Crystalline arrays of molecular rotors with TIPS-trityl and phenolic-trityl stators using phenylene, 1,2-difluorophenylene and pyridine rotators†
Rafael Arcos-Ramosa,
Braulio Rodriguez-Molinab,
E. Gonzalez-Rodrigueza,
Pedro I. Ramirez-Montesc,
Maria Eugenia Ochoac,
Rosa Santillanc,
Norberto Farfán*a and
Miguel A. Garcia-Garibay*d
aFacultad de Química, Departamento de Química Orgánica, Universidad Nacional Autónoma de México, 04510 México, D.F., México. E-mail: norberto.farfan@gmail.com
bInstituto de Química, Universidad Nacional Autónoma de México, Circuito Exterior, Ciudad Universitaria, 04510 México, D.F., México
cDepartamento de Química, Centro de Investigación y Estudios Avanzados del IPN, Apdo. Postal 14-740, México, D.F. 07000, México
dDepartment of Chemistry and Biochemistry, University of California, Los Angeles, California 90095, USA. E-mail: mgg@chem.ucla.edu
First published on 15th June 2015
Abstract
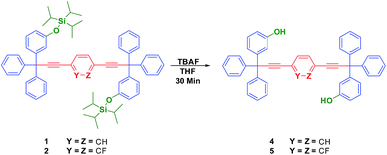
In this work, we describe the synthesis and solid-state characterization of a series of molecular rotors with tri-isopropylsilyloxy-substituted (TIPS) trityl stators axially linked to 1,4-diethynylphenylene, 3,6-diethynyl-1,2-difluorophenylene and 2,5-diethynylpyridine rotators to produce 1,4-bis[(3,3-diphenyl-3-(3′-(tri-isopropylsilyloxy)-phenyl)-prop-1-yn-1-yl)]benzene (1), 1,4-bis[(3,3-diphenyl-3-(3′-(tri-isopropylsilyloxy)-phenyl)-prop-1-yn-1-yl)]-2,3-difluorobenzene (2) and 2,5-bis[(3,3-diphenyl-3-(3′-tri-isopropylsilyloxy)-phenyl)-prop-1-yn-1-yl)]pyridine (3). The subsequent removal of the TIPS protecting group led to their corresponding hydroxyl-substituted trityl derivatives (4) and (5). TIPS- and HO-substituted stators are involved in different inter- and intramolecular interactions (hydrogen bonding, phenyl embraces, C–H–π interactions) that give rise to isomorphic packing motifs that constrained the rotational dynamics in the solid-state to the slow exchange regime.
Introduction
The fascinating and complex operation of biomolecular machines has inspired a large amount of research dedicated to the development of synthetic molecular assemblies that can show a response to external stimuli.1 It has been proposed that the function of artificial molecular machines could possibly emulate those found in nature, such as the skeletal muscle or the ATPase, when based on the collective action of their molecular components.2,3 Keeping in mind that the function of those biomolecular machines occurs in condensed media, during the past few years our research has been focused on the development of amphidynamic crystals: solid materials by design in which specific molecular components experience motion within rigid bulky components that form an ordered crystal lattice.4a We amongst others, have suggested that the precise knowledge about the internal dynamics may offer a new perspective in the field of functional solid materials, for example in Metal Organic Frameworks (MOFs) or Periodic Mesoporous Organosilicas (PMOs). Additionally, those engineered solids may represent a promising entry as molecular switches in condensed media, with potential optoelectronic5 or photonic6 applications.In order to obtain amphidynamic crystals, the current design relies on the synthesis and characterization of molecular rotors, dumbbell-like molecules built up with three elements: rotator (the mobile component), axle and stator which is often the bulky group that acts as the shielding framework for the system.7 Several variations of these components have been explored in the past in order to provide an enhanced dynamic behavior.8 Furthermore, the available theoretical and experimental evidence has shown the enormous influence of crystal packing in the dynamic behavior,9 an interesting example being the BIBCO rotor (1,4-bis-(iodoethynyl)bicyclo[2.2.2]octane), in which the same molecule experiences two distinct rotational dynamics in the solid state.10
Efforts devoted to control the frequency of the internal motion comprise the study of several stator structures, which include substituted triptycenes,8 substituted trityl groups,14 steroids,4c,d and porous solids such as MOFs.11 These molecules tend to pack less densely (often with solvent molecules),12 in a way that facilitates the dynamic behavior in the solid state.13
With that in mind, in a previous report we synthesized a family of compounds with voluminous tert-butyldiphenylsilyl-protected (TBDPS) triphenylmethyl stators to evaluate their effect on the rotator dynamics.14 From these studies, we reported the use of large TBDPS groups to promote rotational dynamics; however, the intramolecular interactions between the phenyl rings of these bulky groups and the rotator shut down the awaited internal motion. Additionally, in such a previous set of molecular rotors the severe disorder of the tert-butylsilyl groups and the low stability and quality of the crystals prevented a detailed analysis of the molecular structure.
While fast rotations could indeed be interesting, slow molecular dynamics can also induce large effects as those resulting from the large conformational changes of proteins.15 Those premises guided us to develop a new series of compounds using similar protecting groups. Consequently, here we report the synthesis, characterization and crystallization of molecular rotors 1, 2 and 3 (Fig. 1). In comparison with the TBPDS-protected stators, the TIPS groups produced stable crystalline samples that allowed the detailed study of the molecular structure and packing environment. We also employed different rotators in order to gain further insights about the stability, crystal growth and intermolecular interactions in these molecules.
Results and discussion
Synthesis and solution characterization
We employed tri-isopropylsilyloxy-substituted trityl groups as stators due to their moderate size and good stability under different conditions. These TIPS-groups were placed in meta-position following the convergent protocol (Fig. 1).14 The stators with one TIPS unit per molecule provided samples with good stability, and the subsequent increase in the number of TIPS units disfavoured crystallinity, forming oily (2 TIPS per stator) and liquid samples (3 TIPS per stator) (see ESI†). Those compounds were fully characterized but difficult to crystallize and will not be described in detail here.Next, we have focused in the introduction of polar substituents in the central part with the aim to explore isomorphic crystallization to create compounds capable of providing physical and chemical properties that change as a result of reorienting dipoles under the influence of external fields. The selected pyridine and difluorobenzene rotators contain calculated dipoles of ca. 1.81 and 2.37 D, respectively.16 Molecular rotors with TIPS-substituted trityl stators 1–3 were synthesized using Sonogashira cross coupling conditions with Pd(PPh3)2Cl2 and CuI as catalysts. The TIPS-substituted trityl 7 (see ESI†) was reacted with 1,4-diiodobenzene, 2,3-difluoro-1,4-diiodobenzene‡ and 2,5-dibromopyridine to produce molecular rotors 1 (77%), 2 (82%), and 3 (81%) in good yields as shown in Scheme 1. The synthesis of molecular rotors 1–3 was corroborated by solution NMR experiments, where the 1H-NMR spectrum of 1 showed a singlet at δ = 7.49 ppm, corresponding to the hydrogen atoms of the phenylene rotator. For the fluorinated derivative 2, the spectrum showed a multiplet between δ = 7.21–7.19 ppm for the two hydrogens of the aromatic rotator, due to the hetero-(H–F) and homonuclear (H–H) coupling. In the proton spectrum of 3, the three signals at δ = 8.66, 7.76 and 7.35 ppm evidenced the incorporation of the pyridine rotator. Additionally, the 29Si-NMR verified the presence of the TIPS groups, with single signals between δ = 16.0–16.2 ppm for all the compounds. The observed peaks at m/z 9555.5301 (1), 991.5111 (2) and 959.5661 (3) from high resolution mass spectrometry analyses were in accordance with the expected molecular ions (Scheme 1).
Solid-state characterization
 | ||
| Fig. 2 Molecular structures of compound (a) 1 and (b) 2, thermal ellipsoids are drawn at 30% probability level for all atoms other than H. | ||
 | ||
| Fig. 3 Crystal packing of the isomorphic packing in compounds 1 and 2, viewed down the crystallographic c axis. | ||
The asymmetric unit of both 1 and 2 contains only half of the molecule and the other half is generated by an inversion center that lies on the center of the 1,4-diethynylphenylene rotator. This is somehow expected considering that molecules having inversion symmetry occupy Wickoff positions of Ci symmetry 99% of the times according to a study based on CSD.17 In the case of compound 1, the TIPS-group in the stator was disordered over two sites (occupancy 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50). On the other hand, compound 2 did not show disorder in the TIPS-group, but instead we observed a disorder of the fluorinated rotator over two positions, which was modeled with a 50
50). On the other hand, compound 2 did not show disorder in the TIPS-group, but instead we observed a disorder of the fluorinated rotator over two positions, which was modeled with a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 occupancy. This prompted us to further investigate the nature of the disorder, given that solid state NMR can distinguish between static and dynamic disorder among partially occupied sites. This aspect will be discussed in a subsequent section.
50 occupancy. This prompted us to further investigate the nature of the disorder, given that solid state NMR can distinguish between static and dynamic disorder among partially occupied sites. This aspect will be discussed in a subsequent section.
The crystal packing of compounds 1 and 2 can be described as columnar arrangement of parallel displaced array of molecular rotors along the crystallographic c axis (more accurately described considering the rotation and translation involved by the presence of a c glide plane). These columnar arrangements are held together by various edge to face interactions that could be classified as parallel fourth-fold phenyl embraces13 between three neighboring molecules in which two aromatic rings of one stator interact with two different rings of the other two stators (Fig. 3).
As an initial assessment of the factors that might affect the solid state dynamics, we performed a search of close contacts around the rotator in 1 as illustrated in the cross section view in Fig. 4. This analysis revealed a crowded environment around the rotator with two isopropyl fragments (in green) above and below of the rotator (in red), with methyl-phenyl distances of 3.001 and 3.473 Å, respectively. Additionally, up to two edge to face interactions of this phenylene with neighboring stators (in blue) could be observed, both showing CH-centroid distances of 2.953 Å. Similar distances were found in compound 2. These distances are indicative of a small crystal cavity which may suggest high activation barriers to rotation that would be discussed in the following sections.
In agreement with the X-ray diffraction data that indicates only one half of the molecule per asymmetric unit, the solid-state 13C-NMR spectra of 1 and 2 showed signals that accounted only for half of their respective molecules, demonstrating that the two halves of the molecular rotors are crystallographically equivalent, sharp peaks confirmed the crystallinity of the samples (Fig. 5). Noticeable differences in the chemical shifts can be observed in signals closer to the central aromatic ring when comparing crystalline molecular rotors 1 and 2 due the effect of the halogen atoms. The signals were assigned by chemical shifts analysis and by comparison with the solution NMR data. Given the low resolution obtained at 7.05 T, we obtained overlapped signals of the central carbons of 1 and 2 with the trityl carbons between 135–125 ppm, which made it impractical to extract dynamic information and prevented further VT experiments.
As a qualitative complement, we carried out 13C CPMAS dipolar dephasing experiments that are designed to remove all protonated signals except those involved in a dynamic process with an exchange frequency higher than the C–H heteronuclear coupling (ca. 20 kHz). Given the fact that all the CH signals were absent in this spectrum, we concluded that only a slow exchange process is taking place. We were able to confirm those results and obtain an upper limit to rotation by acquiring a solid state 2H NMR spectrum using a deuterated analog 1-d4. Based on the 2H line shape at room temperature, we observed that any motion present in compounds 1 or 2 would take place at frequencies not higher than 10 kHz (see ESI† for synthetic and spectroscopic details).
Our spectroscopic results were supported by a computational analysis where the rotational coordinate of the rotator was explored using molecular mechanics methods, since these are known to provide reasonable conformational analysis while being computationally accessible for large systems. The COMPASS force field with fine convergence criteria was used for all the geometry optimizations. Notably, the computed energy minimum structure is very close to the experimental X-ray structure. Angular displacements of 10° led to the energy profile shown in Fig. 6. Given the crystallographic symmetry of the phenylene group, the analyses were restricted to half a rotational cycle, i.e. 180°. To better represent the local environment of a rotor in the crystal, a cluster of three neighbouring molecules was extracted from the X-ray data.
In this conformation, an edge-to-face cooperative stacking is observed. Since electron correlation effects (and thus, dispersive forces) are not explicitly computed in molecular mechanics, the stabilizing interaction is expected to be mainly electrostatic in nature. In the case of compound 1 when the angular displacement approaches 50° a local maximum of ca. 18 kcal mol−1 is reached. Close exploration of this conformer indicates some steric interactions with the isopropyl groups. Nevertheless, a maximum in potential energy of 27 kcal mol−1 was calculated when the phenylene group explores 90° rotation. Even though this very high calculated energy could be lowered taking into account additional conformational changes of the surrounding molecules, we anticipate that the resulting energy would be still high compared with similar molecular rotors showing barriers to rotation in the range of 12–14 kcal mol−1,15 evidencing that angular displacements in 1 or 2 result in an energetically costly conformation resulting from the destabilization of the edge-to-face and the interactions with the bulky TIPS groups.
 | ||
| Fig. 7 Crystal packing of solvates 4 in (a) acetone 4a, (b) DMSO 4c and (c) methanol 4b, view along b axis. | ||
After removal of protecting groups, three solvates of 4 in acetone (4a), methanol (4b) and DMSO (4c) were characterized by means of single crystal X-ray diffraction experiments, all of them in the P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group trapping two solvent molecules per molecule of 4. Crystallization of solvates from compound 5 were obtained but they had poor quality for X-ray single crystal studies and they are not described here. A comparison between the molecular geometries of compound 4 in solvates 4a, 4b, and 4c, revealed only slight changes, in all of them the groups are placed in an anti-fashion with respect to the alkyne axis (Fig. 7). The three molecules show slight deviations from the geometry of an ideal diethynylphenylene fragment19 with the differences in their conformations (see ESI†) arising from interactions present in each solid form, and most important, reflect the complementary effect between classic hydrogen bonding and phenyl embraces, resulting in a conformational adjustment in the crystal packing.20,21
space group trapping two solvent molecules per molecule of 4. Crystallization of solvates from compound 5 were obtained but they had poor quality for X-ray single crystal studies and they are not described here. A comparison between the molecular geometries of compound 4 in solvates 4a, 4b, and 4c, revealed only slight changes, in all of them the groups are placed in an anti-fashion with respect to the alkyne axis (Fig. 7). The three molecules show slight deviations from the geometry of an ideal diethynylphenylene fragment19 with the differences in their conformations (see ESI†) arising from interactions present in each solid form, and most important, reflect the complementary effect between classic hydrogen bonding and phenyl embraces, resulting in a conformational adjustment in the crystal packing.20,21
The similarities between acetone and DMSO solvates allow to treat these two solids as one, in both forms there is a balance between hydrogen bonding (O1–H⋯O2) and parallel fourth-phenyl embrace (P4PE) in which the ring that participates in the C–H⋯π interaction is distorted from the ideal T geometry presumably to favor the hydrogen bond. The absence of this P4PE in the methanol clathrate could be attributed to the stabilization that provides the sigma cooperativity, that accompanies the homodromic cycle of four hydrogen bonds, corresponding to a graph set notation R44(8).
Conclusions
In conclusion, three new molecular rotors 1, 2 and 3 based on TIPS-substituted trityl stators and two deprotected rotors 4 and 5 were synthesized and characterized in solution and in the solid state. Isomorphic crystal arrays of compounds 1 and 2 were obtained upon slow evaporation, whereas compound 3 decomposes over time. Moreover, the analyses of three different solvates 4a–c of molecular rotor 4 showed the complementarity between classic hydrogen bonds and phenyl embraces.Regarding the molecular dynamics of 1 and 2, we were able to correlate the results from 13C and 2H solid state NMR with X-ray and computational models to conclude that very high barriers to rotation are the result of the steric effect with the TIPS groups and the stabilizing edge to face interactions with the surrounding phenyl groups, only allowing rotational dynamics with frequencies lower than 10 kHz.
Experimental
General
All the reactions were carried out under an inert atmosphere (Nitrogen or Argon). The starting materials were purchased from commercial suppliers and used as received. Tetrahydrofuran (THF) was distilled from sodium-benzophenone in a continuous still under an atmosphere of nitrogen. Analytical thin-layer chromatography (TLC) was performed using pre-coated TLC-sheets ALUGRAM Xtra SIL G/UV254 silica gel 60 with fluorescent indicator. Flash column chromatography was performed using silica gel (230–400 mesh) as the stationary phase. Melting points were determined on a melting point apparatus using open glass capillaries and are uncorrected. The IR spectra were recorded with the attenuated total reflectance as solid samples, or using KBr pellets and the units are stated as cm−1. Mass spectrometry data were collected using Fast Atom Bombardment (+) technique. High resolution mass spectrometry data were obtained using Atmospheric Pressure Chemical Ionization (APCI) with a Time-of-Flight (TOF) detector. Thermal analyses were realized on a Calorimeter DSC821e and Thermobalance TGA/SDTA851e. Solution 1H-NMR spectra were recorded at 270, 300, 400 and 500 MHz and solution 13C-NMR spectra were recorded at 67.9, 75.5, 100 and 125 MHz. All the chemical shifts (δ) are reported in ppm with respect to TMS using residual solvent as reference. The coupling constants (J) are reported in Hz. Multiplicities signals are reported as: singlet (s), doublet (d), doublet–doublet (dd), doublet-triplet (dt), sextuplet (sext), multiplet (m). NMR solid state experiments were obtained on a Bruker 300 Avance II spectrometer operating at a frequency of 46.07 MHz, using a Bruker 4 mm wide-line probe. Single crystal X-ray diffraction analyses of 1, 2 and 4a were performed on a Enraf Nonius Kappa-CCD (λ MoKα = 0.71073 Å, graphite monochromator, T = 298 K-CCD), at 173(K). The collection of 4b and 4c was done at 173 (K), on a Bruker SMART APEX II CCD Platform (UCLA). All reflection data set were corrected for Lorentz and polarization effects. The crystals were mounted on conventional MicroLoops™. The first structure solution was obtained using the SHELXS-97 (ref. 22) or SIR2004 (ref. 23) programs and then the SHELXL-97 program was applied for refinement and output data. All software manipulations were done under the WinGX24 environment program set. All heavy atoms were found by Fourier map difference and refined anisotropically. X-ray powder diffraction analyses were realized on a Siemens D5000 Kα Cu α = 1.5404 Å, 35 kV, 30 mA. X-ray powder diffraction analyses were realized on a Siemens D5000 Kα Cu α = 1.5404 Å, 35 kV, 30 mA.Synthetic procedure for [(3-tri-isopropylsilyloxy)phenyl]3,3-diphenyl-propyne (7)
Hydrochloric acid was slowly bubbled to a solution of diphenyl[3-(tri-isopropylsilyloxy)phenyl]methanol II (1.00 g, 2.3 mmol), in CH2Cl2 (50 ml) at room temperature. After 5 h stirring, the solvent was removed at reduce pressure and the solid obtained was redissolved in benzene (25 ml), then, ethynylmagnesium bromide (5.1 ml, 0.7 M in THF, 3.6 mmol) was added and the mixture was stirred for 48 h. After this time, the reaction was quenched by addition of saturated NH4Cl, and the organic phase was extracted twice with CH2Cl2, the combined organic portions were dried over anhydrous Na2SO4. Purification on silica gel with hexane yielded compound 7 as yellow oil (0.60 g, 59%). IR (KBr) (ν, cm−1): 3061, 2945, 2867, 2345, 1597, 1484, 1429, 1386, 1276, 1145, 976, 882, 766, 700, 640, 569, 509, 459. 1H-NMR (400 MHz, CDCl3) δ: 7.38–7.26 (10H, m), 7.23 (1H, t, J = 7.9 Hz), 7.03 (1H, d, J = 7.9 Hz), 6.87 (1H, dd, J = 7.5, 2.2 Hz), 2.73 (1H, s), 1.24–1.13 (3H, m), 1.08 (18H, d, J = 6.6 Hz). 13C-NMR (100 MHz, CDCl3) δ: 155.9, 146.2, 145.0, 129.2, 129.1, 128.1, 127.0, 122.0, 121.2, 118.8, 89.9, 73.3, 55.5, 18.0, 12.7. 29Si-NMR (79.42 MHz, CDCl3) δ: 16.1 ppm. HRMS (APCI-TOF), calcd for C30H37OSi 441.2608. Found 441.2612. Error 0.99 ppm. MS (DIP): 440 (M+, 1), 432 (19), 389 (100), 371 (3) 293 (1), 259 (2), 223 (6), 165 (5), 103 (4), 75 (5). Anal. calcd for C30H36OSi: C, 81.76; H, 8.23. Found: C, 81.61; H, 8.03%.General synthetic procedure for molecular rotors with the protected trityl stators, exemplified by 1,4-bis[(3,3-diphenyl-3-(3′-(tri-isopropylsilyloxy)-phenyl)-prop-1-yn-1-yl)]benzene (1)
A mixture of alkyne [(3-tri-isopropylsilyloxy)phenyl]3,3-diphenyl-propyne 7 (0.30 g, 0.7 mmol), 1,4-diiodobenzene (0.11 g, 0.3 mmol), Pd(PPh3)2Cl2 (0.02 g, 0.03 mmol), CuI (0.01 g, 0.07 mmol) and (iPr)2NH (0.5 ml) in THF (25 ml) previously degassed was refluxed for 2.5 h under N2 atmosphere. After this time, the reaction was cooled down to room temperature and quenched with saturated NH4Cl. The organic phase was extracted twice with CH2Cl2 and the combined organic portions were dried over anhydrous Na2SO4. The solvent was removed at reduced pressure followed by column chromatography purification on neutral alumina, eluting with hexane/ethyl ether (99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) afforded molecular rotor 1 as a crystalline solid (0.25 g, 77%). Melting point: 166–168 °C. FTIR-ATR (ν, cm−1): 3061, 2944, 2866, 1596, 1485, 1446, 1385, 1277, 1182, 1142, 1073, 1002, 988, 881, 787, 765, 698, 640, 509, 458. 1H-NMR (270 MHz, CDCl3) δ: 7.49 (2H, s), 7.39–7.29 (10H, m), 7.22 (1H, t, J = 8.4 Hz), 6.98 (1H, d, J = 8.0 Hz), 6.88–6.80 (2H, m), 1.22–1.09 (3H, m), 1.05 (18H, d, J = 6.0 Hz). 13C-NMR (67.9 MHz, CDCl3) δ: 156.0, 146.6, 145.4, 131.6, 129.3, 129.1, 128.1, 127.0, 123.3, 122.0, 121.2, 118.8, 97.4, 84.7, 56.2, 18.0, 12.7. 29Si-NMR (53.67 MHz, CDCl3) δ: 16.00. HRMS (APCI-TOF), calcd for C66H75O2Si2 955.5300. Found: 955.5301. Error 0.09 ppm. Anal. calcd for C66H74O2Si2: C, 82.97; H, 7.81. Found: C, 82.64, H, 7.69%.
1) afforded molecular rotor 1 as a crystalline solid (0.25 g, 77%). Melting point: 166–168 °C. FTIR-ATR (ν, cm−1): 3061, 2944, 2866, 1596, 1485, 1446, 1385, 1277, 1182, 1142, 1073, 1002, 988, 881, 787, 765, 698, 640, 509, 458. 1H-NMR (270 MHz, CDCl3) δ: 7.49 (2H, s), 7.39–7.29 (10H, m), 7.22 (1H, t, J = 8.4 Hz), 6.98 (1H, d, J = 8.0 Hz), 6.88–6.80 (2H, m), 1.22–1.09 (3H, m), 1.05 (18H, d, J = 6.0 Hz). 13C-NMR (67.9 MHz, CDCl3) δ: 156.0, 146.6, 145.4, 131.6, 129.3, 129.1, 128.1, 127.0, 123.3, 122.0, 121.2, 118.8, 97.4, 84.7, 56.2, 18.0, 12.7. 29Si-NMR (53.67 MHz, CDCl3) δ: 16.00. HRMS (APCI-TOF), calcd for C66H75O2Si2 955.5300. Found: 955.5301. Error 0.09 ppm. Anal. calcd for C66H74O2Si2: C, 82.97; H, 7.81. Found: C, 82.64, H, 7.69%.
1,4-Bis[(3,3-diphenyl-3-(3′-(tri-isopropylsilyloxy)-phenyl)-prop-1-yn-1-yl)]benzene-d4 (1-d4)
Alkyne [(3-tri-isopropylsilyloxy)phenyl]3,3-diphenyl-propyne 7 (0.12 g, 0.30 mmol), 1,4-diiodobenzene-d4 (0.45 g, 0.10 mmol), Pd(PPh3)2Cl2 (0.01 g, 0.01 mmol), CuI (0.005 g, 0.03 mmol) and (iPr)2NH (0.5 ml) in THF (25 ml) previously degassed were refluxed for 8 h under N2 atmosphere. Purification by column chromatography on silica gel, eluting with hexane/ethyl ether (99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) afforded molecular rotor 1-d4 as a white crystalline solid (0.11 g, 82%). Melting point: 166–168 °C. FTIR-ATR (ν, cm−1): 2944, 2866, 2362, 1596, 1483, 1443, 1283, 1184, 1133, 989, 921, 881, 760, 697, 647, 574, 564. 1H-NMR (500 MHz, CDCl3) δ: 7.35–7.23 (20H, m), 7.17 (2H, t, J = 8.3 Hz), 6.92 (2H, d, J = 7.9 Hz), 6.83–6.79 (4H, m), 1.14–1.06 (6H, m), 1.00 (36H, d, J = 6.1 Hz). 13C-NMR (125 MHz, CDCl3) δ: 155.9, 146.6, 145.3, 131.2 (t, JC–D = 26 Hz), 129.3, 129.0, 128.1, 127.0, 123.1, 122.0, 121.2, 118.8, 97.4, 84.7, 56.1, 18.0, 12.7. 29Si-NMR (99.36 MHz, CDCl3) δ: 16.12. HRMS (APCI-TOF), Calcd for C66H71D4O2Si2 959.5613. Found: 959.5661. Error 0.9859 ppm. Anal. calcd for C66H70D4O2Si2: C, 82.62; H, 8.19. Found: C, 81.71; H, 7.41%.
1) afforded molecular rotor 1-d4 as a white crystalline solid (0.11 g, 82%). Melting point: 166–168 °C. FTIR-ATR (ν, cm−1): 2944, 2866, 2362, 1596, 1483, 1443, 1283, 1184, 1133, 989, 921, 881, 760, 697, 647, 574, 564. 1H-NMR (500 MHz, CDCl3) δ: 7.35–7.23 (20H, m), 7.17 (2H, t, J = 8.3 Hz), 6.92 (2H, d, J = 7.9 Hz), 6.83–6.79 (4H, m), 1.14–1.06 (6H, m), 1.00 (36H, d, J = 6.1 Hz). 13C-NMR (125 MHz, CDCl3) δ: 155.9, 146.6, 145.3, 131.2 (t, JC–D = 26 Hz), 129.3, 129.0, 128.1, 127.0, 123.1, 122.0, 121.2, 118.8, 97.4, 84.7, 56.1, 18.0, 12.7. 29Si-NMR (99.36 MHz, CDCl3) δ: 16.12. HRMS (APCI-TOF), Calcd for C66H71D4O2Si2 959.5613. Found: 959.5661. Error 0.9859 ppm. Anal. calcd for C66H70D4O2Si2: C, 82.62; H, 8.19. Found: C, 81.71; H, 7.41%.
1,4-Bis[(3,3-diphenyl-3-(3′-(tri-isopropylsilyloxy)-phenyl)-prop-1-yn-1-yl)]-2,3-difluorobenzene (2)
Alkyne [(3-tri-isopropylsilyloxy)phenyl]3,3-diphenyl-propyne 7 (0.17 g, 0.3 mmol), 2,3-difluoro-1,4-diiodobenzene (0.07 g, 0.10 mmol), Pd(PPh3)2Cl2 (0.02 g, 0.01 mmol), CuI (0.01 g, 0.03 mmol) and (iPr)2NH (0.5 ml) in THF (25 ml) previously degassed were refluxed for 8 h under N2 atmosphere. Purification by column chromatography on silica gel, eluting with hexane/ethyl ether (99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) afforded molecular rotor 2 as a white crystalline solid (0.11 g, 82%). Melting point: 132–133 °C. FTIR-ATR (ν, cm−1): 3063, 3029, 2945, 2892, 2866, 2233, 1595, 1578, 1474, 1446, 1433, 1281, 1260, 1235, 1146, 1003, 990, 876, 761, 696, 683, 639. 1H-NMR (300 MHz, CDCl3) δ: 7.33–7.24 (22H, m), 7.20 (2H, d, J = 7.9 Hz), 7.16 (2H, d, J = 2.6 Hz), 6.94 (2H, dd, J = 7.9, 1.0 Hz), 6.82 (2H, dd, J = 7.5, 2.7 Hz), 6.79–6.76 (2H, m), 1.18–1.09 (6H, m), 1.00 (36H, d, J = 6.1 Hz). 13C-NMR (75.5 MHz, CDCl3) δ: 155.9, 151.4 (dd, J = 14.5, 254.8 Hz), 146.0, 144.7, 129.2, 129.1, 128.2, 127.4, 127.1, 122.0, 121.1, 118.9, 114.1 (J = 7.2 Hz), 84.1, 77.4, 56.4, 17.9, 12.6. 29Si-NMR (53.67 MHz, CDCl3) δ: 16.23 ppm. 19F-NMR δ: −400.66 ppm. HRMS (APCI-TOF), calcd for C66H73F2O2Si2 991.5113 found: 991.5111. Error 0.13 ppm. Anal. calcd for: C66H73F2O2Si2: C, 79.65; H, 7.32. Found: C, 79.96; H, 7.035%.
1) afforded molecular rotor 2 as a white crystalline solid (0.11 g, 82%). Melting point: 132–133 °C. FTIR-ATR (ν, cm−1): 3063, 3029, 2945, 2892, 2866, 2233, 1595, 1578, 1474, 1446, 1433, 1281, 1260, 1235, 1146, 1003, 990, 876, 761, 696, 683, 639. 1H-NMR (300 MHz, CDCl3) δ: 7.33–7.24 (22H, m), 7.20 (2H, d, J = 7.9 Hz), 7.16 (2H, d, J = 2.6 Hz), 6.94 (2H, dd, J = 7.9, 1.0 Hz), 6.82 (2H, dd, J = 7.5, 2.7 Hz), 6.79–6.76 (2H, m), 1.18–1.09 (6H, m), 1.00 (36H, d, J = 6.1 Hz). 13C-NMR (75.5 MHz, CDCl3) δ: 155.9, 151.4 (dd, J = 14.5, 254.8 Hz), 146.0, 144.7, 129.2, 129.1, 128.2, 127.4, 127.1, 122.0, 121.1, 118.9, 114.1 (J = 7.2 Hz), 84.1, 77.4, 56.4, 17.9, 12.6. 29Si-NMR (53.67 MHz, CDCl3) δ: 16.23 ppm. 19F-NMR δ: −400.66 ppm. HRMS (APCI-TOF), calcd for C66H73F2O2Si2 991.5113 found: 991.5111. Error 0.13 ppm. Anal. calcd for: C66H73F2O2Si2: C, 79.65; H, 7.32. Found: C, 79.96; H, 7.035%.
2,5-Bis[(3,3-diphenyl-3-(3′-(tri-isopropylsilyloxy)-phenyl)-prop-1-yn-1-yl)]pyridine (3)
Alkyne [(3-tri-isopropylsilyloxy)phenyl]3,3-diphenyl-propyne 7 (0.16 g, 0.40 mmol), 2,5-dibromopyridine (0.43 g, 0.20 mmol), Pd(PPh3)2Cl2 (0.02 g, 0.02 mmol), CuI (0.01 g, 0.04 mmol) and (iPr)2NH (0.5 ml) in THF (25 ml) previously degassed was refluxed for 2 h under N2 atmosphere. Purification by column chromatography on neutral alumina, eluting with hexane/ethyl ether (99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) afforded molecular rotor 3 as colorless oil (0.14 g, 81%). FTIR-ATR film (ν, cm−1): 3027, 2238, 1951, 1809, 1595, 1581, 1539, 1489, 1467, 1445, 1362, 1270, 1180, 1100, 1064, 1030, 982, 852, 773, 690, 660. 1H-NMR (300 MHz, CDCl3) δ: 8.66 (1H, d, J = 2.8 Hz), 7.76 (1H, dd, J = 8.4, 2.8 Hz), 7.35 (1H, d, J = 8.4 Hz), 7.32–7.26 (10H, m), 7.19 (1H, t, J = 8.3 Hz), 7.01 (1H, d, J = 8.3 Hz), 6.81 (1H, dd, J = 8.3, 2.6 Hz), 6.71 (1H, t, J = 2.5 Hz), 1.15–1.04 (3H, m), 0.99 (18H, d, J = 6.0 Hz). 13C-NMR (75.5 MHz, CDCl3) δ: 155.9, 151.2, 146.0, 144.7, 142.2, 138.8, 135.6, 129.3, 128.4, 128.2, 127.1, 122.1, 120.0, 118.9, 97.1, 83.8, 56.1, 18.0, 12.7. 29Si-NMR (53.67 MHz, CDCl3) δ: 16.20. HRMS (APCI-TOF), calcd for C65H74NO2Si2 959.5613. Found: 959.5661. Error 1.9 ppm. Anal. calcd for C65H73NO2Si2: C, 81.62; H, 7.69; N, 1.46. Found: C, 81.80; H, 7.83; N, 1.90%.
1) afforded molecular rotor 3 as colorless oil (0.14 g, 81%). FTIR-ATR film (ν, cm−1): 3027, 2238, 1951, 1809, 1595, 1581, 1539, 1489, 1467, 1445, 1362, 1270, 1180, 1100, 1064, 1030, 982, 852, 773, 690, 660. 1H-NMR (300 MHz, CDCl3) δ: 8.66 (1H, d, J = 2.8 Hz), 7.76 (1H, dd, J = 8.4, 2.8 Hz), 7.35 (1H, d, J = 8.4 Hz), 7.32–7.26 (10H, m), 7.19 (1H, t, J = 8.3 Hz), 7.01 (1H, d, J = 8.3 Hz), 6.81 (1H, dd, J = 8.3, 2.6 Hz), 6.71 (1H, t, J = 2.5 Hz), 1.15–1.04 (3H, m), 0.99 (18H, d, J = 6.0 Hz). 13C-NMR (75.5 MHz, CDCl3) δ: 155.9, 151.2, 146.0, 144.7, 142.2, 138.8, 135.6, 129.3, 128.4, 128.2, 127.1, 122.1, 120.0, 118.9, 97.1, 83.8, 56.1, 18.0, 12.7. 29Si-NMR (53.67 MHz, CDCl3) δ: 16.20. HRMS (APCI-TOF), calcd for C65H74NO2Si2 959.5613. Found: 959.5661. Error 1.9 ppm. Anal. calcd for C65H73NO2Si2: C, 81.62; H, 7.69; N, 1.46. Found: C, 81.80; H, 7.83; N, 1.90%.
General synthetic procedure for the deprotection reaction exemplified with 1,4-bis-(([3-hydroxy-diphenyl]-phenylmethyl)-ethynyl)-phenylene (4)
Compound 1 (0.13 g, 0.14 mmol) and tetrabutylammonium fluoride hydrate (0.06 g, 0.20 mmol) were stirred in THF (50 mL) for 0.5 h. The resulting solid was filtered off and washed with water. The organic phase was extracted twice with CH2Cl2 (80 ml). The organic portions were dried over anhydrous Na2SO4, evaporated to dryness and purified by column chromatography on silica gel, using hexane/ethyl ether (7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) as eluent, to afford 4 (0.08 g, 88%). Melting point: 250–252 °C. FTIR-ATR (ν, cm−1): 3429, 3059, 2930, 2228, 1598, 1491, 1448, 1260, 1223, 1031, 842, 755, 699. 1H-NMR (270 MHz, CDCl3) δ: 7.43 (2H, s), 7.34–7.20 (10H, m), 7.16 (1H, t, J = 7.9 Hz), 6.86–6.70 (3H, m), 5.60 (1H, s). 13C-NMR (67.9 MHz, CDCl3) δ: 158.2, 147.5, 146.1, 132.5, 130.0, 129.9, 129.0, 127.9, 124.2, 121.2, 117.2, 114.9, 98.3, 85.6, 57.0. HRMS (APCI-TOF), calcd for C48H35O2 643.2631. Found: 643.2623. Error 1.33 ppm. MS (DIP) m/z: 643 ([M+ + 1, 52), 642 (M+, 100), 565 (23), 360 (21), 359 (49), 283 (47), 281 (59), 265 (43), 252 (20); Anal. calcd for C48H34O2: C, 89.69; H, 5.33. Found: C, 89.36; H, 5.90%.
3) as eluent, to afford 4 (0.08 g, 88%). Melting point: 250–252 °C. FTIR-ATR (ν, cm−1): 3429, 3059, 2930, 2228, 1598, 1491, 1448, 1260, 1223, 1031, 842, 755, 699. 1H-NMR (270 MHz, CDCl3) δ: 7.43 (2H, s), 7.34–7.20 (10H, m), 7.16 (1H, t, J = 7.9 Hz), 6.86–6.70 (3H, m), 5.60 (1H, s). 13C-NMR (67.9 MHz, CDCl3) δ: 158.2, 147.5, 146.1, 132.5, 130.0, 129.9, 129.0, 127.9, 124.2, 121.2, 117.2, 114.9, 98.3, 85.6, 57.0. HRMS (APCI-TOF), calcd for C48H35O2 643.2631. Found: 643.2623. Error 1.33 ppm. MS (DIP) m/z: 643 ([M+ + 1, 52), 642 (M+, 100), 565 (23), 360 (21), 359 (49), 283 (47), 281 (59), 265 (43), 252 (20); Anal. calcd for C48H34O2: C, 89.69; H, 5.33. Found: C, 89.36; H, 5.90%.
1,4-Bis-(([3-hydroxy-diphenyl]-phenylmethyl)-ethynyl)-2,3-difluorobenzene (5)
Compound 2 (0.02 g, 0.02 mmol), tetrabutylammonium fluoride hydrate (0.01 g, 0.03 mmol) and THF (10 ml). Purification on silica gel afforded 5 as a white crystalline solid (0.01 g, 88%). Melting point: 270–272 °C. FTIR-ATR (ν, cm−1): 3359, 2928, 2867, 1611, 1500, 1450, 1287, 1237, 1052, 1017, 870, 819, 623, 570 1H-NMR (500 MHz, CDCl3) δ: 7.32 (s), 7.31 (s), 7.29–7.26 (m), 7.18 (t, J = 8.0 Hz), 7.15 (d, J = 2.0 Hz), 6.87 (d, J = 8.0 Hz), 6.78 (t, J = 2.0 Hz), 6.76 (dd, J = 2.0, 8.0 Hz). 13C-NMR (125 MHz, CDCl3) δ: 155.5, 151.4 (dd, J = 254.8, 14.9 Hz), 150.4, 146.6, 144.5, 129.4, 129.2, 128.2, 127.4, 127.2, 121.9, 116.4, 114.2, 103.5, 77.6, 56.4, 31.1. 19F-NMR δ: -400.62 ppm, HRMS (APCI-TOF), calcd for C48H33F2O2 679.2443. Found 679.2449. Error 0.93 ppm. Anal. calcd for C48H32F2O2: C, 84.94; H, 4.75. Found: C, 85.13; H, 5.04%.Acknowledgements
The authors thank to UNAM for financial support, to CONACYT for granting scholarships to R. Arcos-Ramos (223403) and P. Ramirez-Montes, M.A. Leyva-Ramírez for X-Ray data collection and to G. Cuéllar for HRMS and Mauricio Maldonado-Dominguez for computational studies.Notes and references
- (a) G. T. Carrol, G. London, T. Fernández-Landaluce, P. Rudolf and B. L. Feringa, ACS Nano, 2011, 6, 622 CrossRef PubMed; (b) C. Gao, S. Silva, X. Ma, H. Tian, A. Credi and M. Venturi, Chem.–Eur. J., 1992, 18, 16911 CrossRef PubMed; (c) A. C. Fahrenbach, S. C. Warren, J. T. Incorvati, A. J. Avestro, J. C. Bernes, J. F. Stoddart and B. A. Grzybowski, Adv. Mater., 2013, 25, 331 CrossRef CAS PubMed; (d) M. Wolffs, J. L. J. van Velthoven, X. Lou, R. A. A. Bovee, M. Pouderoijen, J. L. J. Dongen, A. P. H. J. Schenning and E. Meijer, Chem.–Eur. J., 2012, 18, 15057 CrossRef CAS PubMed.
- (a) S. D. Karlen and M. Garcia-Garibay, Top. Curr. Chem., 2006, 262, 179 Search PubMed; (b) C. S. Vogelsberg and M. A. Garcia-Garibay, Chem. Soc. Rev., 2012, 41, 1892 RSC; (c) A. Coskun, M. Banaszak, D. Astumian and J. F. Stoddart, Chem. Soc. Rev., 2012, 41, 19 RSC.
- (a) M. Yamada, M. Kondo, J. I. Mayima, Y. Yu, M. Kinoshita, C. J. Barret and T. Ikeda, Angew. Chem., Int. Ed., 2008, 47, 4986 CrossRef CAS PubMed; (b) T. Yasuda, K. Tanabe, T. Tsuji, K. K. Coti, I. Aprahamian, J. F. Stoddart and T. Kato, Chem. Commun., 2010, 46, 1224 RSC; (c) J. Bernál, D. A. Leigh, M. Lumboska, S. M. Mendoza, E. M. Pérez, P. Rudolf, G. Teobaldi and F. Zerbetto, Nat. Mater., 2005, 4, 704 CrossRef PubMed; (d) R. Eelkema, M. M. Pollard, J. Vicario, N. Katsonis, B. Serrano-Ramón, C. W. M. Bastiaansen, D. J. Broer and B. L. Feringa, Nature, 2006, 440, 163 CrossRef CAS PubMed; (e) B. K. Juluri, A. S. Kumar, Y. Liu, T. Ye, Y. W. Yang, A. H. Flood, L. Fang, J. F. Stoddart, P. S. Weiss and T. J. Huang, ACS Nano, 2009, 3, 291 CrossRef CAS PubMed; (f) L. Kobr, K. Zhao, Y. Shen, A. Comotti, S. Bracco, R. K. Shoemaker, P. Sozzani, N. A. Clark, J. C. Price, C. T. Rogers and J. Michl, J. Am. Chem. Soc., 2012, 134, 10122 CrossRef CAS PubMed; (g) Y. Zhang, W. Zhang, S. Li, Q. Ye, H. Cai, F. Deng, R. Xiong and S. Huang, J. Am. Chem. Soc., 2012, 134, 11044 CrossRef CAS PubMed; (h) D. L. Casher, L. Kobr and J. Michl, Langmuir, 2012, 28, 1625 CrossRef CAS PubMed; (i) M. Xue and K. L. Wang, Sensors, 2012, 12, 11612 CrossRef CAS PubMed; (j) G. Du, E. Moulin, N. Jouault, E. Buhler and N. Giuseppone, Angew. Chem., Ed. Int., 2012, 51, 12504 CrossRef CAS PubMed.
- (a) T. A. V. Khuong, J. E. Nuñez, C. E. Godinez and M. A. Garcia-Garibay, Acc. Chem. Res., 2006, 39, 413 CrossRef CAS PubMed; (b) P. D. Jarowski, K. N. Houk and M. A. Garcia-Garibay, J. Am. Chem. Soc., 2007, 129, 3110 CrossRef CAS PubMed; (c) B. Rodriguez-Molina, N. Farfan, M. Romero, J. M. Méndez-Stivalet, R. Santillan and M. A. Garcia-Garibay, J. Am. Chem. Soc., 2011, 133, 7820 CrossRef PubMed; (d) D. Czajkowska-Szczykowska, B. Rodriguez-Molina, N. E. Magaña-Vergara, R. Santillan, J. W. Morzycki and M. A. Garcia-Garibay, J. Org. Chem., 2012, 77, 9970 CrossRef CAS PubMed; (e) W. Setaka, S. Higa and K. Yamaguchi, Org. Biomol. Chem., 2014, 12, 3354 RSC; (f) W. Setaka and K. Yamaguchi, J. Am. Chem. Soc., 2013, 135, 14560 CrossRef CAS PubMed.
- (a) J. Michl and E. C. H. Sykes, ACS Nano, 2009, 3, 1042 CrossRef CAS PubMed; (b) R. D. Horansky, T. F. Magnera, J. C. Price and J. Michl, Artificial Dipolar Molecular Rotors in Controlled Nanoscale Motion, Lecture Notes in Physics 711, ed. H. Linke and A. Mansson, Springer, Berlin Heidelber, 2007, p. 303 Search PubMed.
- (a) B. E. A. Salech and M. C. Teich, Fundamentals of Photonics, Wiley Interscience, NY, 1991 CrossRef PubMed; (b) L. M. Blinov, Electro-Optic and Magneto-Optical Properties of Liquid Crystals, NY, 1983 Search PubMed; (c) R. E. Newnham, Properties of Materials. Anisotropy, Symmetry and Structure, Oxford University Press, NY, 2005 Search PubMed.
- G. S. Kottas, L. I. Clarke, D. Horinek and J. Michl, Chem. Rev., 2005, 105, 1281 CrossRef CAS PubMed.
- C. E. Godinez and M. A. Garcia-Garibay, Cryst. Growth Des., 2009, 9, 3124 Search PubMed.
- S. D. Karlen, H. Reyes, R. E. Taylor, S. I. Khan, M. F. Hawthorne and M. A. Garcia-Garibay, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 14973 CrossRef CAS PubMed.
- (a) C. Lemouchi, C. S. Vogelsberg, L. Zorina, S. Simonov, P. Batail, S. Brown and M. A. Garcia-Garibay, J. Am. Chem. Soc., 2011, 133, 6371 CrossRef CAS PubMed; (b) C. Lemouchi, K. Iliopoulos, L. Zorina, S. Simonov, P. Wzietek, T. Cauchy, A. Rodríguez-Fortea, E. Canadell, J. Kaleta, J. Michl, D. Gindre, M. Chrysos and P. Batail, J. Am. Chem. Soc., 2013, 135, 9366 CrossRef CAS PubMed.
- W. Morris, R. E. Taylor, C. Dybowski, O. M. Yaghi and M. A. Garcia Garibay, J. Mol. Struct., 2011, 1004, 94 CrossRef CAS PubMed.
- D. V. Soldatov, Chem. Crystallogr., 2006, 36, 747 CrossRef CAS PubMed.
- I. Dance and M. Scudder, CrystEngComm, 2009, 11, 2233 RSC.
- R. Arcos-Ramos, B. Rodriguez-Molina, M. Romero, J. M. Méndez-Stivalet, M. E. Ochoa, P. I. Ramirez-Montes, R. Santillan, M. A. Garcia-Garibay and N. Farfán, J. Org. Chem., 2012, 77, 6887 CrossRef CAS PubMed.
- (a) P. D. Jarowski, K. N. Houk and M. A. Garcia-Garibay, J. Am. Chem. Soc., 2007, 129, 3110 CrossRef CAS PubMed; (b) Z. Dominguez, H. Dang, J. M. Strouse and M. A. Garcia-Garibay, J. Am. Chem. Soc., 2002, 124, 7719 CrossRef CAS PubMed.
- (a) B. Rodriguez-Molina, M. E. Ochoa, N. Farfán, R. Santillan and M. A. Garcia-Garibay, J. Org. Chem., 2009, 74, 8554 CrossRef CAS PubMed; (b) B. Rodriguez-Molina, M. E. Ochoa, M. Romero, S. I. Khan, N. Farfán, R. Santillan and M. A. Garcia-Garibay, Cryst. Growth Des., 2013, 13, 5107 CrossRef CAS.
- E. Pidcock, W. D. S. Motherwell and J. C. Cole, Acta Crystallogr., Sect. B: Struct. Sci., 2003, 59, 634 Search PubMed.
- C. Ratcliffe, Part E. Properties of the Crystalline State Chapter 24 Rotational and Translational Dynamics in NMR Crystallography, ed. R. K. Harris, R. E. Wasylishen and M. J. Duer, John Wiley and Sons Ltd., UK, 2009 Search PubMed.
- S. Toyota, Chem. Rev., 2010, 110, 5398 CrossRef CAS PubMed.
- A. J. Cruz-Cabeza and J. Bernstein, Chem. Rev., 2013, 114, 2170 CrossRef PubMed.
- (a) P. I. Ramirez-Montes, M. E. Ochoa, R. Santillan, D. J. Ramírez and N. Farfán, Cryst. Growth Des., 2014, 14, 4681 CrossRef CAS; (b) A. J. Cruz-Cabeza, J. W. Liebeschuetz and F. H. Allen, CrystEngComm, 2012, 14, 6707 Search PubMed; (c) I. Dance, Supramolecular Inorganic Chemistry, in Perspectives in Supramolecular Chemistry, ed. G. R. Desijaru, John Wiley & Sons, Ltd, 1996, 137 Search PubMed; (d) C. P. Brock and R. P. Minton, J. Am. Chem. Soc., 1989, 111, 4586 CrossRef CAS.
- G. M. Sheldrick, Acta Crystallogr., 2008, A64, 112 CrossRef PubMed.
- A. Altomare, M. C. Burla, M. Camalli, A. G. Moliterni, G. Polidori and R. Spagna, J. Appl. Crystallogr., 1999, 32, 115 CrossRef CAS.
- L. J. Farrugia, J. Appl. Crystallogr., 1999, 32, 837 CrossRef CAS.
Footnotes |
| † Electronic supplementary information (ESI) available: Synthetic procedures, 1H and 13C spectral data, ss NMR data, PXRD data, DSC and TGA. Crystallographic data for the structures in this paper has been deposited with the Cambridge Crystallographic Data Centre as supplementary publication nos. CCDC 943819 (4a-acetone), 943820 (4b-methanol), 943821 (4c-DMSO), 943496 (2) and 943497 (1), these data can be obtained free of charge from the Cambridge Crystallographic Data Centre via www.ccdc.cam.uk/data_request/cif. See DOI: 10.1039/c5ra07422c |
| ‡ The rotator of 2,3-difluoro-1,4-diiodobenzene was obtained modifying a described procedure. Colourless needles (5.3 g, 70%). Melting point (hexanes): 52–54 °C. 1H-NMR [400 MHz, CDCl3] (δ, ppm): 7.25 (m 2H). 13C-NMR [100 MHz, CDCl3] (δ, ppm): 150.3 (dd, J = 250, 20 Hz), 135.1, 82.5. T. Ruasis and M. Schlosser, Eur. J. Org. Chem., 2002, 19, 3351. |
| This journal is © The Royal Society of Chemistry 2015 |