Formation and phase transition of hydrogel in a zwitterionic/anionic surfactant system
Haiming Fan*ab,
Shuzhi Zhaoa,
Bingcheng Lia,
Haijian Fana,
Wanli Kanga and
Jianbin Huang*ac
aCollege of Petroleum Engineering, China University of Petroleum (East China), Qingdao 266580, Shandong Province, P. R. China. E-mail: HaimingFan@126.com; Fax: +86-532-86981701; Tel: +86-532-86983051
bState Key Laboratory of Oil and Gas Reservoir Geology and Exploitation, Southwest Petroleum University, Chengdu 610500, Sichuan Province, P. R. China
cBeijing National Laboratory for Molecular Sciences (BNLMS), State Key Laboratory for Structural Chemistry of Unstable and Stable Species, College of Chemistry, Peking University, Beijing 100871, P. R. China. E-mail: JBHuang@pku.edu.cn; Fax: +86-10-62751708; Tel: +86-10-62753557
First published on 5th March 2015
Abstract
The phase behavior and microstructure in a mixture of the zwitterionic surfactant N-hexadecyl-N,N-dimethyl-3-ammonio-1-propane sulfonate (HDPS) and anionic surfactant sodium dodecylsulfate (SDS) were studied. Analysis of its macroscopic appearance, tube inversion testing and rheological measurements were employed to characterize its phase behavior, and it was found that a hydrogel formed in an appropriate total concentration (CT) and molar percentage of SDS (XSDS) at 25 °C for HDPS/SDS systems. Microstructures in the hydrogel were identified to be long wormlike micelles and small spherical vesicles, using transmission electron microscopy (TEM). The coexistence of wormlike micelles and small vesicles brings an appropriate packing parameter (p), which indicates that the wormlike micelles reached a sufficient length and degree of entanglement to form the three-dimensional elastic hydrogel. The HDPS/SDS hydrogel transforms into a viscoelastic sol upon increasing the temperature, and the determined gel–sol transition temperature (Tg–s) has been determined to be around 30 °C, using optical and rheological methods. Besides, adding salt causes the wormlike micelles to lengthen and the rheological properties of the solution to change, such that it may even induce a sol–gel phase transition in the mixed zwitterionic and anionic surfactant system.
Introduction
Hydrogels have attracted considerable growing interest because of their unique features and potential applications as, for example, sensors or optical components,1,2 scaffolds for tissue engineering,3,4 templates for nanomaterial synthesis5–8 and carriers for drug release and delivery.9,10 The formation of hydrogels with biopolymers, such as collagen, hyaluronic acid, fibrin, alginate, F-actin and chitosan, has been well-documented, as well as with synthetic polymers generated from derivates of poly(hydroxyethyl methacrylate) (PHEMA), poly(ethylene glycol) (PEG), poly(vinyl alcohol), poly(acrylic acid), poly(methacrylic acid), polyacrylamide and other derivatives.11,12 Gelation with polymeric gelators is believed to occur by the chemical and/or physical cross-linking of polymeric chains, leading to the formation of a highly intertwined three-dimensional network, which restrains water molecules by surface tension. On the other hand, low-molecular mass hydrogelators create three-dimensional network structures through molecular self-assemblies driven by noncovalent physical interactions, and gelation results from a balance of solubilization and crystallization.13–15 A wide variety of molecular species have been discovered to act as hydrogelators over the last few decades, including amino acid derivates, polypeptides, carbohydrate derivates, bile acid, lipids and surfactants. Compared to polymeric hydrogelators, these small molecule hydrogelators offer the advantage of easy control over the gel properties, by changing parameters such as the temperature, pH value or salinity, or through mechanical agitation. Furthermore, they are easier to degrade than most polymers, which benefits biological applications.Surfactants are one particularly interesting class of hydrogelators.15–18 Surfactant molecules are often quite soluble in water, and they self-assemble into various aggregates, such as wormlike micelles,19–23 vesicles,24–26 lamellar structures,27–29 nanofibers and microtubes30–36 in aqueous solution, above the so-called critical micelle concentration. These aggregates have shown the ability to build stable hydrogels. For example, Gradzielski et al.25 studied the gel phase formed in tetradecyldimethylamine oxide (TDMAO)/tetradecyltrimethyl-ammonium bromide (TTABr) mixed systems, and the formation of hydrogel was attributed to densely packed monodisperse and unilamellar vesicles. González et al.34 investigated the effect of pH on the phase behavior of sodium dodecylsulfate (SDS)/lysine mixtures, and it was found that the gels were formed by the entanglement of fibers that were hundreds of micrometers long. Lin et al.35 found that elastic hydrogel formed in a mixture of 1-hexadecyl-3-methylimidazolium bromide (C16MIMBr) and sodium salicylate (NaSal) and attributed this to the crystallization of wormlike micelles. And, more importantly, to achieve the required balance of solubilization and crystallization for hydrogel formation, the interactions between the aggregates could be modulated and optimized by carefully adjusting environmental factors (e.g., concentration, composition, temperature, pH, salt, etc.) and the surfactant molecular structures (e.g., the nature of the headgroup, the length and architecture of the hydrophobic tail, and the spacer length for Gemini and Bola surfactants).
Zwitterionic surfactants are interesting molecules, owing to their low toxicity, high foam stability and resistance to hard water.19,20,36–47 In previous works, Kumar et al.19 found that elastic hydrogel was formed by the zwitterionic surfactant erucyl dimethyl amidopropyl betaine (EDAB, with an unsaturated C22 tail) and Chu et al.20 also found that hydrogel was formed by concentrated 3-(N-erucamido-propyl-N,N-dimethyl ammonium) propane-sulfonate (EDAS, with an unsaturated C22 tail) in the presence of NaCl. Long-chain zwitterionic surfactants are difficult to dissolve in water; although the unsaturated carbon–carbon double bond and amido group in the hydrophobic tail of EDAB and EDAS can enhance the solubility, they can also cause the surfactant to lose gelation activity easily by oxidation and hydrolysis.36 For short-chain zwitterionic surfactants, it has been reported by Lopez-Diaz et al., Qiao et al. and Fan et al. that a viscoelastic solution with wormlike micelles formed in the aqueous mixtures of N-dodecyl-N,N-dimethyl-3-ammonio-1-propane sulfonate (DDPS, with a saturated C12 tail) or tetradecyl dimethyl-ammonium propane sulfonate (TDPS, with a saturated C14 tail) and SDS.39–41 Comparing these reported phase behavior and microstructure results of short-chain and long-chain zwitterionic surfactant solutions, it can be seen that the microstructures are all wormlike micelles, but the wormlike micelles formed by long-chain zwitterionic surfactants are longer, which led to the surprising result that the solutions behave like an elastic gel with an infinite relaxation time and can support their own weight. Herein, the phase behavior in the mixed system of the zwitterionic surfactant N-hexadecyl-N,N-dimethyl-3-ammonio-1-propane sulfonate (HDPS, with a saturated C16 tail) and anionic surfactant sodium dodecylsulfate (SDS) has been studied. This selection involves two important considerations, namely (i) using a medium length saturated chain zwitterionic surfactant (HDPS) to avoid insolubility and instability, and (ii) adding an anionic surfactant to promote the formation of long wormlike micelles. It was found that hydrogel could be formed in HDPS/SDS systems by changing the total concentration (CT) and molar percentage of SDS (XSDS). The microstructures in the hydrogel were a combination of long wormlike micelles and small vesicles, which is why hydrogel could be found in HDPS/SDS surfactant systems but not in TDPS/SDS and DDPS/SDS surfactant systems. Moreover, the effects of temperature and salt on the sol–gel phase transition were investigated to understand the general rules for hydrogel formation in mixed zwitterionic/anionic surfactant systems.
Experimental
Materials
N-Hexadecyl-N,N-dimethyl-3-ammonio-1-propane sulfonate (HDPS, J&K Chemical, ≥99%), N-tetradecyl-N,N-dimethyl-3-ammonio-1-propane sulfonate (TDPS, J&K Chemical, ≥99%), N-dodecyl-N,N-dimethyl-3-ammonio-1-propane sulfonate (DDPS, Fluka, ≥97%), sodium dodecylsulfate (SDS, Alfa Aesar, >99%) and NaCl (Beijing Chemical Co., A.R. grade) were used as received. The water used was bi-distilled from potassium permanganate containing deionized water to remove traces of organic compounds.Sample preparation
Samples were prepared by mixing the individual surfactant aqueous solution directly in a test tube at certain total concentrations and molar percentages of SDS. The molar percentage of SDS, XSDS, is defined as XSDS = [SDS]/CT, where CT (mmol L−1) is the total concentration of surfactants in the system. Then the desired amount of inorganic salt was added to the tube. After sealing, these samples were vortex-mixed and equilibrated at high temperature (∼70 °C) for 1 h to ensure complete solubility and uniformity. The resulting mixture was maintained in a 25 °C thermostatic bath for at least 72 h before measurements were taken. All measurements were performed at 25 °C unless otherwise specified.Rheology measurements
The rheological properties of the samples were measured with a conventional rheometer (Physica MCR301, Anton Paar, Graz). A cone-plate sensor with a plate diameter of 49.959 mm, a cone angle of 1° and a default gap of 0.047 mm was used. A chamber that covered the sample was used to avoid evaporation. Frequency sweep measurements were carried out from 0.05 to 100 rad s−1 in the linear viscoelastic region determined via dynamic strain sweep measurements.Transmission electron microscopy (TEM)
Micrographs were obtained with a JEM-100CX II transmission electron microscope via the negative-staining method using uranyl acetate.Transmittance measurements
The transmittance measurements were performed by using a Turbiscan MA2000 (Formulaction, France) with a pulsed near-infrared light source (λ = 880 nm) to measure the average transmittance.48 The gel in a test tube was placed in the instrument and the light transmitted from the sample was then measured periodically along their heights at different temperatures. The results are presented as the transmittance changes as a function of sample height.Results and discussion
Phase behavior and hydrogel formation
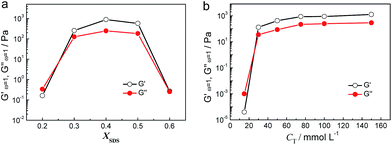
The phase behavior of aqueous mixtures of HDPS and SDS at a total concentration of 100 mmol L−1 (CT) was investigated for six different molar percentages of SDS (XSDS), as shown in Fig. 1. It was found that samples at XSDS = 0.1 and XSDS = 0.2 were transparent viscoelastic isotropic solutions, which are the typical features of samples containing wormlike micelles and similar to previously reported results in TDPS/SDS surfactant systems.39–41 At XSDS = 0.6, the sample is an opalescent viscoelastic solution with birefringence viewed under a crossed polarizer, characterizing the existence of a lamellar phase.49–51 These samples all flowed to the bottom in the tube inversion test. However, when XSDS ranged from 0.3 to 0.5, the samples were opalescent hydrogels that could support their own weight in inverted tubes. It is worth noting that we also studied the phase behavior of TDPS/SDS and DDPS/SDS surfactant systems under the same conditions or in more concentrated solutions, and hydrogel could only be observed in the HDPS/SDS surfactant system.The formation of hydrogel is further proved by dynamic rheological measurements, and the HDPS/SDS mixture at XSDS = 0.4 was chosen as an illustration of this (Fig. 2). It can be seen that the viscoelastic behavior, defined by the elastic modulus (G′) and the viscous modulus (G′′), changes slightly with an increase in the frequency (ω) and the complex viscosity (|η*|) varies as |η*| ∼ ω−1. The value of G′ (∼800 Pa) was also always larger than that of G′′ (∼200 Pa) over the entire investigated frequency range. From a rheological standpoint, these above results indicate that the sample exhibited elastic gel rheological behavior with infinite relaxation time.52 The effect of the molar percentage of SDS, XSDS, and total concentration on the G′ and G′′ of the HDPS/SDS mixed surfactant solutions was also studied. As is shown in Fig. 3a, the value of G′ surpassed that of G′′ and values over 100 Pa were observed for both at XSDS = 0.3–0.5 and a total concentration of 100 mmol L−1, which demonstrated that the hydrogel was formed and was in agreement with the macroscopic appearance results. Fig. 3b shows the values of G′ and G′′ as a function of the total concentration, and it has been found that the hydrogel can be obtained at concentrations higher than 30 mmol L−1 for XSDS = 0.4.
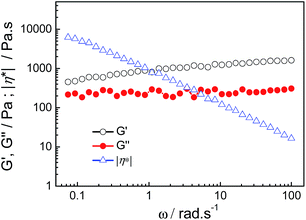 | ||
| Fig. 2 Frequency dependence of the elastic modulus (G′), viscous modulus (G′′) and the complex viscosity (|η*|) of the HDPS/SDS hydrogel (CT = 100 mmol L−1, XSDS = 0.4). | ||
Transmission electron microscopy (TEM) was employed to study the microstructures in the HDPS/SDS hydrogel. As is shown in Fig. 4, a large number of long wormlike micelles and small spherical vesicles were observed, which can provide a deeper understanding for the above phase behavior results. Firstly, the mechanisms for the formation of the aggregate microstructures and their transformation can be described by the well-known theory of the packing parameter p, proposed by Israelachvili et al.,53,54 which has been widely and successfully used to explain aggregate microstructures in surfactant solutions; 1/3 ≤ p < 1/2 corresponds to cylindrical micelles, 1/2 ≤ p < 1 corresponds to bilayer structures and p = 1 corresponds to planar extended bilayers. The synergistic interactions between zwitterionic and anionic surfactants reduces the repulsion force between surfactant headgroups and then increases the aggregate packing parameter p. Hence, the wormlike micelles in the HDPS/SDS surfactant system grow in contour length and the aggregate microstructures undergo transitions from wormlike micelles to the coexistence of wormlike micelle/vesicle and then to lamellar structures upon increasing the XSDS, as can be observed in Fig. 1. Secondly, the coexistence of wormlike micelles and small vesicles brings an appropriate packing parameter p, which indicates that the wormlike micelles reached a sufficient length and degree of entanglement to form the three-dimensional elastic hydrogel. Thirdly, the packing parameter p also increases with an increase in the hydrophobic tail length of the zwitterionic surfactants. Thus, the wormlike micelles formed in HDPS/SDS mixtures are longer than those in TDPS/SDS and DDPS/SDS mixtures under the same conditions. That is why hydrogel can be found in HDPS/SDS surfactant systems but not in TDPS/SDS and DDPS/SDS surfactant systems.
Thermo-reversible sol–gel phase transition
The hydrogel formed in the HDPS/SDS system underwent a gel–sol transition similar to that of other surfactant-based hydrogels and the microstructures changed from entangled long wormlike micelles to short rodlike micelles upon increasing the temperature.17,18 This phase transition process was first studied using an optical analyzer, the Turbiscan MA2000. The hydrogel was equilibrated at different temperatures for 2 h and the transmittance changes were measured as a function of sample height for the HDPS/SDS hydrogel at CT = 100 mmol L−1 and XSDS = 0.3. It can be seen from Fig. 5a that, at temperatures below 28 °C, the sample was an opalescent hydrogel and the transmittance approached zero. As the temperature was increased to 29 °C, the sample became clearer and more light was transmitted, and it also exhibited the characteristic hydrogel behavior of supporting its own weight in the inverted tubes. When the temperature was higher than 30 °C, the solution was a transparent fluid and about 90% incident light was transmitted. It can be inferred that the system was in a sol state in this regime. The transmittances at 20 mm sample height are presented as a function of temperature in a stepwise heating–cooling cycle (Fig. 5b). It is clearly seen that the transmittance at each temperature can be reproduced simply by heating or cooling, which indicates that the sol–gel phase transition is thermo-reversible. Herein, the gel–sol transition temperature (Tg–s) has been established to be 30 °C for the HDPS/SDS hydrogel at CT = 100 mmol L−1 and XSDS = 0.3 from the viewpoint of the optical method. | ||
| Fig. 5 The transmittance for the HDPS/SDS hydrogel (CT = 100 mmol L−1, XSDS = 0.3) at different sample heights (a) and temperatures (b). | ||
The gel–sol phase transition of the above sample was further studied in an oscillatory rheological experiment. The elastic modulus G′ was continuously measured at 6.28 rad s−1 while heating at each temperature for 400 s and then increasing to the next temperature at a rate of 0.01 °C s−1. This procedure aimed to give the sample enough time to fully equilibrate at each temperature value and reduce the degree of water evaporation by shortening the measurement time. Then, the measured G′ was normalized, as plotted in Fig. 6. For the HDPS/SDS hydrogel at CT = 100 mmol L−1 and XSDS = 0.3, the normalized elastic modulus (G′N) decreased slightly but was still larger than 0.5 below 29 °C, which implies that the gel structure was maintained. At 30 °C, G′N dropped dramatically, by over three orders of magnitude, suggesting that the gel underwent the transition to sol. All of these results are in good agreement with the gel–sol transition monitored using the optical method. As reported by many researchers, the gel–sol transition temperature (Tg–s) can be determined from the sharp decrease of the G′N curve in the rheological measurements.55,56 The obtained Tg–s values, at XSDS = 0.3, 0.4 and 0.5, were 30 °C, 31 °C and 29 °C for 100 mmol L−1 HDPS/SDS hydrogel, respectively.
Salt-induced sol–gel phase transition
The hydrogel formed in the HDPS/SDS surfactant system was produced as a result of the existence of wormlike micelles of sufficient contour length. Therefore, the controlling factors for wormlike micelle growth are expected to affect the HDPS/SDS solution properties. It is well known that salt is such an external stimulus.57–59 Therefore, the effect of NaCl on the rheological properties of the HDPS/SDS hydrogel at CT = 50 mmol L−1 and XSDS = 0.4 was studied. As Fig. 7 shows, the elastic modulus (G′) and viscous modulus (G′′) increase with the NaCl concentration. It was suggested that the hydrogel strength was gradually enhanced with the increase of the NaCl concentration. Further investigations showed that salt can also induce the sol to gel phase transition. Fig. 8 shows the elastic modulus (G′) and viscous modulus (G′′) as a function of the angular frequency (ω) for the HDPS/SDS mixed surfactant solution (CT = 15 mmol L−1, XSDS = 0.4) without and with 100 mmol L−1 NaCl. It can be seen that the rheological responses of the HDPS/SDS mixed solution (CT = 15 mmol L−1, XSDS = 0.4) reflected its viscous nature, with G′′ being much higher than G′ over the entire measured frequency range. Moreover, G′ was smaller than 0.0001 Pa. However, after the addition of 100 mmol L−1 NaCl, the sample was predominantly elastic, with the value of G′ exceeding 100 Pa, and being larger than that of G′′ over the entire frequency range. These results imply that the HDPS/SDS mixed solution at CT = 15 mmol L−1 and XSDS = 0.4 was in a sol state but changed to a gel state with the addition of NaCl. | ||
| Fig. 8 Frequency dependence of the elastic modulus (G′) and viscous modulus (G′′) of the HDAPS/SDS mixed solution (CT = 15 mmol L−1, XSDS = 0.4) without and with 100 mmol L−1 NaCl. | ||
For the HDPS/SDS mixed solution, the aggregate surface is negatively charged. In the presence of NaCl, the binding Na+ can continually reduce the repulsion force between surfactant headgroups and increase the critical packing parameter p of the HDPS/SDS system. As a result, adding salt causes the wormlike micelles to lengthen and the rheological properties of the solution to change, such that it may even induce a sol–gel phase transition in the mixed zwitterionic and anionic surfactant system.
As is mentioned above, only viscoelastic wormlike micelles can be detected in the TDPS/SDS mixed surfactant system by adjusting the surfactant concentration and molar ratio at temperatures ranging from 25 °C to 5 °C. And it was found that, upon adding NaCl into the TDPS/SDS mixed solution, the system has the ability to form gel upon cooling, e.g., the gel–sol transition temperature (Tg–s) is about 17 °C for the TDPS/SDS system at CT = 100 mmol L−1 and XSDS = 0.4 in the presence of 100 mmol L−1 NaCl. Fig. 9 shows the effect of NaCl on the rheological responses of the TDPS/SDS system at 15 °C. The dynamic stress sweep experiment of the TDPS/SDS solution (CT = 100 mmol L−1, XSDS = 0.4) only showed a viscous modulus (Fig. 9a). Upon increasing the concentration of NaCl, an elastic modulus was revealed and both moduli increased. When the NaCl concentration was more than 100 mmol L−1, the value of the elastic modulus (G′) surpassed that of the viscous modulus (G′′) and G′ was larger than 100 Pa (Fig. 9b). Moreover, the changes of G′ and G′′ with frequency were very similar to those of the HDPS/SDS hydrogel for the TDPS/SDS solution with 100 mmol L−1 NaCl. All of these results also confirm the phenomenon of the salt-induced sol–gel transition for mixed zwitterionic and anionic surfactant systems.
Conclusions
In the present study, the phase behavior and microstructures were studied in a mixed system of the zwitterionic surfactant N-hexadecyl-N,N-dimethyl-3-ammonio-1-propane sulfonate (HDPS, with a saturated C16 tail) and anionic surfactant sodium dodecylsulfate (SDS). Wormlike micelles and small vesicles were demonstrated to coexist in the HDPS/SDS system, and the wormlike micelles could become elongated and entangled with one another to form a three-dimensional elastic hydrogel. Using a medium length saturated chain zwitterionic surfactant (HDPS) as the hydrogelator avoids the issues of insolubility in water and instability arising from the oxidation and hydrolysis of long chain zwitterionic surfactants containing an unsaturated carbon–carbon double bond and amido group, as noted in the previous literature.19,20,36 By varying the temperature and adjusting the salt concentration, the wormlike micelle growth can be controlled and the formation or destruction of the hydrogel can be induced. Thus the mixed HDPS/SDS surfactant solution can be easily switched between gel and sol by the dual stimulus–response processes. These results provide a template for the synthesis of controllable nanomaterials and controlled release carriers for drug release and delivery. It is expected that this study may provide a better understanding of the general rules for surfactant-based hydrogel formation and advance hydrogel applications in related fields.Acknowledgements
This work is supported by Open Fund of State Key Laboratory of Oil and Gas Reservoir Geology and Exploitation (PLN1302), National Basic Research Program of China (973 Program, 2013CB933800), the NSFC program of China (51104169, 21273286, 21273003, 21473005), Fok Ying Tung Education Foundation (141047) and the Program for Changjiang Scholars and Innovative Research Team in University (IRT1294).Notes and references
- H. J. van der Linden, S. Herber, W. Olthuisa and P. Bergveld, Analyst, 2003, 128, 325–331 RSC.
- Z. Q. Tou, T. W. Koh and C. C. Chan, Sens. Actuators, B, 2014, 202, 185–193 CrossRef CAS PubMed.
- A. L. Butcher, G. S. Offeddu and M. L. Oyen, Trends Biotechnol., 2014, 32, 564–570 CrossRef CAS PubMed.
- C. C. Huang, S. Ravindran, Z. Y. Yin and A. George, Biomaterials, 2014, 35, 5316–5326 CrossRef CAS PubMed.
- A. Döring, W. Birnbauma and D. Kuckling, Chem. Soc. Rev., 2013, 42, 7391–7420 RSC.
- D. Das, T. Kara and P. K. Das, Soft Matter, 2012, 8, 2348–2365 RSC.
- Y. Qiao, Y. Y. Lin, S. F. Zhang and J. B. Huang, Chem.–Eur. J., 2011, 17, 5180–5187 CrossRef CAS PubMed.
- Y. Y. Lin, Y. Qiao, C. Gao, P. F. Tang, Y. Liu, Z. B. Li, Y. Yan and J. B. Huang, Chem. Mater., 2010, 22, 6711–6717 CrossRef CAS.
- J. C. Tiller, Angew. Chem., Int. Ed., 2003, 42, 3072–3075 CrossRef CAS PubMed.
- A. R. Fajardo, M. B. Silva, L. C. Lopes, J. F. Piai, A. F. Rubira and E. C. Muniz, RSC Adv., 2002, 2, 11095–11103 RSC.
- Polymer Gels and Networks, ed. Y. Osada and A. R. Khokhlov, Marcel Dekker, New York, USA, 2002 Search PubMed.
- Y. G. Kim, C. H. Lee and Y. C. Bae, Fluid Phase Equilib., 2014, 361, 200–207 CrossRef CAS PubMed.
- Molecular Gels, ed. P. Terech and R. G. Wiess, Kluwer Academic Publishers, The Netherlands, 2004 Search PubMed.
- L. A. Estroff and A. D. Hamilton, Chem. Rev., 2004, 104, 1201–1217 CrossRef CAS PubMed.
- M. Kodamaa and S. Sekib, Adv. Colloid Interface Sci., 1991, 35, 1–30 CrossRef.
- H. Hoffmann and W. Ulbricht, Curr. Opin. Colloid Interface Sci., 1996, 1, 726–739 CrossRef CAS.
- K. Trickett and J. Eastoe, Adv. Colloid Interface Sci., 2008, 144, 66–74 CrossRef CAS PubMed.
- S. R. Raghavan and J. F. Douglas, Soft Matter, 2012, 8, 8539–8546 RSC.
- R. Kumar, G. C. Kalur, L. Ziserman, D. Danino and S. R. Raghavan, Langmuir, 2007, 23, 12849–12856 CrossRef CAS PubMed.
- Z. L. Chu and Y. J. Feng, Chem. Commun., 2011, 47, 7191–7193 RSC.
- L. Zhao, K. Wang, L. M. Xu, Y. Liu, S. Zhang, Z. B. Li, Y. Yan and J. B. Huang, Soft Matter, 2012, 8, 9079–9085 RSC.
- Y. Y. Lin, X. Han, J. B. Huang, H. L. Fu and C. L. Yu, J. Colloid Interface Sci., 2009, 330, 449–455 CrossRef CAS PubMed.
- H. S. Lu, L. Wang and Z. Y. Huang, RSC Adv., 2014, 4, 51519–51527 RSC.
- H. Hoffmann, C. Thunig, P. Schmiedel and U. Munkert, Faraday Discuss., 1995, 101, 319–333 RSC.
- M. Gradzielski, M. Bergmeier, H. Hoffmann, M. Muller and I. Grillo, J. Phys. Chem. B, 2000, 104, 11594–11597 CrossRef CAS.
- M. Yang, J. C. Hao and H. G. Li, RSC Adv., 2014, 4, 40595–40605 RSC.
- N. Duerr-Auster, J. Kohlbrecher, T. Zuercher, R. Gunde, P. Fischer and E. Windhab, Langmuir, 2007, 23, 12827–12834 CrossRef CAS PubMed.
- Y. Kawabata, A. Matsuno, T. Shinoda and T. Kato, J. Phys. Chem. B, 2009, 113, 5686–5689 CrossRef CAS PubMed.
- P. F. Long and J. C. Hao, Soft Matter, 2010, 6, 4350–4356 RSC.
- I. Nakazawa, M. Masuda, Y. Okada, T. Hanada, K. Yase, M. Asai and T. Shimizu, Langmuir, 1999, 15, 4757–4764 CrossRef CAS.
- T. Imae, N. Hayashi, T. Matsumoto, T. Tada and M. Furusaka, J. Colloid Interface Sci., 2000, 225, 285–290 CrossRef CAS PubMed.
- D. Berthier, T. Buffeteau, J. M. Leger, R. Oda and I. Huc, J. Am. Chem. Soc., 2002, 124, 13486–13494 CrossRef CAS PubMed.
- Y. T. Wang, X. Xin, W. Z. Li, C. Y. Jia, L. Wang, J. L. Shen and G. Y. Xu, J. Colloid Interface Sci., 2014, 431, 82–89 CrossRef CAS PubMed.
- Y. I. González and E. W. Kaler, Langmuir, 2005, 21, 7191–7199 CrossRef PubMed.
- Y. Y. Lin, Y. Qiao, Y. Yan and J. B. Huang, Soft Matter, 2009, 5, 3047–3053 RSC.
- Z. L. Chu, Y. J. Feng, H. Q. Sun, Z. Q. Li, X. W. Song, Y. G. Han and H. Y. Wang, Soft Matter, 2011, 7, 4485–4489 RSC.
- L. X. Jiang, Y. Yan and J. B. Huang, Soft Matter, 2011, 7, 10417–10423 RSC.
- K. Golemanov, N. D. Denkov, S. Tcholakova, M. Vethamuthu and A. Lips, Langmuir, 2008, 24, 9956–9961 CrossRef CAS PubMed.
- D. Lopez-Diaz, E. Sarmiento-Gomez, C. Garza and R. Castillo, J. Colloid Interface Sci., 2010, 348, 152–158 CrossRef CAS PubMed.
- Y. Qiao, Y. Y. Lin, Y. J. Wang, Z. B. Li and J. B. Huang, Langmuir, 2011, 27, 1718–1723 CrossRef CAS PubMed.
- H. M. Fan, X. Y. Wu, S. R. Liu, Y. J. Wang, Y. Li and W. L. Kang, Acta Chim. Sin., 2011, 69, 1997–2002 CAS.
- S. K. Mehta, K. Kaur, Rishu and K. K. Bhasin, J. Colloid Interface Sci., 2009, 336, 322–328 CrossRef CAS PubMed.
- L. Brinchi, R. Germani, P. Di Profio, L. Marte, G. Savelli, R. Oda and D. Berti, J. Colloid Interface Sci., 2010, 346, 100–106 CrossRef CAS PubMed.
- X. Z. Wang, W. L. Kang, X. C. Meng, H. M. Fan, H. Xu, J. B. Fu and Y. N. Zhang, Acta Phys.-Chim. Sin., 2012, 28, 2285–2290 CAS.
- J. H. Zhao, C. L. Dai, Q. F. Ding, M. Y. Du, H. S. Feng, Z. Y. Wei, A. Chen and M. W. Zhao, RSC Adv., 2015, 5, 13993–14001 RSC.
- M. Tiecco, G. Cardinali, L. Roscini, R. Germani and L. Corte, Colloids Surf., B, 2013, 111, 407–417 CrossRef CAS PubMed.
- D. Saul, G. J. T. Tiddy, B. A. Wheeler, P. A. Wheeler and E. Willis, J. Chem. Soc., Faraday Trans. 1, 1974, 70, 163–170 RSC.
- O. Mengual, G. Meunier, I. Cayré, K. Puech and P. Snabre, Talanta, 1999, 50, 445–456 CrossRef CAS.
- Y. Yan, H. Hoffmann, Y. Talmon, A. Makarsky and W. Richter, J. Phys. Chem. B, 2007, 111, 6374–6382 CrossRef CAS PubMed.
- Y. W. Shen, H. Hoffmann and J. C. Hao, Langmuir, 2009, 25, 10540–10547 CrossRef CAS PubMed.
- H. M. Fan, B. C. Li, Y. Yan, J. B. Huang and W. L. Kang, Soft Matter, 2014, 10, 4506–4512 RSC.
- R. G. Larson, The Structure and Rheology of Complex Fluids, Oxford University Press, New York, USA, 1999 Search PubMed.
- J. N. Israelachvili, D. J. Mitchell and B. W. Ninham, J. Chem. Soc., Faraday Trans. 1, 1976, 272, 1525–1567 Search PubMed.
- R. A. Khalil and A. A. Zarari, Appl. Surf. Sci., 2014, 318, 85–89 CrossRef CAS PubMed.
- L. Li, H. Shan, Y. Yue, Y. C. Lam, K. C. Tam and X. Hu, Langmuir, 2002, 18, 7291–7298 CrossRef CAS.
- M. Tomšič, F. Prossnigg and O. Glatter, J. Colloid Interface Sci., 2008, 322, 41–50 CrossRef PubMed.
- P. A. Hassan, S. R. Raghavan and E. W. Kaler, Langmuir, 2002, 18, 2543–2548 CrossRef CAS.
- H. M. Fan, Y. Yan, Z. C. Li, Y. Xu, L. X. Jiang, L. M. Xu, B. Zhang and J. B. Huang, J. Colloid Interface Sci., 2010, 348, 491–497 CrossRef CAS PubMed.
- P. Koshy, V. K. Aswal, M. Venkatesh and P. A. Hassan, Soft Matter, 2011, 7, 4778–4786 RSC.
| This journal is © The Royal Society of Chemistry 2015 |