Regio- and diastereoselectivity of the 1,3-dipolar cycloaddition of α-aryl nitrone with methacrolein. A theoretical investigation†
Wafaa Benchouk and
Sidi Mohamed Mekelleche*
Laboratory of Applied Thermodynamics and Molecular Modelling, Department of Chemistry, Faculty of Science, University of Tlemcen, PB 119, Tlemcen, 13000, Algeria. E-mail: sm_mekelleche@mail.univ-tlemcen.dz
First published on 18th February 2015
Abstract
The mechanism, regio- and diastereoselectivity of the 1,3-dipolar cycloaddition of N-iso-propyl,α-(4-trifluoromethyl)-phenyl nitrone with methacrolein yielding the isoxazolidine cycloadduct [V. Bãdoiu, E. P. Kündig, Org. Biomol. Chem., 2012, 10, 114] has been studied at the B3LYP/6-31G(d) level of theory. The two conformations s-cis and s-trans of methacrolein are considered in the cyclisation modes and all the possible regio-(ortho/meta) and stereo-(endo/exo) isomeric channels to provide the different diastereoisomers were thoroughly investigated. The free activation enthalpies, calculated with the MPW1B95/6-31G(d) method, in dichloromethane show that the meta-endo channel corresponding to the cyclisation mode between the nitrone and the s-cis methacrolein giving the RR/SS diastereoisomers is the most favoured reaction channel as expected experimentally. Intrinsic reaction coordinate (IRC) calculations and the topological analysis of the electron localization function (ELF) of some relevant points of the IRC curve show that the meta-endo favoured channel takes place via a one-step non-concerted mechanism. Charge transfer calculations performed in the transition states in combination with the calculation of DFT-based reactivity indices of the reactants reveal a low polar character of the studied reaction.
Introduction
The synthetic utility of the 1,3-dipolar cycloaddition (13DC) reaction is evident from the number and scope of targets that can be prepared by this chemistry.1 As one of the most thoroughly investigated 1,3-dipoles, nitrones have been successfully employed to generate nitrogen- and oxygen-based functionality from cycloadducts as well as the potential to introduce new and multiple chiral centers stereoselectively.1The most common 13DC reaction of nitrones is the formation of an isoxazolidine using alkene dipolarophiles (see Scheme 1), although other multiply bonded systems may also be used (alkynes, allenes, isocyanates, nitriles, thiocarbonyls, etc.). Isoxazolidines compounds have attracted interest as a result of biological activity, including anti-HIV and cytotoxicity, resulting from their ability to act as nucleoside analogs.2 In addition, they have served as synthetic precursors to other classes of natural products, such as 1,3-amino alcohols, β-lactams, and alkaloids with physiological activity.3 The isoxazolidine cycloadduct contains up to three new chiral centers (see Scheme 1) and, as with other 1,3-dipoles, the highly ordered transition state often allows the regio- and stereochemical preference of a given nitrone to be predicted.1,4 This prediction is achieved through a consideration of steric and electronic factors.
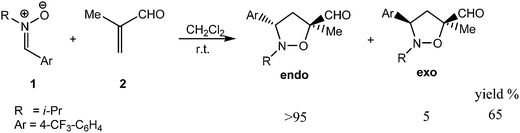
The 13DC reaction of nitrones with dipolarophiles can produce both endo and exo isoxazolidine cycloadducts;5 this nomenclature is well-known from the Diels–Alder (DA) reaction.6 If the dipolarophile, or the 1,3-dipole, contains chiral center(s), the approach toward one of the faces of the 1,3-dipole or dipolarophile can be discriminated, leading to a diastereoselective reaction. The term enantioselectivity will only be applied when optically active products are obtained from achiral or racemic starting materials.5 The selectivity in the 13DC reaction is therefore primarily controlled by the structure of the substrates or by application of chiral Lewis acids (LAs). In this context, the use of metal-based catalysts, organo-catalysts in modern 13DC asymmetric organic synthesis have been expanding uninterruptedly during the last decades.7 Several experimental studies on the regio-, enantio- and endo/exo diastereoselective synthesis of 13DC reactions of nitrones can be found in the literature.8 Nitrones bearing various substituents at the nitrogen atom of the nitrone were synthesized by Bãdoiu et al.9 in order to expand the range of transformations that can be carried out on the isoxazolidine core following the 13DC reaction. In this series, the N-i-Pr (see Scheme 2) and -t-Bu nitrones, respectively, gave the products in moderate yields despite long reaction times and a two-fold excess of methacrolein.
Several experimental works supported by theoretical approaches to investigate the regio-, stereo- and diastereoselectivity of the 13DC of nitrones can be found in the literature.10–13 The regioselectivity and endo/exo selectivities of the 13DC reaction of 1-pyrroline-1-oxide to methyl cinnamate and benzylidene acetophenone were rationalized through both experimental and theoretical findings by Acharjee et al.11 Flores et al.12 undertook a theoretical study of 13DCs of phenylvinylsulfone and several nitrones in order to explain the experimental results, including the regiochemistry, diastereoselectivity, and kinetic control. The reaction mechanism of the 13DC of C,N-diphenylnitrone with unsaturated valerolactone under thermal and LA-mediated conditions has been studied by Śnieżek et al.13 using DFT and MP3 methods. Benchouk et al.14 studied the role of LA catalysts on the 13DC of N-benzylideneaniline N-oxide nitrone with acrolein using DFT calculations. The regioselectivity of the 13DC of C-(methoxycarbonyl)-N-methyl nitrone with methyl acrylate and vinyl acetate have been analysed by Merino et al.10b and Benchouk et al.15 using several theoretical approaches.
In the present work, we present a theoretical study of the regio-, stereo- and diastereoselectivity of the non-catalyzed 13DC of N-iso-propyl,α-(4-trifluoromethyl)-phenyl nitrone 1 and methacrolein 2, experimentally studied by Bãdoiu et al.9 (see Scheme 2). We note that the best yields of diastereoselectivities (in favour of the endo isomer) have been obtained in the case of non-catalyzed reaction (in comparison of the catalyzed one) using the N-i-Pr and t-Bu nitrones and exclusively the 3,5-substituted regioisomers were isolated (see Scheme 2).9 Firstly, in order to justify the most favoured cyclisation approach, we have performed a density functional theory (DFT) study with the aim to localize the stationary points for reactants, transition structures (TSs), cycloadducts (CAs) of all the possible regio- and stereoisomeric channels on the potential energy surface (PES). Then, the reaction mechanism is discussed and analysed on the basis of the intrinsic reaction coordinate (IRC) calculations and the topological analysis of the electron localization function (ELF). Finally, a complementary study based on reactivity indexes defined within the conceptual DFT is used to analyze the polar (vs. non polar) character of the studied 13DC reaction.
Results and discussion
The α-aryl nitrone 1 exists in both Z and E conformations. B3LYP/6-31G(d) calculations show that the Z conformation is more stable than the E conformation by 7.9 kcal mol−1 (see S2 and S3 of the ESI†). This finding is in accordance with experimental outcome since 1H NMR analysis at room temperature showed that only the Z isomer present in solution.9 For this reason, only the Z conformation of the α-aryl nitrone 1 will be considered in the 13DC reaction under study. Liu et al.16 performed theoretical and experimental analyses of the conformational switches in TSs of some DA and 13DC cycloadditions. Barba et al.17 showed that the preference of the s-cis/s-trans conformation of methacrolein 2 in DA and 13DC cycloadditions derives from a delicate balance between the larger stability of the s-trans conformer of the ground-state of methacrolein and the larger reactivity of the corresponding s-cis conformation. B3LYP/6-31G(d) calculations show that the s-trans conformation of methacrolein 2-trans is lower in energy than the 2-cis conformation by 2.9 kcal mol−1 (see S4 of the ESI†). Both the s-cis and s-trans conformations of methacrolein (see Schemes 3 and 4) have been considered in the present study. | ||
| Scheme 3 Regio- and stereoisomeric channels corresponding to the 13DC reaction of α-arylnitrone 1 with methacrolein 2-cis. | ||
 | ||
| Scheme 4 Regio- and stereoisomeric channels corresponding to the 13DC reaction of α-arylnitrone 1 with methacrolein 2-trans. | ||
In the present theoretical study of the regio- and diastereoselectivity of the 13DC reaction, two cyclisation modes were investigated. The cyclisation mode A (CM-A) corresponding to the 13DC reaction between α-aryl nitrone 1 with methacrolein 2-cis (Scheme 3) and the cyclisation mode B (CM-B) corresponding to the 13DC reaction between α-aryl nitrone 1 with methacrolein 2-trans (Scheme 4). Due to the asymmetry of the dipole and the dipolarophile, in the 13DC reaction of the α-aryl nitrone 1 with methacrolein 2-cis/trans, several reaction channels are feasible. The formation of eight isomeric isoxazolidines cycloadducts for CM-A and eight isomeric isoxazolidines cycloadducts for CM-B can be related to the two regioisomeric channels ortho and meta and the two stereoisomeric approaches endo and exo (Schemes 3 and 4). The experimental findings due to Bãdoiu et al.9 indicate that this cycloaddition reaction is characterized by a complete endo stereoselectivity with the unique formation of the regioisomer associated with the formation of the O1–C5 and C3–C4 sigma bonds (Schemes 3 and 4). Bãdoiu and co-workers have also found, for this non-catalyzed reaction, that the enantioselectivity is moderate and a racemic mixture of the (3R,5R) and (3S,5S) enantiomers was obtained. In order to explain the origin of the regio- and diastereoselectivity experimentally observed, sixteen reaction channels were investigated and analysed (Schemes 3 and 4). In this study, we have considered the two regioisomeric channels, namely ortho and meta, corresponding to the formation of the 3,4 and 3,5-disubstituted isoxazolidines, respectively. We note that the regioselectivity is referred to the attack of the oxygen atom of the α-aryl nitrone 1 to the carbon atom of the C–C double bond of methacrolein. Due to the mutual orientation of reactants, two stereoisomeric approaches can be considered, namely, endo and exo, corresponding to the position of the carbonyl group of methacrolein 2 towards to the nitrogen atom of α-aryl nitrone 1. The presence of the four possible diastereoisomeric cycloadducts identified in the reaction is fully justified by the existence of two chiral centers for each regioisomer. Consequently, a total of sixteen reaction channels have been investigated. Eight TSs for CM-A, namely, TS1n-SS-c, TS1n-RR-c, TS1x-RS-c, TS1x-SR-c, TS2n-RR-c, TS2n-SS-c, TS2x-RS-c and TS2x-SR-c and the corresponding isoxazolidines cycloadducts CA1n-SS-c, CA1n-RR-c, CA1x-RS-c, CA1x-SR-c, CA2n-RR-c, CA2n-SS-c, CA2x-RS-c and CA2x-SR-c (see Scheme 3 and pages S5–S20 of the ESI†) and eight TSs for CM-B, namely, TS1n-SS-t, TS1n-RR-t, TS1x-RS-t, TS1x-SR-t, TS2n-RR-t, TS2n-SS-t, TS2x-RS-t and TS2x-SR-t and the corresponding isoxazolidines cycloadducts CA1n-SS-t, CA1n-RR-t, CA1x-RS-t, CA1x-SR-t, CA2n-RR-t, CA2n-SS-t, CA2x-RS-t and CA2x-SR-t (see Scheme 4 and pages S21–S36 of the ESI†) were located and characterized on the PES. The calculated energies of all the stationary points in gas phase and in dichloromethane (DCM) are summarized in Table 1. It turns out the most favourable reaction pathway corresponds to the formation of the endo stereoisomeric isoxazolidines cycloadducts CA1n-SS-c and CA1n-RR-c enantiomers, via TS1n-SS-c and TS1n-RR-c, respectively. We note that for the CM-A, the TS1n-SS-c and TS1n-RR-c TSs are located 15.4 kcal mol−1 above the reagents in the gas phase. The energy differences between TS1n-SS-c/TS1n-RR-c and the other couples of enantiomers are 5.9 kcal mol−1 for TS1x-RS-c/TS1x-SR-c, 1.2 kcal mol−1 for TS2n-SS-c/TS2n-RR-c and 7.2 kcal mol−1 for TS2x-RSc/TS2x-SR-c. For the CM-B, the most favourable reaction pathway corresponds to the formation of the endo CA1n-SS-t and CA1n-RR-t cycloadducts, via TS1n-SS-t and TS1n-RR-t, respectively. Indeed, the TS1n-SS-t and TS1n-RR-t TSs are located 20.2 kcal mol−1 above the reagents in the gas phase. The energy difference between TS1n-SS-t/TS1n-RR-t and the other couples of enantiomers are 2.6 kcal mol−1 for TS1x-RS-t/TS1x-SR-t, 1.7 kcal mol−1 for TS2n-SS-t/TS2n-RR-t and 3.6 kcal mol−1 for TS2x-RS-t/TS2x-SR-t. It turns out that the CM-A is more favoured than CM-B. Indeed, the TS1n-SS-c/TS1n-RR-c are lower in energy than TS1n-SS-t/TS1n-RR-t by 4.8 kcal mol−1, indicating a clear preference of the meta-endo reaction channel via CM-A which is in agreement with experimental findings.9
| Gas phase | Dichloromethane | |||
|---|---|---|---|---|
| E | ΔEa | E | ΔEa | |
| a Relative to reactants. | ||||
| Cyclisation Mode A | ||||
| 1 | −855.852632 | −855.868923 | ||
| 2-cis | −231.228858 | −231.235911 | ||
| TS1n-SS-c | −1087.056973 | 15.4 | −1087.075835 | 18.2 |
| TS1n-RR-c | −1087.056973 | 15.4 | −1087.075835 | 18.2 |
| TS1x-RS-c | −1087.047561 | 21.3 | −1087.068575 | 22.8 |
| TS1x-SR-c | −1087.047561 | 21.3 | −1087.068575 | 22.8 |
| TS2n-RR-c | −1087.054962 | 16.6 | −1087.072847 | 20.1 |
| TS2n-SS-c | −1087.054962 | 16.6 | −1087.072847 | 20.1 |
| TS2x-RS-c | −1087.045425 | 22.6 | −1087.066997 | 23.7 |
| TS2x-SR-c | −1087.045425 | 22.6 | −1087.066997 | 23.7 |
| CA1n-SS-c | −1087.103057 | −13.5 | −1087.121751 | −10.6 |
| CA1n-RR-c | −1087.103057 | −13.5 | −1087.121751 | −10.6 |
| CA1x-RS-c | −1087.101302 | −12.4 | −1087.120545 | −9.9 |
| CA1x-SR-c | −1087.101302 | −12.4 | −1087.120547 | −9.9 |
| CA2n-RR-c | −1087.093715 | −7.7 | −1087.112313 | −4.7 |
| CA2n-SS-c | −1087.093715 | −7.7 | −1087.112313 | −4.7 |
| CA2x-RS-c | −1087.093189 | −7.3 | −1087.112755 | −5.0 |
| CA2x-SR-c | −1087.093189 | −7.3 | −1087.112755 | −5.0 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Cyclisation Mode B | ||||
| 1 | −855.852632 | −855.868923 | ||
| 2-trans | −231.233543 | −231.240636 | ||
| TS1n-SS-t | −1087.053972 | 20.2 | −1087.075862 | 21.1 |
| TS1n-RR-t | −1087.053972 | 20.2 | −1087.075862 | 21.1 |
| TS1x-RS-t | −1087.049825 | 22.8 | −1087.071497 | 23.9 |
| TS1x-SR-t | −1087.049825 | 22.8 | −1087.071498 | 23.9 |
| TS2n-RR-t | −1087.051335 | 21.9 | −1087.072337 | 23.4 |
| TS2n-SS-t | −1087.051335 | 21.9 | −1087.072337 | 23.4 |
| TS2x-RS-t | −1087.048214 | 23.8 | −1087.069159 | 25.4 |
| TS2x-SR-t | −1087.048214 | 23.8 | −1087.069159 | 25.4 |
| CA1n-SS-t | −1087.101744 | −9.8 | −1087.121365 | −7.4 |
| CA1n-RR-t | −1087.101744 | −9.8 | −1087.121365 | −7.4 |
| CA1x-RS-t | −1087.101181 | −9.4 | −1087.121480 | −7.5 |
| CA1x-SR-t | −1087.101181 | −9.4 | −1087.121479 | −7.5 |
| CA2n-RR-t | −1087.095235 | −5.7 | −1087.114731 | −3.2 |
| CA2n-SS-t | −1087.095235 | −5.7 | −1087.114732 | −3.2 |
| CA2x-RS-t | −1087.094966 | −5.5 | −1087.114233 | −2.9 |
| CA2x-SR-t | −1087.094966 | −5.5 | −1087.114233 | −2.9 |
Solvent effects on cycloaddition reactions are well-known and they have received considerable attention.1,18,19 With the inclusion of solvent effects (see Table 1), the TSs and CAs are more stabilized than reactants and the meta-endo reaction channel via CM-A is still the favoured one although the activation energy associated with TS1n-SS-c/TS1n-RR-c is increased from 15.4 kcal mol−1 (in gas phase) to 18.2 kcal mol−1 (in DCM).
It is well-recognized that the B3LYP DFT functional is not adequate for thermochemical calculations and the MPW1PW91/6-311+G(d,p) and MPW1B95/6-31(d) computational levels were found to give a reasonably good agreement with the experimental kinetics and thermodynamic parameters for chemical reactions.20,21 The relative Gibbs free energies in DCM for the sixteen reaction channels were computed at the MPW1B95/6-31(d) level of theory using B3LYP/6-31G(d) optimized geometries and the obtained results are summarized in Table 2. The analyses of the tabulated results show that the meta-endo reaction channel via CM-A yielding to the formation of the CA1n-SS-c/CA1n-RR-c isomers is favoured both kinetically and thermodynamically. We note that this favoured reaction channel is remarkably exothermic by 19.5 kcal mol−1.
| H (a.u.) | ΔHa (kcal mol−1) | S (cal K−1 mol−1) | ΔSa (cal K−1 mol−1) | G (a.u.) | ΔGa (Kcal mol−1) | |
|---|---|---|---|---|---|---|
| a Relative to reactants. | ||||||
| Cyclisation Mode A | ||||||
| 1 | −855.309925 | 118.9 | −855.366395 | |||
| 2-cis | −231.019909 | 73.3 | −231.054730 | |||
| TS1n-SS-c | −1086.307791 | 13.8 | 144.0 | −48.2 | −1086.376193 | 28.2 |
| TS1n-RR-c | −1086.307791 | 13.8 | 144.0 | −48.2 | −1086.376193 | 28.2 |
| TS1x-RS-c | −1086.301136 | 18.0 | 141.5 | −50.6 | −1086.368381 | 33.1 |
| TS1x-SR-c | −1086.301136 | 18.0 | 141.5 | −50.6 | −1086.368381 | 33.1 |
| TS2n-RR-c | −1086.306374 | 14.7 | 142.7 | −49.5 | −1086.374158 | 29.5 |
| TS2n-SS-c | −1086.306374 | 14.7 | 142.7 | −49.5 | −1086.374158 | 29.5 |
| TS2x-RS-c | −1086.300008 | 18.7 | 144.4 | −47.8 | −1086.368599 | 33.0 |
| TS2x-SR-c | −1086.300009 | 18.7 | 144.4 | −47.8 | −1086.368598 | 33.0 |
| CA1n-SS-c | −1086.360914 | −19.5 | 141.4 | −50.7 | −1086.428111 | −4.4 |
| CA1n-RR-c | −1086.360914 | −19.5 | 141.4 | −50.7 | −1086.428111 | −4.4 |
| CA1x-RS-c | −1086.360058 | −19.0 | 142.5 | −49.7 | −1086.427743 | −4.2 |
| CA1x-SR-c | −1086.360054 | −19.0 | 142.4 | −49.7 | −1086.427720 | −4.1 |
| CA2n-RR-c | −1086.352762 | −14.4 | 142.5 | −49.6 | −1086.420486 | 0.4 |
| CA2n-SS-c | −1086.352762 | −14.4 | 142.5 | −49.6 | −1086.420486 | 0.4 |
| CA2x-RS-c | −1086.353005 | −14.5 | 141.6 | −50.5 | −1086.420301 | 0.5 |
| CA2x-SR-c | −1086.353005 | −14.5 | 141.6 | −50.5 | −1086.420301 | 0.5 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Cyclisation Mode B | ||||||
| 1 | −855.309925 | 118.9 | −855.366395 | |||
| 2-trans | −231.024436 | 72.6 | −231.058921 | |||
| TS1n-SS-t | −1086.307338 | 17.0 | 145.4 | −46.0 | −1086.376416 | 30.7 |
| TS1n-RR-t | −1086.307339 | 17.0 | 145.4 | −46.0 | −1086.376420 | 30.7 |
| TS1x-RS-t | −1086.303120 | 19.6 | 145.8 | −45.6 | −1086.372417 | 33.2 |
| TS1x-SR-t | −1086.303120 | 19.6 | 145.7 | −45.7 | −1086.372359 | 33.2 |
| TS2n-RR-t | −1086.305426 | 18.2 | 145.3 | −46.2 | −1086.374444 | 31.9 |
| TS2n-SS-t | −1086.305427 | 18.2 | 145.3 | −46.2 | −1086.374446 | 31.9 |
| TS2x-RS-t | −1086.302206 | 20.2 | 144.5 | −46.9 | −1086.370860 | 34.2 |
| TS2x-SR-t | −1086.302206 | 20.2 | 144.5 | −46.9 | −1086.370861 | 34.2 |
| CA1n-SS-t | −1086.360931 | −16.7 | 142.6 | −48.8 | −1086.42870 | −2.1 |
| CA1n-RR-t | −1086.360931 | −16.7 | 142.6 | −48.8 | −1086.428705 | −2.1 |
| CA1x-RS-t | −1086.359664 | −15.9 | 144.7 | −46.8 | −1086.428399 | −1.9 |
| CA1x-SR-t | −1086.359665 | −15.9 | 144.7 | −46.8 | −1086.428402 | −1.9 |
| CA2n-RR-t | −1086.355063 | −13.0 | 141.1 | −50.3 | −1086.422120 | 2.0 |
| CA2n-SS-t | −1086.355062 | −13.0 | 141.1 | −50.3 | −1086.422114 | 2.0 |
| CA2x-RS-t | −1086.355060 | −13.0 | 142.6 | −48.9 | −1086.422801 | 1.6 |
| CA2x-SR-t | −1086.355060 | −13.0 | 142.6 | −48.9 | −1086.422801 | 1.6 |
The geometries of the eight TSs (four for CM-A and four for CM-B) prepared using CYLView,22 are given in Fig. 1. Obviously, for each couple of enantiomers, the bond lengths for the two forming sigma bonds are identical. The extent of bond formation along a reaction pathway is provided by the concept of bond order (BO).23 At the TSs associated to the meta-endo reaction channel via CM-A, the BO values of the two forming bonds are: 0.21 (O1–C5) and 0.48 (C3–C4) for TS1n-SS-c/TS1n-RR-c, 0.25 (O1–C5) and 0.48 (C3–C4) for TS1x-RS-c/TS1x-SR-c indicating that the C3–C4 sigma bond is more advanced than the O1–C5 sigma bond. By contrast, at the TSs associated to the ortho-endo reaction channel via CM-A, the BO values of the two forming bonds are: 0.60 (O1–C4) and 0.22 (C3–C5) for TS2n-SS-c/TS2n-RR-c, 0.57 (O1–C4) and 0.25 (C3–C5) for TS2x-RS-c/TS2x-SR-c indicating that the O1–C4 sigma bond is more advanced than C3–C5 sigma bond. On the other hand, it is well known that when a 13DC cycloaddition presents highly asynchronous TSs, diradical structures could in principle be involved. The stability test for equilibrium geometries of the transition state TS1n-SS-c of the asynchronous mechanism was performed using the “STABLE” Gaussian keyword. The output indicates that “The wave function is stable under the perturbations considered”. Consequently, we can conclude the inexistence of diradical TS structures.
 | ||
| Fig. 1 B3LYP/6-31G(d) geometries of the transition structures involved in the 13DC reaction between the α-aryl nitrone 1 and methacrolein 2 via CM-A and CM-B. Lengths are given in Angstroms. | ||
IRC calculations indicate that the 13DC cycloaddition of α-aryl nitrone 1 with methacrolein 2-cis follows a one-step mechanism and the eventuality of a stepwise mechanism is excluded. Indeed, the optimization of the last structure on the IRC curve in the forward direction gives a structure identical to that of the cycloadduct, indicating the absence of a stable reaction intermediate. Topological analysis of the ELF along the reaction pathway in 13DC and DA cycloaddition21b,24 can be also used as a valuable tool to understand the bonding changes along the reaction channel. Silvi25 proposed the topological approach of the chemical bonding which enables a position space partition of the electron density in terms of basins of attractor whose chemical significance is given by their location with respect to the nuclei. Indeed, there are two types of basins: core and valence. Core basins correspond to the inner atomic shell density while valence basin density is organized around and between the core basins. The latter are characterized by the number of core basin with which they have a boundary; this number is called the synaptic order. There are therefore monosynaptic basins corresponding to electron lone pairs or non-bonding regions, labeled V(A), while disynaptic basins to conventional two-center bonds, connect the core of two nuclei A and B and, thus, correspond to a bonding region between A and B and are labeled as V(A,B). These graphical representations of molecules in terms of localization domains are very helpful since they provide a direct access to the chemical understanding. In order to explain the bond formation in this 13DC reaction, a topological analysis of the ELF of some relevant points of the IRC curve (forward direction) of the most favorable meta-endo reaction channel via CM-A associated with the reaction between the α-aryl nitrone 1 and methacrolein 2-cis was performed. The ELF valence basins and their corresponding N populations of the relevant points TS1n-SS-c, P-I, P-II, P-III, P-IV and CA1n-SS-c are given in Table 3. The schematic representation of the mono- and disynaptic basins for the five considered points is given in Fig. 2 and the cartesian coordinates of the structures corresponding to P-I, P-II, P-III and P-IV are given in pages S37–S40 of the ESI.† Interesting conclusions can be drawn from the ELF analysis: (i) the presence of two monosynaptic basins, V(C3) and V(C4), integrating 0.63 e and 0.34 e, respectively at the TS1n-SS-c. (ii) A disynaptic basin V(C3,C4), N = 1.58 e is formed after the TS and it corresponds to the first new C3–C4 sigma bond (P-I). (iii) The presence of new V(C5) monosynaptic basins, integrating 0.26 e is observed at P-II. (iv) A disynaptic basin V(O1,C5), N = 0.92 e, is formed in a late stage (P-III). (v) The two disynaptic basins associated with two single bonds formed in this 13DC reaction have reached an electron density of 1.83 e (C1–C6) and 1.09 e (O1–C5) at the P-IV just before the formation of the cycloadduct CA1n-SS-c. Consequently, the studied 13DC reaction, involving asymmetric reagents, occurs via a one-step non-concerted mechanism.
| TS1n-SS-c | P-I | P-II | P-III | P-IV | CA1n-SS-c | ||
|---|---|---|---|---|---|---|---|
| d (Å) | C3–C4 | 2.02 | 1.70 | 1.61 | 1.57 | 1.56 | 1.55 |
| O1–C5 | 2.38 | 2.18 | 1.88 | 1.58 | 1.50 | 1.47 | |
| BO | C3–C4 | 0.48 | 0.81 | 0.90 | 0.95 | 0.96 | 0.97 |
| O1–C5 | 0.21 | 0.38 | 0.58 | 0.79 | 0.83 | 0.87 | |
| N (e) | V(C3,C4) | — | 1.58 | 1.73 | 1.80 | 1.83 | 1.85 |
| V(O1,C5) | — | — | — | 0.92 | 1.09 | 1.19 |
 | ||
| Fig. 2 Schematic representation of the mono- and disynaptic ELF basins for some relevant points on the IRC curve of the meta-endo channel via CM-A. The hydrogen atoms are omitted for clarity. | ||
Another important aspect of cycloaddition reactions is the analysis of the polarity of the process. The natural population analysis (NPA) is a suitable tool to evaluate the charge transfer (CT) at the TSs. The natural charges at the TSs were shared between the α-aryl nitrone 1 and methacrolein 2-cis. The calculations show that the electron flux takes place from 1 to 2-cis for all the eight TSs for meta-endo reaction channel via CM-A and the CT values are as follows: 0.07 e at TS1n-SS-c/TS1n-RR-c, 0.05 e at TS1x-RS-c/TS1x-SR-c, 0.11 e at TS2n-RR-c/TS2n-SS-c, 0.09 e at TS2x-RS-c/TS2x-SR-c. These negligible CTs point out to a low polar cycloaddition process. These findings were confirmed by the calculation of DFT-based reactivity indices of the isolated reagents. In Table 4, we reported the energies of the frontier molecular orbitals HOMO, εH, and LUMO, εL, and the global properties (electronic chemical potential, μ, chemical hardness, η, Parr's electrophilicity index, ω) of the α-aryl nitrone 1 and methacrolein 2-cis. The electrophilicity index of the α-aryl nitrone 1, ω = 1.70 eV, allows to classify this species as a strong electrophile within the electrophilicity scale.26 This value is slightly lower than that of methacrolein 2-cis, ω = 1.75 eV. The very low electrophilicity difference, Δω = 0.05 eV, between the two reactants puts in evidence the very low polar character of the 13DC reaction under study. In conclusion, for the favoured meta-endo reaction channel via CM-A, the relatively high activation energy in solution, 18.2 kcal mol−1, explains the experimental findings, indicating that the studied 13DC reaction occurs at room temperature. We note that 13DC reactions with activation barriers of about 5–8 kcal mol−1 are known to take place at low temperatures.
| εH | εL | μ | η | ω | |
|---|---|---|---|---|---|
| 1 | −0.2154 | −0.0624 | −0.1389 | 0.1530 | 1.70 |
| 2-cis | −0.2551 | −0.0611 | −0.1581 | 0.1941 | 1.75 |
Conclusion
In the present work, the mechanism, regio- and diastereoselectivity of the 13DC of the α-aryl nitrone 1 and methacrolein 2-cis/trans to yield the isoxazolidine cycloadduct have been studied at the B3LYP/6-31G(d) level of theory. The sixteen reaction channels associated to different cyclisation modes of the reagents have been thoroughly elaborated and analysed. DFT calculations performed both in gas phase and in DCM, show that the meta-endo reaction channel via CM-A, yielding the (3R,5R) and (3S,5S) endo cycloadducts, is favoured both kinetically and thermodynamically. These results are in good agreement with experimental findings. The IRC calculations combined to ELF analysis show that the studied 13DC reaction follows a one-step non-concerted mechanism. The relatively high activation energy for the favoured meta-endo reaction channel via CM-A explains the fact that this reaction takes place at room temperature and it is not favoured at low temperatures. The low CT at TS1n-SS-c and the low electrophilicity difference between the reagents 1 and 2-cis explain the low polarity of the studied 13DC reaction.Computational methods
All calculations were carried out with the Gaussian 09 suite of programs.27 DFT calculations were performed using Becke's three-parameter hybrid exchange functional in combination with the gradient corrected correlation functional of Lee et al. (B3LYP)28 together with the standard 6-31G(d) basis set.29 The optimizations were carried out using the Berny analytical gradient optimization method.30 The stationary points were characterized by frequency calculations in order to verify that TSs had one and only one imaginary frequency. The IRC path31 was traced in order to check the energy profiles connecting each TS to the two associated minima (reactants and cycloadducts) using the second order González–Schlegel integration method.32 Thermochemical properties (enthalpy, entropy, and Gibbs free energy) were calculated according to the standard equations of statistical thermodynamics33 at 298.15 K. For the thermochemical evaluation, theoretical calculations are performed with the Truhlar's MPW1B95 density functional,20 together with the standard 6-31G(d) basis set. The electronic structures of stationary points were analysed by the natural bond orbital (NBO) method34 and the topological analysis of the electron localization function (ELF), η(r).25,35 The ELF study was performed with the TopMod program.36 Solvent effects have been considered at the same level of theory by geometry single point of the gas-phase structures using a self-consistent reaction field (SCRF)37 based on the continuum solvation model (SMD) of the Truhlar's group.38 Since this cycloaddition is carried out in DCM, we have selected its dielectric constant at 298.0 K, ε = 8.93. The global electrophilicity index, ω,39 which measures the stabilization energy when the system acquires an additional electronic charge ΔN from the environment, has been given by the following simple expression, ω = μ2/2η,39 in terms of the electronic chemical potential μ and the chemical hardness η. Both quantities may be approached in terms of the one electron energies of the frontier molecular orbital HOMO, εH, and LUMO, εL, as μ ≈ (εH + εL)/2 and η ≈ εL − εH, respectively.40 The reactivity indices were computed from the B3LYP/6-31G(d) HOMO and LUMO energies at the ground state of the reactants.Acknowledgements
Financial support from the Ministry of Higher Education and Scientific Research of the Algerian Government (project CNEPRU E02020110003) is gratefully acknowledged.References
- A. Padwa and W. H. Pearson, The Chemistry of Heterocyclic Compounds, Synthetic Applications of 1,3-Dipolar Cycloaddition Chemistry Toward Heterocycles and Natural Products, John Wiley & Sons, 2002 Search PubMed.
- (a) P. Merino, T. Tejero, F. J. Unzurrunzaga, S. Franco, U. Chiacchio, M. G. Saita, D. Iannazzo, A. Piperno and G. Romeo, Tetrahedron: Asymmetry, 2005, 16, 3865 CrossRef CAS PubMed; (b) V. Mannucci, F. M. Cordero, A. Piperno, G. Romeo and A. Brandi, Tetrahedron: Asymmetry, 2008, 19, 1204 CrossRef CAS PubMed; (c) R. Romeo, S. V. Giofre, B. Macchi, E. Balestrieri, A. Mastino, P. Merino, C. Carnovale, G. Romeo and U. Chiacchio, ChemMedChem, 2012, 7, 565 CrossRef CAS PubMed.
- (a) T. Kiguchi, M. Shirakawa, R. Honda, I. Ninomiya and T. Naito, Tetrahedron, 1998, 54, 15589 CrossRef CAS; (b) F. Cardona, G. Moreno, F. Guarna, P. Vogel, C. Schuetz, P. Merino and A. Goti, J. Org. Chem., 2005, 70, 6552 CrossRef CAS PubMed; (c) I. Delso, T. Tejero, A. Goti and P. Merino, Tetrahedron, 2010, 66, 1220 CrossRef CAS PubMed; (d) J. Peng, D. Jiang, W. Lin and Y. Chen, Org. Biomol. Chem., 2007, 5, 1391 RSC; (e) M. M. Andrade, M. T. Barros and R. C. Pinto, Clean and Sustainable Methodologies for the Synthesis of Isoxazolidines, in Heterocyclic Targets in Advanced Organic Synthesis,ed. M. C. Carreiras and J. Marco-Contelles, Research Signpost, Trivandrum, 2002, vol. 51, p. 67 Search PubMed.
- T. Saito, T. Yamada, S. Miyazaki and T. Otani, Tetrahedron Lett., 2004, 45, 9585 CrossRef CAS PubMed.
- K. V. Gothelf and K. V. Jørgensen, Chem. Rev., 1998, 98, 863 CrossRef CAS PubMed.
- R. B. Woodward and R. Hoffmann, The Conservation of Orbital Symmetry, Verlag Chemie, Weinheim, 1970 Search PubMed.
- (a) D. Carmona, M. P. Lamata, F. Viguri, R. Rodriguez, L. A. Oro, F. J. Lahoz, A. I. Balana, T. Tejero and P. Merino, J. Am. Chem. Soc., 2005, 127, 13386 CrossRef CAS PubMed; (b) D. Carmona, M. P. Lamata, F. Viguri, J. Ferrer, N. Garcia, F. J. Lahoz, M. L. Martin and L. A. Oro, Eur. J. Inorg. Chem., 2006, 3155 CrossRef CAS; (c) D. Carmona, M. P. Lamata, F. Viguri, R. Rodriguez, F. J. Lahoz and L. A. Oro, Chem.–Eur. J., 2007, 13, 9746 CrossRef CAS PubMed; (d) T. Hashimoto, M. Omote, Y. Hato, T. Kano and K. Maruoka, Chem.–Asian. J., 2008, 3, 407 CrossRef CAS PubMed; (e) D. Carmona, M. P. Lamata, F. Viguri, R. Rodriguez, F. J. Lahoz, M. J. Fabra and L. A. Oro, Tetrahedron: Asymmetry, 2009, 20, 1197 CrossRef CAS PubMed; (f) M. Lemay, J. Trant and W. W. Ogilvie, Tetrahedron, 2007, 63, 11644 CrossRef CAS PubMed; (g) S. S. Chow, M. Nevalainen, C. A. Evans and C. W. Johannes, Tetrahedron Lett., 2007, 48, 277 CrossRef CAS PubMed.
- (a) M. Frederickson, Tetrahedron, 1997, 5, 403 CrossRef; (b) S. Karlsson and H. E. Högberg, Org. Prep. Proced. Int., 2001, 33, 103 CrossRef CAS; (c) T. B. Nguyen, A. Martel, C. Gaulon, R. Dhal and G. Dujardin, Org. Prep. Proced. Int., 2010, 42, 387 CrossRef CAS; (d) H. Pellissier, Tetrahedron, 2012, 68, 2197 CrossRef CAS PubMed; (e) Y. Xing and N. Wang, Coord. Chem. Rev., 2012, 256, 938 CrossRef CAS PubMed; (f) W. Wang and K. S. Rein, Tetrahedron Lett., 2013, 54, 1866 CrossRef CAS PubMed.
- V. Bãdoiu and E. P. Kündig, Org. Biomol. Chem., 2012, 10, 114 Search PubMed.
- (a) K. V. Gothelf, R. G. Hazell and K. A. Jørgensen, J. Org. Chem., 1996, 61, 346 CrossRef CAS; (b) P. Merino, J. Revuelta, T. Tejero, U. Chiacchio, A. Rescifinab and G. Romeo, Tetrahedron, 2003, 59, 3581 CrossRef CAS; (c) P. Astolfi, P. Bruni, L. Greci, P. Stipa, L. Righi and C. Rizzoli, Eur. J. Org. Chem., 2003, 2626 CrossRef CAS; (d) C. T. Wong, Tetrahedron, 2013, 69, 6634 CrossRef CAS PubMed.
- N. Acharjee, T. K. Das, A. Banerji, M. Banerjee and T. Prangé, J. Phys. Org. Chem., 2010, 23, 1187 CrossRef CAS.
- M. Flores, P. García, N. M. Garrido, C. T. Nieto, P. Basabe, I. S. Marcos, F. Sanz-González, J. M. Goodman and D. Díez, Tetrahedron: Asymmetry, 2012, 23, 76 CrossRef CAS PubMed.
- M. Śnieżek, S. Stecko, I. Panfil, B. Furman, Z. Urbańczyk-Lipkowska and M. Chmielewski, Tetrahedron: Asymmetry, 2013, 24, 89 CrossRef PubMed.
- L. R. Domingo, W. Benchouk and S. M. Mekelleche, Tetrahedron, 2007, 63, 4464 CrossRef CAS PubMed.
- W. Benchouk and S. M. Mekelleche, J. Mol. Struct.: THEOCHEM, 2008, 852, 46 CrossRef CAS PubMed.
- J. Liu, S. Niwayama, Y. You and K. N. Houk, J. Org. Chem., 1998, 63, 1064 CrossRef CAS.
- C. Barba, D. Carmona, J. I. García, M. P. Lamata, J. A. Mayoral, L. Salvatella and F. Viguri, J. Org. Chem., 2006, 71, 9831 CrossRef CAS PubMed.
- C. Reichardt and T. Welton, Solvents and Solvent Effects, in Organic Chemistry, 4th edn, Wiley-VCH, 2011 Search PubMed.
- W. Benchouk, S. M. Mekelleche, B. Silvi, M. J. Aurell and L. R. Domingo, J. Phys. Org. Chem., 2011, 24, 611 CrossRef CAS.
- Y. Zhao and D. G. Truhlar, J. Phys. Chem. A, 2004, 108, 6908 CrossRef CAS.
- (a) A. Shiroudi and E. Zahedi, Prog. React. Kinet. Mech., 2012, 37, 277 CrossRef CAS; (b) L. R. Domingo, P. Pérez and J. A. Sáez, RSC Adv., 2013, 3, 7520 RSC.
- C. Y. Legault, CYLView, 1.0b, Université de Sherbrooke, Canada, 2009 http://www.cylview.org.
- K. B. Wiberg, Tetrahedron, 1968, 24, 1083 CrossRef CAS.
- (a) S. Berski, J. Andres, B. Silvi and L. R. Domingo, J. Phys. Chem. A, 2006, 110, 13939 CrossRef CAS PubMed; (b) V. Polo, J. Andres, S. Berski, L. R. Domingo and B. Silvi, J. Phys. Chem. A, 2008, 112, 7128 CrossRef CAS PubMed; (c) W. Benchouk, S. M. Mekelleche, M. J. Aurell and L. R. Domingo, Tetrahedron, 2009, 65, 4644 CrossRef CAS PubMed; (d) L. R. Domingo, E. Chamorro and P. Pérez, Org. Biomol. Chem., 2010, 8, 5495 RSC; (e) L. R. Domingo, P. Pérez and J. A. Sáez, Org. Biomol. Chem., 2012, 10, 3841 RSC; (f) L. R. Domingo, P. Pérez and J. A. Sáez, Tetrahedron, 2013, 69, 107 CrossRef CAS PubMed; (g) L. Rhyman, P. Ramasami, J. A. Joule, J. A. Sáez and L. R. Domingo, RSC Adv., 2013, 3, 447 RSC; (h) L. R. Domingo, P. Pérez and J. A. Sáez, Org. Biomol. Chem., 2013, 11, 4350 RSC; (i) L. R. Domingo, J. A. Sáez, J. A. Joule, L. Rhyman and P. Ramasami, J. Org. Chem., 2013, 78, 1621 CrossRef CAS PubMed.
- B. Silvi, J. Mol. Struct., 2002, 614, 3 CrossRef CAS.
- L. R. Domingo, M. J. Aurell, P. Perez and R. Contreras, Tetrahedron, 2002, 58, 4417 CrossRef CAS.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, V. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision A.1, Gaussian, Inc., Wallingford CT, 2009 Search PubMed.
- (a) A. D. Becke, J. Chem. Phys., 1993, 98, 5648 CrossRef CAS PubMed; (b) C. Lee, W. Yang and R. G. Parr, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 37, 785 CrossRef CAS.
- W. J. Hehre, L. Radom, Pv. R. Schleyer and J. A. Pople, Ab Initio Molecular Orbital Theory, Wiley, 1986 Search PubMed.
- (a) H. B. Schlegel, J. Comput. Chem., 1982, 3, 214 CrossRef CAS; (b) H. B. Schlegel, Geometry Optimization on Potential Energy Surface, in Modern Electronic Structure Theory, ed. D. R. Yarkony, World Scientific Publishing, 1994 Search PubMed.
- K. J. Fukui, Phys. Chem., 1970, 74, 4161 CrossRef CAS.
- (a) C. González and H. B. Schlegel, J. Phys. Chem., 1990, 94, 5523 CrossRef; (b) C. González and H. B. Schlegel, J. Chem. Phys., 1991, 95, 5853 CrossRef PubMed.
- C. J. Cramer, Essentials of Computational Chemistry Theories and Models, Wiley Hoboken, 2004 Search PubMed.
- (a) A. E. Reed, L. A. Curtiss and F. Weinhold, Chem. Rev., 1988, 88, 899 CrossRef CAS; (b) A. E. Reed, R. B. Weinstock and F. Weinhold, J. Chem. Phys., 1985, 83, 735 CrossRef CAS PubMed.
- (a) A. Savin, A. D. Becke, J. Flad, R. Nesper, H. Preuss and H. G. Vonschnering, Angew. Chem., Int. Ed. Engl., 1991, 30, 409 CrossRef; (b) B. Silvi and A. Savin, Nature, 1994, 371, 683 CrossRef CAS; (c) A. Savin, B. Silvi and F. Colonna, Can. J. Chem., 1996, 74, 1088 CrossRef CAS; (d) A. Savin, R. Nesper, S. Wengert and T. F. Fassler, Angew. Chem., Int. Ed., 1997, 36, 1808 CrossRef CAS.
- S. Noury, X. Krokidis, F. Fuster and B. Silvi, Comput. Chem., 1999, 23, 597 CrossRef CAS.
- (a) J. Tomasi and M. Persico, Chem. Rev., 1994, 94, 2027 CrossRef CAS; (b) B. Y. Simkin and I. Sheikhet, Quantum Chemical and Statistical Theory of Solutions-A Computational Approach, Ellis Horwood, 1995 Search PubMed.
- (a) A. V. Marenich, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2009, 113, 6378 CrossRef CAS PubMed; (b) R. F. Ribeiro, A. V. Marenich, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2011, 115, 14556 CrossRef CAS PubMed.
- R. G. Parr, L. von Szentpaly and S. J. Liu, Am. Chem. Soc., 1999, 121, 1922 CrossRef CAS.
- (a) R. G. Parr and R. G. Pearson, J. Am. Chem. Soc., 1983, 105, 7512 CrossRef CAS; (b) R. G. Parr and W. Yang, Density Functional Theory of Atoms and Molecules, Oxford University Press, 1989 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra17285j |
| This journal is © The Royal Society of Chemistry 2015 |