Photocatalytic degradation of imidacloprid in soil: application of response surface methodology for the optimization of parameters†
Abstract
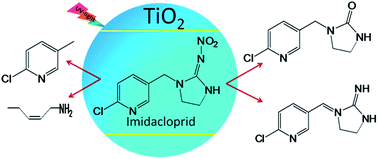
The photocatalytic mineralization of imidacloprid (IMI) in soil to inorganic ions and the formation of various intermediates using TiO2 as the photocatalyst have been investigated under UV light. Various parameters, viz., catalyst concentration, soil depth and pH, intensity of light and initial concentration of IMI were optimized theoretically by using a central composite design based on a response surface methodology and were correlated with experimental results. The statistical analysis from the modelling results indicates that the degradation efficiency of IMI is affected by the depth of soil and the intensity of light, but the effects of the pH and the initial concentration of imidacloprid are more dominant. The optimum conditions obtained for the maximum degradation of imidacloprid were at pH = 3, intensity of UV light = 30 W m−2, soil depth = 0.2 cm and initial concentration of imidacloprid = 10 mg kg−1 of soil. Under these optimum conditions, the highest degradation efficiency of 83% was achieved after 18 h of UV light irradiation. The identification of various photoproduced intermediates of IMI by LC-MS analysis revealed its degradation, whereas the increase in the formation of inorganic ions with time of UV light irradiation confirms its mineralization.

 Please wait while we load your content...
Please wait while we load your content...