the BFCTO sample (50 mg) was dispersed uniformly into 50 mL of RhB solution (5 mg L−1) and the irradiation time is 180 min, and the conversions are calculated according to Fig. 5, which are 87.35%, 79.57% and 41.35% for BFCTO-1.00, BFCTO-1.50 and BFCTO-2.00, respectively. Consequently, the detailed TON and TOF values were listed in Table 1.
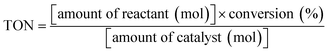 | (1) |
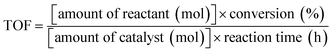 | (2) |
| Samples | TON | TOF (h−1) |
|---|---|---|
| BFCTO-1.00 | 0.0160 | 0.0053 |
| BFCTO-1.50 | 0.0146 | 0.0049 |
| BFCTO-2.00 | 0.0076 | 0.0025 |
4. Conclusion
In summary, we have explored a facial hydrothermal method for the synthesis of various morphology BFCTO nanocrystals by adjusting alkaline concentration. This tunable morphology single-crystalline BFCTO semiconductor is a novel visible light-driven photocatalyst. Meanwhile, we obtained the truncated tetragonal bipyramid BFCTO nanocrystal with high-index exposed {117} facets at 2.00 M NaOH concentration and the corresponding remnant magnetization enhanced. As the {117} facet increases, the intrinsic absorption edge has an obvious red-shift and the corresponding bandgap is shorten. Remarkably, the specific surface area normalization of degradation rate of BFCTO-1.50 showed considerably higher than BFCTO-1.00 and BFCTO-2.00.Acknowledgements
This work was supported by the National Basic Research Program of China (973 Program, 2012CB922001), the Natural Science Foundation of China (51102224), the Key Research Program of Chinese Academy of Sciences (Grant no. KGZD-EW-T06) Anhui Provincial Natural Science Foundation (1408085QE84), and the Fundamental Research Fund for the Central Universities (WK 2060140014). Dr Lu appreciates the support from AFOSR and DTRA (HDTRA12221).Notes and references
- H. L. Wang, L. S. Zhang, Z. G. Chen, J. Q. Hu, S. J. Li, Z. H. Wang, J. S. Liu and X. C. Wang, Chem. Soc. Rev., 2014, 43, 5234 RSC.
- X. C. Wang, K. Maeda, A. Thomas, K. Takanabe, G. Xin, J. M. Carlsson, K. Domen and M. Antonietti, Nat. Mater., 2009, 8, 76 CrossRef CAS PubMed.
- W. Zhao, W. H. Ma, C. C. Chen, J. C. Zhao and Z. G. Shuai, J. Am. Chem. Soc., 2004, 126, 4782 CrossRef CAS PubMed.
- S. Sakthivel and H. Kisch, Angew. Chem., Int. Ed., 2003, 42, 4908 CrossRef CAS PubMed.
- A. Mills, R. H. Davies and D. Worsley, Chem. Soc. Rev., 1993, 22, 417 RSC.
- T. R. Gordon, M. Cargnello, T. Paik, F. Mangolini, R. T. Weber, P. Fornasiero and C. B. Murray, J. Am. Chem. Soc., 2012, 134, 6751 CrossRef CAS PubMed.
- H. G. Yang, G. Liu, S. Z. Qiao, C. H. Sun, Y. G. Jin, S. C. Smith, J. Zou, H. M. Cheng and G. Q. Lu, J. Am. Chem. Soc., 2009, 131, 4078 CrossRef CAS PubMed.
- S. W. Liu, J. G. Yu and M. Jaroniec, Chem. Mater., 2011, 23, 4085 CrossRef CAS.
- M. A. El-Sayed, Acc. Chem. Res., 2004, 37, 326 CrossRef CAS PubMed.
- S. Kan, T. Mokari, E. Rothenberg and U. Banin, Nat. Mater., 2003, 2, 155 CrossRef CAS PubMed.
- A. Demortiere, P. Panissod, B. P. Pichon, G. Pourroy, D. Guillon, B. Donnio and S. Begin-Colin, Nanoscale, 2011, 3, 225 RSC.
- C. N. R. Rao, G. U. Kulkarni, P. J. Thomas and P. P. Edwards, Chem.–Eur. J., 2002, 8, 29 CrossRef.
- Z. B. Jiao, Y. Zhang, H. C. Yu, G. X. Lu, J. H. Ye and Y. P. Bi, Chem. Commun., 2013, 49, 636 RSC.
- X. Wang, C. Lin, B. J. Zhang, Y. Q. Jiang, L. Zhang, Z. X. Xie and L. S. Zhang, J. Mater. Chem. A, 2013, 1, 282 CAS.
- F. Li, J. Xu, L. Chen, B. B. Ni, X. N. Li, Z. P. Fu and Y. L. Lu, J. Mater. Chem. A, 2013, 1, 225 CAS.
- T. Ohno, K. Sarukawa and M. Matsumura, New J. Chem., 2002, 26, 1167 RSC.
- G. Liu, C. H. Sun, H. G. Yang, S. C. Smith, L. Z. Wang, G. Q. Lu and H. M. Cheng, Chem. Commun., 2010, 46, 755 RSC.
- Y. Zhang, B. Deng, T. R. Zhang, D. M. Gao and A. W. Xu, J. Phys. Chem. C, 2010, 114, 5073 CAS.
- S. V. Yanina and K. M. Rosso, Science, 2008, 320, 218 CrossRef CAS PubMed.
- G. Liu, J. C. Yu, G. Q. Lu and H. M. Cheng, Chem. Commun., 2011, 47, 6763 RSC.
- S. Li, Y. H. Lin, B. P. Zhang, Y. Wang and C. W. Nan, J. Phys. Chem. C, 2010, 114, 2903 CAS.
- J. Wang, Z. Fu, R. Peng, M. Liu, S. Sun, H. Huang, L. Li, R. J. Knize and Y. Lu, Mater. Horiz., 2015, 2, 232 RSC.
- S. Sun, Y. Huang, G. Wang, J. Wang, Z. Fu, R. Peng, R. J. Knize and Y. Lu, Nanoscale, 2014, 6, 13494 RSC.
- X. Y. Mao, W. Wang, X. B. Chen and Y. L. Lu, Appl. Phys. Lett., 2009, 95, 082901 CrossRef.
- N. A. Spaldin and M. Fiebig, Science, 2005, 309, 391 CrossRef CAS PubMed.
- J. Ma, J. M. Hu, Z. Li and C. W. Nan, Adv. Mater., 2011, 23, 1062 CrossRef CAS PubMed.
- S. M. Sun, W. Z. Wang, H. L. Xu, L. Zhou, M. Shang and L. Zhang, J. Phys. Chem. C, 2008, 112, 17835 CAS.
- X. N. Li, Z. Ju, F. Li, Y. Huang, Y. M. Xie, Z. P. Fu, R. J. Knize and Y. L. Lu, J. Mater. Chem. A, 2014, 2, 13366 CAS.
- T. J. Park, G. C. Papaefthymiou, A. J. Viescas, A. R. Moodenbaugh and S. S. Wong, Nano Lett., 2007, 7, 766 CrossRef CAS PubMed.
- X. F. Du, Y. L. Xu, H. X. Ma, J. Wang and X. F. Li, J. Am. Ceram. Soc., 2007, 90, 1382 CrossRef CAS.
- S. M. Selbach, T. Tybell, M. A. Einarsrud and T. Grande, Chem. Mater., 2007, 19, 6478 CrossRef CAS.
- M. Cargnello, T. R. Gordon and C. B. Murray, Chem. Rev., 2014, 114, 9319 CrossRef CAS PubMed.
- Y. Chang and H. C. Zeng, Cryst. Growth Des., 2004, 4, 397 CAS.
- Q. J. Ruan and W. D. Zhang, J. Phys. Chem. C, 2009, 113, 4168 CAS.
- J. T. Han, Y. H. Huang, X. J. Wu, C. L. Wu, W. Wei, B. Peng, W. Huang and J. B. Goodenough, Adv. Mater., 2006, 18, 2145 CrossRef CAS.
- J. T. Han, Y. H. Huang, R. J. Jia, G. C. Shan, R. Q. Guo and W. Huang, J. Cryst. Growth, 2006, 294, 469 CrossRef CAS.
- H. B. Jiang, Q. Cuan, C. Z. Wen, J. Xing, D. Wu, X. Q. Gong, C. Li and H. G. Yang, Angew. Chem., Int. Ed. Engl., 2011, 50, 3764 CrossRef CAS PubMed.
- G. Naresh and T. K. Mandal, ACS Appl. Mater. Interfaces, 2014, 6, 21000 CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Refinement XRD patterns of BFCTO-2.00, Zero-field-cooled (ZFC) and field-cooled (FC) magnetization curves of all samples, nitrogen adsorption–desorption isotherms of all samples. See DOI: 10.1039/c5ra07435e |
| This journal is © The Royal Society of Chemistry 2015 |