Synthesis of high strength monolithic alumina aerogels at ambient pressure†
Fengchao Cao,
Lili Ren* and
Xueai Li
School of Chemistry & Chemical Engineering, Southeast University, Nanjing, 211189, China. E-mail: liliren@seu.edu.cn
First published on 5th February 2015
Abstract
Alumina aerogels have many fascinating properties, such as high temperature stability and high surface area, which result in them having great potential applications. However, their mechanical properties are very poor, which greatly limits their practical application and commercialization. In this communication we successfully synthesized monolithic alumina aerogels via ambient pressure drying by using attapulgite (ATP) as a reinforcing agent. The resulting attapulgite/alumina composite aerogels exhibit strong mechanical properties. TEM/SEM analysis showed that alumina particles firmly adhered to the surface of the rod shaped crystal of attapulgite and attapulgite played a supporting role as the skeleton in the structure of the composite aerogels.
Aerogels are highly porous solid materials derived from gels formed when the gas substitutes the liquid component within the pores of the gel,1–3 which make them possess many excellent properties suitable for a range of applications, such as thermal super-insulators, adsorbents, catalyst carriers and sensors.4 But the applications of the aerogels are greatly limited because of the lack of a suitable drying method and the poor mechanical properties of the aerogels, especially inorganic oxide aerogels, which easily break into fragments even under a small stress.5,6 Therefore, improving the strength of the aerogels and devising a new drying method are the main focuses of aerogel research. Parmenter et al.7 synthesized ceramic fiber/silica aerogels by supercritical ethanol drying, and reported that the monolithic composites have stronger mechanical properties than single silica aerogels. Yang et al.8 prepared an inorganic fiber-reinforced silica aerogel by supercritical drying, which showed high compressive strength, without sacrificing its thermal conductivity. Fu et al.9 fabricated activated fiber/carbon aerogel (ACF/CA) composites by supercritical drying and found that the fiber could fortify the structure of the carbon aerogel. Our research group has studied aerogel drying methods thoroughly and successfully prepared monolithic inorganic oxide aerogels through an organic solvent sublimation drying method (OSSD).10 Alumina aerogels possess better thermal and chemical stability in addition to the basic properties of aerogels, which make them have great potential applications, such as high temperature resistant materials and catalyst supports with high surface area.11 Therefore, studying the preparation of alumina aerogel has attracted increasing attention.12–14 However, alumina aerogels are also friable and have poor mechanical properties,4 which make their practical application greatly restricted.
Attapulgite (ATP) is a type of rare natural hydrated magnesium aluminum silicate clay mineral with a layered-chain and fibrous crystal structure.15 Owing to its intrinsic properties, such as special crystal structure and superficial character, ATP possesses many fine properties, such as being adsorptive and adiabatic.15 Recently, ATP has been widely used as an inorganic filler added into a polymer or rubber matrix, and brings great improvement to the properties of the composites, such as tensile strength,16,17 and compressive strength.18 This is mainly attributed to the fibrous and layered-chain structure.19 Additionally, the hydrophilic fibrous structure is typically compatible with hydrophilic polymers.
Therefore, in this communication we proposed to add ATP into the structure of an alumina aerogel, so as to improve its strength through the support effect of the special structure of ATP.
During preparation, we chose AlCl3·6H2O as precursor. Acidic conditions favor hydrolysis, so we used hydrochloric acid (1 M) as acid catalyst. The propylene oxide (PO) was used as gelation inducing agent. It can consume the protons from the hydrated metal species and drive the sol–gel polymerization reaction.20,21 The sol–gel mechanism could be described as follows:
(1) Hydrolysis:
| [Al(H2O)6]3+ → [Al(OH)x(H2O)6−x](3−x)+ + xH+ | (1) |
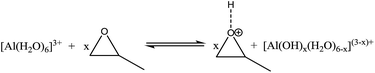 | (2) |
(2) Condensation:
| 2[Al(OH)x(H2O)6−x](3−x)+ → H2O + [(H2O)6−x(OH)x−1Al–O–Al(OH)x−1(H2O)6−x]2(3−x)+ | (3) |
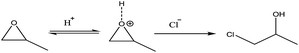 | (4) |
Monolithic alumina aerogel cannot be obtained (Fig. 1a) via ambient pressure drying because of large capillary force.22 While it could be easily prepared when a little ATP was added. We prepared various shapes of ATP/alumina aerogel composites according to different demands (Fig. 1b), and they all kept intact surface. In order to further investigate the effect of ATP, ATP/alumina aerogel composites with different ATP weight ratios were synthesized by ambient pressure drying method. Because the monolithic pure alumina aerogel was not obtained via ambient pressure drying, its compression test cannot be carried out. Fig. 1c shows the compressive stress–displacement curves of ATP/Al2O3 aerogel composites with different attapulgite weight ratios. Fig. 1d shows that the compressive strength significantly increased when a little ATP was added. With the ATP content increasing from 10% to 26%, the compressive strength increases from 40.99 MPa to 75.44 MPa. While as the ATP content continued increasing, the compressive strength began to decrease slightly, and crack-free ATP/alumina aerogel composites cannot be obtained when the content of ATP increased to 66 wt%. The fragility of inorganic oxide aerogel is a well-recognized defect for its practical application. Our research shows that no matter how much the ATP content is, the compressive strength of ATP/Al2O3 aerogel composites is far higher than that of pure alumina aerogels.
Fig. 2 shows the TEM and SEM images of ATP, Al2O3, and ATP/Al2O3 respectively. From Fig. 2 we can see that ATP exists as bundles of rod shaped crystal, the skeleton of Al2O3 aerogels is formed by the random accumulation of colloidal particles (ranging from a few tens to a few hundred nanometers), and the skeleton of ATP/Al2O3 aerogels is formed by the random assemble of rod unit (growing by alumina particles adhering to rod ATP). The rod-like crystal structure of ATP favored the formation of supporting framework, which can effectively improve the mechanical properties of alumina aerogels. That is to say the ATP offers a supporting framework as an initiate skeleton, and the alumina particles can firmly adhere to it and gradually grow up to 3D network (see Fig. 2f), then the total structure of alumina aerogel composites are fortified.
Fig. 3 shows the pore size distribution and N2 adsorption–desorption isotherms for alumina aerogel (a) and ATP/Al2O3 aerogel composite (b, wATP = 26%). The isotherms are type IV with H2 hysteresis loops characteristic of mesoporous materials. The most probable pore size (BJH) and specific surface areas (BET) of the pure alumina and ATP/alumina aerogel (wATP = 26%) were 12.12 nm and 13.89 nm, 295.18 m2 g−1 and 242.86 m2 g−1, respectively. The parameters of the two kinds of aerogels are similar, but ATP/Al2O3 aerogel exhibits as a crack-free monolith. It indicates that the structure of alumina aerogel is strengthened and can withstand the tensile forces of liquid after adding a little ATP. The SEM and TEM images of Al2O3, and ATP/Al2O3 reveal the distinct textures and indicate that the main channels of ATP/Al2O3 (ranging from a few tens to a few hundred nanometers) are effected by the random accumulation of rod unit (see Fig. 2d and f).
 | ||
| Fig. 3 Nitrogen sorption isotherms and Barrett–Joyner–Halenda (BJH) pore size distribution of pure alumina aerogel (a) and ATP/alumina aerogel composite (wATP = 26%) (b). | ||
Based on the micro-morphology of alumina particles and ATP unit, we propose the following mechanism to explain the improvement for mechanical property of the alumina aerogel by adding ATP (Fig. 4). The rod ATP in the sol allows the alumina particles condensing onto it. As condensation proceeds, alumina particles are growing up to secondary particles and the skeleton of ATP/Al2O3 aerogels is forming by the random assemble of rod unit (growing by alumina particles adhering to rod ATP). The alumina colloidal particles firmly adhere to rod ATP and gradually grow up to 3D network, and then the total structure gets fortified. Due to the formation of reinforced framework, the wet gel can withstand the destructive capillary pressure during ambient pressure drying. The structure of the ATP/Al2O3 aerogel remains intact and crack-free during the entire drying process.
 | ||
| Fig. 4 Schematic of the formation of alumina aerogel (a) and ATP/alumina aerogel composites (b) at ambient pressure condition. | ||
Fig. 5 shows the XRD patterns and FTIR spectra of Al2O3 aerogel, ATP/Al2O3 aerogel and ATP. Combined with the IR spectra and XRD patterns, the new characteristic bands and peaks are not found in all the IR spectra and XRD patterns for the ATP/Al2O3 aerogel composite material. This reveals that there is no new form generated when ATP adds into Al2O3, that is to say the addition of ATP will not change the origin nature of alumina aerogel (see Table S1, Fig. S1 and S2†). In addition, low content and evenly dispersed of each component lead some characteristic peaks to shrink or disappear. Compared with traditional supercritical drying, ambient pressure drying is extremely convenient and cost-saving by adding ATP into alumina aerogel.
 | ||
| Fig. 5 The XRD patterns (a) and FTIR spectra (b) of Al2O3 aerogel (black line), ATP/Al2O3 aerogel (red line) and ATP (blue line). | ||
Conclusions
In summary, the monolithic reinforced ATP/Al2O3 aerogel composite was successfully synthesized under ambient pressure by adding ATP into alumina aerogel matrix. The compressive tests show that compressive strength of ATP/alumina aerogel (wATP = 26%) can reach up to 75.44 MPa. Owning to its fortified structure, ATP/Al2O3 wet gel can be dried directly by ambient condition without time-consuming and costly solvent substitution. It is of vital significance for the preparation and application of alumina aerogel, and this approach may also be used for the preparation of other inorganic oxide aerogels.Acknowledgements
This work was financially supported by National Nature Science Foundation of China (21206018) and the Foundation of Ministry of Education of China (11YJCZH139).Notes and references
- S. S. Kistler, Nature, 1931, 127, 741 CrossRef CAS.
- J. Fricke and T. Tillotson, Thin Solid Films, 1997, 297, 212–223 CrossRef CAS.
- H. D. Gesser and P. C. Goswami, Chem. Rev., 1989, 89, 765–788 CrossRef CAS.
- A. C. Pierre and G. M. Pajonk, Chem. Rev., 2002, 102, 4243–4265 CrossRef CAS PubMed.
- J. Yang, S. Li, Y. Luo, L. Yan and F. Wang, Carbon, 2011, 49, 1542–1549 CrossRef CAS PubMed.
- A. Karout, P. Buisson, A. Perrard and A. C. Pierre, J. Sol–Gel Sci. Technol., 2005, 36, 163–171 CrossRef CAS.
- K. E. Parmenter and F. Milstein, J. Non-Cryst. Solids, 1998, 223, 179–189 CrossRef CAS.
- X. Yang, Y. Sun, D. Shi and J. Liu, Mater. Sci. Eng., A, 2011, 528, 4830–4836 CrossRef PubMed.
- R. W. Fu, B. Zheng, J. Liu, S. Weiss, J. Y. Ying, M. S. Dresselhaus, G. Dresselhaus, J. H. Satcher and T. F. Baumann, J. Mater. Res., 2003, 18, 2765–2773 CrossRef CAS.
- L. Ren, S. Cui, F. Cao and Q. Guo, Angew. Chem., Int. Ed., 2014, 53, 10147–10149 CrossRef CAS PubMed.
- J. F. Poco, J. H. Satcher and L. W. Hrubesh, J. Non-Cryst. Solids, 2001, 285, 57–63 CrossRef CAS.
- Z. Sun, T. Zheng, Q. Bo, M. Du and W. Forsling, J. Colloid Interface Sci., 2008, 319, 247–251 CrossRef CAS PubMed.
- L. Wu, Y. Huang, Z. Wang, L. Liu and H. Xu, Appl. Surf. Sci., 2010, 256, 5973–5977 CrossRef CAS PubMed.
- W. Bao, F. Guo, H. Zou, S. Gan, X. Xu and K. Zheng, Powder Technol., 2013, 249, 220–224 CrossRef CAS PubMed.
- W. L. Haden and I. A. Schwint, Ind. Eng. Chem. Res., 1967, 59, 58–69 CrossRef CAS.
- B. Pan, Q. Yue, J. Ren, H. Wang, L. Jian, J. Zhang and S. Yang, Polym. Test., 2006, 25, 384–391 CrossRef CAS PubMed.
- J. Wang and D. Chen, J. Nanomater., 2013, 2013, 1–11 Search PubMed.
- G. Gao, G. Du, Y. Cheng and J. Fu, J. Mater. Chem. B, 2014, 2, 1539–1548 RSC.
- C. Chen, J. Phys. Chem. Solids, 2008, 69, 1411–1414 CrossRef CAS PubMed.
- T. F. Baumann, A. E. Gash, S. C. Chinn, A. M. Sawvel, R. S. Maxwell and J. H. Satcher, Chem. Mater., 2005, 17, 395–401 CrossRef CAS.
- A. E. Gash, T. M. Tillotson, J. H. Satcher, J. F. Poco, L. W. Hrubesh and R. L. Simpson, Chem. Mater., 2001, 13, 999–1007 CrossRef CAS.
- L. L. Hench and J. K. West, Chem. Rev., 1990, 90, 33–72 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra00424a |
| This journal is © The Royal Society of Chemistry 2015 |


