C–H reductive elimination during the reaction of cycloplatinated(II) complexes with pyridine-2-thione: kinetic follow up†
Marzieh Dadkhah
Aseman
a,
S. Masoud
Nabavizadeh
*a,
Hamid R.
Shahsavari
b and
Mehdi
Rashidi
*a
aDepartment of Chemistry, College of Sciences, Shiraz University, Shiraz 71467-13565, Iran. E-mail: nabavi@chem.susc.ac.ir; rashidi@chem.susc.ac.ir; Fax: +98 713 646 0788; Tel: +98 713 613 7110
bDepartment of Chemistry, Institute for Advanced Studies in Basic Sciences, Gava Zang, 45195-1159, Zanjan, Iran
First published on 23rd February 2015
Abstract
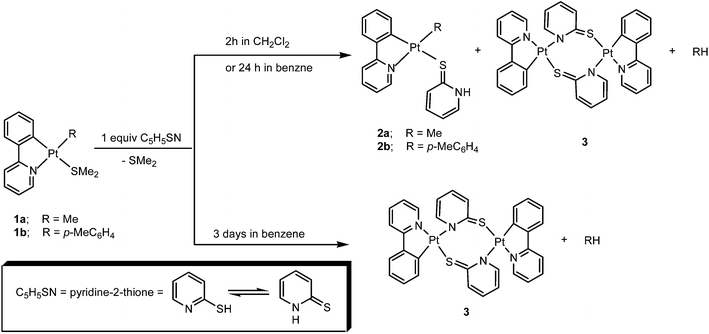
Substitution reactions of the labile SMe2 ligand in the cycloplatinated(II) complexes [PtR(ppy)(SMe2)], 1, in which ppy = 2-phenylpyridinate and R = Me, 1a, or p-MeC6H4, 1b, by pyridine-2-thione, C5H5SN, were studied. When each of the complexes 1 was treated with 1 equiv. C5H5SN, existing as a mixture of thiol (N⁁SH) and thione (HN⁁S) tautomers, a mixture containing the S-bound thiol complex [PtR(ppy)(η1-S-S⁁NH)] (R = Me, 2a, or R = p-MeC6H4, 2b) and the dimeric complex [Pt(ppy)(N⁁S)]2, 3 (having two bridging deprotonated pyridine-2-thione (N⁁S) ligands), was observed along with free R–H. This mixture finally led to pure complex 3 after 3 days. Pure samples of the complexes 2a and 3 were obtained from the abovementioned 2a+3 mixture by using flash chromatography on silica gel. Kinetics of the reactions were investigated by UV-vis spectroscopy (complexes 1 have a MLCT band in the visible region which was used to easily follow the reactions) and 1H NMR spectroscopy. On the basis of the results, a mechanism was proposed for the related reactions.
1. Introduction
The chemistry of cyclometalated complexes has long been the subject of intense study.1–4 Among the cyclometalated complexes, cycloplatinated complexes have attracted a great deal of attention as a result of their highly versatile obvious applications in many fields.5–8 Monoalkyl cycloplatinated complexes with a solvent or labile ligand have been reported only rarely.9–16 These complexes reportedly exhibit various reactivities toward fundamental reactions such as oxidative addition and substitution reactions.17–22The rich reactive features of complexes with heterodonor ligands, such as 2-diphenylphosphinopyridine (pyPPh2)21,23–26 or pyridine-2-thione, C5H5SN,27–38 have been widely studied for application in different aspects. The latter is an unsymmetrical bidentate ligand with a nitrogen donor atom and a sulfur donor atom. Its pKa values30 suggest that the parent molecule exists in solution as a mixture containing the thione–thiol tautomers (Ia and Ib in Fig. 1, respectively). This ligand can be deprotonated27,31 easily to obtain the thiolate form N⁁S (C5H4SN), indicated as Ic in Fig. 1.37 Although the thione–thiol tautomeric equilibrium has been shown generally to favor the thione form, the electronic differentiation associated with the hard nitrogen and soft sulfur donors in this reagent directs its reactivity and coordination behavior when it binds to metal (Fig. 2).37,39 The reported monomeric platinum(II) complexes containing pyridine-2-thione have been shown to be S-bound with pendant pyridyl group,40 such as trans-[Pt(η1-S-C5H4NS)2(PPh3)2] or N⁁S chelating, such as [Pt(η2-N,S-C5H4NS)(PPh3)2]PF6,41 with no related example of the N-bound thiol complex.
In the present study, we describe reaction of the cycloplatinated(II) complexes [PtR(ppy)(SMe2)], 1, (R = Me; 1a, R = p-MeC6H4, 1b, and ppy = deprotonated 2-phenylpyridine) with pyridine-2-thione (C5H5SN) showing that the monodentate S-bound complexes [PtR(ppy)(η1-S-S⁁NH)], 2, with NH pendant group, are formed along with a dimmeric complex [Pt(ppy)(N⁁S)]2, 3, resulting from C–H bond reductive elimination. We have also followed up the kinetics of these reactions by using UV-vis spectroscopy and 1H NMR spectroscopy in different solvents and suggested a mechanism for the related reactions.
2. Experimental section
The 1H NMR spectra of the complexes were recorded on a Bruker Avance DPX 250 MHz or 400 MHz spectrometer, and TMS (0.00) was used as an external reference. All the chemical shifts and coupling constants are given in units of ppm and Hz, respectively. Kinetic studies were carried out by using a Perkin-Elmer Lambda 25 spectrophotometer with temperature control using an EYELA NCB-3100 constant-temperature bath. Pyridine-2-thione (C5H5SN) was purchased from commercial sources and the starting complexes, [PtMe(ppy)(SMe2)], 1a,10 [Pt(p-MeC6H4)(ppy)(SMe2)], 1b,10 and [Pt(ppy)Cl(DMSO)],42 in which DMSO is dimethyl sulfoxide, were prepared as reported. The NMR labeling for ppy and C5H5SN ligands are shown in Scheme 1.43,442.1. Reaction of [PtMe(ppy)(SMe2)], 1a, with C5H5SN
To a solution of [PtMe(ppy)(SMe2)], 1a, (100 mg, 0.23 mmol) in dichloromethane (or benzene) was added 1 equiv. pyridine-2-thione (26.1 mg, 0.23 mmol) at room temperature. The mixture was stirred for 2 h (or 24 h in benzene) and the solvent was then removed under reduced pressure. The residue was triturated with n-hexane to give a pale red solid, which was separated and dried under vacuum. The product was identified as a mixture of [PtMe(ppy)(η1-S-S⁁NH)], 2a, and [Pt(ppy)(N⁁S)]2, 3, with the ratio 2a/3 being 1/2. Total yield: 76 mg, 69%. Mixture of the compounds 2a and 3 were separated by flash chromatography with good yields on silica gel eluted with 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 CH2Cl2/MeOH, to afford pure [PtMe(ppy)(η1-S-S⁁NH)], 2a, and [Pt(ppy)(N⁁S)]2, 3, identified as follows:
2 CH2Cl2/MeOH, to afford pure [PtMe(ppy)(η1-S-S⁁NH)], 2a, and [Pt(ppy)(N⁁S)]2, 3, identified as follows:
The complex 3 has been reported to be synthesized by the reaction of (Bu4N)[PtCl2(ppy)] with C5H5SN and was characterized in solid state by single crystal X-ray crystallography.45 In the present work, [Pt(ppy)Cl(DMSO)] (100 mg, 0.22 mmol) was added to an ethanolic solution of sodium pyridine-2-thiolate, NaC5H4NS (see below), under inert atmosphere condition. 1H NMR data in CDCl3, δ 8.09 (d, 3JPtH6 = 19.1 Hz, 3JH6H5 = 5.6 Hz, CH group adjacent to coordinated N atom, 2H6ppy), 7.47 (d, 3JPtH3′ = 32.8 Hz, 3JH3′H4′ = 5.7 Hz, CH group adjacent to coordinated C atom, 2H3′ppy), 7.53 (t, 2H), 7.49 (d, 2H), 7.86 (d, 3JPtH6 = 16.2 Hz, 3JH3H4 = 7.5 Hz, 2H6N⁁S), 7.13 (t, 4H), 6.91 (t, 4H), 6.74 (t, 2H), 6.71 (t, 2H), 6.43 (t, 2H). Other notes related to complex 3:
(1) Single crystal of dimer 3 was grown in a concentrated benzene solution of the 2a + 3 mixture product obtained from mixing of complex 1a and C5H5SN by slow diffusion of diethylether and its structure was confirmed by single crystal X-ray analysis (see Fig. 3).
(2) NaC5H4NS (as used in direct synthesis of complex 3, see above) was prepared by dissolving of sodium (6 mg 0.25 mmol) in 10 mL of absolute ethanol following by treatment with pyridine-2-thione (25 mg, 0.22 mmol) to immediately give a red solution that after stirring for 6 h at room temperature a red solid was precipitated which was separated and dried under vacuum. Yield: 73 mg, 74%. This compound gave 1H NMR data similar to the reported data in DMSO-d6.45
(3) The complex 3 is not stable in CDCl3 (or CH2Cl2) and slowly forms [Pt(ppy)(N⁁S)(Cl)]2 complex,45 giving 1H NMR data in CDCl3: δ 9.54 (d, 3JPtH6 = 20.8 Hz, 3JH6H5 = 5.6 Hz, CH group adjacent to coordinated N atom, 2H6ppy), 8.15 (d, 3JPtH3′ = 28.2 Hz, 3JH3′H4′ = 6.2 Hz, CH group adjacent to coordinated C atom, 2H3′ppy), 7.56 (t, 2H), 7.37 (t, 2H), 7.29 (d, 2H), 7.17 (t, 4H), 6.97 (t, 4H), 6.90 (t, 2H), 6.71 (t, 2H), 6.77 (t, 2H). These 1H NMR data are similar to those reported for the complex [Pt(ppy)(N⁁S)(Cl)]2 in DMSO-d6.45
(4) When the reaction of [PtMe(ppy)(SMe2)], 1a, with pyridine-2-thione in benzene was stirred for 3 days, only dimer 3 as a pure product was formed.
(5) 1H NMR data for the complex 3 in C6D6: ppy ligand, δ 7.49 (m, 3JH3′H4′ = 5.3 Hz, 4JH3′H5′ = 1.0 Hz, 3JPtH3′ = 34.2 Hz, CH group adjacent to coordinated C atom, 1H3′ppy), 8.09 (m, 3JH6H5 = 6.0 Hz, 4JH6H4 = 1.0 Hz, 3JPtH6 = 20.4 Hz, CH group adjacent to coordinated N atom, 1H6ppy); 7.84 (d, 3JH5H6 = 7.8 Hz, br Pt sat, CH group adjacent to coordinated N atom, 1H6N⁁S).
2.2. Monitoring reaction of 1a with C5H5SN by 1H NMR spectroscopy
To a small sample (10 mg, 0.023 mmol) of 1a dissolved in C6D6 (0.75 mL) in a sealed NMR tube, 1 equiv. pyridine-2-thione (2.6 mg, 0.023 mmol) was added. NMR spectra of the solution at 27 °C were recorded several times showing gradual conversion to a mixture containing the complexes 2a and 3 and free CH4.2.3. Reaction of [Pt(p-MeC6H4)(ppy)(SMe2)], 1b, with C5H5SN
To a solution of [Pt(p-MeC6H4)(ppy)(SMe2)], 1b, (100 mg, 0.19 mmol) in dichloromethane (15 mL) was added 1 equiv. pyridine-2-thione (22.1 mg, 0.19 mmol) at room temperature. The mixture was stirred for 5 h, the solvent was then removed under reduced pressure and the residue was triturated with n-hexane to give a pale red solid, which was separated and dried under vacuum. The product was identified as a mixture of [Pt(p-MeC6H4)(ppy)(η1-S-S⁁NH)], 2b, and [Pt(ppy)(N⁁S)]2, 3, with the ratio 2b/3 being 1/1. Total yield: 68 mg, 65%. Due to low solubility, the compounds formed in this reaction could not be separated and therefore elemental analysis was not useful. 1H NMR data in CDCl3, 2b: δ 2.29 (s, Me of p-tolyl, 3H), aromatic protons: p-tolyl ligand, 6.88 (d, 3JHmHo = 7.6 Hz, 2Hm), 7.17 (d, 3JHoHm = 7.6 Hz, 2Ho); ppy ligand, 6.41 (d, 3JH3′H4′ = 7.5 Hz, 3JPtH3′ = 24.6 Hz, CH group adjacent to coordinated C atom, 1H3′ppy), 8.60 (d, 3JH6H5 = 6.8 Hz, 3JPtH6 = 16.1, CH group adjacent to coordinated N atom, 1H6ppy); η1-S-S⁁NH ligand, 9.07 (d, 3JH3H4 = 5.6 Hz, 1H3). 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8.07 (d, 3JPtH6 = 19.2 Hz, 3JH6H5 = 5.6 Hz, CH group adjacent to coordinated N atom, 1H6ppy), 7.49 (d, 3JPtH3′ = 32.0 Hz, 3JH3′H4′ = 5.8 Hz, CH group adjacent to coordinated C atom 1H3′ppy), 7.89 (d, 3JPtH6 = 16.0 Hz, 3JH3H4 = 7.5 Hz, H6N⁁S), 6.50–7.60 (other aromatic protons of ppy and S⁁N groups).
8.07 (d, 3JPtH6 = 19.2 Hz, 3JH6H5 = 5.6 Hz, CH group adjacent to coordinated N atom, 1H6ppy), 7.49 (d, 3JPtH3′ = 32.0 Hz, 3JH3′H4′ = 5.8 Hz, CH group adjacent to coordinated C atom 1H3′ppy), 7.89 (d, 3JPtH6 = 16.0 Hz, 3JH3H4 = 7.5 Hz, H6N⁁S), 6.50–7.60 (other aromatic protons of ppy and S⁁N groups).
2.4. Kinetic study
In a typical experiment, a solution of complex 1a in dichloromethane (3 mL, 3 × 10−4 M) in a cuvette was thermostated at 25 °C and a 0.01 molar solution (90 µL) of pyridine-2-thione was added using a microsyringe. After rapid stirring, the absorbance at λ = 500 nm was monitored.2.5. Theoretical methods
Geometry optimizations were performed with the program suite Gaussian03 at the DFT/B3LYP level.46 The effective core potential of Hay and Wadt with a double-ξ valence basis set (LANL2DZ)47,48 was chosen to describe Pt and the 6-31G* basis set was used for other atoms. To evaluate and ensure the optimized structures of the molecules, frequency calculations were carried out using analytical second derivatives.3. Results and discussion
3.1. Synthesis and characterization of the complexes
As shown in Scheme 2, reaction of the Pt(II) starting complexes [PtR(ppy)(SMe2)], 1, in which R = Me (1a) or p-MeC6H4 (1b) and ppy = deprotonated 2-phenylpyridine, with pyridine-2-thione (C5H5SN) in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio, followed by the replacement of the labile ligand SMe2 to give a mixture containing the S-bound thione Pt(II) complex [PtR(ppy)(η1-S-S⁁NH)], 2, in which R = Me (2a) or p-MeC6H4 (2b), the dimeric complex [Pt(ppy)(N⁁S)]2, 3, with two bridging deprotonated pyridine-2-thione (N⁁S) ligands, and R–H, i.e. CH4 (in case where R = Me) or MeC6H5 (in case where R = p-MeC6H4). When reaction of 1 with C5H5SN is allowed to stir for 3 days in benzene, only 3 along with free R–H are formed.
1 molar ratio, followed by the replacement of the labile ligand SMe2 to give a mixture containing the S-bound thione Pt(II) complex [PtR(ppy)(η1-S-S⁁NH)], 2, in which R = Me (2a) or p-MeC6H4 (2b), the dimeric complex [Pt(ppy)(N⁁S)]2, 3, with two bridging deprotonated pyridine-2-thione (N⁁S) ligands, and R–H, i.e. CH4 (in case where R = Me) or MeC6H5 (in case where R = p-MeC6H4). When reaction of 1 with C5H5SN is allowed to stir for 3 days in benzene, only 3 along with free R–H are formed.
The products were characterized by 1H NMR and X-ray crystallography (for the complex 3) and full data are collected in the Experimental Section. In the 1H NMR spectrum of complex [PtMe(ppy)(η1-S-S⁁NH)], 2a, in C6D6 at room temperature, the methylplatinum resonance occurred at δ = 2.36 as a singlet which coupled to 195Pt with 2JPtH = 69.9 Hz (the value in the range expected for a methylplatinum(II) complex with methyl being trans to nitrogen49–52). The CH group adjacent to ligating C atom of the ppy ligand, i.e. H3′, appeared as a doublet of doublets at δ = 7.79 with 3JH3′H4′ = 7.4 Hz and 4JH3′H5′ = 1.1 Hz, and with 3JPtH3′ = 24.5 Hz, while the hydrogen related to the CH group adjacent to ligating N atom of the ppy ligand, i.e. H6, as expected was appeared further down field as a doublet of doublets at δ = 8.76 with 3JH6H5 = 5.7 Hz and 4JH6H4 = 1.8 Hz, and with 3JPtH6 = 16.2 Hz. The H3 proton of the η1-S-S⁁NH ligand appeared as a doublet at δ = 7.92 with 3JH3H4 = 5.5 Hz, with no observable coupling to Pt center probably due to the related pyridine plane being perpendicular to square geometry of the complex.18 Notice also that formation of CH4 is confirmed by the observation of a singlet at 0.16. The complex [Pt(ppy)(N⁁S)]2, 3, is also characterized with signals due to H3′ppy, H6N⁁S and H6ppy at 7.49 (3JPtH3′ = 34.2 Hz), 7.84 (br Pt sat) and 8.09 (3JPtH6 = 20.4 Hz) ppm, respectively.
In the 1H NMR spectrum of complex [Pt(p-MeC6H4)(ppy)(η1-S-S⁁NH)], 2b, in CDCl3 at room temperature, a singlet signal was observed at δ = 2.29 for the methyl group on the para-tolyl ligand. The meta and ortho protons of the para-tolyl ligand appeared as two doublets at δ = 6.88 and 7.17, respectively, each with 3JHmHo = 7.6 Hz. A doublet signal at δ = 6.41 with 3JH3′H4′ = 7.5 Hz, accompanied by platinum satellites (3JPtH3′ = 24.6 Hz), is attributed to hydrogen atom of C–H group adjacent to the coordinated C atom of ppy ligand, i.e. H3′. A doublet signal at δ = 8.60 (with 3JH6H5 = 6.8 Hz and 3JPtH6 = 16.1 Hz) is assigned to CH group adjacent to coordinated N atom of ppy ligand, i.e. H6. The H3 proton of the η1-S-S⁁NH group appeared as a doublet at δ = 9.07 with 3JH3H4 = 5.6 Hz. The dimer complex, i.e. [Pt(ppy)(N⁁S)]2, 3, with similar 1H NMR data as mentioned above (see also Experimental section), was also detected. Besides, formation of R–H (i.e. MeC6H5) is confirmed by the observation of a singlet at 2.31.
The dimeric complex [Pt(ppy)(N⁁S)]2, 3, was also prepared by direct route by reaction of the complex [Pt(ppy)Cl(DMSO)]42 with an ethanolic solution of sodium pyridine-2-thiolate (NaC5H4NS) under inert atmosphere condition and a purified sample of it (as checked by microanalysis) was characterized by 1H NMR spectroscopy. In the 1H NMR spectrum of complex 3 in CDCl3, the CH group adjacent to coordinated N atom of ppy appeared at δ = 8.09 as a doublet (3JH6H5 = 5.6 Hz) which coupled to 195Pt with 3JPtH6 = 19.1 Hz. The resonance of CH group adjacent to coordinated C atom of ppy is occurred at δ = 7.47 as a doublet with Pt satellite (3JPtH3′ = 32.8 Hz, 3JH3′H4′ = 5.7 Hz) and the CH group adjacent to coordinated N atom of N⁁S chelate was observed at δ = 7.86 as a doublet signal (3JH3H4 = 7.5 Hz, 3JPtH6 = 16.2 Hz), which confirmed the coordination of N⁁S as chelate. The complex 3 was also characterized by single-crystal X-ray diffraction analysis as illustrated in Fig. 3.45
3.2. Kinetics and mechanism of the reactions
On the basis of the data obtained from UV-vis and 1H NMR spectroscopic studies (see below), a mechanism (being depicted in Scheme 3) for reaction of the complex [PtR(ppy)(SMe2)], 1, with 1 equiv. pyridine-2-thione (C5H5SN) is proposed. The reaction is suggested to involve initial displacement of the SMe2 ligand by C5H5SN, during which S donor site of the thione tautomer (HN⁁S) attacks Pt center of the complex 1 to displace the SMe2 ligand, giving either the product 2a (or 2b) or its isomeric form, 2a′ (or 2b′), in which the py ring has twisted by 90° as compared to that in 2a (or 2b); in the latter case, N–H group has the opportunity to interact with the Pt center to form Pt⋯H–N hydrogen bonding as confirmed by 1H NMR studies (vide infra). The complex 2a′ (or 2b′) then performs an intramolecular N–H oxidative addition to form the intermediate I (or I′) following by CH3–H (or MeC6H5) reductive elimination to give the complex 3.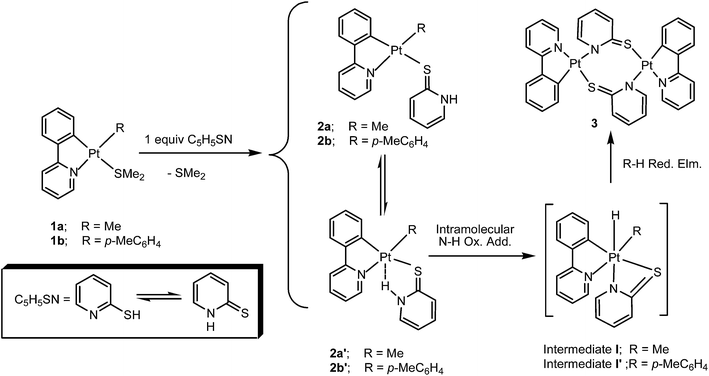 | ||
| Scheme 3 Suggested mechanism for reaction of the complex [PtR(ppy)(SMe2)], 1, with pyridine-2-thione. | ||
The abs-time data at λ = 500 (i.e. formation of the product 3) is fitted to the equation At = A + (A0 − A)exp(−kt), and the calculated rate constants at different temperatures are given in Table 1. The activation parameters were also obtained from measurement at different temperatures using Eyring equation (Fig. 6). Rates of the reactions are sensitive to the nature of R ligand and it is lower when the tolyl complex 1b is involved as compared to the case where the methyl complex 1a is used. The intermediate 2b′ then performs an intramolecular N–H oxidative addition reaction to form intermediate I′ (see Scheme 3) following by C–H reductive elimination to form toluene and the dimer complex 3.
| R | 102k s−1 at different temperatures | ΔH‡/kJ mol−1 | ΔS‡/J K−1 mol−1 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 5 °C | 10 °C | 15 °C | 20 °C | 25 °C | 30 °C | 35 °C | |||
| a Estimated errors in k values are ±5%. b The value in benzene 102k = 0.32 s−1. | |||||||||
| Me | 0.16 | 0.28 | 0.53 | 1.05b | 1.14 | 1.16 | 53.4 ± 3.9 | −105 ± 13 | |
| p-MeC6H4 | 0.012 | 0.019 | 0.027 | 0.039 | 0.052 | 50.8 ± 1.9 | −142 ± 6 | ||
 | ||
| Fig. 6 Eyring plots for reductive elimination of R–H and formation of dimer 3 from (a) complex 2a′ (R = Me) and (b) 2b′ (R = p-MeC6H4) in CH2Cl2. | ||
As is shown in Table 1, ΔH‡ value for methane reductive elimination from the intermediate I is 53.4 kJ mol−1 being considerably lower than the values of 107.4 and 70 kJ mol−1 reported for CH4 reductive elimination from Pt(IV) complexes [PtMe3(H)(dppe)] (dppe = 1,2-bis(diphenylphosphino)ethane)55 and [PtMe(H)(Cl)2(PMe3)2],56 respectively. This lower energy barrier found for [PtMe(H)(ppy)(S⁁N)] may be related to higher ring strain, presented in four-membered S⁁N ring in Pt(IV) intermediate I.
The ratio of 2a′ to 1a, immediately after addition of pyridine-2-thione to 1a, is close to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. As the time was passing on, the signals due to the starting material 1a was disappearing while those due to the complex 2a′ were growing and later started to fade away and meanwhile comparatively weak signals due to the complexes 2a and 3 (being out of the range of Fig. 7) were also observed in the first stages. As the reaction was progressed, the complex 2a′ was disappearing while the final complexes 2a and 3 were forming. As mentioned before, after 3 days the dimer 3 was solely obtained in pure form.
2. As the time was passing on, the signals due to the starting material 1a was disappearing while those due to the complex 2a′ were growing and later started to fade away and meanwhile comparatively weak signals due to the complexes 2a and 3 (being out of the range of Fig. 7) were also observed in the first stages. As the reaction was progressed, the complex 2a′ was disappearing while the final complexes 2a and 3 were forming. As mentioned before, after 3 days the dimer 3 was solely obtained in pure form.
Appearance of the corresponding signal for 2a at δ = 2.36 was used to measure rate of formation of the complex 2a, which was found to be 1.51(0.05) × 10−4 s−1. The rate of formation of CH4 (at δ = 0.16) which is equal to disappearance of the intermediate I and formation of the dimer 3 is measured to be 3.0 × 10−3 s−1, very close to value of 3.2 × 10−3 s−1 obtained from UV-vis study (vide supra).
We suggest that the formation of the complex 3 proceeds through an intramolecular N–H oxidative addition, involving the η1-S-S⁁NH ligand in 2a′, to the platinum center58,59 to form the platinum(IV) hydride intermediate [PtMe(H)(ppy)(S⁁N)], I, followed by reductive elimination of methane to form the complex 3. Our attempts to observe the intermediate I (by following the reaction at low temperatures) were not successful. However, evidence in favor of formation of this hypothesized Pt(IV) complex is the methane production, appearing as a singlet signal at δ = 0.16.60–62 Formation of the dimeric complex [Pt(ppy)(N⁁S)]2, 3, was confirmed by the observation of a doublet signal at δ = 7.84 with 3JH6H5 = 7.6 Hz, due to H6 of the N⁁S bridge, that experiences coupling with Pt with the satellites being broad and so the related coupling value was not measurable. Formation of the dimer 3 was also confirmed by X-ray crystal determination of suitable crystal of 3 obtained from crystallization of the mixture. Formation of the product complex [PtMe(ppy)(η1-S-S⁁NH)], 2a, in which pyridine-2-thione adopts the thione form,63–66 suggests a pathway including direct substitution of labile SMe2 by sulfur of thiolate ligand. Same experiment for the reaction of 1a with pyridine-2-thione was also carried out in CD2Cl2 as NMR solvent and found that the reaction in dichloromethane is faster than in benzene (in agreement with data obtained from the UV-vis spectroscopy, see Table 1).
3.3. Geometry optimizations
To get some insights in structures of the synthesized complexes 2 and 3, calculations in the DFT method were carried out and their geometries were optimized using the B3LYP function. Optimized geometric parameters of the complexes 2 and 3 are given in Table 2 and the DFT-optimized structures for these complexes are shown in Fig. 8. The computed structural details are in good agreement with corresponding experimental parameters obtained for the related complexes. Mean error for the bond lengths is 0.026 Å for the complex 3. One reason for the small bond length discrepancy may be due to differences in the geometrical parameters calculated in the gas phase with those obtained experimentally in the solid state.| 2a | 2b | 3 | 3 (exp) | |||
|---|---|---|---|---|---|---|
| Pt–S(S⁁NH) | 2.514 | Pt–S(S⁁NH) | 2.521 | Pt2–N3(N⁁S) | 2.233 | 2.144(10) |
| Pt–C(Me) | 2.064 | Pt–C(Ar) | 2.024 | Pt2–S2(N⁁S) | 2.370 | 2.286(3) |
| Pt–N(ppy) | 2.193 | Pt–N(ppy) | 2.190 | Pt2–C22(ppy) | 2.009 | 1.977(11) |
| Pt–C(ppy) | 2.012 | Pt–C(ppy) | 2.165 | Pt2–N2(ppy) | 2.093 | 2.064(9) |
| C(Me)–Pt–S(S⁁NH) | 91.4 | C(Ar)–Pt–S(S⁁NH) | 173.4 | Pt1–Pt2 | 3.009 | 2.8513(11) |
| S(S⁁NH)–Pt–N(ppy) | 95.0 | S(S⁁NH)–Pt–N(ppy) | 94.9 | C22(ppy)–Pt2–S2(N⁁S) | 95.0 | 95.3(3) |
| N(ppy)–Pt–C(ppy) | 79.7 | N(ppy)–Pt–C(ppy) | 79.6 | S2(N⁁S)–Pt2–N3(N⁁S) | 89.1 | 90.5(2) |
| C(ppy)–Pt–C(Me) | 93.9 | C(ppy)–Pt–C(Ar) | 95.4 | N2(ppy)–Pt2–N3(N⁁S) | 94.9 | 93.4(4) |
The complex 2a contains a square planar platinum(II) center being connected to the Me group, ortho C and N atoms of the ppy ligand and S atom of the S⁁NH. The two carbon ligating atoms are mutually cis in the square-planar structure, as expected, and largest deviation from ideal geometry is the angle N(ppy)-Pt-C(ppy) = 79.7, which is associated with the Pt(ppy) chelate ring. N atom of the S⁁NH ligand is not coordinated and is positioned opposite to the platinum center. Similar to 2a, the complex 2b has also a square-planar stereochemistry at platinum and the p-tolyl ligand lies roughly orthogonal to the square plane and to the ppy ligand. The Pt–S (S⁁NH) distances are 2.514 and 2.521 Å in 2a and 2b, respectively. The dimeric complex 3 has a square planar stereochemistry at each Pt center and has a head-to-tail configuration with PtSNPt coordination, in which S atom of one of the SN ligands is in a cis arrangement with N atom of the other SN ligand (Fig. 8); the coordination geometry around each platinum center is completed by nitrogen and ortho C atoms of the ppy ligand. This complex with a head-to-tail arrangement of the two SN ligands has been formulated as containing a Pt–Pt distance of 3.009 Å which is close to the experimental value of 2.8513(11) Å. This separation is amongst the shortest Pt–Pt interactions observed in binuclear platinum complexes bridged with two ligands.45 The calculated bond distance Pt–N(N⁁S) = 2.233 Å is slightly longer than Pt–N(ppy) = 2.093 Å due to a higher trans influence of the coordinating C atom of ppy as compared to coordinating S donor atom of N⁁S. These values are comparable with experimental values of 2.144(10) and 2.064(9) Å for Pt2–N3(N⁁S) and Pt2–N2(ppy), respectively. The two nitrogen atoms are cis disposed to one another (N(ppy)PtN(N⁁S) = 94.9°), indicating that angles around the Pt center are rather close to the ideal angle of 90°. The experimental value of this bond angle is equal to 93.4(4)°.
3.4. Energy profile for the product formation
To gain more insights into the species involved in the reaction of complexes 1 with C5H5SN (see Scheme 3), DFT calculations were carried out to show structures and energies for the related complexes in CH2Cl2 solution (Fig. 9 and 10). | ||
| Fig. 9 Calculated structures and relative energies of species involved in reaction of the complex 1a with pyridine-2-thione. | ||
 | ||
| Fig. 10 Calculated structures and relative energies of species involved in reaction of the complex 1b with pyridine-2-thione. | ||
As shown in Fig. 9, replacement of labile ligand SMe2 in the cycloplatinated(II) complex 1a by pyridine-2-thione (C5H5SN) first gives the complex [PtMe(ppy)(η1-S-S⁁NH)], 2a, having a NH pendant group, with 33.1 kJ mol−1 lower in energy with respect to that for 1a. This step is facilitated by strong trans–influence of the metalated C atom of ppy ligand which helps in weakening the Pt–SMe2 bond. The complex 2a is obtained in equilibrium with its isomeric form, 2a′, in which the pyridine ring has twisted by 90° as compared to that in 2a to form Pt⋯H–N hydrogen bonding (as confirmed by 1H NMR studies). The complex 2a′ is more stable than 2a by 14.8 kJ mol−1. In the next step, the complex 2a′ attends in an intramolecular N–H oxidative addition reaction to give the organoplatinum(IV) hydride complex [PtHMe(ppy)(N⁁S)], I, followed by reductive elimination of methane to give the dimer 3. The final product 3 is 139.0 kJ mol−1 more stable than 1a complying with the experimental finding.
In the related reaction profile (see Fig. 10), the complex 1b reacts with pyridine-2-thione to form the S-bonded complexes 2b and 2b′, which are more stable than 1b by 29.9 and 43.3 kJ mol−1, respectively. Coordination of pyridine-2-thione entering ligand through the N atom is not proposed, because the corresponding isomer [Pt(p-MeC6H4)(ppy)(η1-N-N⁁SH)] was calculated to be 42.4 kJ mol−1 less stable than 2b′. Oxidative addition of N–H bond to Pt center of the complex [Pt(p-MeC6H4)(ppy)(η1-S-S⁁NH)], 2b′, gives the Pt(IV) intermediate complex [PtH(p-MeC6H4)(ppy)(N⁁S)], I′, with 12.9 kJ mol−1 higher in energy than 1b. At the final step, reductive elimination of toluene gives dimer 3 which is more stable than 1b by 126.2 kJ mol−1.
4. Conclusion
Reaction of the cycloplatinated(II) complexes [PtR(ppy)(SMe2)], 1, with pyridine-2-thione is suggested to proceed via displacement of the labile SMe2 ligand. In this reaction pyridine-2-thione, which exists as a mixture of tautomers thiol (N⁁SH) and thione (HN⁁S), attacks Pt center of the complex 1 from S side of the thione form to give the four coordinated square-planar complex 2a (or 2b) and its isomer 2a′ (or 2b′), with a general formula [PtR(ppy)(η1-S-S⁁NH)]. DFT calculations show that this behavior is much preferred to the case of attacking from N side of the thiol form. The following points are considered for the reaction:(1) Suggestion in the first step is consistent with the “soft” S atom (from the thione tautomer, HN⁁S) being preferred to be connected to “soft” Pt(II) center as compared to that for the “hard” nature of N atom (from the thiol tautomer, N⁁SH).
(2) Formation of intramolecular Pt⋯H–N hydrogen bonding in one of the product isomers of the first step, i.e. the complex [PtR(ppy)(η1-S-S⁁NH)], 2a′, (as detected by monitoring the reaction using 1H NMR spectroscopy) is suggested to provide an extra stabilization as compared with the product isomer 2a.
(3) As was determined by UV-vis spectroscopy (see Table 1), R–H reductive elimination from the complex 2a′ or 2b′ to form the dimer complex 3 is significantly faster when R = Me (with k = 1.05 × 10−2 s−1 at 25 °C, in CH2Cl2) as compared to that when R = p-MeC6H4 (with k = 0.027 × 10−2 s−1 at 25 °C, in CH2Cl2). We attributed this to the easier reductive elimination of CH4 as compare with toluene.
(4) The rate of reaction of [PtMe(ppy)(SMe2)], 1a, with pyridine-2-thione is significantly faster in CH2Cl2 (k = 1.05 × 10−2 s−1 at 25 °C) than in benzene (k = 0.32 × 10−2 s−1 at 25 °C). We attribute this to the possibility that CH2Cl2 solvent molecules make the species more stable, by solvation, than when solvent molecules are the less polar benzene.
We finally observed the formation of two complexes, the S bounded complex [PtR(ppy)(η1-S-S⁁NH)], 2, and the dimeric complex [Pt(ppy)(N⁁S)]2, 3, along with the evolution of free R–H. These observations led us to believe that an intramolecular N–H oxidative addition reaction must have been taken place in the complex [PtR(ppy)(η1-S-S⁁NH)], 2a′ (or 2b′), to give the Pt(IV) intermediate complex [PtHR(ppy)(N⁁S)], I (or I′), in which the reductive elimination of R–H bond is needed to produce complex 3.
Acknowledgements
We thank the Iran National Science Foundation (Grant no. 92028194 and 93026027), the Shiraz University Research Council and Institute for Advanced Studies in Basic Sciences for financial support.References
- B. Butschke and H. Schwarz, Chem. Sci., 2012, 3, 308–326 RSC.
- M. Albrecht, Chem. Rev., 2009, 110, 576–623 CrossRef PubMed.
- Y. Chi and P.-T. Chou, Chem. Soc. Rev., 2010, 39, 638–655 RSC.
- M. Lersch and M. Tilset, Chem. Rev., 2005, 105, 2471–2526 CrossRef CAS PubMed.
- Y. Tanaka, K. M.-C. Wong and V. W.-W. Yam, Chem. Sci., 2012, 3, 1185–1191 RSC.
- Á. Díez, E. Lalinde and M. T. Moreno, Coord. Chem. Rev., 2011, 255, 2426–2447 CrossRef PubMed.
- M.-E. Moret, Top. Organomet. Chem., 2011, 35, 157–184 CrossRef CAS.
- J. A. G. Williams, Chem. Soc. Rev., 2009, 38, 1783–1801 RSC.
- J. R. Berenguer, E. Lalinde, M. T. Moreno, S. Sánchez and J. Torroba, Inorg. Chem., 2012, 51, 11665–11679 CrossRef CAS PubMed.
- S. M. Nabavizadeh, M. G. Haghighi, A. R. Esmaeilbeig, F. Raoof, Z. Mandegani, S. Jamali, M. Rashidi and R. J. Puddephatt, Organometallics, 2010, 29, 4893–4899 CrossRef CAS.
- A. Zucca, G. L. Petretto, S. Stoccoro, M. A. Cinellu, M. Manassero, C. Manassero and G. Minghetti, Organometallics, 2009, 28, 2150–2159 CrossRef CAS.
- T. Yagyu, J.-I. Ohashi and M. Maeda, Organometallics, 2007, 26, 2383–2391 CrossRef CAS.
- C. Anderson, M. Crespo, J. Morris and J. M. Tanski, J. Organomet. Chem., 2006, 691, 5635–5641 CrossRef CAS PubMed.
- S. Stoccoro, A. Zucca, G. L. Petretto, M. A. Cinellu, G. Minghetti and M. Manassero, J. Organomet. Chem., 2006, 691, 4135–4146 CrossRef CAS PubMed.
- J. S. Owen, J. A. Labinger and J. E. Bercaw, J. Am. Chem. Soc., 2004, 126, 8247–8255 CrossRef CAS PubMed.
- A. Zucca, A. Doppiu, M. A. Cinellu, S. Stoccoro, G. Minghetti and M. Manassero, Organometallics, 2002, 21, 783–785 CrossRef CAS.
- S. M. Nabavizadeh, H. Amini, F. Jame, S. Khosraviolya, H. R. Shahsavari, F. Niroomand Hosseini and M. Rashidi, J. Organomet. Chem., 2012, 698, 53–61 CrossRef CAS PubMed.
- S. M. Nabavizadeh, H. R. Shahsavari, M. Namdar and M. Rashidi, J. Organomet. Chem., 2011, 696, 3564–3571 CrossRef CAS PubMed.
- S. Jamali, R. Czerwieniec, R. Kia, Z. Jamshidi and M. Zabel, Dalton Trans., 2011, 40, 9123–9130 RSC.
- S. M. Nabavizadeh, H. Amini, H. R. Shahsavari, M. Namdar, M. Rashidi, R. Kia, B. Hemmateenejad, M. Nekoeinia, A. Ariafard, F. Niroomand Hosseini, A. Gharavi, A. Khalafi-Nezhad, M. T. Sharbati and F. Panahi, Organometallics, 2011, 30, 1466–1477 CrossRef CAS.
- S. Jamali, Z. Mazloomi, S. M. Nabavizadeh, D. Milić, R. Kia and M. Rashidi, Inorg. Chem., 2010, 49, 2721–2726 CrossRef CAS PubMed.
- S. Jamali, S. M. Nabavizadeh and M. Rashidi, Inorg. Chem., 2008, 47, 5441–5452 CrossRef CAS PubMed.
- B. Shafaatian, A. Akbari, S. M. Nabavizadeh, F. W. Heinemann and M. Rashidi, Dalton Trans., 2007, 4715–4725 RSC.
- M. Rashidi, M. C. Jennings and R. J. Puddephatt, Organometallics, 2003, 22, 2612–2618 CrossRef CAS.
- P. Espinet and K. Soulantica, Coord. Chem. Rev., 1999, 193, 499–556 CrossRef.
- Z.-Z. Zhang and H. Cheng, Coord. Chem. Rev., 1996, 147, 1–39 CrossRef CAS.
- S. Miranda, E. Cerrada, A. Luquin, A. Mendía and M. Laguna, Polyhedron, 2012, 43, 15–21 CrossRef CAS PubMed.
- G. Dyson, A. Hamilton, B. Mitchell and G. R. Owen, Dalton Trans., 2009, 6120–6126 RSC.
- X.-Y. Tang, H.-X. Li, J.-X. Chen, Z.-G. Ren and J.-P. Lang, Coord. Chem. Rev., 2008, 252, 2026–2049 CrossRef CAS PubMed.
- F. S. Nunes, L. D. S. Bonifácio, K. Araki and H. E. Toma, Inorg. Chem., 2006, 45, 94–101 CrossRef CAS PubMed.
- A. Mendía, E. Cerrada, F. J. Arnáiz and M. Laguna, Dalton Trans., 2006, 609–616 RSC.
- C. Vetter, C. Wagner, J. Schmidt and D. Steinborn, Inorg. Chim. Acta, 2006, 359, 4326–4334 CrossRef CAS PubMed.
- H. Fleischer, Coord. Chem. Rev., 2005, 249, 799–827 CrossRef CAS PubMed.
- J. A. García-Vázquez, J. Romero and A. Sousa, Coord. Chem. Rev., 1999, 193, 691–745 CrossRef.
- T. S. Lobana, S. Paul and A. Castineiras, J. Chem. Soc., Dalton Trans., 1999, 1819–1824 RSC.
- E. S. Raper, Coord. Chem. Rev., 1997, 165, 475–567 CrossRef CAS.
- E. S. Raper, Coord. Chem. Rev., 1996, 153, 199–255 CrossRef CAS.
- E. S. Raper, Coord. Chem. Rev., 1985, 61, 115–184 CrossRef CAS.
- G. Cervantes, S. Marchal, M. J. Prieto, J. M. Pérez, V. M. González, C. Alonso and V. Moreno, J. Inorg. Biochem., 1999, 77, 197–203 CrossRef CAS.
- Y. Nakatsu, Y. Nakamura, K. Matsumoto and S. I. Ooi, Inorg. Chim. Acta, 1992, 196, 81–88 CrossRef CAS.
- T. S. Lobana, R. Verma and A. Castineiras, Polyhedron, 1998, 17, 3753–3758 CrossRef CAS.
- N. Godbert, T. Pugliese, I. Aiello, A. Bellusci, A. Crispini and M. Ghedini, Eur. J. Inorg. Chem., 2007, 5105–5111 CrossRef CAS.
- T. S. Lobana, R. Verma, G. Hundal and A. Castineiras, Polyhedron, 2000, 19, 899–906 CrossRef CAS.
- S. M. Nabavizadeh, H. Sepehrpour, R. Kia and A. L. Rheingold, J. Organomet. Chem., 2013, 745, 148–157 CrossRef PubMed.
- T. Koshiyama, A. Omura and M. Kato, Chem. Lett., 2004, 33, 1386–1387 CrossRef CAS.
- M. J. Frisch, N. Rega, G. A. Petersson, G. W. Trucks, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, J. C. Burant, T. Nakajima, Y. Honda, O. Kitao, H. B. Schlegel, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, J. M. Millam, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, G. E. Scuseria, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, S. S. Iyengar, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, M. A. Robb, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, J. Tomasi, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. R. Cheeseman, J. V. Ortiz, Q. Cui, A. G. Baboul, V. Barone, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, J. A. Montgomery Jr, R. L. Martin, D. J. Fox, B. Mennucci, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, T. Vreven, M. W. Wong, M. Cossi, C. Gonzalez, J. A. Pople, K. N. Kudin and G. Scalmani, Gaussian 03, Revision C.02, 2004 Search PubMed.
- P. J. Hay and W. R. Wadt, J. Chem. Phys., 1985, 82, 270–283 CrossRef CAS PubMed.
- C. Lee, W. Yang and R. G. Parr, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 37, 785–788 CrossRef CAS.
- M. G. Haghighi, S. M. Nabavizadeh, M. Rashidi and M. Kubicki, Dalton Trans., 2013, 42, 13369 RSC.
- F. Raoof, A. R. Esmaeilbeig, S. M. Nabavizadeh, F. Niroomand Hosseini and M. Kubicki, Organometallics, 2013, 32, 3850–3858 CrossRef CAS.
- M. D. Aseman, M. Rashidi, S. M. Nabavizadeh and R. J. Puddephatt, Organometallics, 2013, 32, 2593–2598 CrossRef CAS.
- M. G. Haghighi, M. Rashidi, S. M. Nabavizadeh, S. Jamali and R. J. Puddephatt, Dalton Trans., 2010, 39, 11396–11402 RSC.
- S. Stoyanov, I. Petkov, L. Antonov, T. Stoyanova, P. Karagiannidis and P. Aslanidis, Can. J. Chem., 1990, 68, 1482–1489 CrossRef CAS PubMed.
- H.-L. Zhang, S. D. Evans, J. Henderson, R. E. Miles and T. Shen, J. Phys. Chem. B, 2003, 107, 6087–6095 CrossRef.
- D. M. Crumpton-Bregel and K. I. Goldberg, J. Am. Chem. Soc., 2003, 125, 9442–9456 CrossRef CAS PubMed.
- K. L. Bartlett, K. I. Goldberg and W. T. Borden, Organometallics, 2001, 20, 2669–2678 CrossRef CAS.
- A. Martín, J. Chem. Educ., 1999, 76, 578 CrossRef.
- A. Dervisi, R. L. Jenkins, K. M. Abdul Malik, M. B. Hursthouse and S. Coles, Dalton Trans., 2003, 1133–1142 RSC.
- J. Real, A. Polo and J. Duran, Inorg. Chem. Commun., 1998, 1, 457–459 CrossRef CAS.
- G. R. Fulmer, A. J. Miller, N. H. Sherden, H. E. Gottlieb, A. Nudelman, B. M. Stoltz, J. E. Bercaw and K. I. Goldberg, Organometallics, 2010, 29, 2176–2179 CrossRef CAS.
- H. R. Shahsavari, M. Rashidi, S. M. Nabavizadeh, S. Habibzadeh and F. W. Heinemann, Eur. J. Inorg. Chem., 2009, 3814–3820 CrossRef CAS.
- D. M. Crumpton-Bregel and K. I. Goldberg, J. Am. Chem. Soc., 2003, 125, 9442–9456 CrossRef CAS PubMed.
- S. Takemoto, Y. Yamazaki, T. Yamano, D. Mashima and H. Matsuzaka, J. Am. Chem. Soc., 2012, 134, 17027–17035 CrossRef CAS PubMed.
- A. C. Sykes, P. White and M. Brookhart, Organometallics, 2006, 25, 1664–1675 CrossRef PubMed.
- J. Zhao, A. S. Goldman and J. F. Hartwig, Science, 2005, 307, 1080–1082 CrossRef CAS PubMed.
- R. L. Cowan and W. C. Trogler, J. Am. Chem. Soc., 1989, 111, 4750–4761 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra01341k |
| This journal is © The Royal Society of Chemistry 2015 |