Synthetic preparation of novel 3D Si/TiO2–Ti2O3 composite nanorod arrays as anodes in lithium ion batteries†
Yuan Zhonga,
Chuang Yuead,
Binbin Chenc,
Shibo Suna,
Mingsen Zhengc,
Libo Zhaob,
Suntao Wua,
Jing Li*abd,
Junyong Kanga and
Liwei Lind
aDepartment of Physics/Pen-Tung Sah Institute of Micro-Nano Science and Technology, Xiamen University, Xiamen, Fujian 361005, China. E-mail: lijing@xmu.edu.cn
bState Key Laboratory for Manufacturing Systems Engineering, Xi'an Jiaotong University, Xi'an, Shanxi 710049, China
cDepartment of Chemistry, Xiamen University, Xiamen, Fujian 361005, China
dDepartment of Mechanical Engineering, University of California, Berkeley, CA 94720, USA
First published on 17th April 2015
Abstract
A three dimensional (3D) Si/TiO2–Ti2O3 composite has been synthetically prepared on micro-fabricated Si nanorod arrays (NRs) by a solvothermal method. Improved electrochemical performances were achieved with this composite as the anode in lithium ion batteries compared to the individual Si NRs electrode, which benefited from the volume alleviating effect in the TiO2 thin sheath and better conductivity in Ti2O3 grains. This novel nanostructured composite presents great potential in applications in micro/nano-electro-mechanic systems (M/NEMS), photovoltaic and photocatalytic devices.
Introduction
In recent years, 3D lithium ion batteries (LIBs) have received much attention as promising micro/nano energy storage systems with applications in miniaturized electronic devices due to the advantages of high-specific energy, long-cycling life, low cost, environmental benignity and high safety.1–5 The performance of a battery greatly depends on the materials of the anode, cathode and electrolytes.6,7 Among all the electrode materials, silicon, because of its highest theoretical specific capacity of as much as 4200 mA h g−1, low working voltage and high energy density, has become a promising candidate anode in LIBs.8,9 However, the large volume expansion (∼400%) during the repeated Li-ion intercalation/extraction processes and poor electronic conductivity (6.7 × 10−4 S cm−1), hindered the further improvement of its electrochemical performances.10,11 Generally, two common pathways were proposed to solve these problems: (1) designing nanostructures, such as nanoparticles, nanorods/wires and thin films,12–14 which are believed to promote the diffusion of lithium ion and reduce the volume change effect; (2) making nano composites, which would help to enhance the electrical conductivity by introducing high conductivity materials or alleviate volume expansion by compositing with substances with small volume change.15,16TiO2, with the low cost, abundant and nontoxic properties, is commonly used in photo-catalysis, photovoltaic, and electrochemistry system etc.17–19 In recent works, TiO2 also shows promising applications as the anode in LIBs, due to its small volume expansion ratio (<4%) and good cyclic stability during the repeated Li-ion charge/discharge cycles.20–22 Recently, a TiO2 nanowire/Si composite has been fabricated through a solvothermal process followed by magnetron sputtering deposition, which displayed superior electrochemical performances compared with the pure Si electrode.22 But, still, the poor conductivity of both Si and TiO2 is a challenge issue for further improving the composite's electrochemical performances.
In this work, a novel 3D Si/TiO2–Ti2O3 composite was fabricated on Si nanorod (NR) arrays patterned substrates using a combination method of micro-machining and solution synthesis. TiO2 nano grains were synthetically coated around each Si NR and the flower-like Ti2O3 (electronic conductivity of >1.7 × 103 S cm−1, comparable to graphite)23 flakes were successfully grown and uniformly distributed on and in between 3D core–shell Si/TiO2 NRs, which maintained superior electrochemical performances of this electrode due to the volume restriction in TiO2 shell coating and improved electrical conductivity by Ti2O3 flakes. This shape-controlled 3D Si/TiO2–Ti2O3 composite, embracing an improved cycling performance, has a prospect as the anode for micro-LIBs applied in M/NEMS or IC systems.
Experimental
1. Fabrication of 3D Si/TiO2–Ti2O3 composite
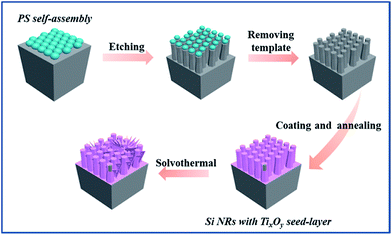
The process of synthesizing 3D Si/TiO2–Ti2O3 composite is illustrated in Scheme 1. Two main parts were included: fabrication of 3D Si substrates by inductive coupled plasma (ICP) dry etching process and the synthesis of TiO2 composite by solvothermal reaction.The fabrication of 3D Si NR arrays with a hexagonal top-view can be referred to our previous work.24 As illustrated in Scheme 1, a compact PS nanospheres (300 nm in diameter) monolayer was spin-coated on a silicon wafer substrate as the template. Then an 8-minute dry etching process was carried out in the ICP system (Alcatel-AMS 200) employing alternative SF6/C4F8 gases under the RF power of 1000 W and 6 mTorr pressure. Finally, the 3D NR arrays were produced by the removal of PS nanosphere templates and following cleaning in DI water and drying in N2.
Subsequently, a TixOy seed-layer was synthesized by dip coating in a titanium(IV) propoxide alcoholic solution (0.75 M) on the above fabricated Si NR arrays followed by an annealing at 450 °C in air for 30 min. A mixture of tetrabutyl titanate (TBT), acetic acid (HAc) (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 in volume ratio) and the Si NRs (in 1 cm × 1 cm pieces) were added into the Teflon-lined (25 mL) autoclave which were then sealed and heated at 200 °C for 30 min. After the above solvothermal process and the following cleaning process by absolute ethyl alcohol for several times and drying overnight at 50 °C,25–27 the novel 3D Si/TiO2–Ti2O3 composite was successfully prepared on the well-prepared Si NRs.
100 in volume ratio) and the Si NRs (in 1 cm × 1 cm pieces) were added into the Teflon-lined (25 mL) autoclave which were then sealed and heated at 200 °C for 30 min. After the above solvothermal process and the following cleaning process by absolute ethyl alcohol for several times and drying overnight at 50 °C,25–27 the novel 3D Si/TiO2–Ti2O3 composite was successfully prepared on the well-prepared Si NRs.
2. Characterizations
Field emission scanning electron microscopy (SEM, Hitachi S-4800) and transmission electron microscopy (TEM, FEI Tecnai F30, 300 kV) were employed to observe the morphologies of the 3D Si/TiO2–Ti2O3 composites. The fine crystal structure and elemental analysis were then worked out by high resolution transmission electron microscopy (HRTEM), X-ray diffraction (XRD, Rigaku Ultima IV, Cu-Kα radiation), selected area electron diffraction (SEAD) and energy dispersive X-ray spectrometer (EDX).3. Electrochemical measurements
Electrochemical performances of the 3D Si/TiO2–Ti2O3 composite were tested by assembling CR-2025 coin cells. The back sides of the well prepared composite substrates (pre-cut into 1 cm × 1 cm pieces) were sputtered consecutively with the 20/200 nm Cr/Au films as the current collector. Then the coin cells were assembled with the 3D Si/TiO2–Ti2O3 composite as the working electrode, lithium metal foil as the counter electrode, a Celgard 2400 membrane as the separator and 1 M LiPF6 which was dissolved in a mixture of vinylene carbonate (VC, 2%), ethylene carbonate (EC) and diethyl carbonate (DEC) as the electrolyte. The whole assembling procedures were performed in a glove box filled with Ar (O2 and H2O content <1 ppm) and a 12 h aging process was carried out before the electrochemical testing of the half-cells. Cyclic voltammetry (CV) tests were conducted on an electrochemical workstation (CHI600A, Chenhua) from 0.01 to 2.5 V vs. Li/Li+ at the scan rate of 0.1 mV s−1. Galvanostatic charge/discharge measurements were recorded within the voltage window of 0.13–2.5 V after an activation process of galvanostatic discharge (Li-ion insertion) for 24 h and subsequent galvanostatic charge (Li-ion extraction) to 2.5 V. All the electrochemical measurements were done at room temperature. Electrochemical Impedance Spectral (EIS) measurements were carried out on a Solartron 1260 electrochemical workstation with amplitude of 5.0 mV and a frequency range of 0.1 Hz to 100 kHz.Results and discussion
1. Morphology and structure characterization
As shown in the top and cross-section SEM images of Fig. 1(a and b) and 2(a and b), the well-arranged Si NRs was successfully produced with a hexagonal top morphology and in a bottle-like shape. The Si NR arrays show the average parallel size of about 130 nm in a height of about 570 nm. After the dip coating and subsequent annealing process, a seed-layer composed of Ti and O contents was uniformly coated around each Si NR from top to bottom as displayed in Fig. 1(c and d) and 2(c and d). Interestingly, as seen in Fig. 1(e and f), after a 30 min solvothermal synthesis process, nanoparticle-like film was homogeneously grown around each Si NR in whole arrays, while flower-like flakes are distributing between Si NRs or extruding above the top surface of Si NR arrays to form a 3D network connecting Si NRs with their neighbors. It is verified that the Ti–O compound layer was successfully synthesized during the solvothermal process by the EDX results, showing the contents of titanium and oxygen as displayed in Fig. 2(e and f). | ||
| Fig. 1 Top and section-view SEM images of (a and b) Si NRs, (c and d) Si NRs coated with a TixOy seed-layer and (e and f) Si/TiO2–Ti2O3 NR array composite. | ||
 | ||
| Fig. 2 Large scale top-view SEM images and the corresponding EDX patterns of (a and b) Si NRs, (c and d) Si NRs coated with a TixOy seed-layer and (e and f) Si/TiO2–Ti2O3 composite NRs. | ||
In order to confirm the crystal structure of the synthesized Ti–O compound around Si NRs, XRD pattern of the Si/TiO2–Ti2O3 composite was measured as illustrated in Fig. 3, in which grazing scan mode was employed in order to eliminate the strong diffraction of the Si substrate. A distinguishable diffraction peak at 53.8° was characterized and may be corresponding to the Ti2O3 (116) or TiO2 (105) plane diffractions with refer to the JCPDS no. 10-0063 and JCPDS no. 21-1272, while another peak at 34.9° can be recognized as the (110) plane diffraction of Ti2O3. Additionally, other two resolved weak diffraction peaks at 25.3° and 55.1° can be assigned to (101) and (211) plane diffractions of anatase TiO2. Therefore, the synthesized Ti–O compound coating layer is polycrystalline and consists of Ti2O3 and TiO2 crystallites. As a comparison, the only Si NR arrays undoubtedly show good single crystallinity as verified in the XRD pattern (normal scan) of Fig. 3 with only strong diffraction peak at 69.1° identified as (400) plane diffraction of Si (JCPDS no. 27-1402).28–30 By the way, it is well accepted that the SiO2 layer would be formed during the synthesis, but it is more like the native oxide in an amorphous state. Therefore, no corresponding diffractions were characterized in the composite's XRD pattern.
To further gain more information about the morphologies and structures of Si/TiO2–Ti2O3 composite, TEM characterizations with the focus on the Ti–O compound were carried out. As seen in the morphology of a randomly selected individual single Si/TiO2–Ti2O3 NR displayed in Fig. 4(a), a dense shell layer well covers around the Si NR with a piece of flake attached beside. The dot-circle pattern, as shown in Fig. 4(b), further confirms the polycrystalline property of the coating layer, while the HRTEM image in Fig. 4(c), corresponding to the area marked in the square of (a), well resolves that the shell layer of Ti–O is majorly composed by anatase (A) TiO2 crystal grains. Part of a Ti–O flower-like piece was characterized in Fig. 4(d), which is obviously composed of flakes (F) and nanoparticles (P). The HRTEM image in the area of the flake “F” as shown in Fig. 4(e) indicates that Ti2O3 crystal grains are the major component, while an individual single nanoparticle (“P”) includes both Ti2O3 and anatase (A) TiO2 crystal grains showing clear grain boundary (GB) between them as evidenced in Fig. 4(f).
The element mapping patterns as shown in Fig. 5(a–d) further indicate the core–shell structure of the Si/TiO2–Ti2O3 NR composite, in which Si NR is the core and Ti–O compound homogeneously exists in the shell.
 | ||
| Fig. 5 TEM image of a Si/TiO2–Ti2O3 NR (a) and the corresponding element mappings (b) O, (c) Ti, and (d) Si. | ||
2. Electrochemical properties
Cyclic voltammetry measurement results as shown in Fig. 6(a and b) were analyzed to explore the electrochemical performance of the Si/TiO2–Ti2O3 composite electrode. As shown in Fig. 6(a), two reduction peaks located at about 0.03 V and 0.16 V can be related to the lithium (Li+) intercalation process. Similarly, the two reversible anodic peaks located at about 0.41 V and 0.54 V are attributed to the lithium (Li+) extraction process. During the first 10 cycles, the corresponding peak current increases with the increase of cycling times, which can be ascribed to the activation of silicon.31,32 The enlargement (within the voltage range of 0.01 V to 2.5 V) in the red block of Fig. 6(a) is illustrated in Fig. 6(b), which clearly displays the reduction (Li+ intercalation) and oxidation (Li+ extraction) peaks of anatase TiO2 at about 1.71 V and 1.98 V (vs. Li+/Li).33,34 It also can be found that as the cycles go on these peaks experience a slight movement around the reduction/oxidation voltage and magnitude decrease in current, which is consistent with previous reports and generally believed to be caused by the amorphization effect and some irreversible reactions.24,35–37 On the other hand, typical charge and discharge curves of Si NRs and Si/TiO2–Ti2O3 NRs electrodes during the 1st, 2nd, 5th and 10th cycle under a current density of 20 μA cm−2 after a galvanostatic discharge (20 μA cm−2) activation process for 24 h in the first cycle are also displayed in Fig. S1(ESI†). The charge plateaus at ∼0.3 and ∼0.5 V can be observed, which are consistent with Li+ intercalation and extraction peaks in the CV curves in Fig. 6(a). In the charge process, a plateau at ∼2 V is characterized, which is consistent with Li+ extraction peaks of anatase TiO2 in the CV curves in Fig. 6(b). The capacity drop after the 1st cycle can be ascribed to the irreversible Li+ storage in active materials or solid electrolyte interface (SEI) film formation. Afterwards, the areal capacity experiences an increase, while the Si/TiO2–Ti2O3 anode presented better and more stable performances.24Fig. 6(c) lays out the rate performance of Si/TiO2–Ti2O3 composite NRs anode compared with those in Si NRs and planar Si electrodes within a voltage window between 0.13 and 2.5 V vs. Li/Li+ after a galvanostatic discharge activation process at a current density of 20 μA cm−2 for 24 h in the first cycle. It can be revealed that the Si/TiO2–Ti2O3 NRs anode held the similar first discharge capacity at about 480 μA h cm−2 as those in Si NRs and planar Si electrodes, but dropped to 290 μA h cm−2 in the second cycle as contrast to the larger reduction to 270 and 150 μA h cm−2 in other two non-composite electrodes either in 3D or planer configurations. Apparently, the Si/TiO2–Ti2O3 NRs anode showed a good capacity stability from current density of 20 to 200 μA cm−2 and presented an even higher capacity increase when returning to 20 μA cm−2 again, which is due to the enhanced Li+ diffusion kinetics during the activation and stabilization process.38,39 Similar results were found in the Si NRs and planar Si electrodes, but the capacities were lower. The improved capacities of Si/TiO2–Ti2O3 NRs anode compared with only Si NRs or planar Si anodes can be understandably ascribed to the contribution of composited Ti–O compounds due to the volume restrict effect in TiO2 shell, the enhanced electronic conductivity contributed by Ti2O3 sheets, and the coating layer's protection from the formation of the SEI layers.40–42 Of course, the TiO2 itself will definitely contribute to the capacity enhancement, but would be in a small portion due to its intrinsic low theoretical capacity compared to that in Si electrode.43,44 The further EIS measurement, as displayed in Fig. S2(ESI†), demonstrates a smaller semicircle diameter in the Si/TiO2–Ti2O3 NRs curve than that in Si NRs electrode within the high-medium frequency region. This indicates that the Si/TiO2–Ti2O3 NRs anode has a smaller charge-transfer resistance and a better conductivity than those in bare Si NRs electrode. The enhanced conductivity may be caused by the existence of highly conductive Ti2O3 flake as a conductive bridge between Si NRs given its conductivity of >1.7 × 103 S cm−1 vs. 10−10 S cm−1 in TiO2.45
Of course, the 3D configuration also plays an important role in enlarging the active surface area and accommodating the restricted volume expansion during cycles. Similarly, as shown in Fig. 6(d), in a larger current density of 300 μA cm−2, the composite electrode presented a capacity increase for long cycles and the areal capacity even increased to 240 μA h cm−2 after the 250th cycle. On the contrary, the Si NRs showed a sharp decrease in capacity, which is understandably caused by the mechanical damage in Si anode imposed by higher current densities.9,46 These results further confirm the protecting and conductive effects of TiO2 and Ti2O3.
3. Post characterizations
The SEM images and XRD pattern of the Si/TiO2–Ti2O3 composite electrode after ten discharge/charge cycles were investigated as shown in Fig. S3(ESI†). The anode was washed by dimethyl carbonate (DMC) and alcohol respectively, and then dried in 40 °C. As illustrated in Fig. S3(a),† the nanorods are well preserved in a large scale and there are still some flowers on the nanorods, which are highlighted by a red block. Also, no apparent mechanical cracks or large volume change can be visualized in the composite Si NRs electrode as seen in Fig. S3(b),† although some parts of TiO2 shell peeled off after the discharge/charge process, which might be caused by the lithium insertion/extraction and/or the cleaning procedures during the disassembly process. It should be noticed that lithium insertion/extraction only happens in the surface region of Si so that the Si NRs configuration is still well maintained after cycles, which is consistent with the previous investigation.24 The corresponding XRD pattern was also measured in a grazing scan mode as displayed in Fig. S3(c).† Two distinguishable diffraction peaks at 56.0° and 56.2° were characterized, which can be recognized as the (2![[4 with combining macron]](https://www.rsc.org/images/entities/char_0034_0304.gif) 1) plane of Ti5O9 and (11 3 1) of Li21Si5. Another peak observed at 37.0° can be ascribed to the (731) plane of Li21Si5 and/or the (2
1) plane of Ti5O9 and (11 3 1) of Li21Si5. Another peak observed at 37.0° can be ascribed to the (731) plane of Li21Si5 and/or the (2![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 0) plane of Ti5O9 (JCPDS no. 18-0747 and JCPDS no. 51-0641). These results once again prove the successful lithium accommodation in the composite electrode.
0) plane of Ti5O9 (JCPDS no. 18-0747 and JCPDS no. 51-0641). These results once again prove the successful lithium accommodation in the composite electrode.
Conclusions
In summary, a 3D Si/TiO2–Ti2O3 NR array composite was fabricated by the combination of ICP dry etching process and a solvothermal synthesis. The improved cycle stability and rate performance were embraced in this composite anode compared with only Si NRs or planar Si electrodes. Under a high current density of 300 μA cm−2, the composite electrode still maintained a much better electrochemistry performance after long cycles. The 3D configuration in Si NRs, volume expansion restriction by TiO2 coating, and the better electrical conductivity of Ti2O3 flakes are believed to contribute to the improved electrochemical performances in this Si-composited anode. The Si-compatible fabrication strategy employed in this work also shows a great prospect in configuration micro-LIBs with applications in M/NEMS or IC devices.Acknowledgements
This work is financially supported by the Science and Technology Project of Fujian Province (2013H0046), Project (sklms2015005) of State Key Laboratory for Manufacturing Systems Engineering (Xi'an Jiaotong University), Fundamental Research Funds for the Central Universities, and the CSC (China Scholarship Council) scholarship under the State Scholarship Fund.Notes and references
- K. Sun, T. S. Wei, B. Y. Ahn, J. Y. Seo, S. J. Dillon and J. A. Lewis, Adv. Mater., 2013, 25, 4539–4543 CrossRef CAS PubMed.
- B. Dunn, C. J. Kim and S. Tolbert, IEEE 23rd International Conference on Micro Electro Mechanical Systems, 2010, 164–167 Search PubMed.
- G. Kim, S. Jeong, J. H. Shin, J. Cho and H. Lee, ACS Nano, 2014, 8, 1907–1912 CrossRef CAS PubMed.
- J. W. Long, B. Dunn, D. R. Rolison and H. S. White, Chem. Rev., 2004, 104, 4463–4492 CrossRef CAS.
- M. Wakihara, Mater. Sci. Eng., R, 2001, 33, 109–134 CrossRef.
- A. K. Shukla and T. P. Kumar, Curr. Sci., 2008, 94, 314–331 CAS.
- J. W. Fergus, J. Power Sources, 2010, 195, 939–954 CrossRef CAS PubMed.
- B. A. Boukamp, G. C. Lesh and R. A. Huggins, J. Electrochem. Soc., 1981, 128, 725–729 CrossRef CAS PubMed.
- M. T. McDowell, S. W. Lee, W. D. Nix and Y. Cui, Adv. Mater., 2013, 25, 4966–4985 CrossRef CAS PubMed.
- J. H. Ryu, J. W. Kim, Y. E. Sung and S. M. Oh, Electrochem. Solid-State Lett., 2004, 7, A306–A309 CrossRef CAS PubMed.
- N. S. Choi, K. H. Yew, H. Kim, S. S. Kim and W. U. Choi, J. Power Sources, 2007, 172, 404–409 CrossRef CAS PubMed.
- M. Y. Ge, J. P. Rong, X. Fang, A. Zhang, Y. H. Lu and C. W. Zhou, Nano Res., 2013, 6, 174–181 CrossRef CAS.
- Y. L. Wang, T. Y. Wang, P. M. Da, M. Xu, H. Wu and G. F. Zheng, Adv. Mater., 2013, 25, 5177–5195 CrossRef CAS PubMed.
- C. J. Yu, X. Li, T. Ma, J. P. Rong, R. J. Zhang, J. Shaffer, Y. H. An, Q. Liu, B. Q. Wei and H. Q. Jiang, Adv. Energy Mater., 2012, 2, 68–73 CrossRef CAS PubMed.
- S. Fang, L. F. Shen, G. Y. Xu, P. Nie, J. Wang, H. Dou and X. G. Zhang, ACS Appl. Mater. Interfaces, 2014, 6, 6497–6503 CAS.
- C. K. Chan, H. Peng, G. Liu, K. McIlwrath, X. F. Zhang, R. A. Huggins and Y. Cui, Nat. Nanotechnol., 2008, 3, 31–35 CrossRef CAS PubMed.
- G. Kim, C. Jo, W. Kim, J. Chun, S. Yoon, J. Lee and W. Choi, Energy Environ. Sci., 2013, 6, 2932–2938 CAS.
- X. B. Chen and S. S. Mao, Chem. Rev., 2007, 107, 2891–2959 CrossRef CAS PubMed.
- T. Ohzuku, Z. Takehara and S. Yoshizawa, Electrochim. Acta, 1979, 24, 219–222 CrossRef CAS.
- D. Deng, M. G. Kim, J. Y. Lee and J. Cho, Energy Environ. Sci., 2009, 2, 818–837 CAS.
- J. H. Jeun, K. Y. Park, D. H. Kim, W. S. Kim, H. C. Kim, B. S. Lee, H. Kim, W. R. Yu, K. Kang and S. H. Hong, Nanoscale, 2013, 5, 8480–8483 RSC.
- Z. Wei, R. S. Li, T. Huang and A. S. Yu, J. Power Sources, 2013, 238, 165–172 CrossRef CAS PubMed.
- S. Tominaka, Chem. Commun., 2012, 48, 7949–7951 RSC.
- C. Yue, Y. J. Yu, J. Yin, T. L. Wong, Y. S. Zang, J. Li and J. Y. Kang, J. Mater. Chem. A, 2013, 1, 7896–7904 CAS.
- C. H. Kwon, H. Shin, J. H. Kim, W. S. Choi and K. H. Yoon, Mater. Chem. Phys., 2004, 86, 78–82 CrossRef CAS PubMed.
- J. F. Ye, W. Liu, J. G. Cai, S. Chen, X. W. Zhao, H. H. Zhou and L. M. Qi, J. Am. Chem. Soc., 2011, 133, 933–940 CrossRef CAS PubMed.
- J. B. Joo, Q. Zhang, I. Lee, M. Dahl, F. Zaera and Y. D. Yin, Adv. Funct. Mater., 2012, 22, 166–174 CrossRef CAS PubMed.
- Y. M. Jiang, K. X. Wang, X. X. Guo, X. Wei, J. F. Wang and J. S. Chen, J. Power Sources, 2012, 214, 298–302 CrossRef CAS PubMed.
- M. Ohwada, K. Kimoto, K. Suenaga, Y. Sato, Y. Ebina and T. Sasaki, J. Phys. Chem. Lett., 2011, 2, 1820–1823 CrossRef CAS.
- M. M. Adachi, M. P. Anantram and K. S. Karim, Nano Lett., 2010, 10, 4093–4098 CrossRef CAS PubMed.
- C. Yue, Y. J. Yu, Z. G. Wu, X. He, J. Y. Wang, J. T. Li, C. Li, S. T. Wu, J. Li and J. Y. Kang, Nanoscale, 2014, 6, 1817 RSC.
- S. H. Ng, J. Z. Wang, D. Wexler, K. Konstantinov, Z. P. Guo and H. K. Liu, Angew. Chem., Int. Ed., 2006, 45, 6896–6899 CrossRef CAS PubMed.
- Z. Chen, D. Q. Zhang, X. L. Wang, X. L. Jia, F. Wei, H. X. Li and Y. F. Lu, Adv. Mater., 2012, 24, 2030–2036 CrossRef CAS PubMed.
- B. Zhao, S. M. Jiang, C. Su, R. Cai, R. Ran, M. O. Tade and Z. P. Shao, J. Mater. Chem. A, 2013, 1, 12310–12320 CAS.
- A. Magasinski, P. Dixon, B. Hertzberg, A. Kvit, J. Ayala and G. Yushin, Nat. Mater., 2010, 9, 353–358 CrossRef CAS PubMed.
- X. P. Gao, H. Y. Zhu, G. L. Pan, S. H. Ye, Y. Lan, F. Wu and D. Y. Song, J. Phys. Chem. B, 2004, 108, 2868–2872 CrossRef CAS.
- D. Bresser, E. Paillard, E. Binetti, S. Krueger, M. Striccoli, M. Winter and S. Passerini, J. Power Sources, 2012, 206, 301–309 CrossRef CAS PubMed.
- L. X. Zeng, F. Y. Xiao, J. C. Wang, S. K. Gao, X. K. Ding and M. D. Wei, J. Mater. Chem., 2012, 22, 14284–14288 RSC.
- Z. S. Wu, W. C. Ren, L. Wen, L. B. Gao, J. P. Zhao, Z. P. Chen, G. M. Zhou, F. Li and H. M. Cheng, ACS Nano, 2010, 4, 3187–3194 CrossRef CAS PubMed.
- D. Yan, Y. Bai, C. Y. Yu, X. G. Li and W. F. Zhang, J. Alloys Compd., 2014, 609, 86–92 CrossRef CAS PubMed.
- S. Tominaka, Inorg. Chem., 2012, 51, 10136–10140 CrossRef CAS PubMed.
- Y. J. Yu, C. Yue, S. B. Sun, W. Lin, H. Su, B. B. Xu, J. T. Li, S. T. Wu, J. Li and J. Y. Kang, ACS Appl. Mater. Interfaces, 2014, 6, 5884–5890 CAS.
- Z. Yang, D. Choi, S. Kerisit, K. M. Rosso, D. Wang, J. Zhang, G. Graff and J. Liu, J. Power Sources, 2009, 192, 588–598 CrossRef CAS PubMed.
- Q. Zhang, W. Zhang, W. Wan, Y. Cui and E. Wang, Nano Lett., 2010, 10, 3243–3249 CrossRef CAS PubMed.
- L. Su, Y. Jing and Z. Zhou, Nanoscale, 2011, 3, 3967–3983 RSC.
- U. Kasavajjula, C. S. Wang and A. J. Appleby, J. Power Sources, 2007, 163, 1003–1039 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra03595c |
| This journal is © The Royal Society of Chemistry 2015 |