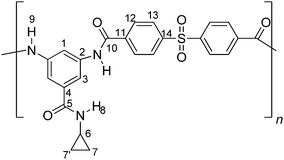
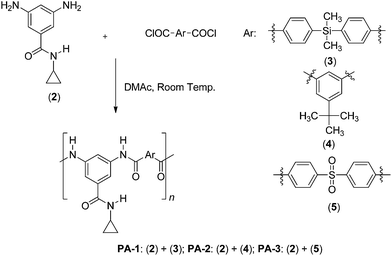
Synthesis and characterization of aromatic poly(amides) based on 3,5-diamino-N-cyclopropylbenzamide
A. Tundidor-Camba*a,
C. A. Terrazaa,
L. H. Taglea,
D. Colla,
I. Ojedaa and
M. Pinob
aOrganic Chemistry Department, Faculty of Chemistry, Pontificia Universidad Católica de Chile, Box 306, Post 22, Santiago, Chile. E-mail: atundido@uc.cl; Tel: +56 223541199
bPhysicochemical Department, Faculty of Chemistry, Pontificia Universidad Católica de Chile, Box 306, Post 22, Santiago, Chile
First published on 23rd February 2015
Abstract
Three aromatic poly(amides) (PAs) were prepared, one of them containing a dimethyldiphenylsilane unit from a new aromatic diamine monomer with a bulky pendant polar group. All PAs were obtained in high yield and the inherent viscosities were in the range of 0.30 and 0.47 dL g−1. The obtained poly(amides) showed excellent solubility in a variety of aprotic polar organic solvents. PAs evidence thermal stability with thermal decomposition temperature (TDT10%) between 300–371 °C and the glass transition temperatures (Tg) values were high (between 215 and 250 °C). PAs containing silicon atom in the main chain captures the highest moles number of water per mole of repeating unit. Contact angles were also tested in order to know the hydrophilicity of the polymer films.
1. Introduction
Thermally stable polymers are important because they can replace metals and ceramics in several fields of science. Specifically, aromatic poly(amides) exhibit excellent mechanical, electrical and chemical properties, making them very interesting for their study as high performance materials.1–3 However, their industrial processing is affected by their remarkable intractability caused by the strong molecular interactions through hydrogen bonding of amide groups.4With the aim to reduce the intermolecular bonding energy, many structural modifications have been made in poly(amides), such as the incorporation of flexible units, 1,3-substituted monomers, bulky pendant groups or heteroatoms in the main chain.5–8 These chemical modifications provide interchain distance, and consequently, higher free volume and solubility, without affecting the thermal properties.9,10 Adamantylcarbamoyl pendant group or other cyclic side group such as fluorene or phenylcyclohexane were effective to improve the solubility of the polymers derivatives.11–13
In other works, and also in our previous works, it was found that the introduction of diphenylsilane units in the polymer backbone improves the solubility and maintains the thermal properties of the polymeric materials.7,14,15 Silicon atom has a lower electronegativity than carbon atom causing a small dipole moment inside the chain, which increases the polarity of the polymeric chain and therefore, its solubility in specific solvents.
Rigid aromatic poly(amides) with other polar groups into the main or side chain are interesting in the reverse osmosis field because it increases the hydrophilicity of the membrane, allowing better diffusion of water through the polymeric matrix.16 Structural rigidity of this sort of polymers is a critical property for the adequate functioning of the membrane.17 Such high stiffness degree is usually associated with high Tg values.
This work reports the synthesis and characterization of a novel aromatic diamine with a cyclopropylcarbamoyl pendant group and three aromatic poly(amides) derived from it. One of them contains a dimethyldiphenylsilane unit in the repeating unit. Solubility, water uptake, contact angle, inherent viscosity and thermal properties of the polymers are also investigated and discussed.
2. Experimental
2.1. Materials
Anhydrous N,N-dimethylacetamide (DMAc), 3,5-dinitrobenzoyl chloride, cyclopropylamine, Pd/C (10% w/w), 4,4′-sulfonyldibenzoic acid and tert-butylisophthalic acid were obtained from Aldrich Chemical (Milwaukee, WI). Bis(4-carboxyphenyl)dimethylsilane was synthesized following a procedure already reported by our research group.18 Diethyl ether and n-hexane were refluxed over metallic sodium under nitrogen atmosphere. All other reagents and solvents were purchased commercially as analytical-grade and used without further purification.2.2. Instrumentation and measurements
IR-TF spectra (KBr pellets) were recorded on a Perkin–Elmer (Fremont CA) 1310 spectrophotometer over the range of 4000–450 cm−1. Melting points (uncorrected) were obtained on a SMP3 Stuart Scientific melting point apparatus. 1H, 13C and 29Si NMR spectra were carried out on a 400 MHz instrument (Bruker AC-200) using DMSO-d6 as solvent and TMS as internal standard. Viscosimetric measurements were made in a Desreux–Bischof type dilution viscosimeter at 25 °C (c = 0.5 g dL−1). Glass transition temperature (Tg) values were obtained with a Mettler-Toledo (Greifensee, Switzerland) DSC 821 calorimetric system (10 °C min−1 under N2 flow). Thermogravimetric analyses were carried out in a Mettler (Switzerland) TA-3000 calorimetric system equipped with a TC-10A processor, and a TG-50 thermobalance with a Mettler MT5 microbalance (temperature range between 25 °C and 900 °C at 10 °C min−1 under N2 flow). Elemental analyses were made on a Fisons EA 1108-CHNS-O equipment. A dataphysics OCA 20 device with a conventional goniometer and high performance video camera, controlled by SCA20 software, was used to measure the optical contact angle. Water sorption measurements were determined gravimetrically at room temperature. Powder polymer samples (0.3 g), previously dried at 120 °C for 24 h, were placed in a closed desiccator containing a saturated aqueous solution of NaNO2, which provided a relative humidity of 65%. The samples were frequently weighed during 15 h and then allowed to humidify for 5 more days until constant weight.2.3. Monomer synthesis and characterization
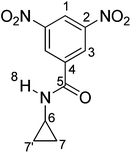
3,5-dinitro-N-cyclopropylbenzamide (1). To a two-necked round-bottomed flask equipped with magnetic stirrer, 3 mL (43.37 mmol) of cyclopropylamine in 125 mL of dry diethyl ether were added. The mixture was stirred under a gentle nitrogen stream. Small amounts (500 mg) of 3,5-dinitrobenzoyl chloride in solid state were added every 5 minutes, until a total of 5 g (21.68 mmol). Suspension was maintained with stirring at room temperature for 30 minutes and then filtered. White precipitate was washed three times with 50 mL of HCl/water mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) and then with water until neutral pH. Finally, the white solid was dried in a vacuum oven. Crude product was recrystallized from dichloromethane to afford 5 g of white needles.
1 v/v) and then with water until neutral pH. Finally, the white solid was dried in a vacuum oven. Crude product was recrystallized from dichloromethane to afford 5 g of white needles.Yield: 92%. M.p.: 158–159 °C. IR (KBr, ν, cm−1): 3289 (N–H); 3099, 3086 (C–H arom.); 2871 (C–H aliph.); 1644 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); 1536, 1343 (NO2); 1457, 1422 (C
O); 1536, 1343 (NO2); 1457, 1422 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C); 730 (1,3,5-arom. subs.). 1H RMN (DMSO-d6, δ, ppm): 0.65 (m, 2H(7,7′)); 0.77 (m, 2H(7,7′)); 2.93 (m, 1H(6)); 8.94 (s, 1H(1)); 9.01 (s, 2H(3)); 9.12 (s, 1H(8)). 13C RMN (DMSO-d6, δ, ppm), 5.97 (C7,7′); 23.76 (C6); 121.05 (C1); 127.73 (C3); 137.21 (C4); 148.47 (C2); 163.46 (C5). Elem. Anal. Calcd. for C10H9N3O5; (251.05): C, 47.81%; H, 3.61%, N, 16.73%. Found: C, 47.18%; H, 3.35%, N, 16.66%.
C); 730 (1,3,5-arom. subs.). 1H RMN (DMSO-d6, δ, ppm): 0.65 (m, 2H(7,7′)); 0.77 (m, 2H(7,7′)); 2.93 (m, 1H(6)); 8.94 (s, 1H(1)); 9.01 (s, 2H(3)); 9.12 (s, 1H(8)). 13C RMN (DMSO-d6, δ, ppm), 5.97 (C7,7′); 23.76 (C6); 121.05 (C1); 127.73 (C3); 137.21 (C4); 148.47 (C2); 163.46 (C5). Elem. Anal. Calcd. for C10H9N3O5; (251.05): C, 47.81%; H, 3.61%, N, 16.73%. Found: C, 47.18%; H, 3.35%, N, 16.66%.
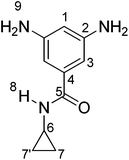
3,5-diamine-N-cyclopropylbenzamide (2). A mixture of 5 g (20 mmol) of 3,5-dinitro-N-cyclopropylbenzamide (1), 10% palladium on activated carbon (0.250 g) and 100 mL of absolute ethanol were placed under an atmosphere of hydrogen (30 psi) and allowed to react with stirring for 10 minutes. When the pressure of the system decreased, the reaction bottle was loaded with 30 psi of additional of hydrogen, until the pressure of hydrogen was kept constant. Catalyst was removed by filtration through a Celite plug, and the solvent removed under reduced pressure to afford 3.5 g of a yellow solid. The crude product was recrystallized from ethanol to afford 3.2 g of yellow crystals.
Yield: 92%. M.p.: 201–202 °C. IR (KBr, ν, cm−1): 3460, 3378, 3341 (NH2); 3236 (N–H); 3080 (C–H arom.); 2998 (C–H aliph.); 1644 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1593 (N–H bending); 1527, 1456 (C
O), 1593 (N–H bending); 1527, 1456 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C); 1283 (C–N bending); 746 (1,3,5-arom. subs.). 1H RMN (DMSO-d6, δ, ppm): 0.51 (m, 2H(7,7′)); 0.62 (m, 2H(7,7′)); 2.77 (m, 1H(6)); 4.81 (s, 4H(9)); 5.93 (s, 1H(1)); 6.18 (s, 2H(3)); 8.00 (s, 1H(8)). 13C RMN (DMSO-d6, δ, ppm), 6.07 (C7,7′); 23.28 (C6); 102.43 (C1); 102.54 (C3); 136.74 (C4); 149.24 (C2); 169.63 (C5). Elem. Anal. Calcd. for C10H13N3O; (191.11): C, 62.81%; H, 6.85%, N, 21.97%. Found: C, 62.16%; H, 6.06%, N, 21.88%.
C); 1283 (C–N bending); 746 (1,3,5-arom. subs.). 1H RMN (DMSO-d6, δ, ppm): 0.51 (m, 2H(7,7′)); 0.62 (m, 2H(7,7′)); 2.77 (m, 1H(6)); 4.81 (s, 4H(9)); 5.93 (s, 1H(1)); 6.18 (s, 2H(3)); 8.00 (s, 1H(8)). 13C RMN (DMSO-d6, δ, ppm), 6.07 (C7,7′); 23.28 (C6); 102.43 (C1); 102.54 (C3); 136.74 (C4); 149.24 (C2); 169.63 (C5). Elem. Anal. Calcd. for C10H13N3O; (191.11): C, 62.81%; H, 6.85%, N, 21.97%. Found: C, 62.16%; H, 6.06%, N, 21.88%.
Aromatic acid dichlorides (3, 4 and 5, Scheme 2). The corresponding dicarboxylic acid (10.0 mmol), 30 mL of thionyl chloride and a few drops of DMF were reacted in a round bottomed flask at 90 °C for 3 h. Thionyl chloride excess was removed from the solution by simple distillation. Crude 3 was washed with dry n-hexane and then recrystallized from n-hexane to afford 2.8 g of crystalline solid. Crude 4 was dissolved in dry n-hexane and then cooled overnight at −30 °C. The solid was filtered and washed with cold n-hexane to afford 2.0 g of crystalline solid. Crude 5 was washed with dry n-hexane and then recrystallized from n-hexane/toluene mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 vol/vol) to afford 2.7 g of white fluffy solid.
1 vol/vol) to afford 2.7 g of white fluffy solid.
4,4'-(dimethylsilanediyl)dibenzoyl chloride (3). Yield: 83%. M.p.: 78–80 °C. IR (KBr, ν, cm−1): 3030 (C–H arom.); 2980 (C–H aliph.); 1774 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1589, 1492 (C
O), 1589, 1492 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C); 1413, 1106 (Si–C arom.); 1375 (Si–C aliph); 832 (arom. p-subst.).
C); 1413, 1106 (Si–C arom.); 1375 (Si–C aliph); 832 (arom. p-subst.).
5-(tert-butyl)isophthaloyl dichloride (4). Yield: 77%. M.p.: 44–45 °C. IR (KBr, ν, cm−1): 3087 (C–H arom.); 2967, 2905 (C–H aliph.); 1757 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1587, 1473 (C
O), 1587, 1473 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C); 1396, 1367 (–C(CH3)3 bending); 752 (1,3,5-arom.).
C); 1396, 1367 (–C(CH3)3 bending); 752 (1,3,5-arom.).
4,4'-sulfonyldibenzoyl chloride (5). Yield: 78%. M.p.: 159–160 °C. IR (KBr, ν, cm−1): 3093, 3041 (C–H arom.); 1782, 1745 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1590, 1575 (C
O), 1590, 1575 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C); 1328, 1154 (S
C); 1328, 1154 (S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bending); 881 (arom. p-subst.).
O bending); 881 (arom. p-subst.).
2.4. Polymer synthesis and characterization
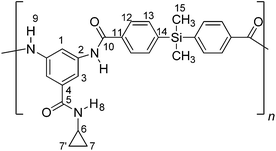
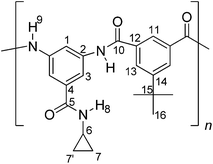
A typical polymerization procedure for the synthesis of poly(amides) was as follows. To a three-necked round-bottomed flask equipped with mechanical stirrer, was added 1.18 mmol of 3,5-diamino-N-cyclopropylbenzamide (2) and 1.5 mL of dry DMAc. When the solution was completed, it was cooled with an ice-water bath and 1.18 mmol of the corresponding acid dichloride (3, 4 or 5) and 1.0 mL of dry DMAc were added. The ice-water bath was removed when acid dichloride was entirely dissolved. Then, the mixture was stirred at room temperature during 6 hours. The resulting mixture was poured into 300 mL of water with stirring. White solid was filtered, washed thoroughly with water and dried at 100 °C during 12 hours. Thus, PA-1 was obtained from acid dichloride 3, PA-2 from acid dichloride 4 and PA-3 using acid dichloride 5.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); 1555, 1538 (C
O); 1555, 1538 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C); 1415, 1093 (Si–C arom.); 1376 (Si–C aliph); 814 (arom. p-subs.); 746 (1,3,5-arom. subs.). 1H NMR (DMSO-d6, δ, ppm), 0.49–0.60 (m, 10H(7,7′,15)); 2.77 (m, 1H(6)); 7.62 (d, J = 7.5 Hz, 4H(13)); 7.80 (s, 2H(3)); 7.89 (d, J = 7.6 Hz, 4H(12)); 8.36 (s, 1H(1)); 8.37 (s, 1H(8)); 10.36 (s, 2H(9)). 13C NMR (DMSO-d6, δ, ppm), 167.3 (C10); 165.1 (C5); 141.3 (C2); 138.6 (C4); 135.2 (C14); 134.9 (C11); 133.4 (C13); 126.4 (C12); 114.9 (C3); 114.8 (C1); 22.6 (C6); 5.2 (C7,7′); 3.4 (C15). 29Si NMR (DMSO-d6, δ, ppm), 8.47. Elem. Anal. Calcd. for [C26H25N3O3Si]n (455.58)n, C, 68.55%; H, 5.53%; N, 9.22%. Found: C, 68.36%; H, 5.43%; N, 9.12%.
C); 1415, 1093 (Si–C arom.); 1376 (Si–C aliph); 814 (arom. p-subs.); 746 (1,3,5-arom. subs.). 1H NMR (DMSO-d6, δ, ppm), 0.49–0.60 (m, 10H(7,7′,15)); 2.77 (m, 1H(6)); 7.62 (d, J = 7.5 Hz, 4H(13)); 7.80 (s, 2H(3)); 7.89 (d, J = 7.6 Hz, 4H(12)); 8.36 (s, 1H(1)); 8.37 (s, 1H(8)); 10.36 (s, 2H(9)). 13C NMR (DMSO-d6, δ, ppm), 167.3 (C10); 165.1 (C5); 141.3 (C2); 138.6 (C4); 135.2 (C14); 134.9 (C11); 133.4 (C13); 126.4 (C12); 114.9 (C3); 114.8 (C1); 22.6 (C6); 5.2 (C7,7′); 3.4 (C15). 29Si NMR (DMSO-d6, δ, ppm), 8.47. Elem. Anal. Calcd. for [C26H25N3O3Si]n (455.58)n, C, 68.55%; H, 5.53%; N, 9.22%. Found: C, 68.36%; H, 5.43%; N, 9.12%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); 1598, 1543 (C
O); 1598, 1543 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C); 739 (1,3,5-arom. subs.). 1H NMR (DMSO-d6, δ, ppm), 0.46 (m, 2H(7,7′)); 0.56 (m, 2H(7,7′)); 1.29 (s, 9H(16)); 2.74 (m, 1H(6)); 7.80 (s, 2H(3)); 8.07 (s, 2H(13)); 8.36 (s, 1H(11)); 8.37 (s, 1H(1)); 8.41 (s, 1H(8)); 10.52 (s, 2H(9)). 13C NMR (DMSO-d6, δ, ppm), 168.2 (C10); 165.8 (C5); 151.8 (C14); 139.5 (C2); 136.1 (C4); 135.1 (C12); 128.1 (C13); 125.2 (C11); 116.4 (C3); 116.2 (C1); 35.4 (C15); 31.4 (C16); 23.6 (C6); 6.1 (C7,7′). Elem. Anal. Calcd. for [C22H23N3O3]n (377.44)n, C, 70.01%; H, 6.14%; N, 11.13%. Found: C, 69.79%; H, 6.09%; N, 11.01%.
C); 739 (1,3,5-arom. subs.). 1H NMR (DMSO-d6, δ, ppm), 0.46 (m, 2H(7,7′)); 0.56 (m, 2H(7,7′)); 1.29 (s, 9H(16)); 2.74 (m, 1H(6)); 7.80 (s, 2H(3)); 8.07 (s, 2H(13)); 8.36 (s, 1H(11)); 8.37 (s, 1H(1)); 8.41 (s, 1H(8)); 10.52 (s, 2H(9)). 13C NMR (DMSO-d6, δ, ppm), 168.2 (C10); 165.8 (C5); 151.8 (C14); 139.5 (C2); 136.1 (C4); 135.1 (C12); 128.1 (C13); 125.2 (C11); 116.4 (C3); 116.2 (C1); 35.4 (C15); 31.4 (C16); 23.6 (C6); 6.1 (C7,7′). Elem. Anal. Calcd. for [C22H23N3O3]n (377.44)n, C, 70.01%; H, 6.14%; N, 11.13%. Found: C, 69.79%; H, 6.09%; N, 11.01%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) 1560, 1543 (C
O) 1560, 1543 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C); 855 (arom. p-subs.); 747 (1,3,5-arom. subs.). 1H NMR (DMSO-d6, δ, ppm), 0.58 (s, 2H(7,7′)); 0.68 (s, 2H(7,7′)); 2.87 (s, 1H(6)); 7.90 (s, 2H(3)); 8.20 (s, 8H(12,13)); 8.45 (s, 1H(1)); 8.50 (s, 1H(8)); 10.71 (s, 2H(9)). 13C NMR (DMSO-d6, δ, ppm), 168.1 (C10); 164.8 (C5); 143.4 (C14); 140.0 (C2); 139.3 (C4); 136.3 (C11); 129.6 (C13); 128.2 (C12); 116.0 (C3); 115.7 (C1); 23.6 (C6); 6.1 (C7,7′). Elem. Anal. Calcd. for [C24H19N3O5S]n (461.49)n, C, 62.46%; H, 4.15%; N, 9.11%. Found: C, 62.16%; H, 4.11%; N, 9.03%.
C); 855 (arom. p-subs.); 747 (1,3,5-arom. subs.). 1H NMR (DMSO-d6, δ, ppm), 0.58 (s, 2H(7,7′)); 0.68 (s, 2H(7,7′)); 2.87 (s, 1H(6)); 7.90 (s, 2H(3)); 8.20 (s, 8H(12,13)); 8.45 (s, 1H(1)); 8.50 (s, 1H(8)); 10.71 (s, 2H(9)). 13C NMR (DMSO-d6, δ, ppm), 168.1 (C10); 164.8 (C5); 143.4 (C14); 140.0 (C2); 139.3 (C4); 136.3 (C11); 129.6 (C13); 128.2 (C12); 116.0 (C3); 115.7 (C1); 23.6 (C6); 6.1 (C7,7′). Elem. Anal. Calcd. for [C24H19N3O5S]n (461.49)n, C, 62.46%; H, 4.15%; N, 9.11%. Found: C, 62.16%; H, 4.11%; N, 9.03%.3. Results and discussion
3.1. Monomer synthesis and characterization
A new diamine, 3,5-diamino-N-cyclopropylbenzamide, was synthesized by reacting 3,5-dinitrobenzoyl chloride with cyclopropylamine in anhydrous diethyl ether, followed by a reduction reaction using hydrogen gas, Pd/C as catalyst and absolute ethanol as solvent (Scheme 1).Dinitro compound 1 was insoluble in the medium reaction (anhydrous diethyl ether), favoring the isolation process. The next step (reduction reaction) was conducted in a dark media to prevent color change due to photodegradation of the diamine 2 (this diamine derivative is sensitive to light, changing from yellow to red color).
The FT-IR spectra in Fig. 1 confirmed the presence of both compounds. The bands at 3289 cm−1 and 1644 cm−1 correspond to stretching of N–H and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups, respectively, while those at 1536 and 1343 cm−1 were assigned to the vibration of the nitro group. Disappearance of these two signals and the appearance of sharp bands at 3460 and 3378 cm−1, corresponding to the stretching of the amino group, confirmed the formation of the monomer.
O groups, respectively, while those at 1536 and 1343 cm−1 were assigned to the vibration of the nitro group. Disappearance of these two signals and the appearance of sharp bands at 3460 and 3378 cm−1, corresponding to the stretching of the amino group, confirmed the formation of the monomer.
The 1H NMR spectrum in Fig. 2 also confirmed the structure of the diamine. The peak at 8.00 ppm was assigned to amidic hydrogen. The singlet at 4.81 ppm, integrating for 4 nuclei corresponds to the hydrogens of the amino groups. Also, a clear pattern of cyclopropyl fragment was possible to observe. The multiplet at 2.77 ppm was assigned to hydrogen H6, while one of the hydrogens H7 appears at 0.62 ppm and the other at 0.51 ppm. The same happened for hydrogens H7′. The difference in chemical shift of the hydrogens H7 and H7′ is due to the axial and equatorial positions of both in the cyclopropyl ring. Meanwhile, the 13C NMR spectrum showed the seven expected carbon signals. The peak at 169.6 ppm corresponds to the carbonyl carbon of the amide group. The carbons C1 and C3 showed similar chemical shifts due to the mesomeric effect induced by the amine group, by shielding the carbon nucleus.
The FT-IR spectra in Fig. 3 confirmed the absence of OH bands in the all aromatic acid dichloride. Characterization by NMR techniques was not necessary in order to continue with the synthesis of polymers.
3.2. Polymer synthesis and characterization
Poly(amides) were prepared in solution, by reacting the appropriate acid dichloride with the diamine in N,N-dimethylacetamide as solvent (Scheme 2).Successful conversion from acid dichloride and diamine to amide was confirmed by the FT-IR spectra, in which the characteristic absorptions due to the amide group moiety around 3440 cm−1 (υN–H) and 1650 cm−1 (υC=O) can be clearly identified (Fig. 4).
Fig. 5 shows the 1H NMR spectrum of PA-1 in DMSO-d6. The singlet at 10.36 ppm confirms the formation of an amide linkage. Other peaks were assigned to hydrogens of the repeating unit. The hydrogens H8 and H1 appeared with a very similar chemical shift, as shown in the enlarged figure. The signals of hydrogens H12 and H13 appear as doublets, characteristic pattern of substitution in the para position of the aromatic ring (AB system). Also, the coupling constant (J) of hydrogens H12 and H13 was determined (J = 7.5 Hz approximately). The chemical shifts of hydrogens H7 and H7′ appear overlapped with the singlet at 0.5 ppm, which corresponds to hydrogens of the methyl groups bound to the silicon atom.
The 13C NMR spectrum of PA-1 (Fig. 6) shows two peaks at 167.3 and 165.1 ppm, corresponding to carbonyl carbon C10 and C5 of the amide groups, respectively, which exhibit different chemical environments. Aliphatic carbon C15 appears with a δ value below zero due to the lower electronegativity of silicon atom respect to carbon. Thus, the respective signal is shifted to higher field. Similar behavior was observed for the methyl hydrogens in the 1H NMR spectra. Chemical shifts of the remaining carbons were in accordance with the proposed structure.
Polymeric samples were also characterized by 29Si NMR. Fig. 7 shows the spectrum of PA-1, where a single signal at −8.47 ppm is observed. This chemical shift is expected when the silicon atom is surrounded by two aromatic rings and two methyl groups.19,20
3.3. Inherent viscosity and solubility
Inherent viscosity was measured at a single point, in N-methyl-2-pyrrolidone (NMP) solution at 25 ± 0.1 °C (0.5 g dL−1). Inherent viscosity values were in the range of 0.30 to 0.47 dL g−1. On the other hand, the solubility of poly(amides) was tested in various organic solvents, and the results are listed in Table 1. Unmodified polyamides such as PPTA and PPMI were insoluble in m-cresol and, in some cases; LiCl was required for their complete dissolution. The chemical modifications made on the new poly(amides) improve solubility, mainly in polar aprotic solvents and m-cresol.| Solubility | |||||||
|---|---|---|---|---|---|---|---|
| a Measured in NMP (0.5 g dL−1 at 25 °C) Solubility: +, Soluble at room temperature; -, Insoluble. *Poly(m-phenylene isophthalamide) used as ref. 21. **Poly(p-phenylene terephthalamide) used as ref. 21.b 5% wt of LiCl was required. | |||||||
| PA | ηinh (dL g−1)a | DMSO | NMP | DMF | DMAc | m-cresol | THF |
| PA-1 | 0.45 | + | + | + | + | + | — |
| PA-2 | 0.47 | + | + | + | + | + | — |
| PA-3 | 0.30 | + | + | + | + | + | — |
| PMPI* | + | + | +b | + | — | — | |
| PPTA** | +b | +b | +b | +b | — | — | |
3.4. Thermal properties
Thermal stability of all poly(amides) was evaluated by TGA and DSC techniques under a nitrogen atmosphere and the results are summarized in Table 2. Fig. 8 shows the TGA curves for all PAs. Three PAs exhibited good thermal stability, with the 10% weight loss temperature (TDT10%) in the range of 300–371 °C.| PA | aTDT10% (°C) | bTg (°C) | cChar Yield (%) |
|---|---|---|---|
| a Thermal decomposition temperature at which 10% weight loss was recovered by TGA at a heating rate of 10 °C min−1 in nitrogen atmosphere.b Glass transition temperature taken at 10 °C min−1 in nitrogen atmosphere.c Residual weight (%) when heated to 800 °C. *Poly(m-phenylene isophthalamide) used as ref. 21. **Poly(p-phenylene terephthalamide) used as ref. 21. | |||
| PA-1 | 371 | 245 | 39 |
| PA-2 | 359 | 215 | 40 |
| PA-3 | 300 | 250 | 37 |
| PMPI* | 280 | ||
| PPTA** | 425 | ||
PA-1 exhibited the highest thermal stability among them, probably due to the presence of the silicon atom, which increases the ionic character of the silicon–carbon bond along the chain.7,22 DTGA curves show a complex degradative process between 300–400 °C. However, a peak near 500 °C, corresponding to the main polymer degradation, was observed. PA-3 exhibited the lower value of TDT10%. Taking into account the values of inherent viscosity, PA-3 showed chains shorter than the other PAs (lower inherent viscosity), which could be associated to the low value of TDT10%.
In all cases, the char yield at 800 °C was nearly to 40%. High aromatic content of all poly(amides) was responsible for the high char yields after calcinations.
DSC analyses were performed in order to obtain the Tg values of all poly(amides) and the results are summarized in Table 2. Fig. 9 shows the DSC curves obtained from the second heating scan. Polymers did not show melting peaks, indicating the amorphous nature of the materials.
One transition was detected in all poly(amides) corresponding to the Tg. PAs showed Tg values greater than 215 °C. PA-1 and PA-3 showed similar Tg values and they were larger than those of PA-2. Regarding the structures of the poly(amides), PA-1 and PA-3 have para-position linkage while PA-2 has meta-position linkage. The chains in PA-2 could be less packed. This, and the fact that repeating unit in PA-2 has the lower aromatic content could be responsible for its lower Tg value. In general, the Tg values were large compared to other structurally modified poly(amides).14,23–25 These results could be interesting for applications in the gas separation membrane field or reverse osmosis membrane field. These technological applications, from the point of view of thermal properties, seek to obtain polymers with high values of the glass transition temperature.26–28
Table 2 also shows the Tg values for PMPI and PPTA. These values are very high, indicating that the structural modifications made on the poly(amides) synthesized in this work allowed to reduce structural rigidity, favoring industrial processability and reducing energy costs in case that mold the material to a temperature above the Tg be necessary.
3.5. Water uptake and contact angle
Water sorption measurements were performed gravimetrically at room temperature using poly(amides) powder samples previously dried at 120 °C for 24 h. Samples were introduced in a closed desiccator containing a relative humidity of 65%. The samples were frequently weighed during 15 h and then allowed to humidify for 5 more days until constant weight.Table 3 shows the percentage of water uptake for each poly(amide). PA-2 had the largest value of water uptake. However, analyzing the moles of water per mol of repeat unit, PA-1 has more moles of water in the repeat unit than PA-2. As the amount of amide groups in both repeating units is the same, the higher water content in the repeating unit of PA-1 could be associated to the presence of the silicon atom. It is known that the affinity of silicon to oxygen is very strong,29 being there an additional interaction between the silicon atom and the water oxygen.
| PA | Water uptake (%)a | mol H2O/mol repeat unit | mol H2O/mol amide | θ (°)b |
|---|---|---|---|---|
| a Measurements were performed using the same conditions as in ref. 30.b Contact angle recorded at a rate of 10 μL s−1 at 25 °C. *Poly(m-phenylene isophthalamide) used as ref. 30. | ||||
| PA-1 | 5.4 | 1.5 | 0.44 | 84 ± 1.8 |
| PA-2 | 6.0 | 1.4 | 0.48 | 69 ± 0.4 |
| PA-3 | 4.5 | 1.2 | 0.34 | 81 ± 2.0 |
| PMPI* | 5.5 | 0.7 | 0.35 | — |
PA-3 had the lowest value of water uptake and the least amount of moles of water per mol of repeating unit. The most plausible explanation for this result is that the sulfone group could form intra and/or intermolecular H-bonds with nearby amide groups, thereby reducing the number of moles of water per mol of amide groups. This fact is related with the large Tg value obtained for PA-3. The greater structural rigidity would hinder penetration of water molecules through the polymer matrix.
For comparison, the results obtained from an unmodified poly(amide), m-phenylene isophthalamide (PMPI),30 were included in Table 3. This poly(amide) is used as membrane for reverse osmosis separation.31 The poly(amides) prepared in this work have similar values of water uptake and exhibit water absorption values larger than 1.2 mol of water per repeating unit, while PMPI only has 0.7. Preliminarily, the prepared poly(amides) could have application in the field of reverse osmosis separation.
Another technique to investigate the wettability of a surface of film samples is by contact angle measurements. Dense films were prepared from all PAs by casting 10% (w/v) filtered N,N-dimethylacetamide polymer solutions onto a glass plate and heating at 60 °C overnight. Then, the films were stripped off and were dried in a vacuum oven at 100 °C overnight and at 150 °C for 48 h. Table 3 also shows contact angles measured for PAs and the image of the drop (Fig. 10) was captured five seconds after placed.
PA-1 and PA-3 had higher contact angles than PA-2, therefore, the surface of PA-2 is the most hydrophilic, since it showed the smallest contact angle. This result would be inconsistent with those obtained in the experiment of water absorption, but morphology of the film sample is a very important issue in wettability properties. The explanation to these results should be sought in the capability that has the chain for adopting different conformations and molecular packing. From this point of view, PA-2 has only two aromatic rings in their repeating unit, while PA-1 and PA-3 have three aromatic rings, therefore PA-2 has the lower aromatic content, which implies a lower hydrophobicity and therefore, more wettability.
4. Conclusions
Based on the synthesis of a novel aromatic diamine containing a bulky pendant polar group, a series of three aromatic poly(amides) were successfully prepared by low temperature solution polycondensation technique. One PAs had a silicon atom in the repeating unit, which possibly contributes to the increased water uptake per mole of the repeating unit. In general, all PAs had water absorption values per repeating unit larger than PMPI. All PAs were thermally stable and the glass transition temperature values were in the range of 215–250 °C. The obtained PAs were characterized by good solubility in aprotic solvents and m-cresol. PA-2 showed the smallest contact angle, indicating the most hydrophilic surface.Acknowledgements
Tundidor-Camba, A. acknowledges the financial assistance by Comisión Nacional de Investigación Científica y Tecnológica, CONICYT, through Project 79130011.References
- J. Lin and D. C. Sherrington, Recent developments in the synthesis, thermostability and liquid crystal properties of aromatic poly(amides), Adv. Polym. Sci., 1994, 111, 177 CrossRef CAS.
- M. Trigo-López, P. Estévez, N. San-José, A. Gómez-Valdemoro, F. C. García and F. Sena, et al. Recent patents on aromatic polyamides, Recent Pat. Mater. Sci., 2009, 2, 190 CrossRef.
- J. K. Fink, High performance polymers, William Andrew Inc., New York, 2008 Search PubMed.
- Y. Imai, Synthesis of novel organic-soluble high-temperature aromatic polymers, High Perform. Polym., 1995, 7, 337 CAS.
- D. J. Liaw, in Macromolecular Nanostructured Materials, ed. N. Ueyama and A. Harada, Kodansha & Springer, Tokyo, 2004, ch. 2.2, pp. 80–100 Search PubMed.
- S.-H. Hsiao and Y.-M. Chang, Synthesis and properties of novel soluble polyamides having ether linkages and laterally attached p-terphenyl units, J. Polym. Sci., Part A: Polym. Chem., 2004, 42, 4056 CrossRef CAS.
- S. F. Thames and K. G. Malone, A Study of Polyesters Synthesized from an Arylsilane and Its Carbon Analog, J. Polym. Sci., Part A: Polym. Chem., 1993, 31, 521 CrossRef CAS.
- A. Tundidor-Camba, C. A. Terraza, L. H. Tagle and D. Coll, Polyamides obtained by direct polycondesation of 4-[4-[9-[4-(4-aminophenoxy)-3-methyl-phenyl] fluoren-9-YL]-2-methyl-phenoxy]aniline with dicarboxylic acids based on a diphenyl-silane moiety, J. Appl. Polym. Sci., 2011, 120, 2381 CrossRef CAS.
- H. Behniafar and M. Ghorbani, New heat stable and processable poly(amideethereimide)s derived from 5-(4-trimellitimidophenoxy)-1-trimellitimido naphthalene and various diamines, Polym. Degrad. Stab., 2008, 93, 608 CrossRef CAS PubMed.
- N. V. Sadavarte, M. R. Halhalli, C. V. Avadhani and P. P. Wadgaonkar, Synthesis and characterization of new polyimides containing pendent pentadecyl chains, Eur. Polym. J., 2009, 45, 582 CrossRef CAS PubMed.
- J. J. Ferreiro, J. G. de la Campa, A. E. Lozano and J. de Abajo, Polyisophthalamides with heteroaromatic pendent rings: synthesis, physical properties, and water uptake, J. Polym. Sci., Part A: Polym. Chem., 2005, 43, 5300 CrossRef CAS.
- D. J. Liaw, B. Y. Liaw and S. H. Lai, Synthesis and characterization of new cardo poly(amides) and polyimides bearing a 4-phenylcyclohexylidene unit, Macromol. Chem. Phys., 2001, 202, 807 CrossRef CAS.
- Y. T. Chern and H. C. Shiue, Low dielectric constants of soluble polyimides based on adamantine, Macromolecules, 1997, 30, 4646 CrossRef CAS.
- C. A. Terraza, L. H. Tagle, C. Contador, A. Tundidor-Camba and C. M. González-Henríquez, Silarylene-containing poly(ether-imide)s obtained by Yamasaki-Higashi phosphorilation techique. Use of bis(4-(4-aminophenoxy)phenyl)ethylmethylsilane, Polym. Bull., 2014, 71(5), 1001 CrossRef PubMed.
- M. Bruma and B. Schulz, Silicon-containing aromatic polymer, J. Macromol. Sci., Polym. Rev., 2001, 41, 1 CrossRef PubMed.
- R. L. Riley, et al., Poly(amide) reverse osmosis membrane fouling and its prevention: oxidation-resistant membrane development, membrane surface smoothing and enhanced membrane hydrophilicity, U. S. Department of the Interior Desalination Research and Development Program Bureau of Reclamation, September, 2000 Search PubMed.
- E. Ferrero, J. F. Espeso, J. G. de la Campa, J. de Abajo and A. E. Lozano, Synthesis and characterization of aromatic poly(amides) containing alkylphthalimido pendent groups, J. Polym. Sci., Part A: Polym. Chem., 2002, 40, 3711 CrossRef CAS.
- C. A. Terraza, L. H. Tagle, D. Mejías, A. Tundidor-Camba, P. Ortiz, D. Muñoz, F. Alavarez and C. M. González-Henríquez, Synthesis and characterization of new poly(amide)s derived from bis(4-(4-aminophenoxy)phenyl)-methylphenylsilana and bis(4-carboxyphenyl)R1R2 silane acids, Polym. Bull., 2013, 70, 773 CrossRef CAS PubMed.
- L. H. Tagle, C. A. Terraza, A. Leiva and F. Devilat, Synthesis, characterization, and thermal studies of polyamides and polyimides containing two silarylene units in the main chain, J. Appl. Polym. Sci., 2008, 110, 2424 CrossRef CAS.
- C. A. Terraza, L. H. Tagle, F. Concha and L. Poblete, Synthesis and characterization of new bi-functional monomers base don germarylene or silarylene units: 4,4′-(R1R2-silylene)bis(phenyl chloroformates) ans 4,4′-bis(R1R2-germylene)bis(phenyl chloroformates), Des. Monomers Polym., 2007, 10(3), 253 CrossRef CAS PubMed.
- J. E. Mark, Polymer Data Handbook, Oxford University Press, 1999, pp. 145, 149, 706 and 707 Search PubMed.
- M. Bruma, E. Hamciuc, I. Sava and N. M. Belomoin, Polymers containing phenylquinoxaline rings, Russ. Chem. Bull., 2004, 53(9), 1813 CrossRef CAS PubMed.
- L. H. Tagle, C. A. Terraza, A. Tundidor-Camba and J. Soto-Salamanca, Oligomeric aliphatic/aromatic poly(amides) containing silicon in the main chain and phthalimide or tetrahalophthalimide moieties as side groups, Polym. Bull., 2014, 71, 287 CrossRef CAS PubMed.
- L. H. Tagle, C. A. Terraza, A. Tundidor-Camba and P. Ortiz, Silicon-containing oligomeric poly(imido-amides) with amino moieties. Synthesis, characterization and thermal studies, RSC Adv., 2014, 4(57), 30197 RSC.
- A. Tundidor-Camba, C. A. Terraza, L. H. Tagle, D. Coll and P. Ortiz, Synthesis of poly(urethane)s based on diphenyl-silane/germane and oxyphenyl units: Structure-properties relationship, J. Appl. Polym. Sci., 2012, 124(2), 1036 CrossRef CAS.
- M. Calle, A. E. Lozano, J. G. de La Campa and J. de Abajo, Novel Aromatic Polyimides Derived from 5′-t-Butyl-2′-pivaloylimino-3,4,3′′,4′′-m-terphenyltetracarboxylic Dianhydride with Potential Application on Gas Separation Processes, Macromolecules, 2010, 43, 2268 CrossRef CAS.
- M. Calle, A. E. Lozano, J. de Abajo, J. G. de la Campa and C. Alvarez, Design of gas separation membranes derived of rigid aromatic polyimides. 1. Polymers from diamines containing di-tert-butyl side groups, J. Membr. Sci., 2010, 365, 145 CrossRef CAS PubMed.
- D. J. Mohan and L. Kullová, A study on the relationship between preparation condition and properties/performance of polyamide TFC membrane by IR, DSC, TGA, and SEM techniques, Desalin. Water Treat., 2013, 51, 586 CrossRef CAS.
- A. P. LeGrand, The surface properties of silicas, John Wiley & Sons, New York, 1998 Search PubMed.
- V. Ayala, E. M. Maya, J. M. Garcia, J. G. de la Campa, A. E. Lozano and J. de Abajo, Synthesis, characterization, and water sorption properties of new aromatic polyamides containing benzimidazole and ethylene oxide moieties, J. Polym. Sci., Part A: Polym. Chem., 2005, 43, 112 CrossRef CAS.
- V. Ramachandhran, B. M. Misra and M. P. S. Ramani, Poly (m-phenylene isophthalamide) membranes for reverse osmosis separations, Int. J. Polym. Mater., 1990, 4(3–4), 157 CrossRef.
| This journal is © The Royal Society of Chemistry 2015 |