Temperature-responsive zinc oxide nanorods arrays grafted with poly(N-isopropylacrylamide) via SI-ATRP
Qian Feng,
Dongyan Tang*,
Haitao Lv,
Weile Zhang and
Wenbo Li
Department of Chemistry, School of Science, Harbin Institute of Technology, Harbin 150001, China. E-mail: dytang@hit.edu.cn
First published on 30th June 2015
Abstract
Well fabricated ZnO nanorods (ZnO NRs) arrays with preferred-orientation that grown on pre-deposited ZnO seed layers substrate were selected to graft thermo-responsive polymers of poly(N-isopropylacrylamide) (PNIPAM) by surface-initiated atom transfer radical polymerization (SI-ATRP). As a controlled/“living” radical polymerization, SI-ATRP could endow the systems with the characteristics of the grafted PNIPAM, meanwhile, with the maintaining of the properties of the ZnO NRs. The structures of ZnO and the grafted polymer with the relatively high molecular weights (Mn, 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300) and narrow molecular weight distributions (Mw/Mn, 1.19) that determined by GPC detection, were characterized by XRD and FT-IR. The graft amount of PNIPAM on ZnO NRs and the interactions between the two components were determined by TG and XPS, respectively. The relatively thin layers of PNIPAM (∼15 nm) formed around NRs via SI-ATRP method were observed by SEM. And the temperature-sensitivity of the grafted nanorods were proved by contact angles measurements. Furthermore, photodegradation of Rhodamine B (Rh-B) by the grafted nanorods revealed that ZnO NRs–PNIPAM exhibited photocatalysis and temperature responsibility characteristics, indicating the significant potential applications with tunable responsiveness by changing the environmental conditions.
300) and narrow molecular weight distributions (Mw/Mn, 1.19) that determined by GPC detection, were characterized by XRD and FT-IR. The graft amount of PNIPAM on ZnO NRs and the interactions between the two components were determined by TG and XPS, respectively. The relatively thin layers of PNIPAM (∼15 nm) formed around NRs via SI-ATRP method were observed by SEM. And the temperature-sensitivity of the grafted nanorods were proved by contact angles measurements. Furthermore, photodegradation of Rhodamine B (Rh-B) by the grafted nanorods revealed that ZnO NRs–PNIPAM exhibited photocatalysis and temperature responsibility characteristics, indicating the significant potential applications with tunable responsiveness by changing the environmental conditions.
Introduction
ZnO, with a wide large band gap of 3.37 eV at 300 K and a large excitation binding energy of 60 meV,1,2 has been considered as an excellent multifunctional semiconductor candidate.3–5 Different morphologies of ZnO nanostructures have salient effects on their properties and applications.6–8 In particular, the large surface-to-volume ratio, direct carrier conduction path and the efficiency of the exaction recombination9 in nanorods of ZnO (ZnO NRs) make them ideal for sensors,10 light emitting diodes,11 optoelectronic devices,12 dye-sensitized solar cells,13 antibacterial agents,14 etc.To expand the field of applications of ZnO nanostructures and to mitigate shortcomings such as poor solubility in aqueous medium, bad biocompatibility and easy agglomeration of nanoparticles, a number of groups have investigated organic/inorganic nanocomposites combining the optical, electronic, and mechanical properties of inorganic nanorods with the solubility, mechanical and chemical stability of organic polymers. Yang et al.15 designed a novel sandwich-structured UV photodetector composed of polyaniline nanowires and ZnO NRs, which only need UV light as power source. Wu et al.16 fabricated 10,12-pentacosadiynoic acid (PCDA)/ZnO composites and demonstrated a colorimetric change with an increase of ZnO concentrations. Among these ZnO based nanocomposites related researches, considering the untunable performance of ZnO or any other restrictions on the applications of semiconductor materials, efforts on the modification of ZnO nanostructures with functions (such as stimuli-responsive), organic components have received much more considerable attentions.
Stimuli-responsive smart polymers have great potentials in many areas due to their remarkable behaviors in response to external stimuli, e.g. temperature, pH, magnetic field and light.17 So it is expected that organic/inorganic nanocomposites would present the tunable property by incorporating stimuli-responsive polymers into. Of the many environmentally sensitive polymers, poly(N-isopropylacrylamide) (PNIPAM), is used most extensively in the functional textiles, intelligent microfluidic, controlled drug release, and thermally responsive filters.18 With a lower critical solution temperature (LCST) in aqueous solution between 30 and 35 °C, PNIPAM could display reversible switchable behaviors between the hydrophilicity and hydrophobicity depending on its detailed structures.19 Hou et al.20 prepared self-assembled graphene–PNIPAM hydrogels with a 3D network structure using a hydrothermal method to exhibit good electrical conductivity, high mechanical strength and reversible stimulus-sensitive volume changes. Chen et al.21 synthesized photoelectrode nanocomposites by the incorporation of TiO2 nanoparticles with pH-stimuli responsive polymer and found that their photocurrent behavior was tunable in response to pH-stimuli.
Relative to the traditional techniques to obtain nanocomposites, such as blending, in situ polymerization, atomic layer deposition (ALD) and self-assembly,22–25 SI-ATRP could precisely control the molecular characteristics of polymer chains tethered to nanoparticle surfaces and provide a densely packed array of polymer chains to engineer the structure and properties of polymer/inorganic interfaces.26,27 As a controlled/“living” radical polymerization, ATRP exhibits better controlling over the chain lengths, and accordingly, the thickness of the grafted polymers.28,29 Such surface modification via SI-ATRP could maintain the properties of semiconductor materials and introduce the surface properties of the grafted polymers. Benjamin et al.30 modified the surface of ZnO nanowires with a variety of polymers, such as poly(methylmethacrylate) and polystyrene, by ATRP and analyzed the grafting density of the surface initiators. Xu et al.31 synthesized magnetic ZnO surface-imprinted polymers by SI-ATRP and demonstrated potential applications in the recognition and separation of antibiotics.
Herein, we provide a practical and flexible approach to modify ZnO NRs with temperature-stimuli responsive PNIPAM via SI-ATRP method and further investigated their temperature-dependent photocatalytic behaviors. The results revealed that ZnO NRs–PNIPAM still maintained photocatalysis and temperature responsibility, indicating the significant potential for using coating with tunable, environmentally responsive phase transformations.
Experimental
Materials
N-Isopropylacrylamide (NIPAM, 98.0%), was purchased from Tokyo Chemical Industry (Tokyo, Japan) and used without further purification. 3-Aminopropyltriethoxysilane (APTES, 99.0%), 2-bromoisobutyryl bromide (BiBB, 98.0%), copper(I) bromide (CuBr, 99.0%), and N,N,N′,N′′,N′′-pentamethyldiethylenetriamine (PMDETA, 99.0%) were all obtained from Aladdin (Beijing, China) and used directly. Ethanol, ammonia, triethylamine, toluene were all purchased from Sinopharm Reagent Co., Ltd (Beijing, China). Ultrapure water was purified by a KSV Minitrough system (∼18.2 MΩ cm).Deposition of ZnO nanorods and the amino-functionalization
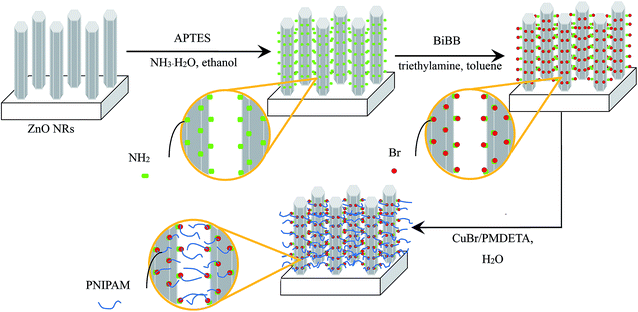
Well-orientated ZnO NRs were prepared by a two-step method including pre-deposition of seed layers of ZnO onto the substrate by LB method and following hydrothermal process of ZnO NRs onto the seed layers, as reported earlier.32Then the ZnO NRs were immersed in a mixture of ethanol (50 mL) and ammonia solution (50 μL). APTES (0.4 mL, 1.7 mmol) was added dropwise and then stirred at 60 °C for 12 h. The amino-functionalized ZnO NRs were rinsed with ethanol to remove the remaining ATPES and then dried in a vacuum at 70 °C overnight.
Synthesis of BiBB-functionalized ZnO nanorods
The above amino-functionalized ZnO NRs were added to BiBB (0.4 mL, 3.2 mmol) in a mixture of toluene (50 mL) and triethylamine (0.45 mL, 3.2 mmol) at 0 °C with constant stirring for 1 h. Then, the mixture was allowed to warm to room temperature and stirred overnight. The BiBB-functionalized ZnO NRs were cleaned with toluene, and dried in a vacuum oven overnight at 70 °C.Grafting of PNIPAM on the surface of ZnO nanorods by SI-ATRP (represented as ZnO NRs–PNIPAM)
SI-ATRP of NIPAM was carried out by immersing the BiBB-functionalized ZnO NRs in a Schlenk flask containing NIPAM (0.7 g, 6.2 mmol), PMDETA (75 μL, 0.4 mmol), and ultrapure water (20 mL). The mixture was degassed by one freeze–pump–thaw cycle. During the frozen state, CuBr (20 mg, 0.14 mmol) was added under the flowing N2. The flask was then subjected to two additional freeze–pump–thaw cycles. To start the reaction, it was warmed to room temperature under nitrogen.33 After 6 h at room temperature, the obtained ZnO NRs–PNIPAM were rinsed thoroughly with ultrapure water and dried in a vacuum oven overnight at 70 °C. The obtained samples were treated with hydrofluoric acid to liberate the grafted PNIPAM chains for further analysis.The general approaches of the synthesis of amino-functionalized, BiBB-functionalized ZnO NRs, and the preparation of ZnO NRs–PNIPAM are shown in Scheme 1.
Characterization
Infrared spectra were collected on a FT-IR spectrometer (FT-IR, AVATER-360B, Nicolet, USA) in the range from 1000 cm−1 to 4000 cm−1.X-ray diffractometer analyses were performed on the D/max-γB X-ray diffractometer (XRD, Rigaku, JP) using Cu Kα radiation in the 2θ range of 30–60° at a voltage of 45 kV and a current of 35 mA.
Molecular weights and molecular weight distributions were determined by a gel permeation chromatograph (GPC, Water, USA) equipped with a Water 1515 pump and a Water 2414 differential refractive index detector. Polystyrene was used as a calibration standard and N,N′-dimethylformamide as the eluent with a flow rate of 1.0 mL min−1.
Surface elemental compositions of ZnO NRs and ZnO NRs–PNIPAM nanocomposites, and the interactions between the two components were characterized and analyzed using X-ray photoelectron spectroscopy (XPS, PHI-5700, Physical Electronics Co., USA) employing an Al Kα X-ray resource (1486.6 eV) and a concentric hemispherical energy electron analyzer operating at 12.5 kV and 250 W with a chamber pressure of 10−6 Torr.
Thermogravimetric analyses were carried out to investigate the graft density of PNIPAM onto the surface of nanorods at a heating rate of 10 °C min−1 from room temperature to 800 °C using thermogravimetric analyze (TG, Pyris 6, Perkin-Elmer, USA).
The morphologies of ZnO NRs and ZnO NRs–PNIPAM nanocomposites were observed by a scanning electron microscope (SEM, HELIOS NANOLAB 600i, FEI, USA).
Thermo-responsive behavior and photocatalytic ability
Contact angles were measured by the sessile drop method at 25 and 45 °C on a contact angle goniometer (CA, JC-200, Shanghai Solon Information Technology, CN) to perform the thermosensitivity of ZnO NRs–PNIPAM nanocomposites. Typically, three drops of the water were placed on the surface of the samples and three readings of the contact angles were taken for each drop. The average of nine readings was used as the final contact angle of each sample. The injection volume was about 2 μL.The degradation experiments of Rhodamine B (Rh-B) aqueous solution (10 mg L−1, 30 mL, pH = 7) under UV light were employed at 25 and 45 °C to evaluate the photocatalytic abilities of ZnO NRs–PNIPAM nanocomposites. Equilibrium experiments were first carried out for two pieces of nanocomposites of the same size (3.0 mg) for 12.0 h to reach adsorption/adsorption equilibrium. The photocatalytic efficiency was calculated by the equation of D = (C0 − Ct)/C0, where D referred to the degradation ratio, C0 and Ct referred to the concentrations of Rh-B solution before and after the irradiation. The concentrations of Rh-B were detected by UV-Vis spectrophotometer (UV-Vis, Beijing Purkinje General Instrument, 760CRT, CN). For comparison, the same method was used for the degradation of Rh-B aqueous solution in the presence of ZnO NRs.
Results and discussions
Materials characterization
The FT-IR spectra of the modified ZnO NRs at various stages are shown in Fig. 1(a) to (d). The strong peak at 460 cm−1 was assigned to the characteristic peak of ZnO34 in Fig. 1(a)–(d) and the absorption peak at 3400 cm−1 corresponded to hydroxyl groups on ZnO NRs in Fig. 1(a). In Fig. 1(b), the new absorption at 1620.1 cm−1 from the amino group could be observed, indicating successful surface modification of ZnO NRs.35 In Fig. 1(c), new peaks from the 2-bromoisobutyramide at 3571, 3384, 1633, 1534, 1475 and 1376 cm−1 demonstrated that the SI-ATRP initiator had successfully been installed on the surface of the BiBB-functionalized ZnO NRs. Strong absorption peaks at 1650 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching) and 1540 cm−1 (N–H bending), and 1460 and 1390 cm−1 (C–H bending) as shown in Fig. 1(d), confirmed the successful graft of PNIPAM via SI-ATRP on the ZnO NRs surfaces.19
O stretching) and 1540 cm−1 (N–H bending), and 1460 and 1390 cm−1 (C–H bending) as shown in Fig. 1(d), confirmed the successful graft of PNIPAM via SI-ATRP on the ZnO NRs surfaces.19
 | ||
| Fig. 1 Fourier transform infrared (FT-IR) spectra of modified ZnO NRs at various stages: (a) unmodified, (b) -amino, (c) -BiBB, and (d) -PNIPAM grafted by SI-ATRP. | ||
Fig. 2 displays the XRD pattern of ZnO NRs and ZnO NRs–PNIPAM nanocomposites. The peaks located at 2θ of 31.68°, 34.48°, 36.22°, and 47.52° were indexed to (100), (002), (101), (102), and (103) diffractions of ZnO, respectively, (JCPDS 36–1451) in curve (a) of Fig. 2. As shown in curve (b) in Fig. 2 of ZnO NRs–PNIPAM nanocomposites, these characteristic peaks also existed, indicating the structures of NRs were unchanged by the SI-ATRP polymerization. Compared with the XRD pattern of ZnO NRs (a), all the peaks of ZnO NRs–PNIPAM in curve (b) had lower intensities. Owing to the encapsulation of ZnO NRs, active sites on the surface of the NRs were coated by PNIPAM partially, resulting in the decline of the above mentioned characteristic peaks.36
The grafted PNIPAM chains were liberated from ZnO NRs–PNIPAM nanocomposites for GPC analysis by treating with hydrofluoric acid. The time evolution of number average molecular weights (Mn), and molecular weight distributions (Mw/Mn) for the cleaved PNIPAM chains is shown in Fig. 3. After 6 h, the PNIPAM had reached Mn of 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 Daltons and a molecular weight distribution (Mw/Mn) of 1.19. A small shoulder appeared in the figure, which might be a result from the impurities or lower molecular weight components. The GPC results exhibited a relatively narrow molecular weight distribution of PNIPAM (around the value of 1.0) on ZnO NRs by controlled/living radical polymerization for our present synthesized procedures.
300 Daltons and a molecular weight distribution (Mw/Mn) of 1.19. A small shoulder appeared in the figure, which might be a result from the impurities or lower molecular weight components. The GPC results exhibited a relatively narrow molecular weight distribution of PNIPAM (around the value of 1.0) on ZnO NRs by controlled/living radical polymerization for our present synthesized procedures.
The wide-scan, N1s, Zn2p and Br3d XPS spectra of ZnO NRs–BiBB and ZnO NRs–PNIPAM nanocomposites are shown in Fig. 4(a)–(c), respectively. Table 1 shows the element concentrations calculated from the XPS detection results. The atomic concentration of C and N in the ZnO NRs–PNIPAM nanocomposites increased from 35.04 to 78.16% and from 1.13 to 8.77%, respectively, while the atomic concentration of O element decreased from 19.14 to 9.83%. This change in C, N and O concentrations on the surface of ZnO NRs was consistent with surface grafting of PNIPAM. Br element in Fig. 4(d) confirmed the present of the ATRP initiators of BiBB on the surface of the ZnO NRs.37 The decrease in Zn from 9.40 to 2.24% indicated some active sites remained even after surface modification of the ZnO NRs–PNIPAM. SI-ATRP in the present work supplied an effective grafting method for polymers with different functions on the surface of the inorganic phase, without sacrificing its original properties.38
 | ||
| Fig. 4 XPS spectra of ZnO NRs–Br and ZnO NRs–PNIPAM: (a) wide-scan, (b) N1s, (c) Zn2p, and (d) Br3d. | ||
| Atomic concentration | |||||
|---|---|---|---|---|---|
| Samples | C (%) | N (%) | O (%) | Zn (%) | Br (%) |
| ZnO NRs–Br | 35.04 | 1.13 | 19.14 | 9.40 | 2.09 |
| ZnO NRs–PNIPAM | 78.16 | 8.77 | 9.83 | 2.24 | 0.00 |
Furthermore, C1s curve fitting spectra of ZnO NRs–PNIPAM are shown in Fig. 5. The three sub-peaks correspond to hydrocarbon (CxHy: 285.5 eV), carbon adjacent to an amide group (C–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O: 286.4 eV), and carbon attached to oxygen and nitrogen (N–C
O: 286.4 eV), and carbon attached to oxygen and nitrogen (N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O: 288.2 eV), respectively.39 The appearance of the three peaks for carbon proved the successful grafting of PNIPAM onto ZnO NRs by the SI-ATRP method.
O: 288.2 eV), respectively.39 The appearance of the three peaks for carbon proved the successful grafting of PNIPAM onto ZnO NRs by the SI-ATRP method.
The grafting amount of the initiator and PNIPAM on the surface of ZnO NRs could be measured by TG analysis.40 Fig. 6 displays the decomposition behavior of ZnO NRs, ZnO NRs–BiBB and ZnO NRs–PNIPAM under a nitrogen atmosphere at different temperatures by TG detection. ZnO NRs show a weight loss of about 4.90% as heating from 50 to 780 °C, in the curve (a), due to the removal of the absorbed water and the decomposition of the hydroxide group. TGA revealed 2.99 wt% difference in weight retention at 780 °C between ZnO NRs and ZnO NRs–BiBB, in the curves (a) and (b), attributed to the degradation of organic components from the grafted initiator on the surface of ZnO NRs, indicating that the grafting amount of the initiator groups on the surface of ZnO NRs was roughly estimated to be 0.100 mmol g−1 calculated by the method in a previous report.41 As shown in the curve (c) in Fig. 6, the weight retention at 780 °C obtained for ZnO NRs–PNIPAM nanocomposites was 73.77%. Using the weight retention at 780 °C of ZnO NRs–BiBB in curve (b) as a reference, the weight content of the grafting polymers was calculated to be 18.34%, indicating the presence of polymer on the surfaces of ZnO NRs. The purpose of this work focused on providing a practical and flexible approach to prepare ZnO NRs–PNIPAM with better photocatalysis performance, as well as temperature responsivity. Too much polymer coated on the NRs would occupy the active sites onto the surfaces of ZnO NRs and sacrificing its original properties. Therefore, it was well demonstrated that PNIPAM molecule chains had been successfully grafted onto ZnO NRs by the SI-ATRP method, in agreement with that of the XPS measurement results of C1s peak of ZnO NRs–PNIPAM, as shown in Fig. 5.42
Fig. 7 displays the morphologies of ZnO NRs and ZnO NRs–PNIPAM nanocomposites by SEM images. The unmodified ZnO NRs had smooth and clean surfaces with typical hexagonal wurtzite structures, shown in Fig. 7(a). These NRs displayed high regularity and crystallinity, suggesting a better photocatalysis performance. In Fig. 7(b) and (c), after the SI-ATRP process to ZnO NRs, the smooth and regular surfaces became rough and their distinct edges changed to indistinct. These phenomena further confirmed the successful grafting of PNIPAM onto the surfaces of ZnO NRs. Compared with the NRs in Fig. 7(a) and (b), the contours of ZnO NRs–PNIPAM nanocomposites expanded and became indistinct due to the grafting of the polymers. Polymers coated on the surfaces of ZnO NRs would interfere the crystallinity of NRs, indicating the reduction of the intensities of all the peaks of ZnO NRs–PNIPAM, that were in agreements with the XRD measurement results shown in Fig. 2. But the grafted ZnO NRs changed their original rod-like structures unobviously, indicating the existence of the characteristic peaks in XRD detections. Furthermore, the diameters of ZnO NRs in Fig. 7(b) were larger than those in Fig. 7(a), implying the formation of a thin layer of polymer with a thickness of approximately 15 nm.
Temperature-sensitive behaviors
To study the temperature-sensitive behaviors of ZnO NRs–PNIPAM nanocomposites, Fig. 8 shows the CA values of the nanocomposites with different contact times at 25 and at 45 °C, respectively. The value of the angles decreased to 39.50° with the increasing of contact time from 0–8 s at 25 °C, while there was no obvious change at 45 °C with the CA values of 121.90°. This phenomenon could be explained by the competition between the intermolecular and intramolecular hydrogen bonds while below and above the LCST values of about 32 °C. Below the LCST, the predominantly intermolecular hydrogen bond between PNIPAM chains and water molecules contributed to the hydrophilicity of PNIPAM. Above the LCST, the intramolecular hydrogen bonding between C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and N–H groups within PNIPAM chains resulted in a compact and collapsed conformation of PNIPAM chains, thus making it difficult for the hydrophilic C
O and N–H groups within PNIPAM chains resulted in a compact and collapsed conformation of PNIPAM chains, thus making it difficult for the hydrophilic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and N–H groups to interact with water molecules. Hence, shown in Fig. 8, this graft ZnO NRs exhibited temperature-sensitive variations between hydrophilicity and hydrophobicity with the change of ambient temperatures. This enabled other functions to be added to the original properties of inorganic materials by surface modification.
O and N–H groups to interact with water molecules. Hence, shown in Fig. 8, this graft ZnO NRs exhibited temperature-sensitive variations between hydrophilicity and hydrophobicity with the change of ambient temperatures. This enabled other functions to be added to the original properties of inorganic materials by surface modification.
Photocatalytic behaviors detections
Photocatalytic activity of ZnO NRs and ZnO NRs–PNIPAM nanocomposites were investigated through the degradation of Rh-B solution at 25 and 45 °C, respectively. Time-dependent absorption spectra of Rh-B solutions under UV light (λ of 365 nm) illumination of ZnO-NRs at 25 °C (with pH of 7.0), ZnO NRs–PNIPAM nanocomposites at 25 °C (with pH of 7.0), and at 45 °C (with pH of 7.0) are shown in Fig. 9(a)–(c). Under UV light illumination for 100 min, the absorption peak corresponding to Rh-B diminished with time, indicating photocatalysis of the ZnO NRs–PNIPAM prepared via SI-ATRP. Degradation ratios of Rh-B solution in the presence of ZnO NRs and ZnO NRs–PNIPAM nanocomposites at 25 and 45 °C are shown in Fig. 10. For ZnO NRs, over 20% Rh-B could be degraded after 100 min of irradiation. A lower extent of Rh-B photodegradation could be obtained of about 14% and 8.0%, for the same irradiation time of ZnO NRs–PNIPAM nanocomposites at 25 and 45 °C, respectively. So, the photocatalytic efficiency of nanocomposites was much lower than that of pure inorganic materials of ZnO NRs, owing mainly to the partial occupation of the active sites on the surface of NRs by SI-ATRP, and suggesting the decline of ZnO content. Whereas, the photocatalytic activity was higher for nanocomposites at 25 °C than that at 45 °C. These results could be reasoned that, at low temperature, the swelling of polymers made them more hydrophilic, and thus the chains of polymers were in a relaxed state.43 The active sites could be exposed, and the Rh-B molecules thus could reach the surface of NRs easily. At higher temperature the collapsed PNIPAM chains block most of the active sites on the surface of NRs, resulting in the reduction of their catalytic abilities (as illustrated in Scheme 2). Hence, the grafting of stimuli-responsive polymers to inorganic materials by the SI-ATRP method, as a feasible and promising method to fabricate valuable catalysis, could provide nanocomposites with better responsibilities over surface modification and further the tunable catalytic properties.36 | ||
| Fig. 10 Degradation percentage of Rh-B solutions in the presence of (a) ZnO NRs, and ZnO NRs–PNIPAM at (b) 25 °C and (c) 45 °C for 100 min, respectively. | ||
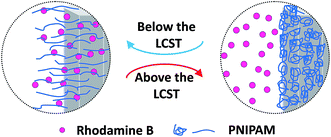 | ||
| Scheme 2 Illustration of the temperature-dependent photodegradation of Rhodamine B with the expanded/collapsed polymer below/above the LCST, thus exposed/blocked the active sites. | ||
Conclusions
Surface-initiated atom transfer radical polymerization (SI-ATRP) was used to graft thermo-responsive PNIPAM onto the surfaces of ZnO nanorods (ZnO NR) arrays with preferred-orientation, to afford novel ZnO NRs-PNIPAM nanocomposites photocatalysts. SI-ATRP ensured grated PNIPAM on the surfaces of ZnO NRs with relatively high molecular weights, narrow molecular weight distribution PNIPAM on the surface of ZnO NRs without sacrificing the active sites of ZnO NRs. The exposed active sites could participate in the photocatalytic reactions, the grafted polymer could endow ZnO NRs temperature responsivity characteristics. The photodegradation of Rhodamine B at lower temperature (25 °C) presented a significant catalysis than that at 45 °C. Moreover, functional PNIPAM grafted onto the surfaces of ZnO NRs could provide nanocomposites with tunable responsiveness by changing the environmental temperatures.Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grand no. 51402073), the Fundamental Research Funds for the Central Universities and Program for Innovation Research of Science in Harbin Institute of Technology, China (no. T201410), and the Excellent Academic Leaders Foundation of Harbin, China (no. 2014RFXXJ017).Notes and references
- C. Klingshirn, Phys. Status Solidi B, 1975, 71, 547 CrossRef CAS PubMed.
- Ü. Özgür, Y. I. Alivov, C. Liu, A. Teke, M. A. Reshchikov, S. Doğan, V. Avrutin, S. J. Cho and H. Morkoç, J. Appl. Phys., 2005, 98, 041301–041404 CrossRef PubMed.
- S. Q. Bi, F. L. Meng, Y. Z. Zheng, X. Han, X. Tao and J. F. Chen, J. Power Sources, 2014, 272, 485–490 CrossRef CAS PubMed.
- D. Iqbal, A. Kostka, A. Bashir, A. Sarfraz, Y. Chen, A. D. Wieck and A. Erbe, ACS Appl. Mater. Interfaces, 2014, 6, 18728–18734 CAS.
- S. K. Arya, S. Saha, J. E. Ramirez-Vick, V. Gupta, S. Bhansali and S. P. Singh, Anal. Chim. Acta, 2012, 737, 1–21 CrossRef CAS PubMed.
- A. H. Jadhav, S. H. Patil, S. D. Sathaye and K. R. Patil, J. Mater. Sci., 2014, 49, 5945–5954 CrossRef CAS.
- S. L. Wang, X. Jia, P. Jiang, H. Fang and W. H. Tang, J. Alloys Compd., 2010, 502, 118–122 CrossRef CAS PubMed.
- K. K. Naik, R. Khare, D. Chakravarty, M. A. More, R. Thapa, D. J. Late and C. S. Rout, Appl. Phys. Lett., 2014, 105, 233101–233106 CrossRef PubMed.
- R. S. Devan, R. A. Patil, J. H. Lin and Y. R. Ma, Adv. Funct. Mater., 2012, 22, 3326–3370 CrossRef CAS PubMed.
- P. Sanguino, T. Monteiro, S. R. Bhattacharyya, C. J. Dias, R. Igreja and R. Franco, Sens. Actuators, B, 2014, 204, 211–217 CrossRef CAS PubMed.
- A. Echresh, C. O. Chey, M. Z. Shoushtari, O. Nur and M. Willander, J. Appl. Phy., 2014, 116, 193104–193112 CrossRef PubMed.
- D. Sett, S. Sarkar and D. Basak, RSC Adv., 2014, 4, 58553–58558 RSC.
- L. Z. Liu, Y. Q. Chen, T. B. Guo, Y. Q. Zhu, Y. Su, C. Jia, M. Q. Wei and Y. F. Cheng, ACS Appl. Mater. Interfaces, 2012, 4, 17–23 CAS.
- R. Kumar, S. Anandan, K. Hembram and T. N. Rao, ACS Mater. Interfaces, 2014, 6, 13138–13148 CrossRef CAS PubMed.
- S. X. Yang, J. Gong and Y. L. Deng, J. Mater. Chem., 2012, 22, 13899–13902 RSC.
- A. Wu, Y. Gu, C. Stavrou, H. Kazerani, J. F. Federici and Z. Iqbal, Sens. Actuators, B, 2014, 203, 320–326 CrossRef CAS PubMed.
- M. R. Islam, Y. F. Gao, X. Li, Q. M. Zhang, M. L. Wei and M. J. Serpe, Chin. Sci. Bull., 2014, 59, 4237–4255 CrossRef CAS.
- J. Ramos, A. Imaz and J. Forcada, Polym. Chem., 2012, 3, 852–856 RSC.
- H. G. Schild, Prog. Polym. Sci., 1992, 17, 163–249 CrossRef CAS.
- C. Y. Hou, Q. H. Zhang, Y. G. Li and H. Z. Wang, Carbon, 2012, 50, 1959–1965 CrossRef CAS PubMed.
- D. Chen and J. H. Li, J. Phys. Chem. C, 2010, 114, 10478–10483 CAS.
- V. B. Schwartz, F. Thétiot, S. Ritz, S. Pütz, L. Choritz, A. Lappas, R. Förch, K. Landfester and U. Jonas, Adv. Funct. Mater., 2012, 22, 2376–2386 CrossRef CAS PubMed.
- Q. Y. Lu, J. J. Zhang, X. F. Liu, Y. Y. Wu, R. Yuan and S. H. Chen, Analyst, 2014, 139, 6556–6562 RSC.
- F. Kayaci, C. Ozgit-Alkgun, I. Donmez, N. Biyikli and T. Uyar, ACS Appl. Mater. Interfaces, 2012, 4, 6185–6194 CAS.
- M. M. L. Arras, C. Schillai and K. D. Jandt, Langmuir, 2014, 30, 14263–14269 CrossRef CAS PubMed.
- C. M. Hui, J. Pietrasik, M. Schmitt, C. Mahoney, J. Choi, M. R. Bockstaller and K. Matyjaszewski, Chem. Mater., 2014, 26, 745–762 CrossRef CAS.
- M. J. Mulvihill, B. L. Rupert, R. He, A. Hochbaum, J. Arnold and P. Yang, J. Am. Chem. Soc., 2005, 127, 16040–16041 CrossRef CAS PubMed.
- J. S. Wang and K. Matyjaszewski, J. Am. Chem. Soc., 1995, 117, 5614–5615 CrossRef CAS.
- K. Matyjaszewski and J. H. Xia, Chem. Rev., 2001, 101, 2921–2990 CrossRef CAS PubMed.
- B. L. Rupert, M. J. Mulvihill and J. Arnold, Chem. Mater., 2006, 18, 5045–5051 CrossRef CAS.
- L. C. Xu, J. M. Pan, J. D. Dai, Z. J. Cao, H. Hang, X. X. Li and Y. S. Yang, RSC Adv., 2012, 2, 5571–5579 RSC.
- Q. Feng, D. Y. Tang, E. Y. Jing, S. Gu and S. Han, J. Alloys Compd., 2013, 578, 228–234 CrossRef CAS PubMed.
- T. Wu, Z. S. Ge and S. Y. Liu, Chem. Mater., 2011, 23, 2370–2380 CrossRef CAS.
- H. Jiang, J. Q. Hu, F. Gu and C. Z. Li, J. Phys. Chem. C, 2008, 112, 12138–12141 CAS.
- K. K. Jena, T. K. Rout, R. Narayan and K. V. S. N. Raju, Polym. Int., 2012, 61, 1101–1106 CrossRef CAS PubMed.
- L. C. Xu, J. M. Pan, Q. F. Xia, F. F. Shi, J. D. Dai, X. Wei and Y. S. Yan, J. Phys. Chem. C, 2012, 116, 25309–25318 CAS.
- X. M. Hou, L. X. Wang and J. C. Hao, Mater. Lett., 2013, 107, 162–165 CrossRef CAS PubMed.
- Z. L. Gong, D. Y. Tang and Y. D. Guo, J. Mater. Chem., 2012, 22, 16872–16879 RSC.
- C. J. Chang and E. H. Kuo, Thin Solid Films, 2010, 519, 1755–1760 CrossRef CAS PubMed.
- P. Pasetto, H. Blas, F. Audouin, C. Boissière, C. Sanchez, M. Save and B. Charleux, Macromolecules, 2009, 42, 5983–5995 CrossRef CAS.
- T. Wu, Y. F. Zhang, X. F. Wang and S. Y. Liu, Chem. Mater., 2008, 20, 101–109 CrossRef CAS.
- L. C. Tan, J. Liu, W. H. Zhou, J. C. Wei and Z. P. Peng, Mater. Sci. Eng., C, 2014, 45, 524–529 CrossRef CAS PubMed.
- M. Chen, M. Dong, R. Havelund, V. R. Regina, R. L. Meyer, F. Besenbacher and P. Kingshott, Chem. Mater., 2010, 22, 4214–4221 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2015 |