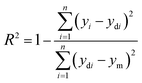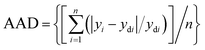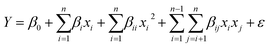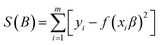DOI:
10.1039/C4RA16134C
(Paper)
RSC Adv., 2015,
5, 21384-21395
Enhance protection of electronic appliances through multivariate modelling and optimization of ceramic core materials in varistor devices
Received
10th December 2014
, Accepted 10th February 2015
First published on 10th February 2015
Abstract
E-waste comprises discarded low quality protected electronic appliances that annually accumulate million tons of hazardous materials in the environment. Protection is provided to control unwanted voltages that usually generate in associated electrical circuits by a multi-junction ceramic in a voltage dependent varistor. The ceramic's microstructure consists of ZnO grains that are surrounded by the narrow boundaries of melted specific additives such as Bi2O3, TiO2 and Sb2O3. In fact, the boundaries manage the quality of protection through a certain volume of intrinsic oxygen vacancies transformation which depends on the amounts of the additives. Since these amounts are the ceramic fabrication's initial input variables, the optimization process is capable of improving the quality of the protection (non-linear coefficient) as an output of the varistor devices. In this work, the fabrication was designed and then experimentally performed to calculate the non-linear coefficients of the produced varistors as actual responses. The responses were used to obtain an appropriate model for the fabrication by different semi-empirical methods. Afterward, the models predicted the optimized amounts of the additives which maximized the quality of the varistors. The predicted condition was fabricated as final varistors that were electrically characterized to compare their nonlinear coefficients as the quality indicator. The comparison demonstrated that the optimized amounts of Bi2O3 (0.5), TiO2 (0.47) and Sb2O3 (0.21) in mol% provided the very high protective varistor with nonlinear coefficients of 28.1. In conclusion, the optimization, which has industrial scale-up potential, warranties the electronic protection that controls global e-waste.
Introduction
Globally, e-waste accumulates millions of tons of hazardous materials such as heavy metals including lead, mercury, cadmium, and halogenated substances into water, soil and air.1 E-waste comprises discarded electronic appliances, of which anything with a plug (even old refrigerators and motorized toothbrushes) are disproportionately abundant because of their short lifespan.2 These electronics are often damaged from repeated exposure to large overvoltages which are generated by electrostatic discharge and electrical overstress such as lightning strikes, power outages, tripped circuits, power transitions, power malfunctions, electromagnetic pulses and inductive spikes in an associated circuit.3,4 However, the electronics are protected by voltage limiting devices such as voltage dependent low voltage varistors and back-to-back zener diodes that are placed in parallel positions with the electronics in an associated electrical circuit.5 The problem is that the diodes are degraded by repeated exposure to large overvoltages due to their low capacity and single p–n junction.6 On the other hand, the varistors tend to be more stable in AC and DC fields over wide ranges of voltage, a few volts to tens of kilovolts, and current from micro-amperes to kilo-amperes. However, the varistors are not sufficiently developed and present several drawbacks such as low non-linear properties, high leakage-current, high breakdown-voltage and high degradation from repeated exposures which come from the microstructure of ceramic cores used in varistors.7,8 In the most common varistor, the microstructure consists of highly conductive n-type ZnO grains that are surrounded by the narrow boundaries of melted specific additives.9,10 The microstructure is fabricated by mixing an appropriate amount of ZnO and the additives (starting powder). Then the mixed powder is pressed and sintered at a temperature under the melting point of ZnO.11,12 Accordingly, the melted additives occupy the ZnO grains boundaries as intergranular layers which navigate the non-linear property by using intrinsic oxygen vacancies transformation.5,13–21 Therefore, the origin of varistor action is attributed to composition of the intergranular layer that depends on many operations such as type, amount and mixing method of the additives in the starting powders as well as the sintering process.19–21 The optimum sintering temperatures and holding time for a Bi2O3 doped ZnO based low voltage varistor were reported from 1200 to 1280 and 1 hour respectively.22 Stable performance throughout the intergranular layers requires homogeneity in terms of its components which is provided by chemical mixing methods (solution coating) of the starting powders.23,24 The non-linearity as quality of the protection primarily depends on chemical compositions of the materials in an intergranular layer which comes from the starting powders.25,26 For instance, Bi2O3 is used as a former which is a crucial parameter for varistor manufacturing, TiO2 prevents the vaporization of Bi2O3 to facilitate ZnO grain growth, and Sb2O3 stabilizes the electrical properties and diminishes the leakage current of the varistor during performance.16,27–31 Among the operators, follow-up in the compositions is very difficult because the layer formulation consists of several high pressure oxides.32 This means the compositions amounts are changed during the sintering process because of many reasons such as component vaporization. Moreover, there are other complexities such as different reactions including formation and decomposition of many phases, kinetics of ZnO grain growth, and densification of melted additives during the ceramic fabrication. On other hand, the additives are not completely independent: therefore, it is very difficult to consider the effect of one additive as a variable on the non-linearity as response while other additives are kept constant in an optimization process.33,34 Thus, multivariate semi-empirical methods such as response surface methodology (RSM) and artificial neural networks (ANNs) have been widely accepted to model and optimize the productive processes.35–37 The multivariate methods consider the effect of two initial ingredients (variables) on the final output product (response) simultaneously and are free of mentioned complexity.38 In addition, the semi-empirical methods have used the responses of designed actual experiments for modelling and then are applied to optimize the process.36–41 In this work, the fabrication of the ZnO–Bi2O3 based low voltage varistor was modelled and optimized by RSM and ANN. In the modelling, the amounts of the starting powders were selected as input variable while the non-linear coefficients of the fabricated varistors were the actual responses. The generated models of both RSM and ANN were validated by particular techniques and then they were used to navigate the fabrication.
Experimental setup
Materials and methods
In this work, ZnO (99.9%), Bi(NO3)3·5H2O (98%, Alfa Aesar), Ti(OC4H9)4 (96%, Alfa Aesar), Antimony acetate (99.99%, Aldrich), and absolute ethanol (Merck) were used to prepare starting powder. To prepare coated ZnO powder, the appropriate amount of Bi(NO3)3·5H2O, antimony acetate and Ti(OC4H9)4 were dissolved in 100 ml of ethanol under continuous stirring for 1 h. Then, the appropriate amount of ZnO powder was slowly added to the solution at 80 °C to obtain the slurry. The slurry was changed to paste with continual heating and magnetic stirring. The paste was dried in an oven at 100 °C overnight. Thereafter, the dried paste was ground and characterized by Field Emission Scanning Electron Microscopy (FESEM) and thermo-gravimetric analysis (TGA) to indicate a coating layer and determine the calcination temperature respectively. The FESEM confirmed the coated ZnO in this stage (Fig. 1). The calcination was conducted at 750 °C for 2 h in air with a heating and cooling rate of 5 °C min−1 to convert coated hydroxide (Bi(OH)2, Ti(OH)2 and Sb(OH)2) to metal oxide (Bi2O3, TiO2 and Sb2O3) in a box furnace (CMTS model HTS 1400). To make the varistor, the proper amount of coated ZnO powders as starting powder was pressed into 10 mm diameter pellets at 200 MPa using a uniaxial presser machine. The compacted pellet was sintered at 1260 °C in air for 1 h, also with heating and cooling rates of 5 °C min−1.38 Both sides of a sintered pellet as ceramic core of the varistor were painted by a silver electrode for DC current–voltage (I–V) characterization. The I–Vs were obtained by scanning the varistors from 0 to 100 volts using a step size of 2.5 which was performed by a Keithley 2400 sourcemeter. The obtained current density (J) and electrical field (E), the I and V were divided by surface of the painted silver electrode (cm2) and thickness of the ceramic core (mm) respectively. The non-linear coefficient of the varistor which comes from I = KVα (α = alpha) was calculated according to eqn (1),| |
 | (1) |
where E1 (V mm−1) and E2 (V mm−1) were obtained at J1 = 0.1 (mA cm−2) and J2 = 1 (mA cm−2), respectively.42 The alpha was used for fitting and learning processes of the semi-empirical methods used to obtain the optimized varistor. The optimized varistor was characterized by X-ray diffraction XRD; (PANanalytica, Philips-X'pert Pro PW3040/60) and field emission scanning electron microscopy (FESEM; JEOL JSM-7200) with energy dispersive X-ray analysis (EDX). The XRD was within the 2θ scan range of 20–80° for the phase analysis.
 |
| | Fig. 1 The morphology of the used ZnO grain in the ceramic core of the low voltage varistor: (a) ZnO powder before coating, (b) ZnO powder after coating and before calcination. | |
RSM experimental design
RSM modelling as a semi-empirical method uses the actual responses which are obtained by a particular experiment of design (EOD). In this case, the design was carried out by a central composite design (CCD) that was embedded in the Design-Expert software version 8.0.7.1, Stat-Ease Inc., USA.28,29 In the design, the amounts of the additives (Bi2O3, TiO2, and Sb2O3) in the ceramic starting powder were considered as the input effective variables. The amounts of the variables were selected to be in the vicinity of their reported range.16,43–47 Table 1 shows the variables in coded symbols as well as the actual values and ranges used in the design. Table 2 illustrates the design of 20 samples which were categorized as follows: factorial points (8 samples), axial points (6 samples), and central points (6 samples). The central points are the replicated samples which were acquired to measure the experimental pure error. In the design, each row shows the process of a varistor's fabrication (Run), which is explained in Section 2.1, while the columns indicate amounts of the additives, and the calculated and model predicted alpha of the fabricated varistor. Therefore, the process in Section 2.1 was carried out for each run in the laboratory. The calculated alphas presented in Table 2 (the actual responses) were used for the RSM fitting process to find the appropriate model which was then applied for optimization of a varistor (Section 3.1).48,49 The fitting process proposed a provisional model which was deeply validated by analysis of variance (ANOVA). The model was then used to track the optimum amounts of additives in the experimental design points as well as to predict the desirable condition that maximizes the alpha of the final varistor.
Table 1 The effective variables in the ceramic's starting powder and their levels used for experimental design
| Effective variables |
Level of the variables |
Unit |
| Coded |
Actual |
The lowest (−α) |
Low (−1) |
Center (0) |
High (+1) |
The highest (+α) |
| x1 |
Bi2O3 |
0.16 |
0.3 |
0.5 |
0.7 |
0.84 |
mol% |
| x2 |
TiO2 |
0.16 |
0.3 |
0.5 |
0.7 |
0.84 |
mol% |
| x3 |
Sb2O3 |
0.13 |
0.2 |
0.3 |
0.4 |
0.47 |
mol% |
Table 2 Experimental design of the varistor's fabrication. The columns show the amounts of Bi2O3, TiO2, Sb2O3, actual and model predicted alpha while the rows are varistors as samples
| Run |
Bi2O3 (mol%) |
TiO2 (mol%) |
Sb2O3 (mol%) |
Observed alpha |
Predicted alpha |
| 1 |
0.3 |
0.3 |
0.2 |
4.1 |
4.6 |
| 2 |
0.7 |
0.3 |
0.2 |
4.3 |
4.7 |
| 3 |
0.3 |
0.7 |
0.2 |
1.0 |
1.4 |
| 4 |
0.7 |
0.7 |
0.2 |
5.5 |
5.3 |
| 5 |
0.3 |
0.3 |
0.4 |
4.0 |
4.4 |
| 6 |
0.7 |
0.3 |
0.4 |
3.5 |
3.3 |
| 7 |
0.3 |
0.7 |
0.4 |
3.9 |
3.6 |
| 8 |
0.7 |
0.7 |
0.4 |
6.6 |
6.3 |
| 9 |
0.164 |
0.5 |
0.3 |
5.7 |
5.2 |
| 10 |
0.836 |
0.5 |
0.3 |
7.2 |
7.5 |
| 11 |
0.5 |
0.164 |
0.3 |
5.9 |
5.3 |
| 12 |
0.5 |
0.836 |
0.3 |
4.8 |
5.1 |
| 13 |
0.5 |
0.5 |
0.132 |
3.0 |
2.5 |
| 14 |
0.5 |
0.5 |
0.468 |
2.9 |
3.1 |
| 15 |
0.5 |
0.5 |
0.3 |
15.3 |
14.5 |
| 16 |
0.5 |
0.5 |
0.3 |
13.6 |
14.5 |
| 17 |
0.5 |
0.5 |
0.3 |
15.3 |
14.5 |
| 18 |
0.5 |
0.5 |
0.3 |
13.6 |
14.5 |
| 19 |
0.5 |
0.5 |
0.3 |
13.6 |
14.5 |
| 20 |
0.5 |
0.5 |
0.3 |
15.3 |
14.5 |
ANN learning
The learning process was carried out to determine the structure of ANNs semi-empirical model by using training and testing data sets. Therefore, the performed experiments in Table 2 were randomly split up into two sets as training and testing data sets using the option available in NeuralPower software version 2.5.50,51 The ANN structure consists of input, hidden, and output layers while the input layer is made of initial variables (additives) and the output layer has only one node as a response (alpha). The learning process determines the number of the node in the hidden layer by using the split data sets. The number of nodes in the hidden layer was obtained by using a trial and error learning calculation which was examined from 1 to ‘15’ nodes. The learning process was initially started with one node in the hidden layer to obtain a network (architecture) with 3 nodes input, 1 node in hidden and 1 node in output layer by a quick propagation algorithm (QP). The nodes in the input and output layers were kept constant during the process while the number of nodes in the hidden layers were varied up to 20. The examination of each node was repeated for 10 times to avoid random correlation due to the random initialization of the weights. Among the repeated examinations, the architecture with the lowest root mean squared error (RMSE) was selected for each node. Therefore, 20 architectures were obtained at the end of the learning process for QP algorithm. As a result of the learning process, the architecture with minimum RMSE was selected as a final topology for calculating the coefficient of determination (R2) and the percentage of absolute average deviation (AAD) (eqn (2) and (3)),| |
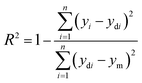 | (2) |
| |
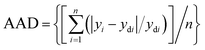 | (3) |
where ‘n’ is the number of points, ‘yi’ is the predicted value, ‘ydi’ is the actual value, and ‘ym’ is the average of the actual values. Therefore, the appropriate topologies were determined by minimum RMSE and ADD while the R2 was at maximum value. The model was used to obtain the importance and optimum narrow level of the additives in the initial powder. In addition, the model predicted the optimum values of the additives to achieve the maximum alpha value.
The semi-empirical methods
A corner of RSM. RSM creates a functional relationship between variable–variable and variables–response(s) by using approximated low-degree polynomial models that consist of the variables and their coefficients. Eqn (4) shows the second-order polynomial which RSM commonly uses for optimization process,| |
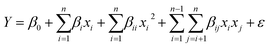 | (4) |
where Y is the response of interest, β0 is a constant term, βi is the coefficient of the linear terms, βii demonstrates the quadratic term coefficient, and βij is the coefficient of the interaction terms. All of the coefficients are unknown. The xi and xj are control variables and “ε” is a random experimental error. The β's are estimated by a fitting process that uses the actual experimental responses. In the fitting process, the responses are fitted to the polynomial (eqn (4)) by sequential model sums of squares (SMSS). SMSS compares the sufficiency of linear, two-factor interaction (2FI), quadratic, and cubic models using the statistical significance of adding new model terms, step-by-step in increasing order.52 The comparison is evaluated using statistical evidence such as the F-value, predicted residual sum of squares (PRESS), adjusted R-squared (RAdj), predicted R-squared (RPred), and probability value (P-value). The PRESS is the sum of the squares of a model's prediction errors. The minimum value of the P-value and PRESS as well as the maximum value of RAdj, RPred, and F-value are considered to determine the provisional model of the process.48,53 The provisional model is usually suggested by the software and is studied in detail using analysis of variance (ANOVA).54 The ANOVA indicates the significance of each term of the model, including the intercept, linear, interaction, and square terms. In fact, the adequacy of the model is certified by ANOVA and then the model is used to navigate the process. The model is able to track the optimum amount of the variables in the experimental design points by canonical and three-dimensional (3D) plots as the surface response. Moreover, the model is capable of predicting the desirable condition that maximizes the yield of the productive process.
ANN description. ANNs are semi-empirical modelling methods which use the actual processing condition and corresponding responses to govern a network to avoid of complexity. The network consists of different layers such as input, hidden, and output which are made of several connected units (nodes). Nodes are simple artificial neurons which mimic a biological neural network makeup. The nodes of an input layer are the effective variables and in an output layer are the responses. In a hidden layer, the number of nodes is determined by a learning process.55,56 In the network, the nodes are connected by a multilayer normal feed-forward or feed-back connection formula.57 To qualify the network, the input layer acts as distributor and sends data via the weights to the nodes of the second layer (hidden layer).58 The weighted data is saved as processing nodes in the hidden layer and then transferred to the output layer by a particular transferred function.59,60 Therefore, the qualified data are passed into the input layer, propagated to a hidden layer and then transferred into the output layer of the network by an iterative procedure.61 The iteration is an act of repeating a process to approach a desired result. After it appears in the first input–output iteration result, the second period is then processed and so on. The network changes the weights in order to reduce the difference between actual and the network's predicted responses at each iteration. The results of an iteration are used as a starting point of the next iteration. For example, when the results of the last iteration become almost equal to the results of a previous iteration, the process will be terminated. The iteration process is continued by a self-similarity method (eqn (5)).61| |
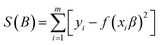 | (5) |
where ‘m’ is an empirical data pair of independent and dependent variables such as (xi, xi) and f(xi, β) in the model curve. In a self-similarity process, the β parameter of f(xi, β) is optimized by minimizing the root mean squared error (RMSE). As a result, the main aim of the learning process is to find the weights for minimizing the RMSE which is obtained from a difference between network predictions and actual responses (eqn (6)).| |
 | (6) |
where ‘n’ is number of the points, yi is the predicted values and ydi is the actual values.
Results and discussion
In this work the fabrication of ZnO–Bi2O3–TiO2–Sb2O3 ceramic that was used as core of low voltage varistors was modelled and optimized to improve the protectiveness of electrical devices and, consequently, e-waste reduction. The modelling processes were carried out by semi-empirical methods such as RSM and ANN. In the processes the initial additives in the varistor's ceramic core starting powder (including Bi2O3, TiO2 and Sb2O3) were input variables while the non-linear property of the fabricated varistors (alpha) was output. The obtained models of the methods used were validated by different techniques and then they were used to navigate the fabrication which included optimization of the input variables to maximize the output as well as to determine the importance of the input variables. As a final conclusion, the models predicted the optimum varistors which were then experimentally fabricated. The electrical characteristics of the varistors were compared to select the final optimized varistor.
RSM modelling and validation
According to the experimental design (Table 2), twenty varistors were fabricated and their I–V characteristics were measured to calculate actual alpha which is presented by Fig. 2. As shown, the maximum alpha belonged to the middle of the selected levels of the additives and that shows the levels were properly selected. To obtain a suitable model, the collected data in Table 2, as experimental design including the amount of the additives and observed alpha respectively, were used as input variables and output response for fitting process by RSM. First, the fitting process were carried out for 2FI, linear, quadratic, and cubic models to obtain Lack of Fit indicators and the standard deviation (Std. Dev.), RAdj, RPred, and RPred (Table 3). Then the results of each model were compared to suggest the provisional model for deep validation (Table 4). As Table 4 indicates the quadratic model was merited to suggest while the cubic model was aliased. As a result, the quadratic model with Std. Dev. (0.8), RAdj (0.973), RPred (0.951), and R2 (0.986) was selected as the provisional model for deep validation.
 |
| | Fig. 2 The obtained actual alpha for 20 varistors in the experimental design while the run numbers 15 to 20 are replication which are in the middle of the selected levels. | |
Table 3 The fitting results of the 2FI, linear, quadratic and cubic models. Std. Dev. is standard deviation
| Source |
Lack of Fit indicator |
Model Summary Statistics |
| F-Value |
P-value |
Std. Dev. |
RAdj |
RPred |
R2 |
PRESS |
| Linear |
46.9 |
0.0003 |
5.3 |
0.0 |
−0.2 |
−0.3 |
617.3 |
| 2FI |
63.0 |
0.0001 |
5.8 |
0.0 |
−0.4 |
−1.5 |
1146.5 |
| Quadratic |
0.5 |
0.7789 |
0.8 |
1.0 |
1.0 |
1.0 |
22.5 |
| Cubic |
0.2 |
0.6830 |
0.9 |
1.0 |
1.0 |
0.9 |
42.2 |
Table 4 The sequential model sum of squares comparison for 2FI, linear, quadratic and cubic models to suggest the provisional
| Source |
F-Value |
P-value |
Remark |
| 2FI vs. linear |
0.1 |
0.9578 |
|
| Quadratic vs. 2FI |
225.8 |
<0.0001 |
Suggested |
| Cubic vs. quadratic |
0.6 |
0.6519 |
Aliased |
The selected provisional model, which is a mathematic equation (eqn (6)), presents the relationship between the inimical additives as input variables as well as the variables and final output (alpha) using estimated coefficients and linking signs (±).
| | |
Y = −55.244 + 68.436x1 + 60.736x2 + 242.182x3 + 23.130x1x2 − 14.612x1x3 + 29.810x2x3 − 72.143x12 − 81.49x22 − 413.012x32
| (7) |
where
Y is alpha;
x1,
x2, and
x3 are linear parameters;
x12,
x22,
x32 are the quadratic terms; and
x1x2,
x1x3, and
x2x3 are the interaction factors which were introduced in
Table 1. The next number in the items are fitting estimated coefficients, which are the weights of the terms, while the linked signs (+, −) determine the synergic and antagonistic behavior of the parameters in the model.
The deep validation of the provisional model was carried out by analysis of variance (ANOVA) which was depicted in Table 5. As shown, the general model fit and lack of fit were correctly significant and not-significant respectively. As a detail of validation, the term's partial sum of squares has confirmed the significance of x1, x1x2, x12, x22, and x32 in the model while x2, x3, x1x3, and x2x3 were not significant, which means they can be removed from the model. Therefore, the modified model could be presented by:
| | |
Y = −55.244 + 68.436x1 + 23.130x1x2 − 72.143x12 − 81.49x22 − 413.012x32
| (8) |
where the linear term of
x1 (Bi
2O
3) and the interaction term of
x1x2 (Bi
2O
3 × TiO
2) have a synergic effect on alpha, while the quadratic terms have an antagonistic effect on the response. Moreover, the importance of the terms is exhibited by the coefficients and their priority:
x32 >
x22 >
x12 >
x1x2 >
x1. As result of the validation, the quadratic model was recognized as the outstanding final model which was then used to navigate the ceramic fabrication process.
Table 5 The model analysis of variance for model fit and lack of fit as well as the importance of the terms in the provisional model
| Source |
F-Value |
p-value |
Remark |
| Model fit |
78.27 |
<0.0001 |
Significant |
| Model lack of fit |
0.48 |
0.7789 |
Not significant |
| x1 |
10.22 |
0.0095 |
Significant |
| x2 |
0.05 |
0.8335 |
Not significant |
| x3 |
0.82 |
0.3852 |
Not significant |
| x1x2 |
10.62 |
0.0086 |
Significant |
| x1x3 |
1.06 |
0.3275 |
Not significant |
| x2x3 |
4.41 |
0.0621 |
Not significant |
| x12 |
185.58 |
<0.0001 |
Significant |
| x22 |
239.73 |
<0.0001 |
Significant |
| x12 |
380.48 |
<0.0001 |
Significant |
| Lack of fit |
0.48 |
0.7789 |
Not significant |
Model application
Levels optimization. The validated model optimized the input variables (Bi2O3, TiO2, and Sb2O3) in range of the experimental design to obtain a varistor with maximum alpha. The optimization was carried out by mathematical derivation of the validated model (eqn (7)) and graphic three-dimensional plots (3D plots) that produced the points and response surface for the additives as well as alpha respectively. The optimized points were obtained by eqn (8)–(10) in which only one parameter is varied.36,37where the variables x1, x2, and x3 in the equations were introduced by Table 2. The calculation point method is very simple; however, its experimental validation test resulted in a large error.On the other hand, the response surface method showed the effect of two variables (additives) on the output (alpha) in a 3D plot (Fig. 3) while the other parameter was kept constant. In this case, there are 3 variables such as TiO2, Bi2O3, and Sb2O3 which are shown as three 3D plots in Fig. 3(a)–(c). Fig. 3(a) shows the simultaneous effect of TiO2 and Bi2O3 on the alpha with a constant amount of Sb2O3. As observed, the increasing amounts of TiO2 and Bi2O3 up to 0.5 mol%, made a synergic effect on the alpha, while the amounts antagonistically operated to reduce the alpha beyond the optimum (0.5 mol%). Therefore, the optimum has been presented by a small surface as response instead of a point that reduced the error of the experimental validation of the varistor. Fig. 3(b) shows the interaction effect of Sb2O3 and Bi2O3 on the alpha with a constant amount of TiO2 which depicts an increased alpha up to 0.5 and 0.3 mol% of Bi2O3 and Sb2O3, respectively. However, at the excess amounts of the additives, the alpha was decreased. Moreover, Fig. 3(c) demonstrates the effects of TiO2 and Sb2O3 with a constant amount of Bi2O3. As shown in the both 3D plots, the maximum value of the alpha appeared at 0.3 mol% Sb2O3 and the amounts of Bi2O3 and TiO2 were confirmed as indicated in Fig. 2(a). The amount of Sb2O3 was synergistically affected from 0.2 to 0.3 mol% on the alpha and then it operated as an antagonistic effect up to 0.4 mol%. The synergic effect of Sb2O3 may be due to densification of the ceramic matrix during the sintering process.62 However, the antagonistic effect beyond optimum might be due to homogeneous segregation of the additives at this concentration.47,63 As a result, the optimum determined a very narrow level of Bi2O3, TiO2 and Sb2O3 and quite small surface response around 14.52 for the alpha.
 |
| | Fig. 3 The graphical presentation of the maximized alpha response surface and optimized amounts of TiO2, Bi2O3 and Sb2O3 as additives in the starting powder of the ceramic core that used in ZnO low voltage varistors. (a) The effect of TiO2 and Bi2O3 on the alpha at constant amount of Sb2O3, (b) effect of Sb2O3 and Bi2O3 on the alpha at constant amount of TiO2 and (c) effect of TiO2 and Sb2O3 on the alpha at constant amount of Bi2O3. | |
The model prediction. The model was able to predict a varistor with maximum non-linearity coefficient (alpha) at high desirability value by using a numerical particular condition which was selected by the experimenter. The desirability is an objective function that uses mathematical methods,64 where the range of the desirability starts from zero for out of the limited area and goes to one at the goal. The desirability of this prediction was 0.92 and that was very close to the goal. The selected options of the particular condition were ‘in range’, ‘minimum’ and maximum for ‘amount of the additives’, ‘standard error’ and ‘alpha’ respectively. The options were facilitated by using the default of the software. The model-predicted values for Bi2O3, TiO2 and Sb2O3 were 0.52, 0.5, and 0.3 mol% at standard error, 0.328, and alpha, 14.52. The suggested values of the additive were used to fabricate the ceramic core in the laboratory for further experimental validation. Both sides of the ceramic were painted with a silver electrode and used as a final varistor for electrical characterization. Fig. 9 shows the results of the characterization which was used to calculate the alpha at J1 = 0.1 (mA cm−2) and J2 = 1 (mA cm−2). The alpha was 15.3 for E1 = 7.4 (V mm−1) and E2 = 8.6 (V mm−1).
ANN modelling and validation
The ANN modelling determined the network structure of the ceramic fabrication by designating the hidden layer artificial node number. The node number was obtained by trial and error learning calculations which were examined from one to 20 nodes. Calculations were initially started with one node in the hidden layer to obtain the architecture with 3 input nodes, 1 node in a hidden and 1 node in an output layer by a QP algorithm. The nodes in the input and output layers were kept constant during the process while the number of nodes in the hidden layers were varied up to 20. The examination of each node was repeated for 10 times to avoid random correlation due to the random initialization of the weights. Among the repeated examination node number, the architecture with the lowest RMSE was selected to compare with other architectures. As Fig. 4 shows, the RMSE of 20 architectures were plotted versus their hidden layer node number at the end of the learning process. As a result of the learning process, the architecture with minimum RMSE was selected as a final topology which was validated by R2 and AAD calculations.
 |
| | Fig. 4 The RMSE of the learned hidden layer of the obtained topologies. The smallest RMSE belonged to the topology that has 15 node in its hidden layer (QP-3-15-1). | |
The AAD calculation of the selected topology (QP-3-15-1) was 1.57 and 6.87 for training and testing data sets respectively, which exhibited a reasonable minimum absolute average deviation. In addition, the scatter plots of actual alpha versus model-predicted alpha of the varistors in the training and testing data sets exhibit the R2 of the QP-3-15-1 topologies (Fig. 5). As shown, the predicted values fitted well with the actual values for the training data set (R2 = 0.991) as well as testing data (0.974) sets which confirmed the validity of the topology (QP-3-15-1). Therefore, the QP-3-15-1 topology was considered as an efficient final model for navigation of the ceramic fabrication (Fig. 6). In the model, the input variables such as Bi2O3, TiO2 and Sb2O3 are connected to the calculated hidden nodes layer by multilayer normal feed-forward and then connect to alpha in output layer. The bias shifts the space of the nonlinearity properties. Therefore, the model was applied to obtain the importance and optimized level of the additives in the starting powder of the ceramic as well as to predict the optimum values of the additives in the ceramic's starting powder to achieve the maximum non-linearity (alpha) for the varistor.
 |
| | Fig. 5 The scatter plots of the actual and predicted alpha of the training and testing data set to visualize the R2 of selected topology (QP-3-15-1). | |
 |
| | Fig. 6 The fabrication model structure of the ceramic core in ZnO based low voltage varistor. The model consists of 3 variables in input layer, 15 nodes in hidden layer and 1 response in output layer (QP-3-15-1), bias shifts the space of the non-linearity properties. | |
Importance of the effective variables. Importance shows the relative effect (%) of the initial additives in starting powder of the ceramic core as input variables on the non-linearity of the varistor as response (Alpha). Therefore, the importance determines the effectiveness of the inputs as well as confirming or rejecting the initial suppositions for effective variables. In this case, the selected ANN model (Q-3-15-1) determined the relative importance of Bi2O3, TiO2 and Sb2O3 in starting powder of the ceramic at optimum condition (Fig. 7). As shown, the relative importance was 36.76% (Sb2O3), 35.55% (TiO2) and 27.69% (Bi2O3). As a result, the selected additive's variables were confirmed as effective input for the ceramic fabrication and none of them was negligible in this work.
 |
| | Fig. 7 The relative importance of Sb2O3 (36.76%), Bi2O3 (35.55%), and TiO2 (27.69%), as the additives used in starting powder of a ceramic core for a ZnO based low voltage varistor. | |
Model applications.
Level optimization. The wide levels of the additives in the starting powder were selected according to previous works which were carried out by traditional methods such as one variable at a time.45 Therefore, the levels were re-designed and optimized by the validated model (QP-3-15-1). For this purpose, the model simulated the effect of two additives on the alpha simultaneously without further requirement of mathematic function and equation knowledge while the other factor was kept constant. The simulated effects are presented as three-dimensional plots (3D plots) in Fig. 8 which demonstrate the surface of the additives' effect on the alpha. Therefore, the optimized narrow levels of the additives were Bi2O3 (0.606–0.836), TiO2 (0.293–0.836) and Sb2O3 (0.154–0.301) in mol% at optimum conditions. These levels were used to predict the optimum point value of the additives that maximized the alpha.
 |
| | Fig. 8 The 3D plots of the simultaneous effect of two additives on the alpha. The red surface response is the desirable alpha and blue color shows the lowest values of the alpha, (a) the effect of Sb2O3 and Bi2O3, (b) the effect of Sb2O3 and TiO2 and (c) the effect of Bi2O3 and TiO2. | |
Model prediction. The model was used to predict the optimum condition in the optimized levels for fabrication of 3 varistors (Table 7). The table shows the optimum point values of the additives and the related alpha for each suggested varistor. The fabricated processes including preparation of the starting powder, pressing, sintering, and electroding were carried out for the three varistors in the laboratory to validate the model prediction. The electrical characterization of the varistors was carried out to calculate the alpha (eqn (1)) which is indicated in Table 6. As shown, the actual alphas were very close to the model prediction which confirmed the model predictability. Therefore, varistor 1 was selected as an optimized case for electrical (E–J) and structural characterization including FESEM, EDX and XRD.
Table 6 The model predicted varistors that consist of the values of the additives in ceramic's starting powder. The rows show the optimum amounts of the additives and the columns indicate the composition in the ceramic core of each varistor; the predicted alpha was suggested by the model and the actual alpha is experimental result
| Additives and alpha |
Varistor 1 |
Varistor 2 |
Varistor 3 |
| Bi2O3 |
0.50 |
0.4611 |
0.4611 |
| TiO2 |
0.47 |
0.468 |
0.437 |
| Sb2O3 |
0.21 |
0.256 |
0.262 |
| Predicted alpha |
27.24 |
27.21 |
26.90 |
| Actual alpha |
28.10 |
27.74 |
26.44 |
The models navigation
In this work, the variables were initially used in wide levels, identical importance, and without any considered points. Therefore, the RSM (eqn (6)) and ANN models (Fig. 6) were used to determine the optimum levels, optimum points, and the importance of the effective variables which is presented by Table 7. As shown, there is a large difference between the values of the obtained alpha from RSM and ANN predicted varistors which are 15.3 and 28.1 respectively. The differences might be due to the selected levels and consequently the used point values of the Sb2O3 in the ceramic starting powder. The Sb2O3 controls the growth of the ZnO grains, which is necessary for low voltage varistors, by decreasing the mobility of grain boundaries by making a fine Sb-rich film on the surface of the ZnO grains.65 Moreover, the high model's importance Sb2O3 confirmed that the alpha was very sensitive to the amount of that while RSM's model (eqn (6)) depicted the antagonistic effect of the additive on the alpha. As a final result of the modelling processes, the ANN-predicted varistor was selected to characterize the microstructure of the ceramic core by using XRD, FESEM and EDX.
Table 7 The results of applying RSM and ANN validated models for additives of ceramic starting powder as input effective variables and non-linear properties of final optimized varistors
| Model |
Bi2O3 |
TiO2 |
Sb2O3 |
Alpha |
| Level (mol%) |
Point (mol%) |
Imp. (%) |
Level (mol%) |
Point (mol%) |
Imp. (%) |
Level (mol%) |
Point (mol%) |
Imp. (%) |
| RSM |
0.4–0.7 |
0.52 |
23 |
0.4–0.6 |
0.5 |
30 |
0.2–0.35 |
0.3 |
47 |
15.3 |
| ANN |
0.61–0.84 |
0.50 |
36.55 |
0.29–0.84 |
0.47 |
27.69 |
0.15–0.30 |
0.21 |
36.76 |
28.1 |
Starting powder of the final varistor. The starting powder of the final varistor ceramic core was prepared according to above methods (Section 2.1). Fig. 9 shows the FESEM morphology of the starting powder which was calcined to produce the coated metal oxides over the ZnO grains. As Fig. 9(a) indicated, the distribution of the coated additives presented great homogeneity which confirmed the ability of the solution coating method for the fabrication. Moreover, the most frequently coated additives particle sizes were within 40 to 50 nm (Fig. 9(b)), as obtained from 100 particles in different images of the calcined powder.
 |
| | Fig. 9 The FESEM morphology of the calcined starting powder of the final varistor ceramic core: (a) the coated additives over ZnO grains, (b) the particles size of the coated additives. | |
Fig. 10 illustrates the limited area EDX spectra of the starting powder element analysis. As shown, the elements Zn, Bi, Ti, and O were detected in the selected area of the powder while antimony (Sb) was not detected although it was detected by XRD (Fig. not shown).
 |
| | Fig. 10 The EDX histogram of the staring powder of final varistor ceramic core. The C peak is related to carbon type used as the base of the sample. | |
The ceramic core of the final varistor. To prepare the ceramic core of the final varistor, the appropriate staring powder was pressed into a particular pellet and then sintered at 1260 °C for one hour. The sintered pellet as a ceramic core was characterized using FESEM, EDX, and XRD. Fig. 11 demonstrates the FESEM morphology of the ceramic core microstructure. Fig. 11(a) illustrates the homogenized ZnO grain sizes which may be due to the appropriate distribution of the additives in the initial powder.66 The size frequency of the ZnO grains is presented by Fig. 11(b) which is in the range of 7 to 26 μm. As observed, the maximum frequency of the sizes was concentrated between 13 and 14 μm which demonstrated the excellent enhancement of grain size in the optimized comparison of the starting powder.
 |
| | Fig. 11 The microstructure of the ceramic core in the low voltage varistor: (a) FESEM micrographs, (b) ZnO grain size distribution. | |
Elemental analysis of the ceramic core was also investigated. An XRD pattern was reported according to the reference code such as 00-005-0664, 00-008-0258, 00-034-0097, and 00-025-1164 (Fig. 12). The XRD detected the element antimony which was not detected by EDX analysis of the starting powder.
 |
| | Fig. 12 The XRD pattern of the ceramic core used in the optimized varistor. | |
Conclusions
In this work, the fabrication of the used ceramic in a voltage dependent varistor was designed and then experiments were performed to calculate its non-linear coefficients as output actual responses. The responses were used to obtain the appropriate model for the fabrication by RSM and ANN semi-empirical methods. The obtained models were carefully validated by mathematical, statistical and experimental evidences. Then the models were used to determine the importance of the effective additives and the selected levels of the initial additives were confirmed in an experimental design along with the optimum points of the additives which were able to maximize the quality of the varistors. Thereafter, the collected results of the two models were compared to select the final varistor. As a result of the comparison, the highest quality protection, 28.1, was provided by the varistor which was made of Bi2O3 (0.5 mol%), TiO2 (0.47 mol%) and Sb2O3 (0.21 mol%). Therefore, the modelling and optimization successfully predicted the quit high protective varistor, free of mathematical and physical complexity, which has industrial scale-up potential to produce highly protected electronics with which to control global e-waste.
Acknowledgements
The authors wish to acknowledge the University of Malaya-Ministry of Higher Education Grant UM.C/625/1/HIR/MOHE/ENG/29, University of Malaya Research Grant (UMRG) RP014C-13AET, Fundamental Research Grant Scheme (FRGS) with project no: FP011-2014A and Science Fund with project no: SF020-2013.
Notes and references
- S. B. Wath, P. Dutt and T. Chakrabarti, Environ. Monit. Assess., 2011, 172, 249–262 CrossRef PubMed.
- B. H. Robinson, Sci. Total Environ., 2009, 408, 183–191 CrossRef CAS PubMed.
- R. B. Standler, Protection of electronic circuits from overvoltages, Courier Dover Publications, 2012 Search PubMed.
- L. R. Avery, in Secondary, Google Patents, no. US4405933 A, 1983.
- R. Einzinger, Appl. Surf. Sci., 1978, 1, 329–340 CrossRef CAS.
- C.-W. Nahm, Mater. Sci. Semicond. Process., 2013, 16, 1308–1315 CrossRef CAS PubMed.
- L. J. Bowen and F. J. Avella, J. Appl. Phys., 1983, 54, 2764–2772 CrossRef CAS PubMed.
- K. Brown, IAEI News, March, 2004.
- F. Greuter and G. Blatter, Semicond. Sci. Technol., 1999, 5, 111 CrossRef.
- E. Olsson, L. Falk, G. Dunlop and R. Österlund, J. Mater. Sci., 1985, 20, 4091–4098 CrossRef CAS.
- H. Ö. Toplan and Y. Karakaş, Ceram. Int., 2001, 27, 761–765 CrossRef CAS.
- D. R. Clarke, J. Am. Ceram. Soc., 1999, 82, 485–502 CrossRef CAS PubMed.
- T. K. Gupta, J. Mater. Res., 1992, 7, 3280–3295 CrossRef CAS.
- J. Wong, J. Appl. Phys., 1980, 51, 4453–4459 CrossRef CAS PubMed.
- W. H. Pan, S. T. Kuo, W. H. Tuan and H. R. Chen, Int. J. Appl. Ceram. Technol., 2010, 7, E80–E88 CrossRef PubMed.
- C. Zhang, D. Zhou, W. Lu and Y. Hu, J. Mater. Sci.: Mater. Electron., 2001, 12, 357–360 CrossRef CAS.
- S. Ezhilvalavan and T. Kutty, Mater. Chem. Phys., 1997, 49, 258–269 CrossRef CAS.
- P. Mantas and J. Baptista, J. Eur. Ceram. Soc., 1995, 15, 605–615 CrossRef CAS.
- K. Eda, J. Appl. Phys., 1978, 49, 2964–2972 CrossRef CAS PubMed.
- T. K. Gupta, J. Am. Ceram. Soc., 1990, 73, 1817–1840 CrossRef CAS PubMed.
- G. Blatter and F. Greuter, Phys. Rev. B: Condens. Matter Mater. Phys., 1986, 33, 3952 CrossRef.
- A. Sedky, T. Ali and S. Mohamed, J. Phys. Chem. Solids, 2011, 73, 505–510 CrossRef PubMed.
- A. Banerjee, T. Ramamohan and M. Patni, Mater. Res. Bull., 2001, 36, 1259–1267 CrossRef CAS.
- M.-h. Wang, C. Yao and N.-f. Zhang, J. Mater. Process. Technol., 2008, 202, 406–411 CrossRef CAS PubMed.
- M. Inada, Jpn. J. Appl. Phys., 1980, 19, 409 CrossRef CAS.
- E. Olsson, G. Dunlop and R. Österlund, J. Am. Ceram. Soc., 1993, 76, 65–71 CrossRef CAS PubMed.
- R. Metz, H. Delalu, J. Vignalou, N. Achard and M. Elkhatib, Mater. Chem. Phys., 2000, 63, 157–162 CrossRef CAS.
- S. Ezhilvalavan and T. R. N. Kutty, Mater. Chem. Phys., 1997, 49, 258–269 CrossRef CAS.
- L. Levinson and H. Philipp, Am. Ceram. Soc. Bull., 1986, 65, 639 CAS.
- C. Zhang, Y. Hu, W. Lu, M. Cao and D. Zhou, J. Eur. Ceram. Soc., 2002, 22, 61–65 CrossRef CAS.
- H. Feng, Z. Peng, X. Fu, Z. Fu, C. Wang, L. Qi and H. Miao, J. Alloys Compd., 2010, 497, 304–307 CrossRef CAS PubMed.
- M. Peiteado, M. De la Rubia, M. Velasco, F. Valle and A. Caballero, J. Eur. Ceram. Soc., 2005, 25, 1675–1680 CrossRef CAS PubMed.
- J. Wong, J. Appl. Phys., 1980, 51, 4453–4459 CrossRef CAS PubMed.
- T. Senda and R. C. Bradt, J. Am. Ceram. Soc., 1990, 73, 106–114 CrossRef CAS PubMed.
- D. C. Montgomery, Design and analysis of experiments, John Wiley & Sons Inc, New York, 2008 Search PubMed.
- Y. Abdollahi, A. Zakaria, A. Abdul Halim, M. Hamid Reza Fard, J. Hossein, K. Shameli, R. Majid, B. Santo and A. Tahereh, Chem. Cent. J., 2012, 6, 1–8 CrossRef PubMed.
- Y. Abdollahi, Z. Azmi, M. Khamirul Amin, S. Kamyar, J. Hossein, R. Majid and A. Tahereh, Chem. Cent. J., 2012, 6, 1–5 CrossRef PubMed.
- Y. Abdollahi, Z. Azmi, R. Aziz, S. Tamili, K. A. Matori, N. Shahrani, N. M. Sidek, M. Dorraj and S. Moosavi, Chem. Cent. J., 2013, 7, 137–143 CrossRef PubMed.
- Y. Abdollahi, Z. Azmi, A. Mina, M. Hamid Reza Fard, M. Mansour Ghaffari, M. Khamirul Amin, J. Hossein and K. Ashkan, Chem. Cent. J., 2013, 7, 1–7 CrossRef PubMed.
- Y. Abdollahi, N. A. Sairi, M. K. Aroua, H. R. F. Masoumi, H. Jahangirian and Y. Alias, J. Ind. Eng. Chem., 2014 DOI:10.1016/j.jiec.2014.10.029.
- Y. Abdollahi, A. Zakaria, N. A. Sairi, K. Amin Matori, H. R. Fard Masoumi, A. R. Sadrolhosseini and H. Jahangirian, Sci. World J., 2014, 2014 Search PubMed.
- S. Bernik, P. ZupanÄiÄ and D. Kolar, J. Eur. Ceram. Soc., 1999, 19, 709–713 CrossRef CAS.
- D. Makovec, D. Kolar and M. Trontelj, Mater. Res. Bull., 1993, 28, 803–811 CrossRef CAS.
- Y. Li, G. Li and Q. Yin, Mater. Sci. Eng., B, 2006, 130, 264–268 CrossRef CAS PubMed.
- Q. Wang, Y. Qin, G. Xu, L. Chen, Y. Li, L. Duan, Z. Li, Y. Li and P. Cui, Ceram. Int., 2008, 34, 1697–1701 CrossRef CAS PubMed.
- D. Xu, X.-n. Cheng, X.-h. Yan, H.-x. Xu and L.-y. Shi, Trans. Nonferrous Met. Soc. China, 2009, 19, 1526–1532 CrossRef CAS.
- L. Cheng, G. Li, K. Yuan, L. Meng and L. Zheng, J. Am. Ceram. Soc., 2012, 95, 1004–1010 CAS.
- M. J. Anderson and P. J. Whitcomb, RSM simplified: optimizing processes using response surface methods for design of experiments, Productivity Press, Virginia, 2005 Search PubMed.
- G. W. Oehlert, WH Freeman, A first course in design and analysis of experiments, New York, 2000, vol. 1 Search PubMed.
- M. Sanchooli and M. Ghaffari Moghaddam, J. Chem. Eng. Jpn., 2012, 45, 373–379 CrossRef CAS.
- M. G. Moghaddam and M. Khajeh, Food Nutr., 2011, 2, 803–808 CrossRef CAS.
- S. Deming, CHEMTECH, 1989, 19, 249–255 Search PubMed.
- P. J. Whitcomb and M. J. Anderson, RSM simplified: optimizing processes using response surface methods for design of experiments, Productivity Press, New York, 2004 Search PubMed.
- R. Sen and T. Swaminathan, Biochem. Eng. J., 2004, 21, 141–148 CrossRef CAS PubMed.
- F. Despagne and D. L. Massart, Analyst, 1998, 123, 157–178 RSC.
- V. Pareek, M. Brungs, A. Adesina and R. Sharma, J. Photochem. Photobiol., A, 2002, 149, 139–146 CrossRef CAS.
- A. Ghaffari, H. Abdollahi, M. Khoshayand, I. S. Bozchalooi, A. Dadgar and M. Rafiee-Tehrani, Int. J. Pharm., 2006, 327, 126–138 CrossRef CAS PubMed.
- D. Baş and İ. H. Boyacı, J. Food Eng., 2007, 78, 846–854 CrossRef PubMed.
- E. Jorjani, S. Chehreh Chelgani and S. Mesroghli, Fuel, 2008, 87, 2727–2734 CrossRef CAS PubMed.
- S. Göb, E. Oliveros, S. H. Bossmann, A. M. Braun, R. Guardani and C. A. Nascimento, Chem. Eng. Process., 1999, 38, 373–382 CrossRef.
- B. B. Mandelbrot, Science, 1967, 156, 636–638 CAS.
- F. Souza, J. Gomes, P. Bueno, M. Cassia-Santos, A. Araujo, E. Leite, E. Longo and J. Varela, Mater. Chem. Phys., 2003, 80, 512–516 CrossRef CAS.
- Y.-W. Lao, S.-T. Kuo and W.-H. Tuan, Journal of Electroceramics, 2007, 19, 187–194 CrossRef CAS.
- R. H. Myers and C. M. Anderson-Cook, Response surface methodology: process and product optimization using designed experiments, Wiley. com, vol. 705, 2009 Search PubMed.
- S. Bernik, N. Daneu and A. Rečnik, J. Eur. Ceram. Soc., 2004, 24, 3703–3708 CrossRef CAS PubMed.
- J. Shi, Q. Cao, Y. Wei and Y. Huang, Mater. Sci. Eng., B, 2003, 99, 344–347 CrossRef.
|
| This journal is © The Royal Society of Chemistry 2015 |
Click here to see how this site uses Cookies. View our privacy policy here.