Characterization and interfacial properties of the surfactant ionic liquid 1-dodecyl-3-methyl imidazolium acetate for enhanced oil recovery
Iago Rodríguez-Palmeiro,
Iria Rodríguez-Escontrela,
Oscar Rodríguez,
Alberto Arce and
Ana Soto*
Department of Chemical Engineering, University of Santiago de Compostela, E-15782, Santiago de Compostela, Spain. E-mail: ana.soto@usc.es
First published on 16th April 2015
Abstract
The use of surfactant ionic liquids has recently gained attention for surfactant-based enhanced oil recovery. The introduction of the surfactant character in ionic liquids combines interesting properties of both types of chemicals, which are relevant for this application. An imidazolium-based surfactant has been synthesized combined with an acetate counter-ion. The aggregation effects in water were evaluated by means of surface tension and electrical conductivity. The dynamic interfacial tensions between the aqueous solutions of the surfactant ionic liquid and crude oil (Saharan blend) were evaluated by the spinning drop method. The effect of different variables has been analyzed, namely the concentration of surfactant ionic liquid, electrolytes (NaCl) and alkalis (NaOH, Na2CO3), and temperature. Formulations of ionic liquids and alkalis were tested for the first time for enhanced oil recovery. The results obtained here improve significantly (at least one order of magnitude, most often two) previous results obtained up to now solely with ionic liquids.
Introduction
Oil extraction presents a series of difficulties due to different resistances to oil flow within the reservoirs. Enhanced Oil Recovery (EOR) techniques are used to improve the oilfield extraction efficiency by reducing such resistances. There are different types, depending on the mechanism of the flowing resistance.1,2 High viscosity can be reduced by increasing the temperature (thermal methods), but nowadays efforts are directed towards chemical methods. This approach is based on the use of additives to improve the water flooding and its capacity to push the oil out of the reservoir. Different chemicals can be used with different purposes. Surfactants are used to reduce the water/oil interfacial and thus facilitate the extraction of oil trapped in the pores. Alkalis are added to produce surfactants in situ, by hydrolysis reaction with acid groups present in the oil. Polymers are used to increase water viscosity and thus its capacity to push the oil out, reducing water digitations and formation of preferential paths. Most times, best results are accomplished combining different chemicals and thus complementing their effects. Surfactant flooding EOR has been studied since the 1970's, and the use of ionic and non-ionic surfactants, the optimization of their formulation and the formation of microemulsion is well-documented, but it is still under investigation.3–6Ionic liquids are gaining attention in the late 1990's as a new type of solvents, with potential to be greener substitutes of organic (volatile) solvents. Indeed, ionic liquids are greener from the point of view of atmospheric emissions, since they have negligible vapor pressures. But soil and aquatic contamination is another issue that has to be evaluated case by case. One of the big claims about ionic liquids is that they are designer solvents: their properties can be tailored to a given application, and so the solvent can be optimized with a judicious choice of ions and functional groups for any given application. In recent years, there has been a boom on research for surfactant ionic liquids, which are ionic liquids with surface active properties.7–9 Conventional ionic surfactants have a chemical structure with a hydrophobic part (often a long alkyl chain) and an ionic moiety acting as hydrophilic or polar part. A surfactant ionic liquid is just an ionic surfactant that is liquid below 100 °C. The surface activity may be associated to the anion, the cation, or both (biamphiphilic or catanionic surfactants).10–12
The possibility of using surfactant ionic liquids in surfactant flooding EOR has been recently raised in the literature.13–20 Despite the novelty (and difficulties) of these components, there is an encouraging interest on the evaluation of their potential for EOR. If surfactant ionic liquids prove to be efficient additives for EOR, there is no doubt that there is room for their application in the oilfield. The suitability of ionic liquids for surfactant EOR is evaluated by means of reduction of interfacial tension between aqueous and oil phases,13–20 but also through the phase diagrams in mixtures with water and oil (that should be a Winsor type III diagram for microemulsion formation),13 adsorption experiments in reservoir-like core materials15 and/or core-flooding experiments.18,19
The role of surfactants in chemical flooding EOR is to reduce the high water/oil interfacial tension to facilitate the pushing out of the oil from the reservoir pores (increase of the capillary number) by the flooding front. Thus, a significant decrease in aqueous/oil interfacial tension is the proof of concept for a feasible application of surfactant ionic liquids in EOR. Recently, the use of some surfactant ionic liquids on dynamic interfacial tension has been investigated by several researchers.14–18 Most efforts focus on the imidazolium family of surfactants, with chloride counter-ion. But some data with other counter-ions and/or pyridinium, ammonium and phosphonium cations can be found. Very recently, a comprehensive study of effect of alkyl chain length and counter-ion effect was published, using imidazolium-based ionic liquids and focusing on the equilibrium interfacial tension reduction between aqueous and crude oil phases.20 In all these articles, the reduction of interfacial tension shown is limited, even concluding that these salts are promising additives for EOR. A step forward, the optimization of the flooding formulation, is required.
In this work, a new surfactant ionic liquid is synthesized by combination of the 1-dodecyl-3-methylimidazolium cation and the acetate anion. The surface active properties are characterized from surface tension and electrical conductivity in aqueous solution, obtaining the cmc and related parameters. Then, dynamic interfacial tensions between aqueous solutions of this surfactant ionic liquid and crude oil are measured. With the aim of gaining some insights on the effects of important process variables (surfactant ionic liquid composition, temperature, water salt content) or the effect of some alkaline additives, a series of experiments for dynamic interfacial tension are carried out. Comparison of the results found here with those obtained in the literature for other ionic liquids previously studied14–18 are established.
Materials and methods
Materials
The ionic liquid 1-dodecyl-3-methylimidazolium acetate, [C12mim][OAc], was synthesized in a single step metathesis reaction. 1-Dodecyl-3-methylimidazolium bromide (Iolitec, >98%) and potassium acetate (Sigma-Aldrich, 99%) were mixed in 2-propanol (Sigma-Aldrich, ≥99.5%) for 48 hours. Insoluble potassium bromide precipitated as a white solid, and the mixture was filtered in order to remove it. 2-Propanol was removed by evaporation in a rotary evaporator. The ionic liquid was dissolved in cold acetone (Sigma-Aldrich, ACS reagent, ≥99.5%) and filtrated again to achieve for total removal of potassium bromide. No precipitation was observed upon addition of 0.1 M aqueous solution of silver nitrate to an aqueous solution of the ionic liquid. The bromide content (<3700 ppm) was measured using an ion chromatograph (Metrohm 861) equipped with a suppressed conductivity detector, a sample processor (Metrohm 838) and a Metrosep A column (250 × 4.0 mm). A carbonate buffer (3.2 mM Na2CO3 and 1.0 mM NaHCO3) was used as the mobile phase at a flow rate of 0.7 mL min−1. The desired ionic liquid was purified in a vacuum line (<0.1 mbar) at 343.15 K for no less than one day. Its water content (<400 ppm) was measured using a titration method (Metrohm 737 Karl-Fischer coulometer).The crude oil (Saharan blend, 45.3 °API, pour point −20 °C, 30.3 wt% atmospheric residue at 370 °C) was kindly supplied by Repsol (Refinery of A Coruña, Spain). Its characterization is shown in Table 1. Double-distilled water was used in all experiments. Sodium chloride (Sigma-Aldrich, ACS reagent, ≥99%) was dried for 24 h at 400 °C before use. Sodium hydroxide (Panreac, flakes, assay, 98.0–100.5%) and sodium carbonate (Sigma-Aldrich, ACS reagent primary standard, anhydrous, 99.95–100.05% dry basis) were used as received.
| Gravity, °API | 45.3 |
| Density, kg L−1 | 0.8003 |
| Sulphur, wt% | 0.1 |
| Pour point, °C | −20 |
| Viscosity at 40 °C, cSt | 1.6 |
| Acid number, mg KOH g−1 | 0.15 |
| Atmos. residue 370 °C, wt% | 30.3 |
Methods
All the mixtures required for the determination of the critical micelle concentration were prepared gravimetrically on a Mettler-Toledo AE 240 analytic balance with a precision of 10−4 g. Surface tensions were measured using a Krüss K11 tensiometer by the Wilhelmy plate method. Krüss accessory reference PL22, which is a folded platinum ‘plate’, was used. The cylindrical shape allows for measurements using smaller samples. The estimated uncertainty in measurement is ±0.1 mN m−1. The temperature was kept constant by means of a cryogenic circulating water bath Selecta Frigiterm with an uncertainly of ±0.05 K. Electric conductivities were measured in a jacketed glass cell with a Basis 30 Crison electric conductivimeter with an accuracy of 0.5%. Temperature in the cell was controlled using a Selecta Ultraterm thermostatic bath with an uncertainly of ±0.1 K.A Krüss SITE 100 spinning drop tensiometer was employed to carry out the measurement of interfacial tensions between the surfactant solutions and the crude oil. The aqueous phases were used to fill the capillary tube as bulk phase, and crude oil (4 μL) was injected in the middle of the tube as drop phase. Rotating velocities between 3000 and 5000 rpm were applied to measure the interfacial tension in all cases but for pure water/crude oil that needed a rotating velocity of 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm. The interfacial tension was calculated according to the following equation:
000 rpm. The interfacial tension was calculated according to the following equation:
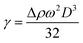 | (1) |
Results and discussion
Aggregation in water
First, the aggregation behavior of the surfactant ionic liquid [C12mim][OAc] in water was evaluated by means of surface tension and electrical conductivity. Surface tension was measured for different concentrations at 298.15 K and results are presented in Fig. 1. The property diminishes with addition of the ionic liquid and then remains constant. The critical micelle concentration, cmc, is this breakpoint, 0.81 mM, identified in Fig. 1. Besides, important parameters can be obtained directly from the surface tension/concentration data such as the surface tension at cmc, γcmc = 36.8 mN m−1, or the pC20 = 3.27 (negative logarithm of the concentration, C20, needed to reduce solvent surface tension in 20 mN m−1). The surface excess concentration of surfactant at saturation, Γm, and the minimum area per surfactant molecule at saturated interface, asm, can be calculated from the experimental data close to (but below) the cmc using the equations:21
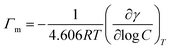 | (2) |
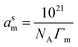 | (3) |
 | ||
| Fig. 1 Surface tension of aqueous solutions of surfactant ionic liquid [C12mim][OAc] at 298.15 K and atmospheric pressure. Symbols are experimental data. Lines present the linear regression of data just below the cmc, to be used in eqn (2), and the average surface tension above the cmc. | ||
The values obtained for all these properties are presented in Table 2 together with literature values9,22–24 for the same surfactant ionic liquid with different counter-ions (chloride, bromide and iodide). The cmc obtained for [C12mim][OAc] is one order of magnitude lower than in the case of all other ionic liquids. The surface tension at cmc is slightly lower than Br− or Cl− but larger than I−. The pC20 with the acetate anion is larger than for the others, indicating a better efficiency of adsorption of the surfactant at the interface. The cmc/C20 ratio is significantly smaller, following the trend OAc− < Cl− < Br− < I−. This indicates that acetate as counter-ion promotes micellization over surface adsorption more than the halides.21 In a similar way, the surface excess concentration at saturation, Γm, is higher for the acetate anion, and thus the area per molecule, asm, is lower, indicating a higher packing of the surfactant at the aqueous/air interface. The results show that asm diminishes following the series: Cl− > Br− > I− > OAc−.
| Ref. | cmc (mM) | γcmc (mN m−1) | pC20 | cmc/C20 | Γm (μmol m−2) | asm (nm2) | |
|---|---|---|---|---|---|---|---|
| a cmc values obtained by: (1) IFT, (2) EC, (3) 1H NMR spectroscopy, (4) fluorescence, (5) EMF. | |||||||
| [C12mim][OAc] | This work | 0.811 | 36.8 | 3.27 | 1.50 | 7.53 | 0.22 |
| 0.462 | |||||||
| [C12mim]Cl | 9 | 151 | ∼40 | — | — | — | — |
| 133 | |||||||
| 74 | |||||||
| 22 | 13.171 | 42.3 | 2.35 | 2.44 | — | 0.72 | |
| 13.472 | |||||||
| 14.04 | |||||||
| 12.85 | |||||||
| 23 | 16.81 | 38.4 | 2.16 | 2.43 | 2.91 | 0.57 | |
| [C12mim]Br | 24 | 8.731 | 38.677 | 2.454 | 2.48 | 3.095 | 0.537 |
| 9.972 | |||||||
| 23 | 10.61 | 36.80 | 2.38 | 2.54 | 3.03 | 0.55 | |
| [C12mim]I | 23 | 4.61 | 31.7 | 2.80 | 2.90 | 4.47 | 0.37 |
| 5.22 | |||||||
Electric conductivity of aqueous solutions of [C12mim][OAc], measured at 288.15, 298.15 and 308.15 K, are plotted in Fig. 2. The experimental data were fitted by least squares nonlinear regression to the model proposed by Carpena and co-workers,25 in order to obtain the cmc and the slopes before and after it:
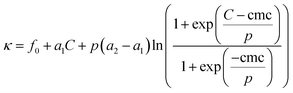 | (4) |
 | ||
| Fig. 2 Electrical conductivity of aqueous solutions of surfactant ionic liquid [C12mim][OAc] at atmospheric pressure. Symbols are experimental data (squares: 288.15 K, circles: 298.15 K, triangles: 308.15 K), lines are regressions for Carpena and co-workers model.25 | ||
| T (K) | f0 | a1 | a2 | p | cmc (mM) | α = a2/a1 | ΔG0mic (kJ mol−1) |
|---|---|---|---|---|---|---|---|
| 288.15 | 0.8197 | 52.6001 | 48.9183 | 0.0008 | 0.39 | 0.93 | −30.45 |
| 298.15 | 1.0079 | 64.0200 | 62.0420 | 0.0007 | 0.46 | 0.97 | −29.88 |
| 308.15 | 1.3458 | 64.0706 | 62.6958 | 0.0008 | 0.38 | 0.98 | −31.09 |
From the values of the cmc and α, the standard Gibbs energy of the micellization process, ΔG0mic, can be calculated as:
ΔG0mic = (2 − α)RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(xcmc) ln(xcmc)
| (5) |
 | ||
| Fig. 3 Gibbs energy of micellization (experimental data from this work and literature22–24) plot against the Gibbs energy of hydration of the counter-ions (calculated by Marcus26). Counter-ions are identified in graph. Line is linear regression of all data. | ||
Dynamic interfacial tension in crude oil/aqueous systems
A value of 19.2 mN m−1 was found for the Saharan crude oil/water interfacial tension. As a starting point, the effect of the concentration of surfactant ionic liquid was evaluated. Three different concentrations were prepared in distilled water: 500, 2000 and 4000 ppm. Dynamic interfacial tension measurements between crude oil and the aqueous solutions were carried out at 298.15 K and atmospheric pressure. As can be seen in Fig. 4, the interfacial tension decreases with concentration of the surfactant ionic liquid, with equilibrium values of 6.91, 2.92 and 2.08 mN m−1 for 500, 2000 and 4000 ppm concentration, respectively. These are also the minimum values except for 2000 ppm, which produced a minimum of 2.77 mN m−1. There is a clear reduction in the interfacial tension with the concentration of [C12mim][OAc], but the efficiency on interfacial tension reduction decreases from a certain surfactant concentration. The same behavior has been found for other surfactant ionic liquids.14–18 Thus, concentration of [C12mim][OAc] was set to 2000 ppm for the following experiments, selected as a reasonable balance between interfacial tension reduction and surfactant spent. | ||
| Fig. 4 Effect of [C12mim][OAc] concentration on the interfacial tension (IFT) between crude oil and water at 298.15 K and atmospheric pressure. | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm provided similar equilibrium values for interfacial tension. Nevertheless, the profiles of the dynamic interfacial tension were different for different salinities. Equilibrium interfacial tension decreased from 2.92 to 1.54 mN m−1, when NaCl concentration was set to 1000 ppm, and stabilized around 1.2 mN m−1 for NaCl concentrations in the mentioned range 2000–40
000 ppm provided similar equilibrium values for interfacial tension. Nevertheless, the profiles of the dynamic interfacial tension were different for different salinities. Equilibrium interfacial tension decreased from 2.92 to 1.54 mN m−1, when NaCl concentration was set to 1000 ppm, and stabilized around 1.2 mN m−1 for NaCl concentrations in the mentioned range 2000–40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm. All dynamic interfacial tension profiles showed minima below the equilibrium values, being for the range 2000–40
000 ppm. All dynamic interfacial tension profiles showed minima below the equilibrium values, being for the range 2000–40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm between 0.4–0.8 mN m−1. In this way, some limit on the interfacial tension reduction by salinity effect was found. Data from the literature14–16 show that increasing NaCl concentration decreases the interfacial tension, but a limit on the effect was only found for 1-dodecyl pyridinium chloride15 and at much higher NaCl concentrations (in the 15–20 wt% range). This may be due to the higher values of the interfacial tension found with those surfactant ionic liquids.
000 ppm between 0.4–0.8 mN m−1. In this way, some limit on the interfacial tension reduction by salinity effect was found. Data from the literature14–16 show that increasing NaCl concentration decreases the interfacial tension, but a limit on the effect was only found for 1-dodecyl pyridinium chloride15 and at much higher NaCl concentrations (in the 15–20 wt% range). This may be due to the higher values of the interfacial tension found with those surfactant ionic liquids.
In order to further check this effect, another alkali (sodium carbonate) was evaluated. Different solutions of [C12mim][OAc] (2000 ppm) in brine (4 wt%) were prepared, varying Na2CO3 concentration from 0 to 1.5 wt%. Dynamic interfacial tensions with crude oil were measured at 298.15 K and the results are presented in Fig. 8. Again, a clear reduction in the interfacial tension is produced, but the composition effect is not clear: addition of 0.25, 0.50, 1.0 or 1.5 wt% of Na2CO3 produced similar equilibrium interfacial tensions between oil and aqueous phase (in the 0.69–0.81 mN m−1 range). In the same way, the minimum interfacial tension in the profiles are all in the 0.29–0.32 mN m−1 range. In any case, best results are found with the use of NaOH as alkali.
Conclusions
The aggregation effects of surfactant ionic liquid [C12mim][OAc] have been evaluated by means of surface tension and electrical conductivity, and the results obtained have been compared with literature values for the same surfactant ionic liquid with halide counter-ions. [C12mim][OAc] presented, in general, lower cmc, better tendency for micellization over adsorption at the interface and lower spontaneity for micellization. Besides, the effect of the counter-ion can be explained by means of their hydration radii, similar to what is known from typical ionic and nonionic surfactants.The dynamic interfacial tensions of aqueous solutions of [C12mim][OAc] with crude oil were evaluated for different formulations and conditions. The results obtained are promising, with a significant lowering of the interfacial tension and stability in the presence of salts (up to 4 wt% NaCl) and alkalis, which further reduce the interfacial tension.
All the previous works found in the literature, testing the use of surfactant ionic liquids to reduce crude oil/aqueous systems interfacial tension, conclude the promising expectative of using these salts for EOR. However, the obtained interfacial tensions are far from the ultra-low values required, barely reaching 1 mN m−1 in most cases.13–18,20 Our results show that combination of surfactant ionic liquids with alkalis is important to further reduce interfacial tension (at least one order of magnitude below the best results from literature14,16), with strong alkalis like NaOH producing larger effects. The enormous number of possibilities due to the tunability of ionic liquids has also to be combined with other aspects classically considered in surfactant EOR to reach the optimal formulation.
Acknowledgements
The authors acknowledge the Ministry of Economy and Competitiveness of Spain for financial support through project CQT2012-33359 (including European Regional Development Fund advanced funding). OR acknowledges financial support from Ministerio de Economia y Competitividad, Spain (Ref RYC-2012-10378).References
- J. G. Speight, Enhanced recovery methods for heavy oil and tar sands, Gulf publishing company, Houston, 2009 Search PubMed
.
- J. J. Sheng, Modern chemical enhanced oil recovery. Theory and practice, Elsevier, Amsterdam, 2011 Search PubMed
.
- J.-L. Salager, A. M. Forgiarini and J. Bullón, J. Surfactants Deterg., 2013, 16, 449–472 CrossRef CAS PubMed
.
- W. Qiao, Y. Cui, Y. Zhu and H. Cai, Fuel, 2012, 102, 746–750 CrossRef CAS PubMed
.
- H. Pei, G. Zhang, J. Ge, L. Jin and L. Ding, Fuel, 2013, 104, 372–378 CrossRef CAS PubMed
.
- A. Bera, T. Kumar, K. Ojha and A. Mandal, Fuel, 2014, 121, 198–207 CrossRef CAS PubMed
.
- J. L. Anderson, V. Pino, E. C. Hagberg, V. V. Sheares and D. W. Armstrong, Chem. Commun., 2003, 2444–2445 RSC
.
- J. Bowers, C. P. Butts, P. J. Martin and M. C. Vergara-Gutierrez, Langmuir, 2004, 20, 2191–2198 CrossRef CAS
.
- M. Blesic, M. H. Marques, N. V. Plechkova, K. R. Seddon, L. P. N. Rebelo and A. Lopes, Green Chem., 2007, 9, 481–490 RSC
.
- J. M. Obliosca, S. D. Arco and M. H. Huang, J. Fluoresc., 2007, 17, 613–618 CrossRef CAS PubMed
.
- M. Blesic, M. Swadzba-Kwasny, J. D. Holbrey, J. N. Canongia Lopes, K. R. Seddon and L. P. N. Rebelo, Phys. Chem. Chem. Phys., 2009, 11, 4260–4268 RSC
.
- K. S. Rao, T. J. Trivedi and A. Kumar, J. Phys. Chem. B, 2012, 116, 14363–14374 CrossRef CAS PubMed
.
- S. Lago, H. Rodriguez, M. K. Khoshkbarchi, A. Soto and A. Arce, RSC Adv., 2012, 2, 9392–9397 RSC
.
- A. Z. Hezave, S. Dorostkar, S. Ayatollahi, M. Nabipour and B. Hemmateenejad, J. Mol. Liq., 2013, 187, 83–89 CrossRef CAS PubMed
.
- A. Z. Hezave, S. Dorostkar, S. Ayatollahi, M. Nabipour and B. Hemmateenejad, Fluid Phase Equilib., 2013, 360, 139–145 CrossRef PubMed
.
- A. Z. Hezave, S. Dorostkar, S. Ayatollahi, M. Nabipour and B. Hemmateenejad, Colloids Surf., A, 2013, 421, 63–71 CrossRef CAS PubMed
.
- A. Z. Hezave, S. Dorostkar, S. Ayatollahi, M. Nabipour and B. Hemmateenejad, J. Dispersion Sci. Technol., 2014, 35, 1483–1491 CrossRef CAS
.
- M. S. Bin-Dahbag, A. A. Al Quraishi, M. S. Benzagouta, M. M. Kinawy, I. M. Al Nashef and E. Al Mushaegeh, J. Pet. Environ. Biotechnol., 2014, 4, 165 Search PubMed
.
- J. F. B. Pereira, R. Costa, N. Foios and J. A. P. Coutinho, Fuel, 2014, 134, 196–200 CrossRef CAS PubMed
.
- S. Sakthivel, S. Velusamy, R. L. Gardas and J. S. Sangwai, Ind. Eng. Chem. Res., 2015, 54, 968–978 CrossRef CAS
.
- M. J. Rosen, Surfactants and interfacial phenomena, John Wiley & Sons Inc., Hoboken, New Jersey, USA, 3rd edn, 2004 Search PubMed
.
- O. A. El Seoud, P. A. R. Pires, T. Abdel-Moghny and E. L. Bastos, J. Colloid Interface Sci., 2007, 313, 296–304 CrossRef CAS PubMed
.
- M. Ao and D. Kim, J. Chem. Eng. Data, 2013, 58, 1529–1534 CrossRef CAS
.
- T. Nazemi and R. Sadeghi, Colloids Surf., A, 2014, 462, 271–279 CrossRef CAS PubMed
.
- P. Carpena, J. Aguiar, P. Bernaola-Galván and C. Carnero Ruiz, Langmuir, 2002, 18, 6054–6058 CrossRef CAS
.
- Y. Marcus, J. Chem. Soc., Faraday Trans., 1991, 87, 2995–2999 RSC
.
- W. Qiao, Y. Cui, Y. Zhu and H. Cai, Fuel, 2012, 102, 746–750 CrossRef CAS PubMed
.
- Z. Xu, A. Y. Shaw, W. Qiao and Z. Li, Energy Sources, Part A, 2012, 34, 1723–1730 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2015 |




