Highly processible and electrochemically active graphene-doped polyacrylic acid/polyaniline allowing the preparation of defect-free thin films for solid-state supercapacitors
Yongguang Wang,
Xiangyu Wang,
Shaochun Tang*,
Sascha Vongehr,
Junaid Ali Syed and
Xiangkang Meng*
National Laboratory of Solid State Microstructures, College of Engineering Applied Sciences, Institute of Materials Engineering, Nanjing University, Jiangsu, P. R. China. E-mail: tangsc@nju.edu.cn; mengxk@nju.edu.cn; Fax: +86-25-83595535; Tel: +86-25-83685585
First published on 8th July 2015
Abstract
In the present work, we report the preparation of graphene (G) doped polyacrylic acid/polyaniline (G-PAA/PANI) composites with excellent processibility for ensuring ultrathin, defect-free and highly flexible films, as well as high electrochemical performance. The weight content of PANI is maximized under the constraint of still allowing defect-free films, and the G content is optimized. Interestingly, we combine two steps that both, if taken in isolation as a strategy, worsen the solubility. The PANI and G contents are optimized to be 20 wt% and 1.3 wt%, respectively. The optimal G-PAA/PANI composite film has a gravimetric capacitance of 399 F g−1 at 10 mV s−1, which is more than twice that of pure PANI nanoparticles. Considering the film thickness of only 50 μm, its specific areal and volumetric capacitances are as high as 1.20 F cm−2 and 240 F cm−3. The film still has a gravimetric capacitance of 342 F g−1 at a high scan rate of 100 mV s−1 (86% of that at 10 mV s−1), which promises great potential for applications needing a rapid charge/discharge. An assembled all-solid-state supercapacitor using two such flexible G-PAA/PANI films provides 93 F g−1; an eighteen-fold improvement over that of a previously reported similar device. The capacitor also exhibits excellent electrochemical stability under different bending angles.
1. Introduction
With portable electronics becoming increasingly prevalent, the corresponding power sources require being lightweight, small, highly efficient and stable under different mechanical deformations.1–3 Flexible all-solid-state supercapacitors (ASSS) promise to meet these requirements because of their mechanical flexibility as well as their high power and energy densities, long-term cycle life, and operational security.4–6 In particular, those with planar shaped designs (based on thin films) have gained increasing interest as mobile power supplies as they can be easily integrated with other electronic components.7–9 Nanostructured carbon and transition metal oxides have been demonstrated as excellent electrode materials for supercapacitors,10–14 but they are not suitable for use in flexible electronics directly owing to their brittleness and powder morphology. For applications, they are usually pressed onto a metal substrate (mainly Ni foam15 and Au foil16) via complex procedures. To enhance the adhesion as well as to improve the conductivity, binders and conductive additives are necessary, which decrease the energy density further.17 In addition, such electrode materials may easily detach and would hardly restore their original shape after being bent. Therefore, ideal electrode materials should not only have high specific capacitances, but also be endowed with properties such as shape conformability and capability for simple, additive-free preparation of flexible ASSS.18Conducting polymers have some advantages such as processibility and shape-conformability19 over carbon and metal oxides for flexible ASSS. They provide an opportunity to fabricate flexible films with controllable thickness and tunable composition. Due to the extensive delocalization of π-electrons, polyaniline (PANI) shows a high conductivity and is the most promising polymer supercapacitor material.14,20 However, pure conducting polymers are insoluble powders when synthesized in water, or brittle films when obtained by electro-deposition.21–23 In order to overcome the poor solubility of PANI, various strategies like doping PANI with macromolecular acids24–26 and the introduction of substituent groups (e.g. sulfonic acid ring-substituted polyanilines27) have been developed based on blending processes or copolymerization reactions. But, homogeneous composites with excellent properties are rather difficult to obtain by these methods. A recent trend is toward embedding PANI as the guest in a polymer host matrix such as polymethyl methacrylate,28 polyvinyl carbazole,29 poly(vinyl alcohol),30 or polycarbonate.31 However, these polymer matrixes are all insulating, which decreases the electroactivity and conductivity of PANI significantly. Generally, the electroactivity of composites increases with the aniline content, but solubility decreases sharply. In particular, continuous, ultrathin, defect-free and highly flexible films are difficult to achieve from composite suspensions by blending or physical doping.
In this work, we develop a novel method to prepare graphene (G) doped polyacrylic acid (PAA)/PANI composite films with both extraordinary mechanical flexibility and high electroactivity. The synthesis starts with an in situ polymerization reaction to obtain a PAA/PANI copolymer, which forms a stable aqueous solution. This is followed by the addition of PANI nanoparticles. The first or second step alone cannot produce a homogeneous suspension with a high PANI content that is also well processed into defect-free films, but the combination of the two steps offers this. The second step adds much PANI in spite of its poor solubility. The addition is maximized under the constraint of still allowing defect-free thin films. G doping increases electron transport between the particles (similar to that of carbon nanotubes’ doping32) while keeping the excellent processibility of the composite suspensions, and the graphene content is adjusted to optimize the electrochemical capacity of the G-PAA/PANI flexible films. The composite films’ properties were evaluated regarding their possible applications in ASSS with a parallel plate assembly, producing a superior flexible capacitor with a specific capacitance of 93 F g−1.
2. Experimental section
2.1. Materials
Polyacrylic acid (PAA) (average molecular weight Mv ∼ 450![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), aniline (ANI), poly(vinyl alcohol) (PVA), ammonium persulphate (APS, (NH4)2S2O8), concentrated hydrochloric acid (HCl), and ethylenediamine (EDA) were purchased from the Sigma-Aldrich Chemical Company. All the chemicals were analytical grade. Aniline was purified by vacuum distillation prior to use. Deionized water with a resistivity exceeding 18.0 MΩ cm from a JL-RO 100 Millipore-Q Plus purifier was used throughout the experiments.
000), aniline (ANI), poly(vinyl alcohol) (PVA), ammonium persulphate (APS, (NH4)2S2O8), concentrated hydrochloric acid (HCl), and ethylenediamine (EDA) were purchased from the Sigma-Aldrich Chemical Company. All the chemicals were analytical grade. Aniline was purified by vacuum distillation prior to use. Deionized water with a resistivity exceeding 18.0 MΩ cm from a JL-RO 100 Millipore-Q Plus purifier was used throughout the experiments.
2.2. Synthesis of PANI nanoparticles
A 0.1 M APS solution was obtained by dissolving 2.28 g APS in 100 ml of 1 M aqueous HCl solution, resulting in acidic conditions with a pH value near zero. Then, 1 ml of distilled aniline was added drop-wise under vigorous stirring at a bath temperature of below 5 °C. After the complete addition of aniline in 30 min, the solution was stirred for another 2 h. The mixture was kept in an air tight flask for 12 h at room temperature (r.t.). A dark bluish gray precipitate was collected by filtration, washed with methanol and 0.1 M HCl, and dried at 80 °C in a vacuum. The obtained PANI powder was stored in a vacuum desiccator for further use. When pH < 2.0, the protonated PANI has good electrical conductivity with metallic characteristics.332.3. Synthesis of PAA/PANI suspensions
A stable PAA/PANI aqueous solution was synthesized by an in situ oxidative polymerization at first. Typically, a mixed solution was prepared by dissolving 0.9 g PAA and 3.25 g APS in 100 ml of 1 M HCl aqueous solution in a 250 ml conical flask under stirring at 0–5 °C. The acidic conditions can also ensure full protonation of PANI. The resulting concentrations of PAA and APS were 0.05 mM (calculated based on the Mv) and 14 mM, respectively. Then, 1 ml of distilled aniline was added drop-wise in 30 minutes under vigorous stirring at a bath temperature of below 5 °C. A stable aqueous mixture was obtained after stirring for another 6 h at the same low temperature. To ensure complete polymerization, the solution was stirred violently for another 24 h at r.t. PANI oligomers and impurities from the oxidative polymerization were removed by dialysis in 0.1 M HCl using a dialysis membrane with a molecular weight cut off of ∼3500 Da. Dialysis was continued until the dialysis solution became colorless again. The pre-prepared PANI nanoparticles were added to obtain PAA/PANI mixtures of different PANI content. The PANI weight percentages per resulting dried films are in a wide range of 1–70 wt%.2.4. Synthesis of graphene nanosheets
Graphene nanosheets were synthesized hydrothermally by reducing graphene oxide obtained from a modified Hummer’s method, as described in our previous work.34 Typically, 500 μl EDA was added to a 2 mg ml−1 graphene oxide suspension under magnetic stirring for 15 min. The mixture was transferred to a 100 ml Teflon-lined autoclave for reaction at 160 °C for 12 h. The resulting products were purified by centrifugation and dried in a freeze dryer for 7 h.2.5. Synthesis of G-PAA/PANI composite films
Dried graphene was added to the 20 wt% PAA/PANI suspension under vigorous stirring. Then the mixture was sonicated continuously for 1 h. Suspensions containing various graphene weight percentages from 0.05 to 5 wt% (relative to the dried film) were obtained. Finally G-PAA/PANI flexible films were prepared by solution casting the suspensions onto a flat substrate and drying at 60 °C for 50 min.2.6. Characterization
The morphology of the composite films was investigated using a Hitachi S-4800 scanning electron microscope (SEM). Fourier-transform infrared (FTIR) spectra of KBr powder-pressed pellets of the G-PAA/PANI composites were recorded on a Perkin Elmer Spectrum-GX spectrometer. X-ray diffraction (XRD) was performed with an Ultima-III X-ray diffractometer. Raman spectra were collected with a JY HR800 laser Raman spectrometer at an excitation wavelength of 632 nm (HeNe laser as the source).2.7. Electrochemical measurements
The electrochemical measurements were carried out using a computer controlled CH1660D electrochemical workstation. To obtain working electrodes, 200 μl of G-PAA/PANI composite suspension was coated onto a Pt plate and dried at 60 °C for 50 min. The mass loading was typically ∼2 mg after drying. Cyclic voltammetry (CV) and galvanostatic charge–discharge (CD) measurements were conducted in a three-electrode cell where a Pt electrode served as the counter electrode and a standard Ag/AgCl electrode as the reference. The electrolyte was 1 M H2SO4 aqueous solution. CV measurements were performed between −0.2 and 1.0 V (vs. Ag/AgCl). The specific gravimetric capacitance (Cg) was calculated by the software according to the integral Cg = ∫idV/(2νmΔV) under the CV curve, where ν is the scan rate, m is the mass of the active material, and ΔV is the operating potential range. The specific areal capacitance (Ca) of each electrode was calculated according to ∫idV/(2νSΔV), where S is the immersed area of electrode and the other parameters are the same as above.A solid gel electrolyte was prepared by pouring 10 g of concentrated H2SO4 into 100 ml of deionized water and then adding 9 g of PVA powder. The mixture was heated to 89 °C under stirring until it became clear. The solution was stirred slowly for 2 h at r.t. in order to eliminate bubbles. The solution was poured into a mold to obtain H2SO4–PVA gel membranes. To assemble flexible solid-state supercapacitors, two pieces of thin Cu foil coated with the G-PAA/PANI composite were used as the electrodes, and the H2SO4–PVA gel membrane was sandwiched in between and thus serves as a solid electrolyte and separator dielectric. Cu foil was thus used as the film substrate and the current collector, as well as making the conductive connections.
3. Results and discussion
Through an oxidative polymerization reaction, the PAA copolymer makes PANI dispersible in water. The capacitance and conductivity of the resulting PAA/PANI films (before the addition of PANI and graphene) both decrease with an increase of CPAA (due to relative reduction of PANI content), but stable solutions cannot be obtained with CPAA below 0.02 mM. Note that the synthesis with a lower CPAA resulted in the generation of precipitates. Thus, we chose the PAA/PANI aqueous solution obtained from CPAA = 0.02 mM, which makes the PANI content ∼1 wt% in the PAA/PANI solution. Fig. 1a shows an aqueous solution containing pure PANI nanoparticles, and the same nanoparticles were added into the PAA/PANI preparation (Fig. 1b). The pure PANI nanoparticles precipitated, while the PAA/PANI allows the added particles to form a homogeneous suspension (no precipitation for several months). When preparing the PAA/PANI by the polymerization reaction, the PANI percentage could not be increased above 1 wt%, while a stable suspension (depicted in Fig. 1b) obtained after the addition of pre-prepared PANI nanoparticles contains 69 wt%. The generation of such a stable suspension allows the fabrication and optimization of thin G-PAA/PANI films, which is discussed later. In other words, this separation of the procedure into two steps allows us to enormously increase the PANI content while preserving the processibility, leading to defect-free, flexible films. While using larger aniline concentrations too early on produces more PANI in the in situ oxidative polymerization, and thus leads to large nanoparticles that will precipitate, the two step method prepares a PAA/PANI matrix in which the added PANI nanoparticles get caught, attaching to the carboxylic functional groups of the PAA.35 The resulting PANI particles being attached with long chains can stay suspended. After adding graphene, the resulting G-PAA/PANI is also still a homogeneous dispersion (Fig. 1c). The suspensions are stable in air for several months when stored in air tight bottles. Fig. 1d and e show the G-PAA/PANI composite films on flexible conductive graphite substrates with different shapes. The bent films do not separate from the substrate (Fig. 1f). Fig. 1g shows a G-PAA/PANI film removed from the Pt substrate, demonstrating excellent flexibility. The cross-sectional SEM image (Fig. 1h) shows that the film has a thickness of about 50 μm.Fig. 2 shows PAA/PANI (before G doping) films containing different weight percentages of PANI per resulting dried film (wPANI) of 10, 16, 20, 32, 37 and 42 wt% on a flat metal substrate. Up to 20 wt%, defect-less surfaces result upon drying (Fig. 2a–c). Further increasing wPANI results in the generation of cracks (Fig. 2e and f). As discussed above, homogeneous and stable suspensions can be obtained at a higher wPANI of 56 and 69 wt%, but more cracks appear in the corresponding films. This is likely due to the fact that there is too little PAA/PANI matrix per added PANI nanoparticles. With less macromolecular matrix holding the film together against the slight contraction due to drying, the films break up.
 | ||
| Fig. 2 SEM photos showing the surface morphology of PAA/PANI composite films with a PANI content of 10, 16, 20, 32, 37, and 42 wt% (a–f respectively). | ||
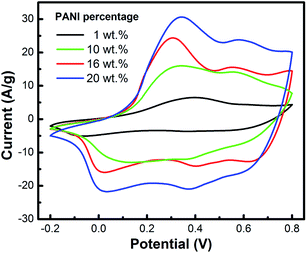
Fig. 3 shows the CV curves of the Pt electrodes coated with defect-free PAA/PANI composite films with different wPANI of 1, 10, 16 and 20 wt% at a scan rate of 50 mV s−1. There are two oxidation–reduction peaks, which are attributed to the structure transitions of the redox of PANI to emeraldine, and from emeraldine to pernigraniline. The area of the CV curve increases with increasing wPANI. These observations suggest that the added PANI is the main active component. The specific capacitances of the film samples with wPANI of 1, 10, 16 and 20 wt% were calculated to be 72, 162, 212, and 296 F g−1, respectively. Note that the PAA/PANI composite films with high wPANI are very susceptible to structure fractures (the cracks) during cycling, as demonstrated by the CV curves for the cycling measurements. Combined with the SEM results, a wPANI of 20 wt% is therefore shown to be the value that provides the optimal balance to ensure a continuous, defect-less and flexible film combined with a high Cg. Interestingly, starting above only 10 wt%, the Cg value of the PAA/PANI composite is already higher than that of the pure PANI nanoparticles (∼150 F g−1),36,37 which of course has 100 wt% PANI. Thus, PAA does not only serve to disperse more PANI in the suspension. It also improves the desired electrochemical properties, similar to the studies that dope PANI with PAA.38 This improvement can be partially attributed to the modification of PANI by the interaction with the PAA chains through an acid–base reaction.
 | ||
| Fig. 3 CV curves of Pt electrodes coated with PAA/PANI composites containing different PANI percentages. | ||
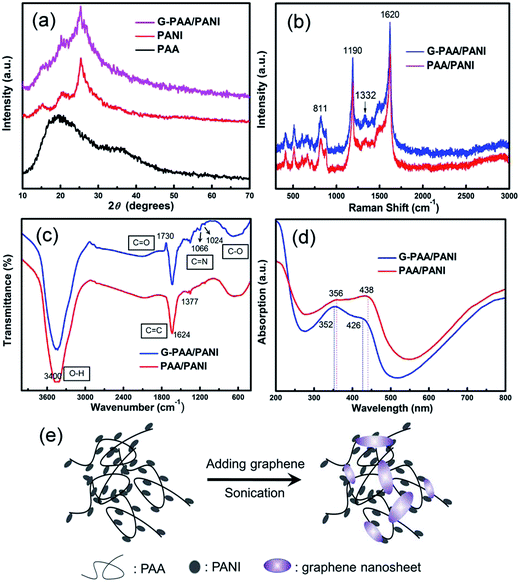
Fig. 4a shows the XRD patterns of the G-PAA/PANI composite (wPANI = 20 wt%, wG = 1.3 wt%). The diffraction peaks at 2θ = 16.34°, 20.41° and 25.32° correspond to PANI. The broad amorphous band with a peak at 18.42° is due to the PAA. The PANI related peaks are not shifted in the PAA/PANI spectrum, which indicates that no other organics were generated through the PAA action on the PANI. The peaks corresponding to graphene are not observable since its content in the composites is fairly low. Graphene is also not found in the Raman spectra (Fig. 4b). A remarkable change is observed in the FTIR characteristic spectra of the PAA/PANI composites with and without graphene (Fig. 4c). In G-PAA/PANI, there are oxygenous groups on the graphene sheets, which can easily interact with the nitrogen atoms in the –NH groups of PANI through hydrogen bonds. Thus, the change is due to an acid–base reaction between the –NH group of PANI and the –COOH groups on graphene. The band at 1730 cm−1 is due to the stretching of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups of PAA, indicating the presence of PAA chains in G-PAA/PANI. In the UV-visible absorption spectra (Fig. 4d), the PAA/PANI composites show two peaks at 356 nm and 438 nm due to π–π* and polaronic transitions.24 After doping with graphene, the characteristic peaks both shift towards shorter wavelengths, confirming a chemical interaction between graphene and PAA/PANI. During the hydrothermal synthesis of graphene nanosheets, some oxygen species such as hydroxyl, carbonyl, and carboxyl remained,39 which promotes the dispersion and combination of graphene sheets with PAA/PANI by chemical interactions. Protonated PANI particles, formed during the in situ polymerization and added after that, are attached to the PAA framework by chemical bonding. Graphene is very light and so it can distribute all over the sample; moreover, the sheets being also very thin allows them to spread out far and connect many PAA/PANI nanoparticles. The dispersion of G-PAA/PANI could be stable for several months. Also, a conductive network is set up in the composite system by the bridges of graphene. The scheme in Fig. 4e illustrates that graphene is closely integrated with the PAA/PANI assembly, allowing the enhancement of electron transfer.
O groups of PAA, indicating the presence of PAA chains in G-PAA/PANI. In the UV-visible absorption spectra (Fig. 4d), the PAA/PANI composites show two peaks at 356 nm and 438 nm due to π–π* and polaronic transitions.24 After doping with graphene, the characteristic peaks both shift towards shorter wavelengths, confirming a chemical interaction between graphene and PAA/PANI. During the hydrothermal synthesis of graphene nanosheets, some oxygen species such as hydroxyl, carbonyl, and carboxyl remained,39 which promotes the dispersion and combination of graphene sheets with PAA/PANI by chemical interactions. Protonated PANI particles, formed during the in situ polymerization and added after that, are attached to the PAA framework by chemical bonding. Graphene is very light and so it can distribute all over the sample; moreover, the sheets being also very thin allows them to spread out far and connect many PAA/PANI nanoparticles. The dispersion of G-PAA/PANI could be stable for several months. Also, a conductive network is set up in the composite system by the bridges of graphene. The scheme in Fig. 4e illustrates that graphene is closely integrated with the PAA/PANI assembly, allowing the enhancement of electron transfer.
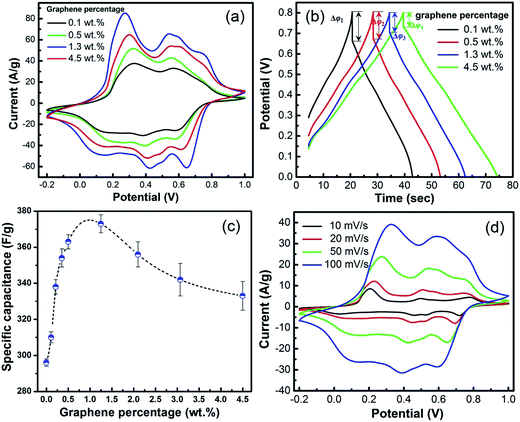
Fig. 5a shows the CV curves of electrodes modified by the G-PAA/PANI composite films with different graphene percentages at a scan rate of 20 mV s−1. No adhesives are necessary to combine the materials with the electrodes. The shape of the curves reveals that the electrochemical capacitances are mainly associated with Faradic pseudo-capacitance. The area of the CV curves and the peak intensities of the oxidation–reduction peaks initially increase rapidly with increasing graphene content. Three redox peaks due to oxidation at about 0.24, 0.48, and 0.70 V (all vs. Ag/AgCl) are observed, and reduction peaks at 0.16, 0.41 and 0.64 V are observed. According to the CV curves, the calculated gravimetric specific capacitances per weight of dried film are 310, 338, 354, 363, 373, 356, 342, and 333 F g−1 at graphene percentages of 0.1, 0.2, 0.36, 0.5, 1.25, 2.1, 3.1, and 4.5 wt%, respectively. Fig. 5b shows the CD characteristics of the G-PAA/PANI film electrodes with various graphene percentages in a potential range of −0.2 to 1.0 V. The CD curves indicate that the electrode can be charged to saturation within 35 seconds at 3 A g−1, and that the voltage can be up to 0.8 V. The discharge time of the G-PAA/PANI with 1.3 wt% graphene (35.0 seconds) is 13.0 seconds longer than that with only 0.1 wt% graphene (22.0 seconds), meaning that is has a higher specific capacitance, which is consistent with the CV results. The capacitance is optimal around 1 wt% (Fig. 5c), at about double that of the pure PANI nanoparticles.
The IR drop Δφ in the CD curves is due to the internal resistance. Galvanostatic measurements show that the IR drop Δφ decreases with increasing G content in the composite, and it is minimal at a high graphene content of 4.5 wt%. This further confirms that G doping increases electron transport between the particles. Graphene nanosheets bridged the PANI particles in the PAA/PANI composites, which reduced the internal resistance. Interestingly, the average specific capacitance does not increase along with the reduction of internal resistance, and reaches a maximum at around 1 wt% of graphene (see Fig. 5c). The specific capacitance is optimized at the point of 1.3 wt%, but not at 4.5 wt%. This can be explained as follows: the performance increases steeply as soon as very little graphene is added, which is usually observed with graphene doping40 and consistent with the fact that it mainly facilitates electron transport. Once conduction between most of the particles is ensured, further addition of graphene no longer improves the connection but merely decreases the content of the electrochemically active material. Therefore, the contribution of the added graphene to the performance improvement reduces at higher contents.
Fig. 5d shows the CV curves of our optimized G-PAA/PANI composite film (1.3 wt% graphene) in 1 M H2SO4 solution at various scan rates. The areas of the curves increase with the scan rate. The Cg values of G-PAA/PANI are much higher than those of the PAA/PANI composites measured at the same scan rates. At 10 mV s−1, the gravimetric capacitance of the optimal G-PAA/PANI is as high as 399 F g−1, which is in the upper range of the previously reported values for flexible polymer based films.14 Considering the film thickness of only 50 μm, the specific areal and volumetric capacitances of the G-PAA/PANI with 1.3 wt% graphene are very high, namely as high as 1.20 F cm−2 and 240 F cm−3. Compared with other modified PANI nanostructures such as paper-like composite films of G-PANI-nanofibers (210 F g−1),41,42 our composite reaches a roughly doubled value, while excellent processibility ensures defect-free films. There is no obvious change in the shape of the CV curves with increasing scan rate and the shape is conserved even at high scan rates. The G-PAA/PANI composite still has a Cg of 342 F g−1 even at 100 mV s−1 (86% of the Cg at 10 mV s−1), making it a very promising candidate for rapid charge/discharge applications. The high Cg at high scan rates confirms that graphene doping facilitates electron transport. The film exhibits a very high volumetric power (1.43 W cm−3), gravimetric power (52 W h kg−1), and energy density (20 mW h cm−3). The fast response confirms the efficient ion and electron transport within the films, which promises the great potential of the G-PAA/PANI composite films in flat, flexible all-solid-state devices.
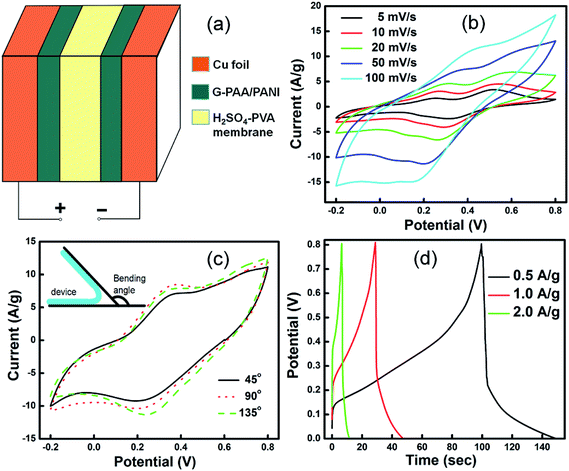
Using the G-PAA/PANI composite film on Cu foil, a flexible all-solid-state capacitor with H2SO4–PVA as the electrolyte was assembled. Fig. 6a shows its sandwich structure. The CV curves of the capacitor (Fig. 6b) show a pseudocapacitive behavior and the curve shape is conserved even at high scan rates. The capacitance of the device reaches 93 F g−1 at a scan rate of 50 mV s−1, which is an eighteen-fold improvement over that of a previously reported similar PAA/PANI device (5.05 F g−1).38 The specific capacitance of the capacitor is almost constant under bending since there is no obvious change in the CV curve shape under bending angles from 0° to 135° (Fig. 6c). Also, this electrochemical behavior is conserved after ten times of repeating the bending cycles, which promises great potential for flexible devices needing an excellent mechanical stability. The galvanostatic CD curves (Fig. 6d) indicate that the capacitor can be charged to saturation within several seconds at 2 A g−1, and that the voltage can be up to 0.8 V. The fast response confirms the efficient ion and electron transport within the films. The potential drops in the CD curves are attributed to a large internal resistance caused by the solid–solid interface between the two electrodes and the H2SO4–PVA gel membrane. The potential drop is generally due to internal resistance mainly from electrical processes at the interfaces,43 and so it is usually larger with solid electrolytes.44
4. Conclusions
In summary, the fabrication of G-PAA/PANI composites with excellent processibility for ensuring continuous, ultrathin, defect-free and highly flexible films, as well as high electrochemical performance is demonstrated. In the preparation process, one of the crucial steps is an in situ polymerization followed by mixing in irregularly shaped pre-prepared PANI nanoparticles, which results in a very high PANI content of 69 wt% without compromising the processibility of the material. At an optimized PANI weight content of 20 wt%, doping with very little (1.3 wt%) graphene nanosheets obtains a superior weight specific capacity. The optimal G-PAA/PANI composite film has a gravimetric capacitance of 399 F g−1 at 10 mV s−1, which is more than twice that of pure PANI nanoparticles. Considering the film thickness of only 50 μm, its specific areal and volumetric capacitances are as high as 1.20 F cm−2 and 240 F cm−3. The film still has 342 F g−1 at a high scan rate of 100 mV s−1, which is 86% of that at 10 mV s−1. An assembled all-solid-state supercapacitor using two such flexible active G-PAA/PANI films provides 93 F g−1, and exhibits an excellent electrochemical stability under different bending angles.Conflict of interest
The authors declare no competing financial interest.Acknowledgements
The authors kindly acknowledge the joint support by the PAPD (no. 50831004), the Fundamental Research Funds for the Central Universities, the Innovation Fund of Jiangsu Province (no. BY2013072-06), the Natural Science Foundation of Jiangsu Province (no. 2012729), the National Natural Science Foundation of China (no. 11374136), and the State Key Program for Basic Research of China (no. 2010CB631004).References
- J. Chmiola, C. Largeot, P. L. Taberna, P. Simon and Y. Gogotsi, Science, 2010, 328, 480–483 CrossRef CAS PubMed.
- L. B. Hu, H. Wu, F. L. Mantia, Y. Yang and Y. Cui, ACS Nano, 2010, 4, 5843–5848 CrossRef CAS PubMed.
- N. Hiroyuki and O. Kenichi, Science, 2008, 319, 737–738 CrossRef PubMed.
- J. Bae, M. K. Song, Y. J. Park, J. M. Kim, M. L. Liu and Z. L. Wang, Angew. Chem., Int. Ed., 2011, 50, 1683–1687 CrossRef CAS PubMed.
- L. H. Bao, J. F. Zang and X. D. Li, Nano Lett., 2011, 11, 1215–1220 CrossRef CAS PubMed.
- X. F. Wang, B. Liu and Q. F. Wang, Adv. Mater., 2013, 25, 1479–1486 CrossRef CAS PubMed.
- C. Z. Meng, C. H. Liu, L. Z. Chen, C. H. Hu and S. S. Fan, Nano Lett., 2010, 10, 4025–4031 CrossRef CAS PubMed.
- B. G. Choi, S. J. Chang, H. W. Kang, C. P. Park, H. J. Kim, W. H. Hong, S. Lee and Y. S. Huh, Nanoscale, 2012, 4, 4983–4988 RSC.
- M. F. El-Kady, V. Strong, S. Dubin and R. B. Kaner, Science, 2012, 335, 1326–1330 CrossRef CAS PubMed.
- Y. M. Dai, S. C. Tang, S. Vongehr and X. K. Meng, ACS Sustainable Chem. Eng., 2014, 2, 692–698 CrossRef CAS.
- Y. W. Zhu, S. Murali and M. D. Stoller, Science, 2011, 332, 1537–1541 CrossRef CAS PubMed.
- A. Rudge, J. Davey, I. Raistrick, S. Gottesfeld and J. P. Ferraris, J. Power Sources, 1994, 47, 89–107 CrossRef CAS.
- N. L. Wu, Mater. Chem. Phys., 2002, 75, 6–11 CrossRef CAS.
- G. A. Snook, P. Kao and A. S. Best, J. Power Sources, 2011, 196, 1–12 CrossRef CAS PubMed.
- X. Zhang, X. Z. Sun, H. T. Zhang, D. C. Zhang and Y. W. Ma, Electrochim. Acta, 2013, 87, 637–644 CrossRef CAS PubMed.
- Y. X. Xu, Z. Y. Lin, X. Q. Huang, Y. Liu, Y. Huang and X. F. Duan, ACS Nano, 2013, 7, 4042–4049 CrossRef CAS PubMed.
- Y. Gu, J. W. Cai, M. Z. He, L. P. Kang, Z. B. Lei and Z. H. Liu, J. Power Sources, 2013, 239, 347–355 CrossRef CAS PubMed.
- D. W. Wang, F. Li, J. P. Zhao, W. C. Ren, Z. G. Chen, J. Tan, Z. S. Wu, I. Gentle, G. Q. Lu and H. M. Cheng, ACS Nano, 2009, 3, 1745–1752 CrossRef CAS PubMed.
- H. Gao and K. Lian, RSC Adv., 2014, 4, 33091–33113 RSC.
- H. H. Zhou, G. Y. Han, Y. M. Xiao, Y. Z. Chang and H. J. Zhai, J. Power Sources, 2014, 263, 259–267 CrossRef CAS PubMed.
- H. Namazi, R. Kabiri and A. Entezami, Eur. Polym. J., 2002, 38, 771–777 CrossRef CAS.
- W. C. Chen, T. C. Wen and A. Gopalan, Synth. Met., 2002, 128, 179–189 CrossRef CAS.
- E. Frackowiak, V. Khomenko, K. Jurewicz, K. Lota and F. Béguin, J. Power Sources, 2006, 153, 413–418 CrossRef CAS PubMed.
- W. B. Zhong, Y. S. Yang and W. T. Yang, Thin Solid Films, 2005, 479, 24–30 CrossRef CAS PubMed.
- X. H. Lu, C. Y. Tan, J. W. Xu and C. B. He, Synth. Met., 2003, 138, 429–440 CrossRef CAS.
- M. Ibrahim and E. Koglin, Acta Chim. Slov., 2005, 52, 159–163 Search PubMed.
- X. L. Wei, Y. Z. Wang, S. M. Long, C. Bobeczko and A. J. Epstein, J. Am. Chem. Soc., 1996, 118, 2545–2555 CrossRef CAS.
- J. L. Cadenas and H. L. Hu, Sol. Energy Mater. Sol. Cells, 1998, 55, 105–112 CrossRef CAS.
- P. Dutta, S. Biswas and S. Kumar, Mater. Res. Bull., 2002, 37, 193–200 CrossRef CAS.
- A. Mirmohseni and G. G. Wallace, Polymer, 2003, 44, 3523–3528 CrossRef CAS.
- P. S. Rao, S. Subrahmanya and D. N Sathyanarayana, Synth. Met., 2004, 143, 323–330 CrossRef CAS PubMed.
- H. Zengin, W. S. Zhou, J. Y. Jin, R. Czerw, D. W. Smith, L. Echegoyen, D. L. Carroll, S. H. Foulger and J. Ballato, Adv. Mater., 2002, 14, 1480–1483 CrossRef CAS.
- P. M. McManus, R. J. Cushman and S. C. Yang, J. Phys. Chem., 1987, 91, 747–750 CrossRef.
- H. Xie, S. C. Tang, Z. L. Gong, S. Vongehr, F. Fang, M. Li and X. K. Meng, RSC Adv., 2014, 4, 61753–61758 RSC.
- Y. H. Geng, Z. C. Sun, J. Li, X. B. Jing, X. H. Wang and F. S. Wang, Polymer, 1999, 40, 5723–5727 CrossRef CAS.
- F. Fusalba, P. Gouérec, D. Villers and D. Bélanger, J. Electrochem. Soc., 2001, 148, A1–A6 CrossRef CAS PubMed.
- K. S. Ryu, K. M. Kim, N. G. Park, Y. J. Park and S. H. Chang, J. Power Sources, 2002, 103, 305–309 CrossRef CAS.
- B. Gupta and R. Prakash, Mater. Sci. Eng., C, 2009, 29, 1746–1751 CrossRef CAS PubMed.
- B. Xue, J. G. Zhu, N. Liu and Y. X. Li, Catal. Commun., 2015, 64, 105–109 CrossRef CAS PubMed.
- J. Li, H. Q. Xie, Y. Li and Z. X. Li, J. Power Sources, 2011, 196, 10775–10781 CrossRef CAS PubMed.
- Z. P. Li, J. Q. Wang, L. Y. Niu, J. F. Sun, P. W. Gong, W. Hong, L. M. Ma and S. G. Yang, J. Power Sources, 2014, 245, 224–231 CrossRef CAS PubMed.
- Q. Wu, Y. X. Xu, Z. Y. Yao, A. R. Liu and G. Q. Shi, ACS Nano, 2010, 4, 1963–1970 CrossRef CAS PubMed.
- K. Pandey, P. Yadav and I. Mukhopadhyay, RSC Adv., 2014, 4, 53740–53751 RSC.
- C. Z. Meng, C. H. Liu, L. Z. Chen, C. H. Hu and S. S. Fan, Nano Lett., 2010, 10, 4025–4031 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |