Assistant effect of poly(methyl methacrylate)-grafted carbon nanotubes on the beta polymorph of poly(vinylidene fluoride) during microinjection†
Feng
Bai
,
Gang
Chen
,
Min
Nie
* and
Qi
Wang
State Key Laboratory of Polymer Materials Engineering, Polymer Research Institute of Sichuan University, Chengdu 610065, China. E-mail: poly.nie@gmail.com; Fax: +86-28-85402465; Tel: +86-28-85405133
First published on 5th June 2015
Abstract
Poly(vinylidene fluoride) (PVDF) with numerous electroactive beta crystals was obtained via melt-processing under the coupled effects of an external flow field generated by microinjection and by dipole–dipole interaction with poly(methyl methacrylate)-grafted carbon nanotubes, which represents a significant route for expanding the potential applications of piezoelectric PVDF devices.
Poly(vinylidene fluoride) (PVDF) is the best all-around electroactive polymer available and exhibits great potential for application in sensors, ferroelectric memories and biomedical fields.1,2 Its unique electroactive properties strongly depend on its crystalline phases. PVDF possesses five polymorphs, among which the α and β phases are the most investigated crystals. For α-PVDF, the polymer chains are piled up in a trans–gauche–trans–gauche(TGTG) conformation, where the fluorine atoms normal to the chain are antiparallel and thus the dipoles neutralize each other, presenting non-polarization; on the contrary, β-PVDF has an all-trans(TT) conformation with the dipoles fully aligned in the same direction and possesses the larger dipole moment and excellent piezo- and ferroelectric properties, i.e., the electroactivity of PVDF is associated with the content of polar β-PVDF.3 However, α-PVDF is the more stable phase and is commonly obtained in normal melt-processing.4,5 Therefore, obtaining the high-level polar β-phase in PVDF materials is an ongoing pursuit.
To date, considerable effort has been devoted to the preferential formation of β-PVDF, such as via mechanical stretching, solution crystallization in polar solvents, melt crystallization under high pressure and electrospinning. Moreover, the incorporation of nanofillers also has been proven to a facile route for the enhancement of β-PVDF and suppression of α-PVDF.6,7 Carbon nanotubes (CNTs) have zigzag carbon atoms, which match with the TT conformation of β-PVDF and so can be expected to be an ideal filler to induce the crystallization of PVDF into its β-phase.8 However, the energy of the TT conformation of PVDF is higher than that of TGTG, and PVDF chains in the TGTG conformation are easily absorbed on the surface of pristine CNTs.9 The direct addition of unmodified CNTs fails due to the increased β-PVDF. Recently, the interactions between the local electric field and PVDF dipoles, such as hydrogen bond–dipole, ion–dipole, and dipole–dipole, were found to facilitate the transformation from the TGTG to the TT conformation, and thus chemically-functionalized CNTs benefit the formation of β-PVDF.10 Manna found that ester-functionalized CNTs promoted α-β transition by the special interaction of the >C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group and the >CF2 group of PVDF.11 Mandal prepared PMMA-functionalized CNTs via a nitrene reaction and fabricated almost full β-PVDF through a solution mixing route.12 However, in the melt state, polymer chains interpenetrate and entangle each other, and the molecular motion is restricted compared to that in solution, thus depressing the transformation from the TGTG to the TT conformation. Therefore, a high content of β-PVDF is not yet obtainable from melt-cooled PVDF/functionalized CNTs composites, even at a high loading of the CNTs,13 although melt processing is a facile and versatile processing way for the preparation of smart devices such as sensors and actuators.
O group and the >CF2 group of PVDF.11 Mandal prepared PMMA-functionalized CNTs via a nitrene reaction and fabricated almost full β-PVDF through a solution mixing route.12 However, in the melt state, polymer chains interpenetrate and entangle each other, and the molecular motion is restricted compared to that in solution, thus depressing the transformation from the TGTG to the TT conformation. Therefore, a high content of β-PVDF is not yet obtainable from melt-cooled PVDF/functionalized CNTs composites, even at a high loading of the CNTs,13 although melt processing is a facile and versatile processing way for the preparation of smart devices such as sensors and actuators.
It has been reported that external flow fields can help polymer chains disentangle and induce coil-stretch transition, which is helpful for the conversion of the helix TGTG conformation to the zigzag TT one.14,15 Due to the lower energy of the TGTG conformation, the conversion is instable and the TT conformation easily relaxes via thermal motion. As expected, the introduction of special interaction can retard the relaxation and stabilize the TT conformation to promote the formation of β-PVDF.4,16 In the present study, poly(methyl methacrylate) (PMMA) was first grafted onto CNTs via a solid-state mechano-chemical process as the interaction between the carbonyl groups of PMMA and PVDF existed and the PVDF/PMMA blends were miscible in the molten state over the whole composition range.17,18 Then, the PMMA-g-CNTs/PVDF nanocomposites were mixed and microinjected, where a very high shear rate (>105 s−1) was imposed on the polymer melts.19 Utilizing the synergetic effects of the functionalized CNTs and the external flow field, high-level β-PVDF was obtained directly through melt processing, and this provided a simple and efficient way to fabricate an electroactive PVDF device (detailed description of the materials, sample preparation and characterizations are provided in the ESI†).
First, a self-designed pan-mill type of equipment was adopted to achieve the direct attachment of PMMA onto the surface of CNTs. This equipment exerted a strong shearing force like a pair of three-dimensional scissors on the milling materials, bringing about a solid mechano-chemical reaction between the polymer and filler.20 In this case, PMMA/CNTs mixtures (9/1) were fed into the solid-state mechano-chemical equipment and the milled powders were extracted with boiling N,N-dimethylformamide (DMF) in a Soxhlet extractor for 48 h to remove free PMMA. The obtained CNTs were named as PMMA-g-CNTs. According to our previous work,21 PMMA underwent chain scission and formed free macroradicals during the pan-milling processing. The radicals could be terminated at the surface of CNTs to allow functionalization of the CNTs with PMMA (see FTIR spectra of PMMA-g-CNTs in ESI†). The content of PMMA grafted onto CNTs was estimated based on the mass loss differences among the pure PMMA, CNTs and PMMA-modified CNTs. As shown in Fig. 1, pristine CNTs showed no mass loss when heated to 600 °C, while pure PMMA was degraded completely. In comparison, PMMA-g-CNTs exhibited 18 wt% weight-loss, suggesting PMMA was successfully grafted onto the CNTs surface. This was also confirmed by the SEM image shown in Fig. 1. Clearly, CNTs were wrapped by some polymer and the surface became uneven.
 | ||
| Fig. 1 TGA weight loss curves for CNTs, PMMA and PMMA-g-CNTs via mechano-chemical reaction. The inset shows the SEM image of the PMMA-g-CNTs. | ||
Subsequently, the compounding of PVDF and 0.5 wt% pristine CNTs or PMMA-g-CNTs was conducted through DMF solution, and the CNTs/PVDF nanocomposites were melt-processed on a Battenfeld MicroPower-5 molding machine (Wittmann Battenfeld GmbH, Austria). Because the concentration of the introduced pristine CNTs and the functionalized CNTs were low (only 0.5%), the processing behaviors of PVDF remained unchanged (The rheological results are described in the ESI†). For comparison, the static samples were also prepared under no stress field, where the samples were melted at 190 °C in a hot stage for 5 min, followed by cooling. Because PVDF, CNTs/PVDF composite and PMMA-CNTs/PVDF composite prepared by static state and microinjection had similar crystallinities, the relative amount of β-PVDF was key for the preparation of an electroactive PVDF device (the DSC results are described in the ESI†). Fig. 2 shows the conformation sensitive bands in the wavenumber range from 600 to 1500 cm−1 for the three samples prepared by static state and microinjection. The characteristic absorption peaks of α-PVDF with the TGTG conformation appear at 764, 796, 976 and 1218 cm−1, while the peaks at 840 and 1274 cm−1 were assigned to the TT conformation of β-PVDF.3 For the static samples shown in Fig. 2a, one can observe strong absorption peaks corresponding to α-PVDF and weak absorption ones representing β-PVDF. On the contrary, the characteristic absorption peaks of β-PVDF become visible under the external flow field generated by microinjection, and the intensities of β-PVDF was higher than that of α-PVDF with the presence of PMMA-g-CNTs, as shown in Fig. 2b. The relative amount of β-PVDF (Fβ) was estimated according to the following equation:22
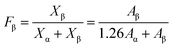 | (1) |
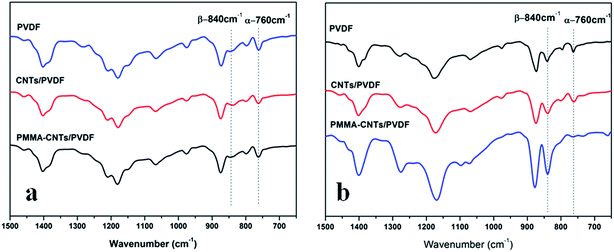 | ||
| Fig. 2 FTIR spectra of PVDF, tCNTs/PVDF composite and PMMA-CNTs/PVDF composite prepared by static state (a) and microinjection (b). | ||
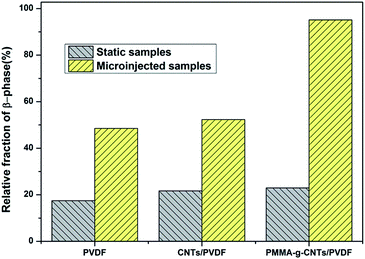 | ||
| Fig. 3 Relative fraction of the β-phase in pure PVDF, CNTs/PVDF nanocomposite and PMMA-CNTs/PVDF nanocomposite prepared via static state and microinjection. | ||
Mechanism: α-form crystal with the TGTG conformation, the most common polymorph, is frequently generated in the melt-cooled condition, while β-PVDF having the TT conformation is not always retained, because the free energy of the former conformation is lower than that of the latter.3,9 The interaction between the local electric field and the PVDF dipoles is widely recognized as an accepted mechanism for the direct formation of β-PVDF,23 where owing to the specific interaction, F atoms are located along one side of the PVDF chain, directly transform the molecular conformation into the TT one and form β-PVDF. In order to prepare high-level β-PVDF, PMMA with a >C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group, which has a strong dipole–dipole interaction with the >CF2 groups of PVDF, was grafted onto the surface of CNTs via a solid mechano-chemical method. However, such interaction only works in the solution where PVDF chains are spread out, which is favorable for the strong interaction between PVDF and PMMA-g-CNTs and the subsequent conversion of α- to β-PVDF.24 Different from β-PVDF in solution, entangled PVDF chains in the melt state are in the coiled state and thus only a few chains can interact with the PMMA-g-CNTs,25 so the functionalized CNTs hardly affected the formation of β-PVDF. Evidently, there is no shift of the C–F stretching in the amorphous phase at 874 cm−1 for the three PVDF samples prepared in the static state. A similar result also was manifested in other research reports.26 On the other hand, PVDF chains are stretched under a high flow field, facilitating the transformation from the helix TGTG conformation to the zigzag TT conformation, but due to the molecular thermal motion, the stretched molecules are compelled to relax into the TGTG conformation at low energy. As a result, numerous α-PVDFs still existed in the microinjected pure PVDF sample. Pristine CNTs with zigzag carbon atoms, which match the TT conformation of β-PVDF, could promote the formation of β-PVDF as the nucleating agent. However, because the CNTs had little specific interaction with PVDF, the TT conformation relaxation was inevitable; and the absorbed energy of the TT conformation on the surface of CNTs was higher than that of the TGTG conformation. Therefore, it is clear that pristine CNT exerted a positive effect on β-PVDF to a small extent. Upon PMMA's functionalization on CNTs, there were strong interactions between the >CF2 groups of PVDF and the CNTs,27 as inferred from the shift of the C–F stretching in the amorphous phase from 874 cm−1 in pure PVDF to higher wavenumber of 877 cm−1 in the PMMA-g-CNTs/PVDF nanocomposites (Fig. 2b). Obviously, the interaction could not only stabilize the TT conformation generated during the microinjection, but it could also aid the conversion of TGTG into the TT conformation. As a result, the formation of β-PVDF was significantly promoted under the synergistic effects of an external flow field and an interaction between PVDF and the functionalized CNTs, and the microinjected PMMA-g-CNTs/PVDF nanocomposite was rich in β-form crystals.
O group, which has a strong dipole–dipole interaction with the >CF2 groups of PVDF, was grafted onto the surface of CNTs via a solid mechano-chemical method. However, such interaction only works in the solution where PVDF chains are spread out, which is favorable for the strong interaction between PVDF and PMMA-g-CNTs and the subsequent conversion of α- to β-PVDF.24 Different from β-PVDF in solution, entangled PVDF chains in the melt state are in the coiled state and thus only a few chains can interact with the PMMA-g-CNTs,25 so the functionalized CNTs hardly affected the formation of β-PVDF. Evidently, there is no shift of the C–F stretching in the amorphous phase at 874 cm−1 for the three PVDF samples prepared in the static state. A similar result also was manifested in other research reports.26 On the other hand, PVDF chains are stretched under a high flow field, facilitating the transformation from the helix TGTG conformation to the zigzag TT conformation, but due to the molecular thermal motion, the stretched molecules are compelled to relax into the TGTG conformation at low energy. As a result, numerous α-PVDFs still existed in the microinjected pure PVDF sample. Pristine CNTs with zigzag carbon atoms, which match the TT conformation of β-PVDF, could promote the formation of β-PVDF as the nucleating agent. However, because the CNTs had little specific interaction with PVDF, the TT conformation relaxation was inevitable; and the absorbed energy of the TT conformation on the surface of CNTs was higher than that of the TGTG conformation. Therefore, it is clear that pristine CNT exerted a positive effect on β-PVDF to a small extent. Upon PMMA's functionalization on CNTs, there were strong interactions between the >CF2 groups of PVDF and the CNTs,27 as inferred from the shift of the C–F stretching in the amorphous phase from 874 cm−1 in pure PVDF to higher wavenumber of 877 cm−1 in the PMMA-g-CNTs/PVDF nanocomposites (Fig. 2b). Obviously, the interaction could not only stabilize the TT conformation generated during the microinjection, but it could also aid the conversion of TGTG into the TT conformation. As a result, the formation of β-PVDF was significantly promoted under the synergistic effects of an external flow field and an interaction between PVDF and the functionalized CNTs, and the microinjected PMMA-g-CNTs/PVDF nanocomposite was rich in β-form crystals.
In summary, high-level β-PVDF nanocomposites were fabricated successfully via microinjection with the help of PMMA-g-CNTs. The results showed that the PVDF chains were stretched under the high flow field during microinjection, so the helix TGTG conformation was transformed into a zigzag TT one. When PMMA-g-CNTs were incorporated into the PVDF matrix, the special interaction between the >C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group of PMMA and the >CF2 groups of the PVDF-stabilized TT conformation generated during the microinjection further facilitated the conversion of the TGTG conformation into the TT conformation. Accordingly, an almost full β-PVDF sample was obtained. Obviously, this melt-processing method for the β-PVDF object is a step towards expanding the potential applications of piezoelectric PVDF devices.
O group of PMMA and the >CF2 groups of the PVDF-stabilized TT conformation generated during the microinjection further facilitated the conversion of the TGTG conformation into the TT conformation. Accordingly, an almost full β-PVDF sample was obtained. Obviously, this melt-processing method for the β-PVDF object is a step towards expanding the potential applications of piezoelectric PVDF devices.
Acknowledgements
This work is financed by the National Natural Science Foundation of China (51421061).Notes and references
- B. Chu, X. Zhou, K. Ren, B. Neese, M. Lin, Q. Wang, F. Bauer and Q. M. Zhang, Science, 2006, 313, 334 CrossRef CAS PubMed.
- G. D. Kang and Y. M. Cao, J. Membr. Sci., 2014, 463, 145 CrossRef CAS.
- P. Martins, A. C. Lopes and S. Lanceros-Mendez, Prog. Polym. Sci., 2014, 39, 683 CrossRef CAS.
- J. Yang, J. Wang, Q. Zhang, F. Chen, H. Deng, K. Wang and Q. Fu, Polymer, 2011, 52, 4970 CrossRef CAS.
- M. Benz, W. B. Euler and O. J. Gregory, Macromolecules, 2002, 35, 2682 CrossRef CAS.
- Y. Wu, S. L. Hsu, C. Honeker, D. J. Bravet and D. S. Williams, J. Phys. Chem. B, 2012, 116, 7379 CrossRef CAS PubMed.
- N. Jia, Q. Xing, G. Xia, J. Sun, R. Song and W. Huang, Mater. Lett., 2015, 139, 212 CrossRef CAS.
- S. Vidhate, J. Chung, V. Vaidyanathan and N. D'Souza, Mater. Lett., 2009, 63, 1771 CrossRef CAS.
- S. Yu, W. Zheng, W. Yu, Y. Zhang, Q. Jiang and Z. Zhao, Macromolecules, 2009, 42, 8870 CrossRef CAS.
- G. Zhong, L. Zhang, R. Su, K. Wang, H. Fong and L. Zhu, Polymer, 2011, 52, 2228 CrossRef CAS.
- S. Manna and A. K. Nandi, J. Phys. Chem. C, 2007, 111, 14670 CAS.
- A. Mandal and A. K. Nandi, J. Mater. Chem., 2011, 21, 15752 RSC.
- G. H. Kim, S. M. Hong and Y. Seo, Phys. Chem. Chem. Phys., 2009, 11, 10506 RSC.
- C. Xing, L. Zhao, J. You, W. Dong, X. Cao and Y. Li, J. Phys. Chem. B, 2012, 116, 8312 CrossRef CAS PubMed.
- Y. Ahn, J. Y. Lim, S. M. Hong, J. Lee, J. Ha, H. J. Choi and Y. Seo, J. Phys. Chem. C, 2013, 117, 11791 CAS.
- Y. L. Liu, Y. Li, J. T. Xu and Z. Q. Fan, ACS Appl. Mater. Interfaces, 2010, 2, 1759 CAS.
- H. Sasaki, P. Kanti Bala, H. Yoshida and E. Ito, Polymer, 1995, 36, 4805 CrossRef CAS.
- H. Song, S. Yang, S. Sun and H. Zhang, Polym.–Plast. Technol. Eng., 2013, 52, 221 CrossRef CAS.
- A. L. Kelly, T. Gough, B. R. Whiteside and P. D. Coates, J. Appl. Polym. Sci., 2009, 114, 864 CrossRef CAS.
- W. Shao, Q. Wang, F. Wang and Y. Chen, Carbon, 2006, 44, 2708 CrossRef CAS.
- H. Xia, Q. Wang, K. Li and G.-H. Hu, J. Appl. Polym. Sci., 2004, 93, 378 CrossRef CAS.
- B. Mohammadi, A. A. Yousefi and S. M. Bellah, Polym. Test., 2007, 26, 42 CrossRef CAS.
- A. Mandal and A. K. Nandi, ACS Appl. Mater. Interfaces, 2013, 5, 747 CAS.
- Y. Li, J. Z. Xu, L. Zhu, G. J. Zhong and Z. M. Li, J. Phys. Chem. B, 2012, 116, 14951 CrossRef CAS PubMed.
- R. Krishnamoorti, W. W. Graessley, A. Zirkel, D. Richter, N. Hadjichristidis, L. J. Fetters and D. J. Lohse, J. Polym. Sci., Part B: Polym. Phys., 2002, 40, 1768 CrossRef CAS.
- K. Ke, P. Pötschke, D. Jehnichen, D. Fischer and B. Voit, Polymer, 2014, 55, 611 CrossRef CAS.
- K. Zhang, J. Y. Lim and H. J. Choi, Diamond Relat. Mater., 2009, 18, 316 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra08397d |
| This journal is © The Royal Society of Chemistry 2015 |
